Wednesday, March 13, 2024 1:40:37 PM
sentiment_stocks, I do wholeheartedly agree that once Flaskworks devices are approved to be implemented into the DCVax-L manufacturing process, that will significantly decrease the cost to manufacture DCVax-L and it will also decrease the cost of DCVax-L for patients.
However, as you know, when Advent, Charles River Laboratories (CRL), or any other CDMO working with NWBio to manufacture DCVax-L, all the patients’ dosages (up to 8 to 10 years of doses) are made upfront in 1 batch process. NWBio will be required by their CDMO to pay for all the doses made for each patient regardless.
I believe that the reasons that NWBio allowed patients to pay for doses in installments in the Specials Program are:
(1) DCVax-L is not covered by the NHS national insurance in the UK, because it has not been approved by the MHRA yet. As a result, patients are having to fund-raise to pay for DCVax-L out-of-pocket. The funds that they are raising are coming in incrementally over time, and NWBio wants these patients to be able to start their treatment on DCVax-L ASAP.
(2) NWBio and Advent wanted as many patients in the UK as possible to be treated in the UK to help with the Flaskworks comparability studies. By allowing these patients to pay for DCVax-L in installments, that was a win-win for the patients, NWBio, Flaskworks and Advent.
However, one fallacy (lie) that shorts and other NWBio detractors like to promote is that DCVax-L will cost too much for regulators to approve, and for patients to afford.
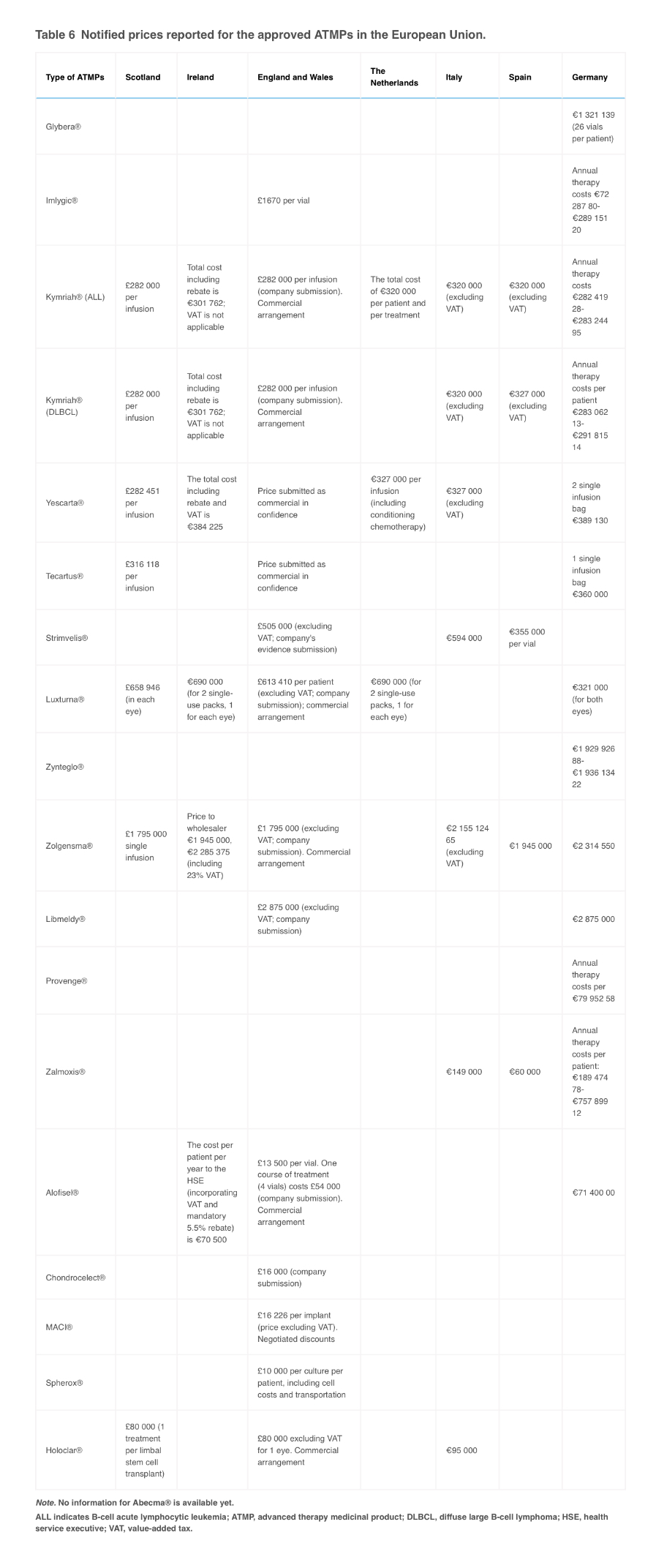
The truth is, even at a total cost of $250,000 for up to 8 to 10 years of DCVax-L doses. DCVax-L would be 1 of the least expensive (cheapest) Advanced Therapy Medical Products (ATMPs) on the market in the UK, US, EU, Canada and the rest of the world (ROW).
