Wednesday, May 31, 2023 4:38:34 PM
iclight,
You are mixed up and you might wish to consider better sources. Your post is curiously composed and misleading. Always remember to verify what you find posted online and seek trustworthy full-context sources.
The P3 data were validated by an independent peer review published in JAMA Oncology.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

The interim PD1 combo data are significant too.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM.
https://www.fda.gov/media/120721/download


https://clinicaltrials.gov/ct2/show/NCT04201873
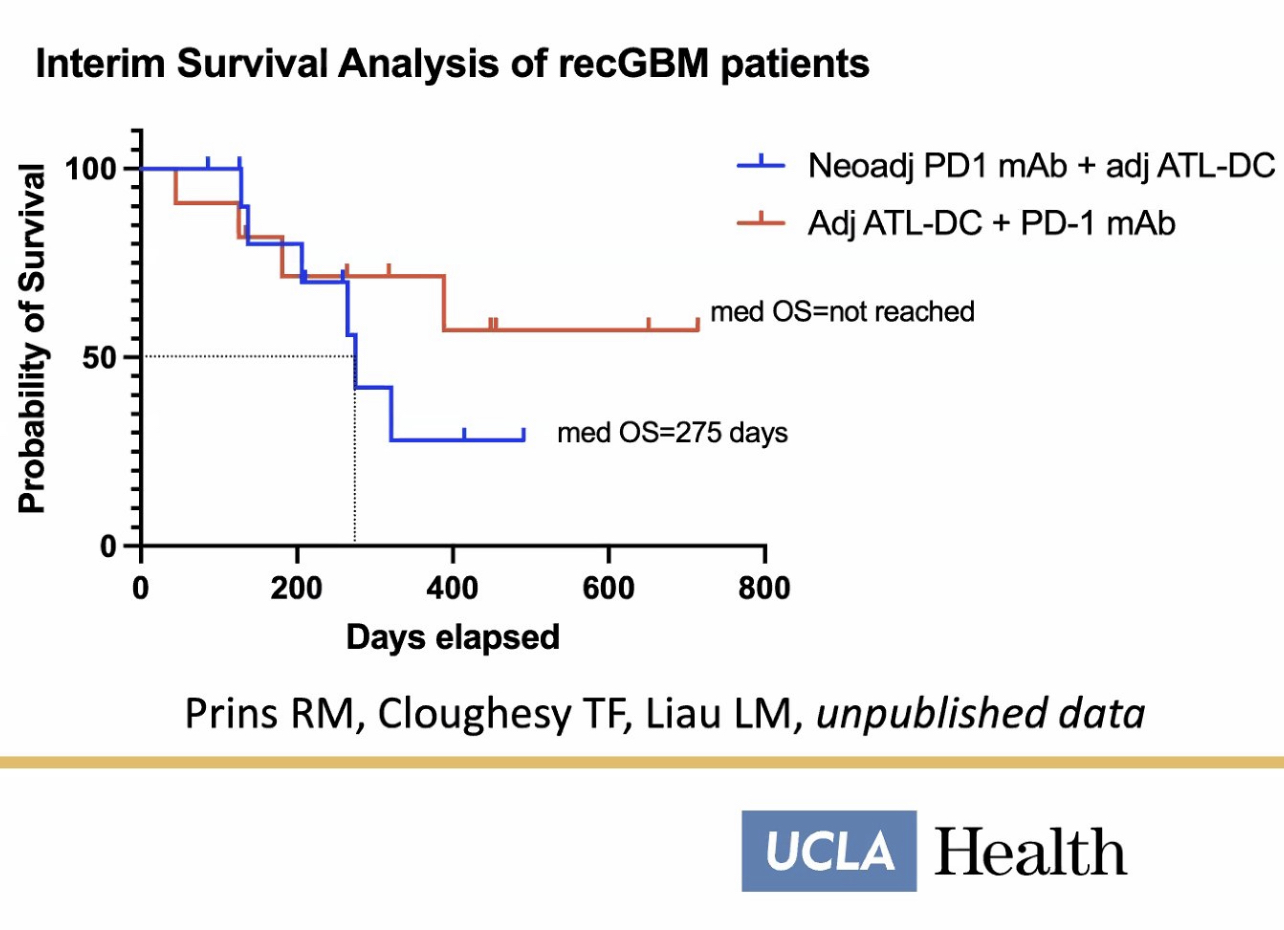
Bosch gave a great presentation recently. Here are some additional data highlights from the P3 and some additional data from compassionate use…
March 2023 presentation by Dr. Bosch:
http://nwbio.com/wp-content/uploads/NWBT-GBM-Summit-slides-3-15-23.pdf
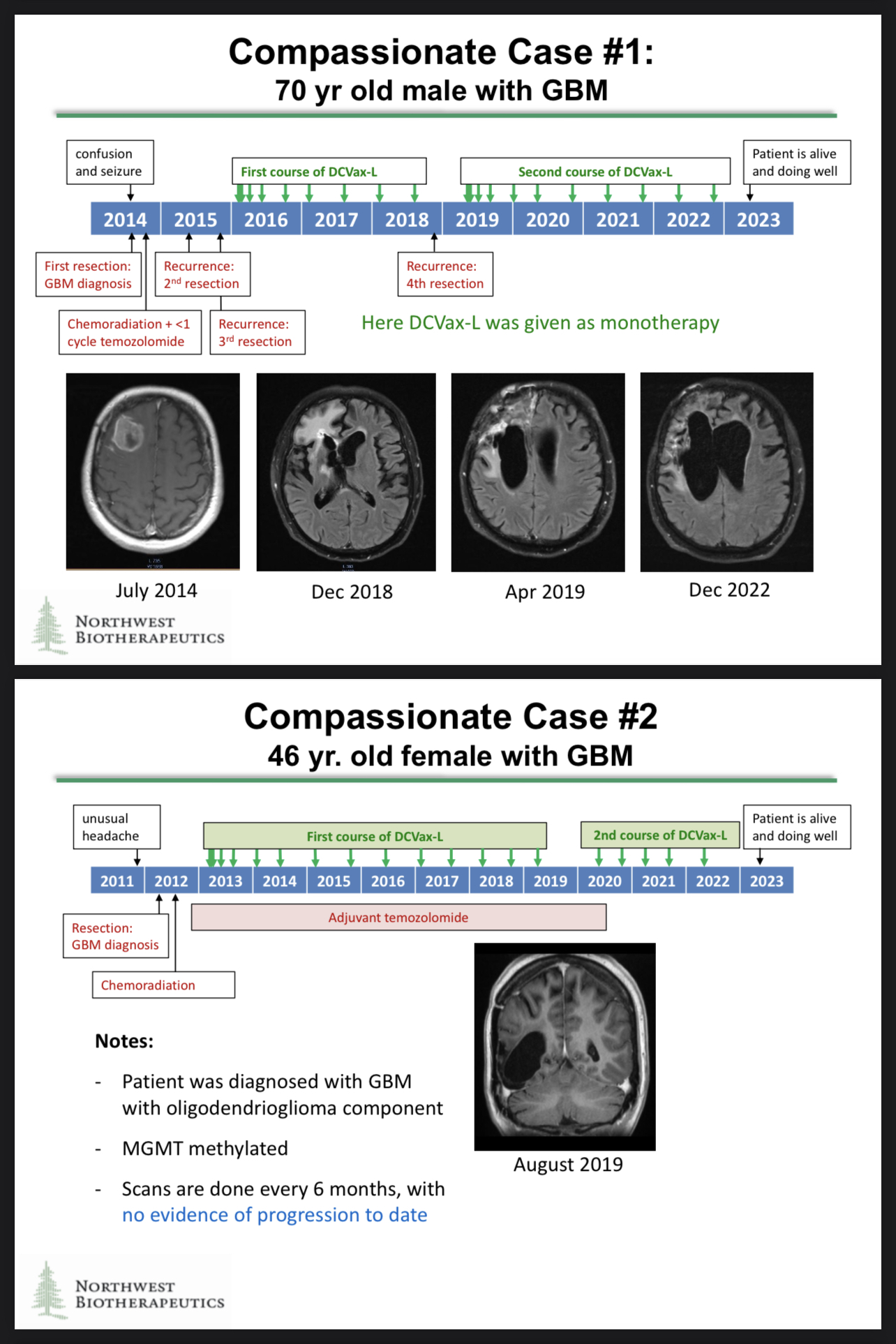

