Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Nidan and bas, yes the analysts are disappointing...must be spending all their time on BP. Oh wait a minute, this is their job, as they have they bio companies come in to talk with them.
Investor, I think this is the link you referred to
https://www.9news.com.au/national/alzheimers-disease-drug-trial-inflammation-dementia-health-news-australia/c62e8702-5bf3-4334-8a19-f53e288d2cda
Another drug.
Fakconer, I mean they look at PAST SP charts
Plex, the company has said for sometime they will partner for distribution for PD and AZ, but go it alone for Rett.
I couldnt make the call but thanks to you and others for reporting!
Falconer, I have warned the shorts that betting against anavex, because no others have succeeded, is certainly reasonable, but not necessarily correct. The error there is called DEFAULT BIAS. Just because something has never happened, assume it CANNOT happen.
They also tend to ignore the fundamental science and just look at
charts of the PAST.
Yeah JJ, wait for Rett and PD. Right now if anavex needs $1million they sell 333,000 shares, significant dilution. When the shares are say $30/share, to raise $1million, then dilution is only 33,300 shares dilution, not even noticeable. You get the idea....I am ignoring the Cantor/LPC financing.
George, great find that Boston Childrens Hospital has 250 patients wth Rett or similar.
Ths means any major hospital has many patients "on Rolodex" to get enrolled for trials. Many more than needed!
This raises interesting scenario. It has been several days now since our results came out. It would be reasonable to assume that Rett parents, and Rett oriented hospitals are ACTIVELY recruiting and enrolling. What happens if there are lots of patients that now want enrolled in our Rett trials?
Meds. In data analysis, generally one does not throw out any data unless it is KNOWN to be erroneous. The average response is the average of ALL the valid data, including super responders and all others.
Now if one wants to acknowlege multiple classes of responders, then the data can be broken up. For example, assuming there are super responders and everyone else. In the anavex case, then one might use the 5 patient "normal" data average, and acknowledge there is a super responder class also. Could this be construed as a negative for anavex? No, as the "normal" responders also improved. Even if the non super responders showed no improvement at all, then the conclusion would be that patients have a 1 in 6 chance of improving Superbly!
But in the case of anavex the average of the whole cohort (6) is fine for stating the overall average response.
Remember, world expert on Rett, Dr. Kaufman's words were the data is "very encouraging". There is no other way to spin that, though some seem to try.
Now anavex has two separate trials reported, AZ and Rett, both of which are very encouraging.
Polar, I am interested in reading what comes out of the RS organizations in the coming weeks, regarding A2-73. When word gets to the RS parent/caregiver, they will be thrilled with the results, given almost no alternatives.
Anavex getting lots of press these days. Word is getting out!
Falconer, thanks for the insights. Nice descriptions of the processes.
SP up1.4% after hours. Somebody wants some.
Talon, I figured, when enrollment 50% PDD was given(SH meeting) that we could reasonably have had full enrollment(assuming same rate) for PDD at end of July. Here we are nearing end of September.
I had been wondering about this. So I just called IR to confirm they would PR full enrollment for Rett and PD, and they said yes they would PR those, and enrollment was not complete.
Dunno whats going on. I can only continue to conclude that we must be close to full for PDD trial at least.
Pcguy, yes you are correct...generally.
I said the same thing, as that was my fear also.
However, now we have results from TWO trials, the original ALZ trial and now Retts. Both show translation. Now ALZ alone showing translation still left considerable room for translation skepticism, and Anavex did not really understand the science originally. But now they understand the mechanisms better, and with proven translation with Rett, A2-73 translation for many CNS diseases now actually seems LIKELY.
Knowing this should make all longs more excited.
Good to be skeptical investing, just letting you know how my thinking has evolved.
Poor Doctor Walter Kaufmann...
PLACE: Rett Finland Conference
SPEAKER: Mr Walter E. Kaufmann (USA) "subject: Behavior" 1 Hour
Months before the Finland Rett conference he committed to speak on "Behavior" for an hour. The PowerPoint presentation was complete.
Then a few weeks before the conference, the company he is CMO for, discovers their drug improves (see Note1) Rett Syndrome symptoms and various chemical imbalances in a group of older Rett patients!
What is a good doctor to do?
Does Dr. Kaufmann:
A) Stick to his prepared presentation about behavior.
-- OR --
B) Discuss the very latest breakthroughs in Rett TREATMENT? (followed by dropping microphone)
-- OR --
C) Some combination of A and B???
OK, a little tongue in cheek, but it will be interesting to see how this plays out.
I am hoping for B, but... who knows.
Another thought. When speaking, one should tailor the speech to the audience.
I wonder which option his audience would choose?
(Note1: Oh yes, there was NO placebo arm.)
BIG BUYs at 10:45 and 3:59. The early one did not move the price but the last one sure did.
Investor, excellent point about GABA and glutamate levels not being subjective measurements! All good news.
Dunno about Acadia...dont follow their news.
