Monday, January 16, 2023 3:02:00 PM
Thanks for your opinion about the regulators.
I agree that the regulators are crucial. In my opinion, the FDA and NIH are quite significant and NIH support for Dr. Liau’s work is noteworthy. The NIH has supported Liau’s work for 20 consecutive years.




https://cancer.ucla.edu/research/ucla-brain-spore/research-projects
https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://nwbio.com/wp-content/uploads/jamaoncology_liau_2022_oi_220066_1668698380.80695.pdf
September 14, 2022 Update of NIH NCI Funding of Dr. Liau
https://connect.uclahealth.org/2022/09/14/brain-cancer-discovery-clinical-trials/
9/14/2022
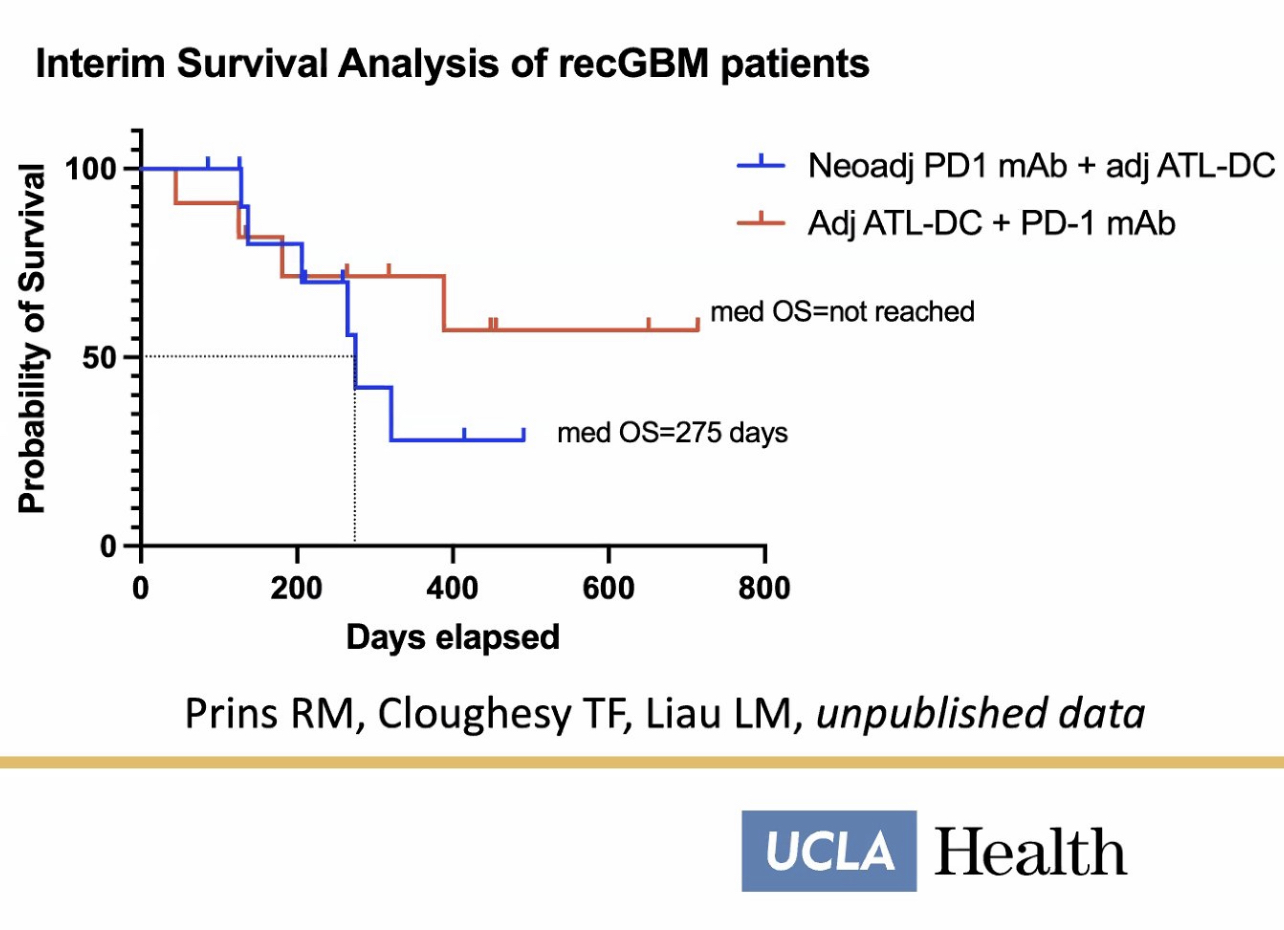
In a trial now underway, a dendritic cell vaccine is made from a participant's own tumor tissue and combined with an anti-PD1 immune checkpoint inhibitor to counter resistance when either treatment is used alone. The checkpoint drugs work by blocking the proteins that stop the immune system from attacking cancer cells.
"What I'm excited about is that we're seeing a growing number of long-term survivors in our patients treated with immunotherapy combinations," Dr. Liau says. "We're seeing some patients with certain combination immunotherapies that are living for many more years than would be expected. Currently, we're trying to find out what combination works best and for which patients."
The NIH is largely responsible for the development of the murcidencel cell-based platform technology as they have always supported and funded Liau’s research. The 20 consecutive years of NIH funding of Dr. Liau have played a direct and crucial role in murcidencel. Together with Merck, NIH is funding the murcidencel doses going into arms TODAY in a study investigating its efficacy in combo with pembrolizumab.
https://clinicaltrials.gov/ct2/show/NCT04201873



