Wednesday, May 31, 2023 8:47:26 PM
iclight,
Thanks for your thoughts about commercial manufacturing. There has been some great news and discussion recently about manufacturing this cell-based pharmaceutical product at large scale. Capacity to manufacture is crucial and the acquisition of the Flaskworks technology was a brilliant move by the company. Thanks for highlighting these topics. I can’t wait to hear more news about manufacturing DCVax-L.
March 2023 Manufacturing News:


https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html

LP said that the UK facility can supply North America, but they will likely use the Memphis facility too depending on money/timing. Either facility should have enough capacity to stand alone, but they could use both.
They have been planning well to build the capacity to address a worldwide demand.
https://p.widencdn.net/ud1zkk/DS-TS-immunology-dendritic-cells-activation-assay
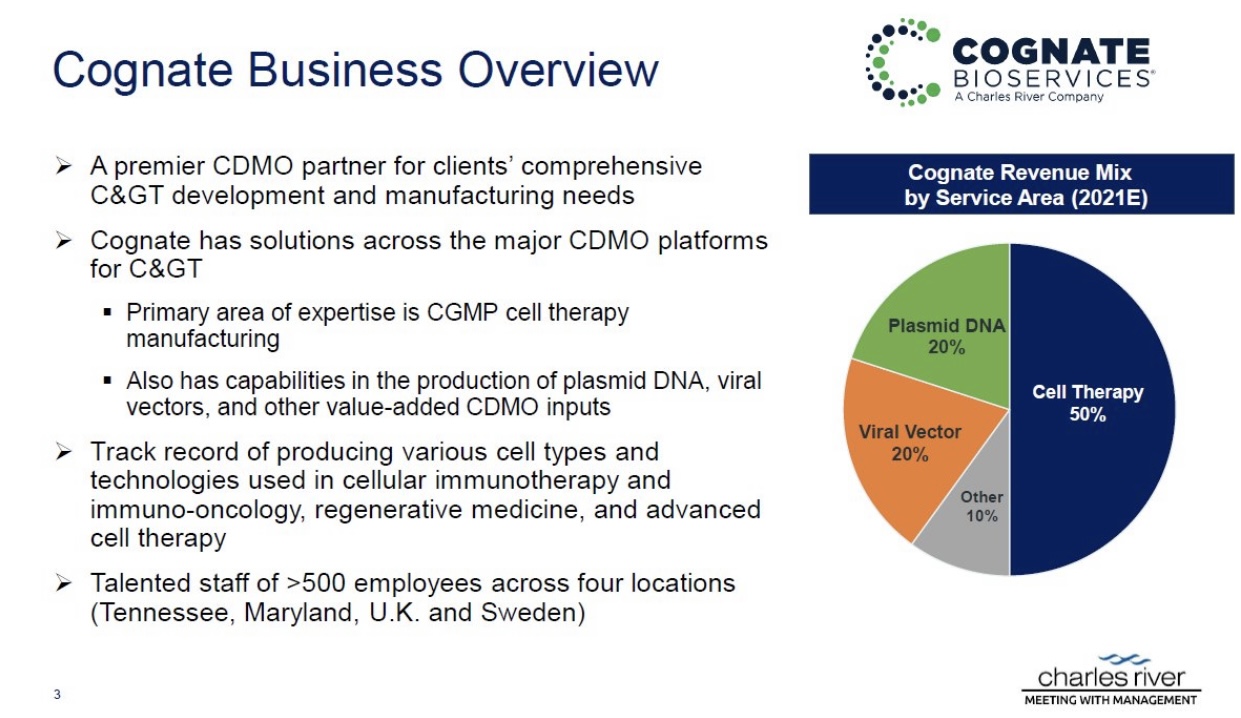
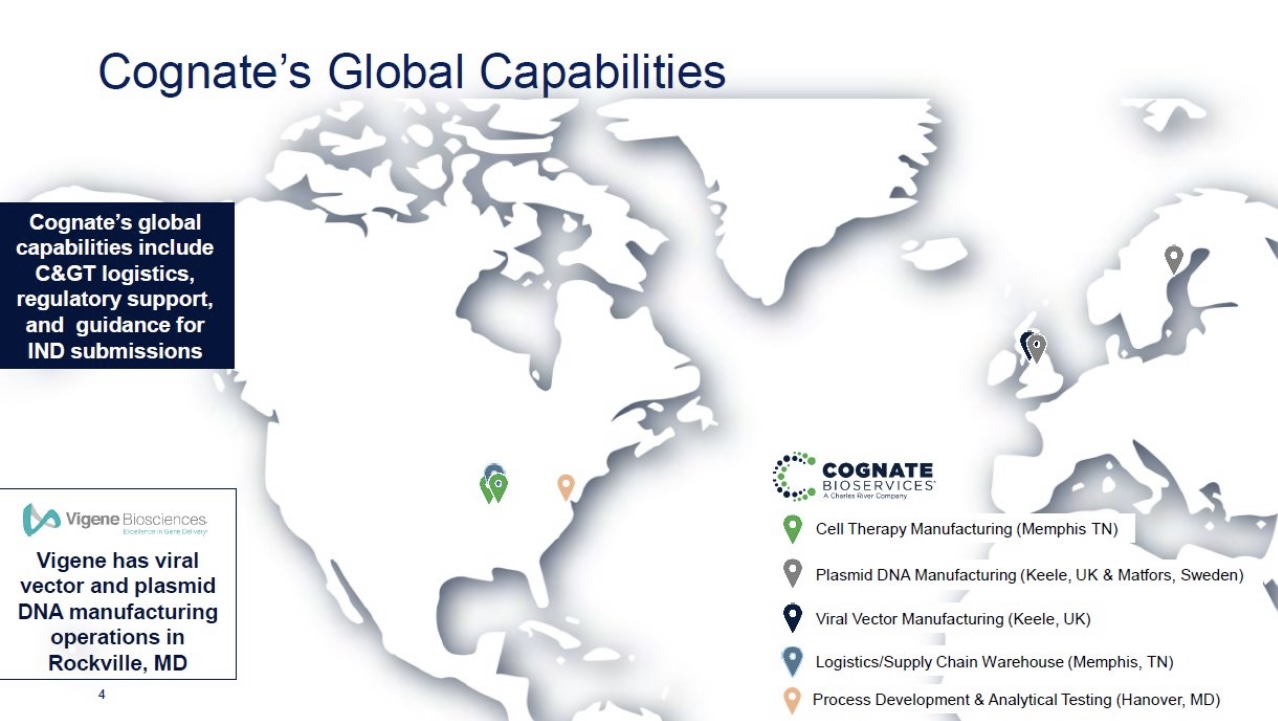
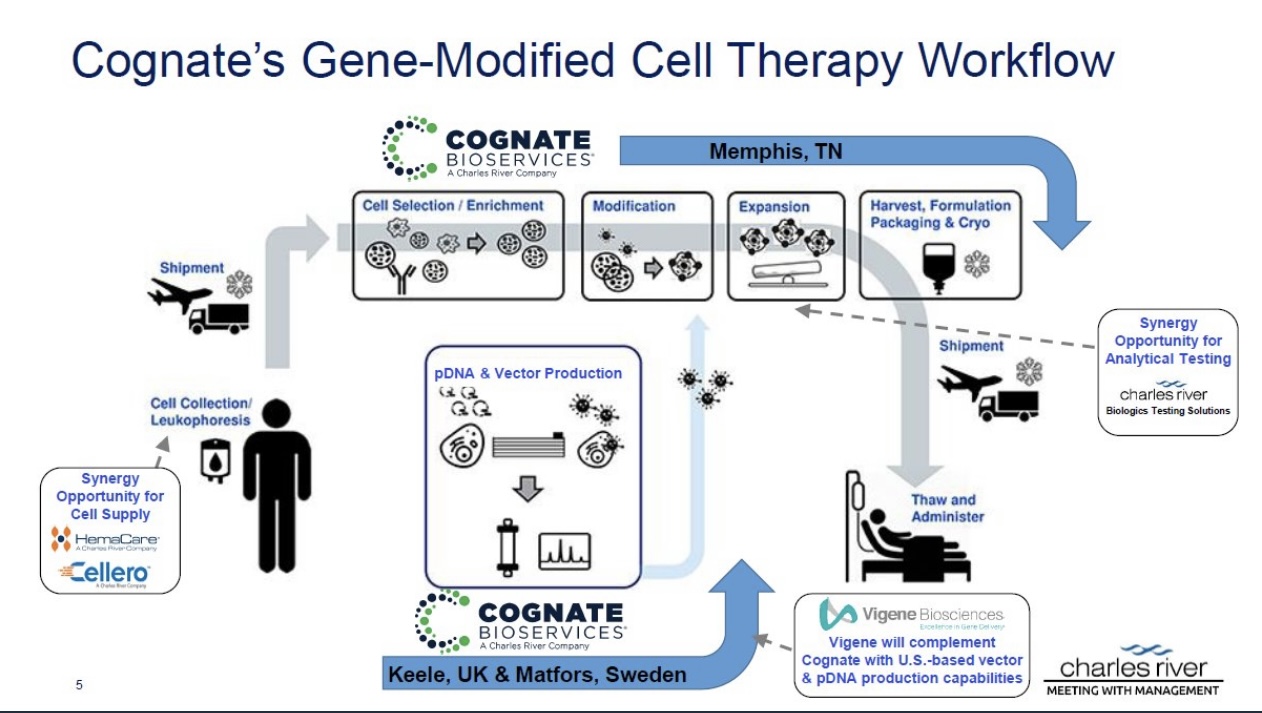

Manufacturing is also crucial when considering the vast possibility of combo therapies and the possibility of treating other cancers beyond GBM.