The P3 was brilliantly executed and is more than adequate all by itself, but the application(s) for approval will require lots of paper clips and plenty of staples for all the extra pages that will be included—there is a voluminous catalog of supportive data that has been collected, analyzed, and validated.
All clinical data are relevant to a regulatory application including those data gathered from other studies, interim analyses, and compassionate use. All the DCVax-L trials have produced substantial safety and efficacy data, and two of the three clinical trials ran for over a decade with survivors alive today.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


ASCO 2023
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171648758



https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter


You are mixed up and you might wish to consider better sources. Your post is curiously composed and misleading. Always remember to verify what you find posted online and seek trustworthy full-context sources.
The P3 data were validated by an independent peer review published in JAMA Oncology.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

The interim PD1 combo data are significant too.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM.
https://www.fda.gov/media/120721/download


https://clinicaltrials.gov/ct2/show/NCT04201873

Bosch gave a great presentation recently. Here are some additional data highlights from the P3 and some additional data from compassionate use…
March 2023 presentation by Dr. Bosch:
http://nwbio.com/wp-content/uploads/NWBT-GBM-Summit-slides-3-15-23.pdf


The P3 was brilliantly executed and is more than adequate all by itself, but the application(s) for approval will require lots of paper clips and plenty of staples for all the extra pages that will be included—there is a voluminous catalog of supportive data that has been collected, analyzed, and validated.
All clinical data are relevant to a regulatory application including those data gathered from other studies, interim analyses, and compassionate use. All the DCVax-L trials have produced substantial safety and efficacy data, and two of the three clinical trials ran for over a decade with survivors alive today.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


ASCO 2023
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171648758



https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter


Bullish
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