Comparison of A273 and Trofinetide Results...
Comparing Trofinetide data to the Anavex data, we can see:
1) A2-73 provided a much stronger signal(16pts), >2x that of Trofinetide(6.7)!
2) The placebo affect accounts for a drop of only 2.3 points. Assuming similar placebo affect with A2-73, then A2-73 still has a very strong signal (still almost 2x better)!
This Trofinetide data is from your (and Steady's) link.
https://n.neurology.org/content/92/16/e1912
Here is the table of interest:
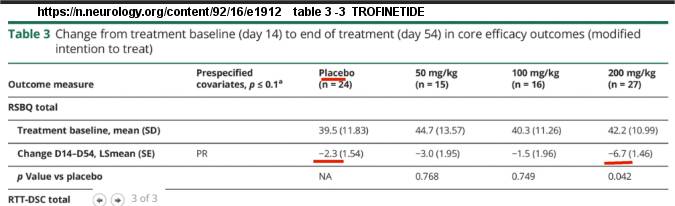
(Note: to view this table in the article...view pop up, at bottom left then select table 3 of 3.)
Caveat: The Trofinetide study had 82 patients with a placebo arm. The A2-73 study had 6 with no placebo.
SteadyT, followup to your good link...
You posted a good link to Trofinetide's Rett study results:
https://n.neurology.org/content/92/16/e1912
This link's Table 3, helps to put the Anavex Rett data in context.
Looking at table 3 (view pop up, at bottom left select table 3 of 3)
we can see the Trofinetide results with placebo and dosages.
Here is the top part of that table:

Note the Trofinetide trial was with 82 participants, and placebo controlled. The first 14 days (D14) obtained baseline data, and the full study was for 54 days (D54).
The 200mg/kg dose arm shows RSBQ score dropping 6.7, while the placebo arm drops 2.3.
See the red underlined interesting data.
Comparing these to the Anavex scores that dropped 16 points, we can see:
1) A2-73 provided a much stronger signal, >2x that of Trofinetide!
2) The placebo affect accounts for a drop of only 2.3 points. Assuming similar placebo affect with A2-73, there is still a very strong signal!
A2-73 DROPPING SCORES 16 POINTS IS STILL GREAT NEWS!
Great research Steady and George. Steady I was curious about those improvement numbers as well. For reference, those are like taking a typical school 100 point test and the scores going up 38% to 55% !!!
Wow, those are huge improvements.
Fireworks at the Rett conference at the end of the month!
Thanks guys.
SportsNut Good call!
Now if you were only so prophetic about SPs! Lol
Investor, remembering your discussions of chances of success and blood concentrations...
Regarding blood concentrations, my opinion is Anavex is following blood concentrations closely, because it is closer to the CNS mechanisms, being already in the blood stream, and should be more correlatable to the responses. That is, oral doses have to go into the digestive system first and that may cause variance in the amount of drug that actually gets to the blood stream, depending on the patient. Blood concentration may be eliminating that variable. Does that make sense?
Regarding your guesstimate of chances of success for A2-73, I always appreciated and admired that you were willing to publish a number! My fear has been the low percentage of mouse studies that were also effective in humans. I have to say this PR goes a long way to alleviating those fears. So now we have apparent efficacy in Rett and ALZ. My guesstimate for chances of success go up 15% with this news. Does your guesstimate improve with this PR?
Detailed Data to be Presented at European Rett Conference in Tampere, Finland, September 27-28, 2019
From the press release. This will go over like fireworks!!
Could also have a full Rett trial enrollment PR any day now (soon or sooner).
Why is Dr.M talking about Rett so much now? Because he can! ALZ is still to come.
Expect enrollments to rapidly increase in all Anavex trials.
JJ my thoughts exactly on Autism. Plus...
Very interested to hear Dr.M mention autism up with AD, PD, and Rett! That is a HUGE market.
HOPE AUTISM TRIAL IS SOON!
Would love to hear him start commenting more about it.
Janney notes typos. Thanks powerwalker and george.
In my notes, Dr.M defined several benefits of A2-73 and I got two of them wrong.
Here they are corrected:
* high hyperphosphorylation of tau
* restores mitochondrial FUNCTION
Guys, thanks for catching those!
Notes from Janney presentation.
I listened to the first 11 minutes of the recorded presentation, looking for interesting items, and I wanted to record exactly Dr.Ms description of autophagy.
Exact Dr.M quotes are in quotes.
My comments and questions are in parentheses.
Going into 4th year of observing dosing of patients with AD (P2A 32coh.)
---(I wonder if there is any other AD drug with 3 years of AD data, except Donepezil? Also, can they be varying the dose? Of course, not higher than current max.)
Sufficient cash for all objectives beyond the next 24 months.
---(I think he means to 24months)
Next Catalysts:
* Rett Study top line data
* PD P2 Demential study top line data
* Clinical data publication of AD P2A "information which has been presented at various scientific meetings."