Don’t miss ASCO 2023! It’s starting this week!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171909324
https://clinicaltrials.gov/ct2/show/NCT04201873
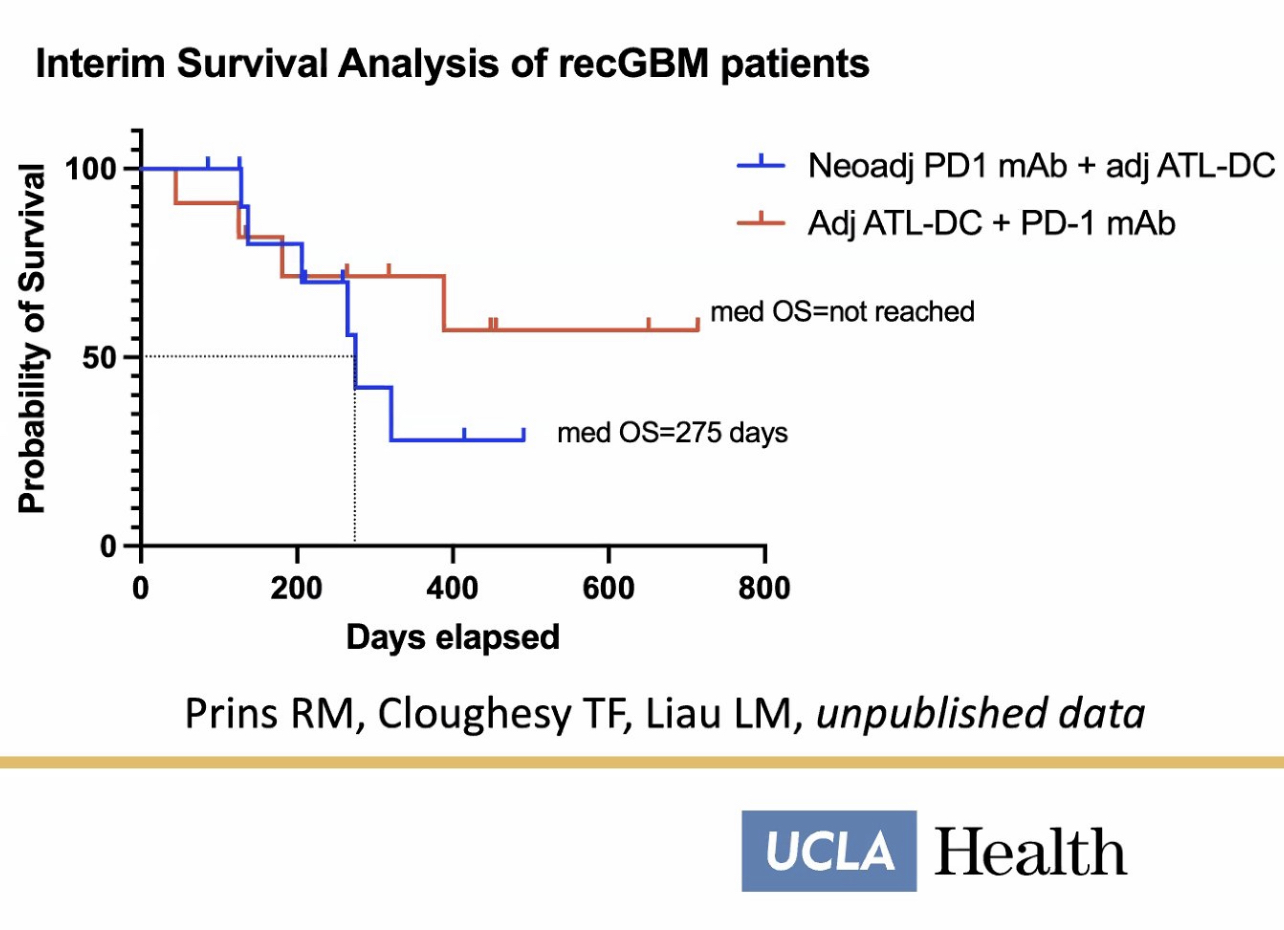

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Thanks for your thoughts about commercial manufacturing. There has been some great news and discussion recently about manufacturing this cell-based pharmaceutical product at large scale. Capacity to manufacture is crucial and the acquisition of the Flaskworks technology was a brilliant move by the company. Thanks for highlighting these topics. I can’t wait to hear more news about manufacturing DCVax-L.
March 2023 Manufacturing News:


https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html

LP said that the UK facility can supply North America, but they will likely use the Memphis facility too depending on money/timing. Either facility should have enough capacity to stand alone, but they could use both.
They have been planning well to build the capacity to address a worldwide demand.
https://p.widencdn.net/ud1zkk/DS-TS-immunology-dendritic-cells-activation-assay




Manufacturing is also crucial when considering the vast possibility of combo therapies and the possibility of treating other cancers beyond GBM.

Don’t miss ASCO 2023! It’s starting this week!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171909324
https://clinicaltrials.gov/ct2/show/NCT04201873


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Bullish
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








