Monday, January 22, 2024 3:32:24 PM
sukus,
As a matter of fact, the freezer facilities at Merck are not the reason that makes believe the potential partnership. There are two main things that are really interesting to me.
First, it was the back and forth contacts between Merck and Timothy Cloughesy after BMY got the combo trial and was ready to start the trial. Then after the last contact with Timothy Cloughesy in March 2021, there was no other contact at least until the end of 2023.
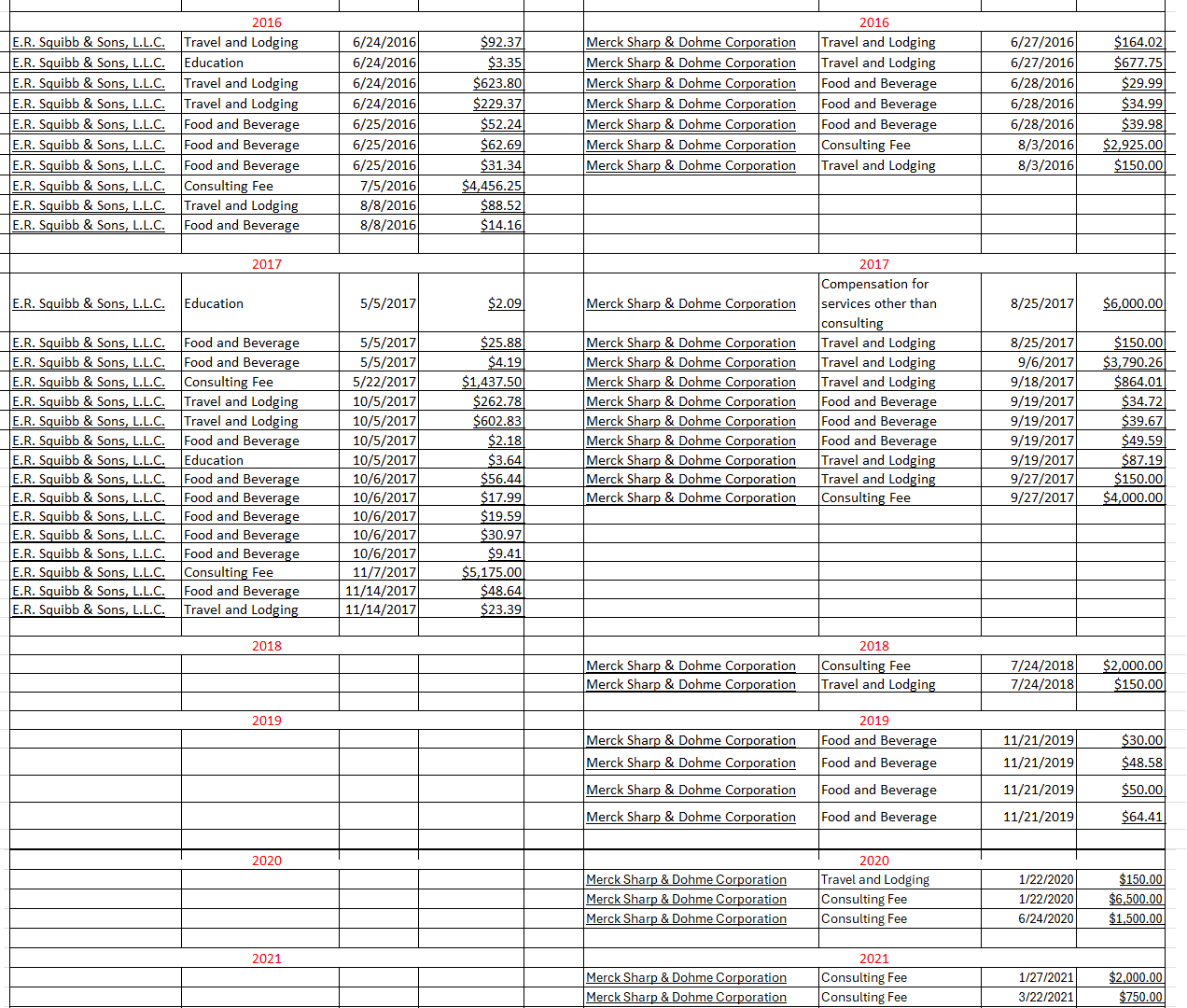
Second, Merck struck the deal with Daiichi claiming it was because of Daiichi's ADCs. Did you check the depth and breadth of the collaboration between Merck and Seagn? Take a look at the data on ADCs from the Seagen's trials and make a comparison with the ones from Daiichi. You will know Merck's true motive.
As a matter of fact, the freezer facilities at Merck are not the reason that makes believe the potential partnership. There are two main things that are really interesting to me.
First, it was the back and forth contacts between Merck and Timothy Cloughesy after BMY got the combo trial and was ready to start the trial. Then after the last contact with Timothy Cloughesy in March 2021, there was no other contact at least until the end of 2023.
Second, Merck struck the deal with Daiichi claiming it was because of Daiichi's ADCs. Did you check the depth and breadth of the collaboration between Merck and Seagn? Take a look at the data on ADCs from the Seagen's trials and make a comparison with the ones from Daiichi. You will know Merck's true motive.

Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








