Monday, April 26, 2021 12:40:06 AM
Thanks Swegen, I appreciate all of your efforts to counter some of the FUD that is being spread on this board, and on Twitter.
I believe that there is a deliberate coordinated and concerted attempt by some here to control the narrative, and steer the discussions here towards meaningless, make-believe problems.
AF was insinuating that NWBio did not have a patent that covers combination trials with DCVax-L plus a checkpoint inhibitor. I posted the link to the patent application for DCVax plus a checkpoint inhibitor. That patent application is still pending, and it has not been granted yet.
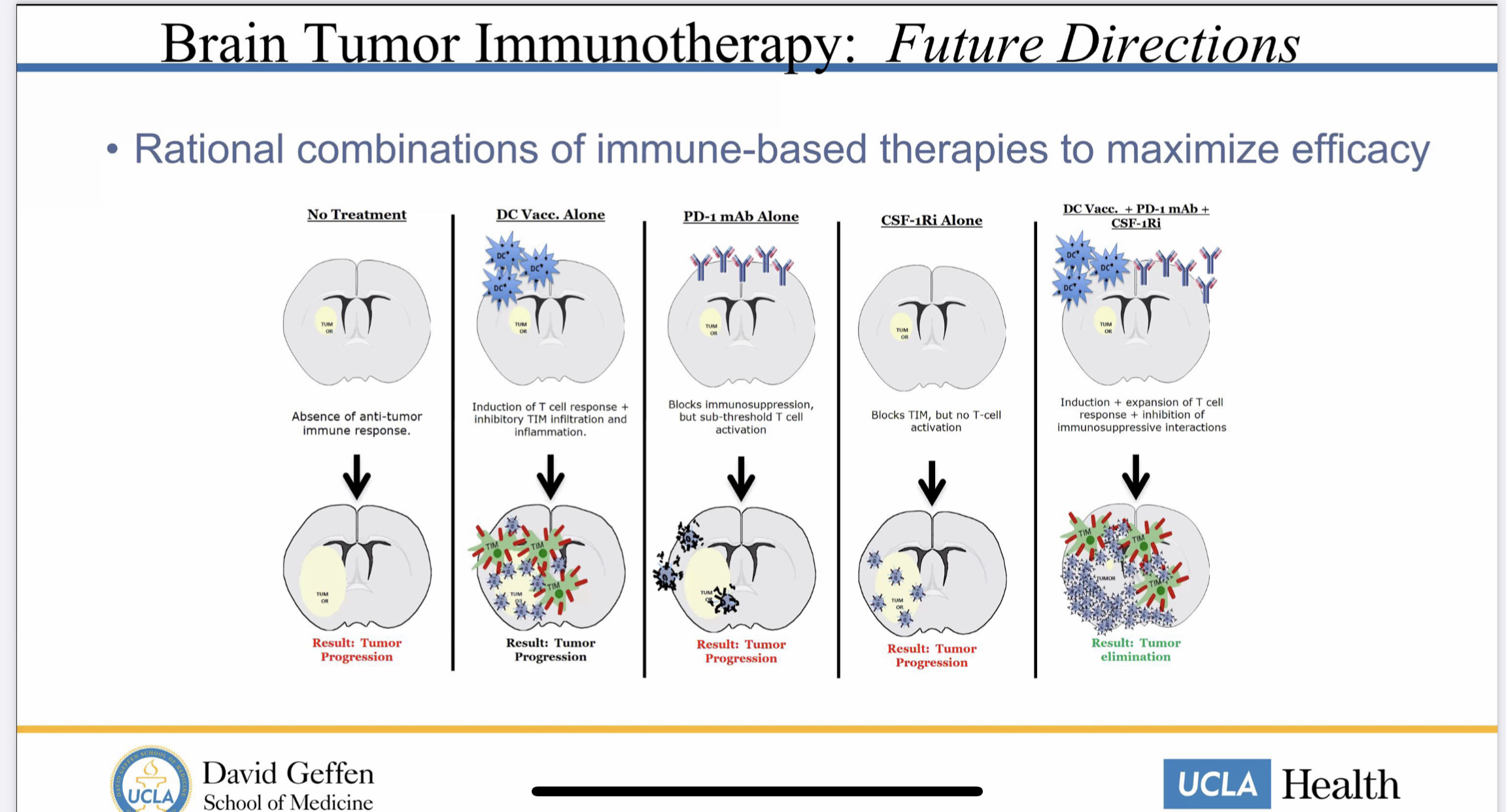
AF was also saying that UCLA was not working with NWBio anymore, and the combination trial with the autologous tumor lysate (ATL) dendritic cell (DC) vaccine and Pembrolizumab (Keytruda) does not involve DCVax-L. However, this argument was refuted by several posts on this board, including some posts by me, Dr. Bala and sentiment_stocks. I have included the slide that Dr. Liau used at UAB, and other presentations, that clearly shows that UCLA is using DCVax-L in combination with a PD-1 inhibitor (Keytruda/Pembrolizumab)below:
Here are some links to a few of those message posts:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=153800290
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=159130450
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160770243
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160881279
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160881478
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160947256
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=161070268
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=161766342
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162233225
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162237566
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162238406
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162259889
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162263186
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162826911
I believe that there is a deliberate coordinated and concerted attempt by some here to control the narrative, and steer the discussions here towards meaningless, make-believe problems.
AF was insinuating that NWBio did not have a patent that covers combination trials with DCVax-L plus a checkpoint inhibitor. I posted the link to the patent application for DCVax plus a checkpoint inhibitor. That patent application is still pending, and it has not been granted yet.
AF was also saying that UCLA was not working with NWBio anymore, and the combination trial with the autologous tumor lysate (ATL) dendritic cell (DC) vaccine and Pembrolizumab (Keytruda) does not involve DCVax-L. However, this argument was refuted by several posts on this board, including some posts by me, Dr. Bala and sentiment_stocks. I have included the slide that Dr. Liau used at UAB, and other presentations, that clearly shows that UCLA is using DCVax-L in combination with a PD-1 inhibitor (Keytruda/Pembrolizumab)below:
Here are some links to a few of those message posts:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=153800290
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=159130450
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160770243
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160881279
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160881478
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=160947256
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=161070268
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=161766342
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162233225
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162237566
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162238406
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162259889
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162263186
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=162826911

Recent NWBO News
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Northwest Biotherapeutics Announces Establishment Of the Company's Own Dedicated Leukapheresis Clinic • PR Newswire (US) • 04/21/2026 01:30:00 PM
- Form EFFECT - Notice of Effectiveness • Edgar (US Regulatory) • 04/21/2026 04:15:08 AM
- Form POS AM - Post-Effective amendments for registration statement • Edgar (US Regulatory) • 04/16/2026 09:25:30 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 04/07/2026 04:30:50 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/31/2026 09:04:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/15/2026 10:06:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/02/2026 10:14:59 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 11/28/2025 09:43:27 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 11/25/2025 10:23:07 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/20/2025 09:26:03 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 11/19/2025 09:15:48 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:44:21 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/31/2025 04:29:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/30/2025 08:40:05 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/24/2025 04:28:38 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 10/14/2025 06:22:26 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/14/2025 09:00:38 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 07/01/2025 09:04:38 PM








