Sunday, April 05, 2020 1:55:38 PM
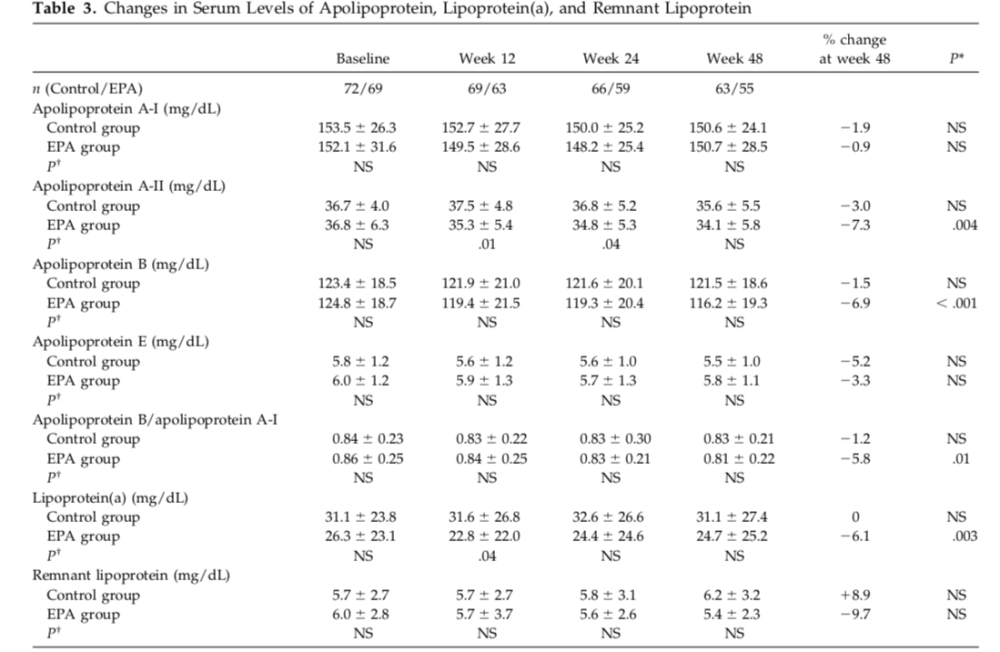
A few people have asked me to comment on this post. I don't have too much to add. mc1988 is exactly right-- there is no statistical change in ApoB levels reported relative to the control group. This is quite clear in Table 3 of Kurabayashi (pasted below).
It is interesting to note that Kurabayashi reports:
"The apolipoprotein B level in the eicosapentaenoic acid group was significantly lower at week 48 compared with the baseline level, but there was no significant difference between the groups."
So to be clear, Kurabayashi found NO statistically significant change at any of the timepoints between the control and treatment groups. But they did find a difference between treatment and baseline at week 48.
So, oddly, the flavor of error is similar to that made in Mori.
However, again, the correct comparison is clearly BETWEEN groups here (and is even more important, IMO). Why? Because the treatment group received E3 plus EPA and the control group received E3 only. So it is critical to compare between groups, because you have to disentangle the effects of E3 from the effects of EPA.
This error on Du's part seems a little more disingenuous than the Mori error, since Mori did not do the between group comparison (as they were not testing that hypothesis), whereas here, Kurabayashi absolutely did the comparison and reported it. It's almost as if she were cherry picking the paper to support her conclusion.
Edit: I am NOT suggesting Du was not acting with honorable intent. I find it likely that she was. What I am suggesting is that she made up her mind that the patent was invalid and was looking for reasons to support this conclusion. I am not a lawyer, but it is my understanding that the method that she went about her reasoning is important, and I think this mistake may add some color to that methodology.
It is interesting to note that Kurabayashi reports:
"The apolipoprotein B level in the eicosapentaenoic acid group was significantly lower at week 48 compared with the baseline level, but there was no significant difference between the groups."
So to be clear, Kurabayashi found NO statistically significant change at any of the timepoints between the control and treatment groups. But they did find a difference between treatment and baseline at week 48.
So, oddly, the flavor of error is similar to that made in Mori.
