Tuesday, January 17, 2023 8:46:34 PM
I hear you.
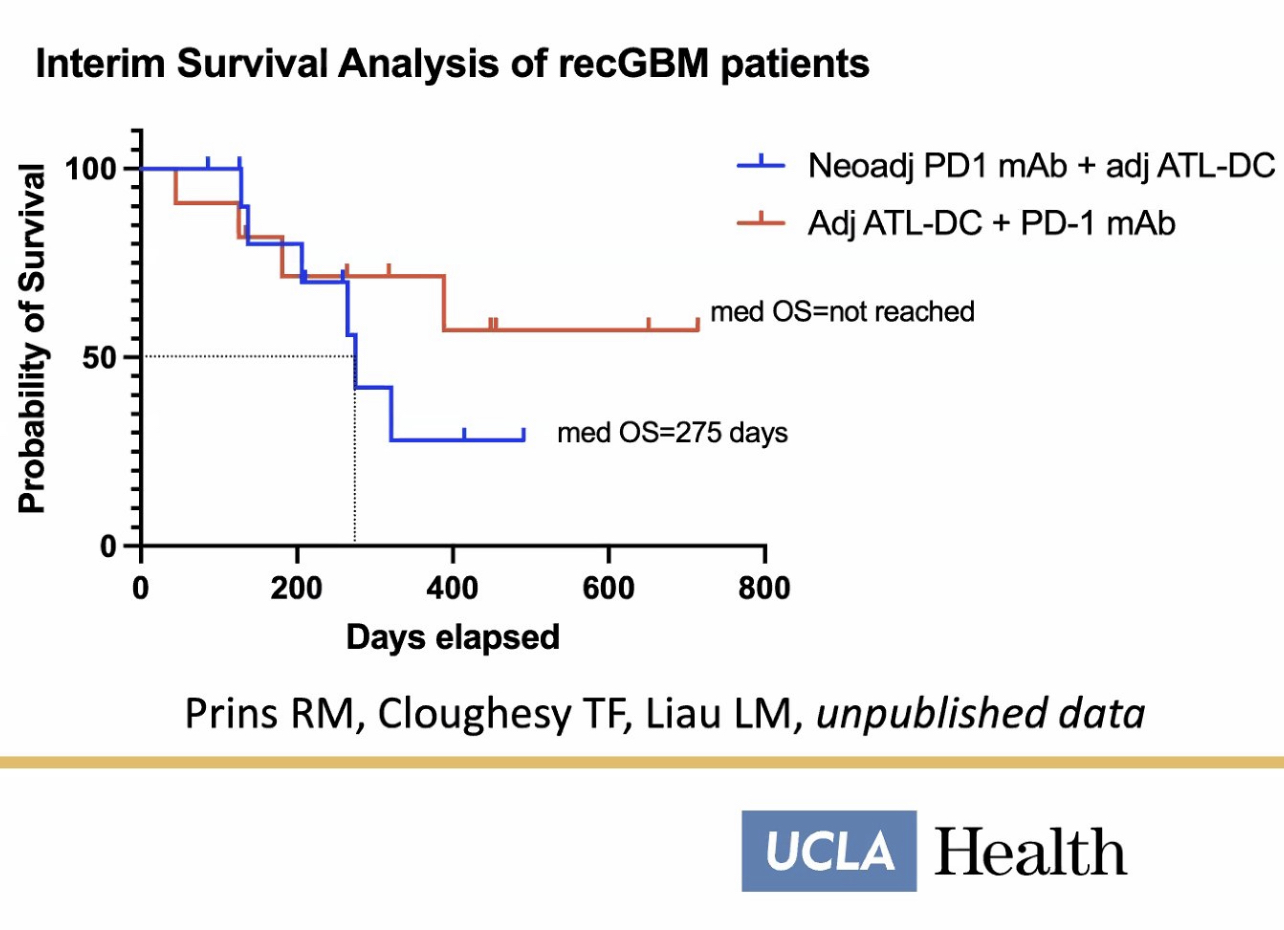
It sounds like you would prefer a completely different trial altogether. Murcidencel has been investigated in three successful trials—only the P3 relied on an ECA. I recommend the pembrolizumab combo study at UCLA supported by the NIH and Merck. The interim survival data are astonishing so far at the midpoint of this study.
https://clinicaltrials.gov/ct2/show/NCT04201873
The combo research is briefly highlighted by Dr. Toms at timestamp 6:30:
The placebo group for the combo trial are all receiving murcidencel (DCVax-L) as if it were SoC already.


https://cancer.ucla.edu/research/ucla-brain-spore/research-projects
Murcidencel (DCVax-L) is discussed beginning at minute 40, to focus on Keytruda (pembrolizumab) plus DCVax in combo at UCLA, skip to minute 45:40





https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/


https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter


The NIH is largely responsible for the development of the murcidencel cell-based platform technology as they have always supported and funded Liau’s research. The 20 consecutive years of NIH funding of Dr. Liau have played a direct and crucial role in murcidencel. Together with Merck, NIH is funding the murcidencel doses going into arms TODAY in a study investigating its efficacy in combo with pembrolizumab.

It sounds like you would prefer a completely different trial altogether. Murcidencel has been investigated in three successful trials—only the P3 relied on an ECA. I recommend the pembrolizumab combo study at UCLA supported by the NIH and Merck. The interim survival data are astonishing so far at the midpoint of this study.
https://clinicaltrials.gov/ct2/show/NCT04201873
The combo research is briefly highlighted by Dr. Toms at timestamp 6:30:
The placebo group for the combo trial are all receiving murcidencel (DCVax-L) as if it were SoC already.



https://cancer.ucla.edu/research/ucla-brain-spore/research-projects
Murcidencel (DCVax-L) is discussed beginning at minute 40, to focus on Keytruda (pembrolizumab) plus DCVax in combo at UCLA, skip to minute 45:40





https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/


https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter



The NIH is largely responsible for the development of the murcidencel cell-based platform technology as they have always supported and funded Liau’s research. The 20 consecutive years of NIH funding of Dr. Liau have played a direct and crucial role in murcidencel. Together with Merck, NIH is funding the murcidencel doses going into arms TODAY in a study investigating its efficacy in combo with pembrolizumab.

Bullish
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