If you assume a patient will use all of their DCVax-L doses over 8 years, the average annual cost would be $31,250. If you assume a patient will use all of their DCVax-L doses over 10 years, the average annual cost would be $25,000 per year.
The regulators will look at the average annual cost of DCVax-L compared to other ATMPs. Even if you assume the malignant brain tumor patients will only use their DCVax-L doses for up to 3 years, instead of 8 to 10 years, the average annual cost of DCVax-L for $250,000, over 3 years would only be $83,333 per year.
Compare that to the cost of the other approved ATMPs in the UK and the EU in the table below, and you will see that even at a total cost of $250,000, DCVax-L is 1 of the least expensive (cheapest) ATMPs:
https://www.valueinhealthjournal.com/action/showFullTableHTML?isHtml=true&tableId=tbl6&pii=S1098-3015%2823%2900005-0
However, as you know, when Advent, Charles River Laboratories (CRL), or any other CDMO working with NWBio to manufacture DCVax-L, all the patients’ dosages (up to 8 to 10 years of doses) are made upfront in 1 batch process. NWBio will be required by their CDMO to pay for all the doses made for each patient regardless.
I believe that the reasons that NWBio allowed patients to pay for doses in installments in the Specials Program are:
(1) DCVax-L is not covered by the NHS national insurance in the UK, because it has not been approved by the MHRA yet. As a result, patients are having to fund-raise to pay for DCVax-L out-of-pocket. The funds that they are raising are coming in incrementally over time, and NWBio wants these patients to be able to start their treatment on DCVax-L ASAP.
(2) NWBio and Advent wanted as many patients in the UK as possible to be treated in the UK to help with the Flaskworks comparability studies. By allowing these patients to pay for DCVax-L in installments, that was a win-win for the patients, NWBio, Flaskworks and Advent.
However, one fallacy (lie) that shorts and other NWBio detractors like to promote is that DCVax-L will cost too much for regulators to approve, and for patients to afford.
The truth is, even at a total cost of $250,000 for up to 8 to 10 years of DCVax-L doses. DCVax-L would be 1 of the least expensive (cheapest) Advanced Therapy Medical Products (ATMPs) on the market in the UK, US, EU, Canada and the rest of the world (ROW).
If you assume a patient will use all of their DCVax-L doses over 8 years, the average annual cost would be $31,250. If you assume a patient will use all of their DCVax-L doses over 10 years, the average annual cost would be $25,000 per year.
The regulators will look at the average annual cost of DCVax-L compared to other ATMPs. Even if you assume the malignant brain tumor patients will only use their DCVax-L doses for up to 3 years, instead of 8 to 10 years, the average annual cost of DCVax-L for $250,000, over 3 years would only be $83,333 per year.
Compare that to the cost of the other approved ATMPs in the UK and the EU in the table below, and you will see that even at a total cost of $250,000, DCVax-L is 1 of the least expensive (cheapest) ATMPs:
https://www.valueinhealthjournal.com/action/showFullTableHTML?isHtml=true&tableId=tbl6&pii=S1098-3015%2823%2900005-0

Bullish
Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM









