Saturday, January 14, 2023 4:51:01 PM
Thanks for the reply.
No problem—it sounds like you would prefer a completely different trial from the P3. Take a look at the combo trial! It has a great placebo design that I bet you will love:
The UCLA combo study deserves more publicity and many people are unaware of its interim survival data.
For those who might not have heard:
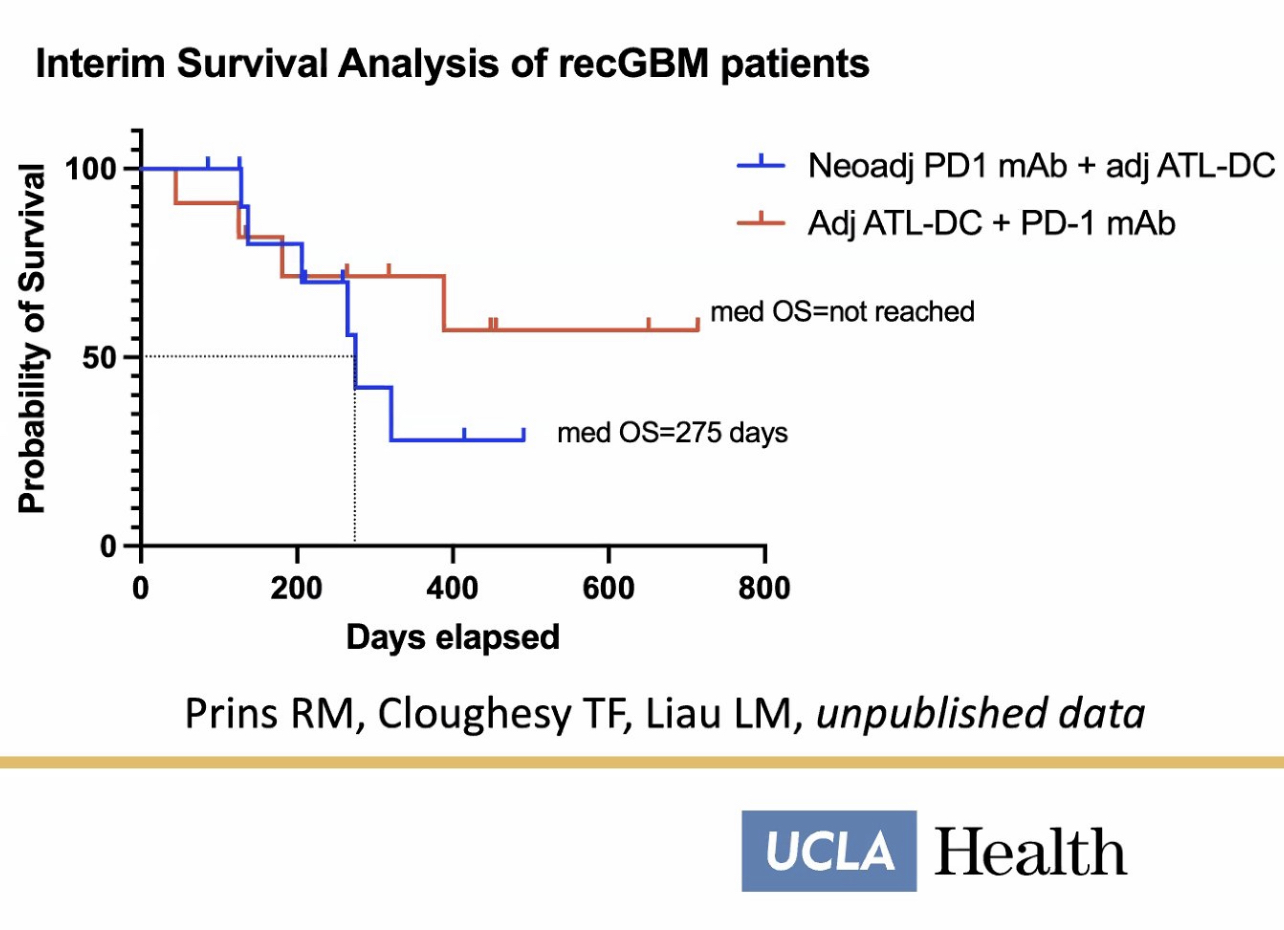
Aside from the well-publicized P3, there is another clinical study for murcidencel + pembrolizumab specifically for rGBM that has reached its midpoint with interim survival data. This study does not involve any chemo or radiation. OS has not been reached yet because patients are alive. This trial is using an internal placebo control group who are all receiving murcidencel—only Keytruda in combo is being investigated.
https://clinicaltrials.gov/ct2/show/NCT04201873






https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/

No problem—it sounds like you would prefer a completely different trial from the P3. Take a look at the combo trial! It has a great placebo design that I bet you will love:
The UCLA combo study deserves more publicity and many people are unaware of its interim survival data.
For those who might not have heard:
Aside from the well-publicized P3, there is another clinical study for murcidencel + pembrolizumab specifically for rGBM that has reached its midpoint with interim survival data. This study does not involve any chemo or radiation. OS has not been reached yet because patients are alive. This trial is using an internal placebo control group who are all receiving murcidencel—only Keytruda in combo is being investigated.
https://clinicaltrials.gov/ct2/show/NCT04201873







https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://connect.uclahealth.org/2021/03/22/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020/

Bullish
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