Dr. Toms briefly highlights the combo study at timestamp 6:30 of the video:

I agree that the regulators are crucial. In my opinion, the FDA and NIH are quite significant and NIH support for Dr. Liau’s work is noteworthy. The NIH has supported Liau’s work for 20 consecutive years.




https://cancer.ucla.edu/research/ucla-brain-spore/research-projects
https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://nwbio.com/wp-content/uploads/jamaoncology_liau_2022_oi_220066_1668698380.80695.pdf
September 14, 2022 Update of NIH NCI Funding of Dr. Liau
https://connect.uclahealth.org/2022/09/14/brain-cancer-discovery-clinical-trials/
9/14/2022
In a trial now underway, a dendritic cell vaccine is made from a participant's own tumor tissue and combined with an anti-PD1 immune checkpoint inhibitor to counter resistance when either treatment is used alone. The checkpoint drugs work by blocking the proteins that stop the immune system from attacking cancer cells.
"What I'm excited about is that we're seeing a growing number of long-term survivors in our patients treated with immunotherapy combinations," Dr. Liau says. "We're seeing some patients with certain combination immunotherapies that are living for many more years than would be expected. Currently, we're trying to find out what combination works best and for which patients."
The NIH is largely responsible for the development of the murcidencel cell-based platform technology as they have always supported and funded Liau’s research. The 20 consecutive years of NIH funding of Dr. Liau have played a direct and crucial role in murcidencel. Together with Merck, NIH is funding the murcidencel doses going into arms TODAY in a study investigating its efficacy in combo with pembrolizumab.
https://clinicaltrials.gov/ct2/show/NCT04201873




Dr. Toms briefly highlights the combo study at timestamp 6:30 of the video:

Bullish
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Northwest Biotherapeutics Appoints Dr. Annalisa Jenkins As Strategic Adviser To Advance Dendritic Cell Cancer Vaccine Platform • PR Newswire (US) • 04/30/2026 04:38:00 PM
- Northwest Biotherapeutics Appoints Dr. Annalisa Jenkins As Strategic Adviser To Advance Dendritic Cell Cancer Vaccine Platform • PR Newswire (US) • 04/30/2026 04:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