---(It sounds like this is the peer reviewed study. I hope it is more than what has already been presented. Maybe it ties theories together including gut microbiota.)
* In discussions to and explore, new indications with our approach, as well as looking at licensing opportunities.
---(New indications could be ERP, or gut signals or something totally new. Implied the licensing is after top line data. Licensing separate bullet?)
...All these diseases have in common Rett, PD, AZ and autism, they all have extensive cellular stress in the body.
---(Wow Autism has made it up to his top 4 to be mentioned. Autism is huge!)
This cellular stress must be addressed, and the body uses the Sigma1 receptor. When the S1 receptor is constantly requested to reduce the stress, it become overused or not sufficiently available any more.
So added S1 activation is needed and that can be given from the outside. And that is what A2-73 is doing. A2-73 provides added stimulation of the S1 gene and S1 protein to do its job.
The beneficial effects, and this has been published, are:
* reduces tau
* high postulation of tau (not sure I got this right...chemistry is not my first language!)
* reduce protein misfolding
* reduces inflammation
* restores mitochondrial dysfunction
In a publication in March of this year, ..."A2-73 has demonstrated to activate an impaired autophagy which is a key clearance process of CNS neurons which cannot just get rid of trash which accumulates in the cells. Because there is no exit for those cells.
The only way the cell can survive is by triggering or activating this recycling process and that is called autophagy."
The publication demonstrated that A2-73 enhanced the autophagy process in animals and cellular experiments, and the animal had extended life and longevity.
Xena, I followed the last half of the presentation looking at the most recent corp presentation doc that Anavex has on the website. The presentation was almost line for line from the doc. Dr.M may have had a few new changes to the presentation, like mentioning marketing, but no bombshells.
Regarding the PD trial he specifically stated it "will finish in due time". Lol
Steady, very well said reply to the post about trying to find responders, being a trick.
Also thanks for the answer to my question this week.
Leo, yes there is no official trial yet. Good points!
Given the recent posts about Rett, 200 girls in AU, rarity of Rett, Avatar AU waiting (so far 5 weeks) for one patient, and Excellence AU trial with "approximately 69 pediatric patients".
This may take a long time to enroll 69 patients!
That said, there are factors that could dramatically speed up enrollment:
1. Trial expansion to rest of world.
2. Positive results from age >18 trial.
Note the PR wording is "approximately 69".
Is this common wording for trial enrollment?
Does it imply if they have trouble getting to say 50 patients, they could finish the trial with only 50?
Lima, good point. AMAZING. I re-read Georgejj's post...
Anavex seminar bullet:
*Outlining HOW and WHY A2-73 may have a beneficial homeostatic effect on brain-gut microbiota axis. (Caps mine)
Yes, in their first report I think anavex did not commit to understanding the cause/effect, but it looks like they have been looking into it and now have an explanation. Another dot connected.
Definitely leading the science, now for trial success!
JJ, when I spoke with the company, a week or two ago, I asked if the full enrollment announcement would be made immediately, or maybe delayed for announcement at a conference. I was told a conf announcement would not be appropriate, the PR would be made immediately.
Makes sense. I dont think there is any "buffer" that is advantageous here. After full PR, everyone understands the timeline.
Brich, thanks very nice follow up...
I think in this period of waiting, we should get as much info from the company as possible. At least we can talk about facts instead of fantasy.
Kund, maybe, but Phds invest a lot of time, money, and effort to earn a Phd. I have heard of corrupt Phds (actually MDs) by themselves that have scammed or hurt clients. But you are suggesting that Dr.M has scammed/mislead a handful of other Phds, that have put their reputation/careers on the line. In my lifetime, I have never heard of that happening.
Leo, true that! But...
I prefer, and appreciate, the company communicating as openly AS POSSIBLE clearing up questions, versus erroneous assumptions and speculation ad infinitum.
Hey Bourbon, I dont think he could say if Pdd or Rett would be announced within a month... depends on if/when patients enroll. Hopefully after full enrollment, they can accurately speak to the results date.
Spoke with company 8/21/2019.....
Tried to get some answers to overview type questions, nothing technical or detailed. The company representative was very open and ready to answer any question that could be commented on.
Has the peer reviewed paper been published, or what is the publication name or time frame for it? Ans. No, it has not been published, and no comment on the publication or time frame.
Will full enrollment announcement be made immediately or at a conference? Ans. It will probably be an immediate PR announcement.
How many employees does the company now have? Ans. Under 20. Between 10-20.
Why was the shelf announcement made after hours right before a holiday? Ans. That was when the process was finished and info was available. The company says there is no intent to use this shelf money now, but it is available in case of good trial results and more trials need to be run.
General discussion...The company continues to try to run lean and frugally. The trials were delayed because Dr.M saw all these large expensive Alz trials failing and he decided to try to do them smarter using PM, even if it delayed the trials. The advantage being the trials would be less expensive and require fewer people. This is continued to be seen as the year of execution, with lots riding on read outs.
TTT good find. Thanks for posting!