However, again, the correct comparison is clearly BETWEEN groups here (and is even more important, IMO). Why? Because the treatment group received E3 plus EPA and the control group received E3 only. So it is critical to compare between groups, because you have to disentangle the effects of E3 from the effects of EPA.
This error on Du's part seems a little more disingenuous than the Mori error, since Mori did not do the between group comparison (as they were not testing that hypothesis), whereas here, Kurabayashi absolutely did the comparison and reported it. It's almost as if she were cherry picking the paper to support her conclusion.
Edit: I am NOT suggesting Du was not acting with honorable intent. I find it likely that she was. What I am suggesting is that she made up her mind that the patent was invalid and was looking for reasons to support this conclusion. I am not a lawyer, but it is my understanding that the method that she went about her reasoning is important, and I think this mistake may add some color to that methodology.

Recent AMRN News
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 04/02/2026 08:30:11 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 03/30/2026 09:57:06 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 03/27/2026 09:00:07 PM
- Amarin Highlights Guideline Recommended Role of Icosapent Ethyl in Managing Cardiovascular Risk Following Release of Updated 2026 ACC/AHA/Multisociety Dyslipidemia Guideline • GlobeNewswire Inc. • 03/18/2026 12:30:00 PM
- New REDUCE-IT Data in Patients at Extreme Cardiovascular Risk and In Vitro Research on the Mechanistic Effects of Eicosapentaenoic Acid (EPA) on Lipoprotein(a) [Lp(a)] Oxidation to be Presented at the American College of Cardiology’s (ACC) Annual Scienti • GlobeNewswire Inc. • 03/16/2026 12:15:00 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 03/02/2026 10:31:01 PM
- Effects of Icosapent Ethyl on Risk and Duration of Hospitalizations and Death in REDUCE-IT® Post Hoc Analysis Published in the European Journal of Preventive Cardiology • GlobeNewswire Inc. • 03/02/2026 01:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/25/2026 12:05:26 PM
- Amarin Reports Fourth Quarter and Full Year 2025 Financial Results • GlobeNewswire Inc. • 02/25/2026 12:00:00 PM
- This American Heart Month Amarin Spotlights the Need to Prioritize Proven Widely Available Yet Underutilized Therapies in the Battle Against Cardiovascular Disease • GlobeNewswire Inc. • 02/23/2026 01:00:00 PM
- Amarin to Report Fourth Quarter and Full Year 2025 Financial Results and Host Conference Call on February 25, 2026 • GlobeNewswire Inc. • 02/11/2026 01:00:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/04/2026 12:00:09 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/04/2026 12:00:07 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/04/2026 12:00:06 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/04/2026 12:00:05 AM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 02/04/2026 12:00:04 AM
- Amarin Applauds Breakthroughs In Therapies For Patients With Elevated Triglycerides; Company’s VASCEPA®/VAZKEPA® (Icosapent Ethyl) Franchise Well Positioned To Benefit Globally From Broadened Category Commercialization • GlobeNewswire Inc. • 01/09/2026 12:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/08/2026 12:05:27 PM
- Amarin Announces Preliminary 2025 Financial Highlights and Operational Accomplishments, Including Achieving Positive Cash Flow; Company Well Positioned to Capture Global Growth Opportunities • GlobeNewswire Inc. • 01/08/2026 12:00:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/06/2026 10:00:21 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/06/2026 10:00:17 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/06/2026 10:00:10 PM
- New REDUCE-IT® Aspirin Analysis Presented at AHA Scientific Sessions 2025 Reinforces VASCEPA®/VAZKEPA® (Icosapent Ethyl) Reduced Cardiovascular Events in High-Risk Patients • GlobeNewswire Inc. • 11/09/2025 09:15:00 PM
- Amarin To Present New Analyses Affirming Robustness of Data from REDUCE-IT and Additional Mechanisms of Action of Eicosapentaenoic Acid (EPA) at American Heart Association (AHA) Scientific Sessions 2025 • GlobeNewswire Inc. • 11/03/2025 01:30:00 PM
- Form 3 - Initial statement of beneficial ownership of securities • Edgar (US Regulatory) • 10/31/2025 08:30:06 PM








