Thursday, April 25, 2019 9:02:59 PM
Isradipine Parkinson’s 3 year $23 million dollar Study completed Nov 2018
Results Imminent
Quote:
STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia on May 7, 2019.
https://scienceofparkinsons.com/2019/02/28/february-2019/#more-52900
There are +1,000,000 Parkinson’s patients and another 1,000,000 hypertensives with a family history of PD
TWO
Pharmaceuticals to supply:
Watson and Elite .
$500per year per patient
Split $1 Billion between Watson Elite and the Pharmacies
Watch what happens to the SP
Watch for leaks
—————————————————————————
Scientists believe isradipine works to prevent the death of dopamine-producing cells and therefore may slow the progression of PD.
Isradipine can affect the function of specialized channels that are present in the types of brain cells that are affected in PD patient. These cells are usually responsible for making dopamine, which is depleted in patients with PD. Isradipine may block the damage caused by the flow of certain chemicals through these channels. Laboratory data has showed that Isradipine may prevent the development of Parkinson-like symptoms in animals.
5 mg of isradipine taken twice daily for 36 months.
https://clinicaltrials.gov/ct2/show/NCT02168842
Elite’s e’s Isradipine is a calcium channel blocker approved for the treatment of high blood pressure and was recently tested as a disease-modifying drug for Parkinson’s Disease.
There is solid scientific basis as well as preclinical and epidemiological data that Elite’s isradipine has the potential to prevent development of parkinsonian signs.
Positive results from a Phase II study have provided support for a Phase III trial, STEADY-PD III, a major step toward a novel treatment to slow the progression of PD.
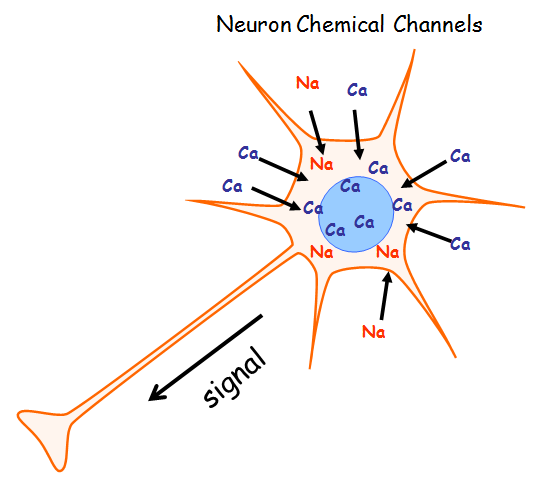
A lack of healthy dopamine neurons in the brain is the major cause of the motor symptoms of Parkinson disease (PD).
Dopaminergic nerve cells are unique in the brain because they have their own pacemaking rhythms.
These special rhythm changes are being driven by the chemical channels that are either sodium or calcium.
Calcium is more energy demanding and with age, these neurons increasingly rely on calcium channels.
What is the science behind it? The neuron cells’ reliance on calcium to drive this activity creates a lot of ‘wear and tear’ on dopamine neurons-aging them more rapidly than other neurons and making them vulnerable.
Elite’s Calcium channel blockers (class of drugs frequently used to treat high blood pressure), have possible neuroprotective effects that can slow or stop Parkinson’s progression.
Population studies have shown that current long-term use of calcium channel blockers has been associated with a significantly reduced risk of Parkinson disease.
STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia May 7, 2019.
STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia on the 4th-10th May, 2019.
https://investorshub.advfn.com/uimage/uploads/2019/4/2/eifnxGg4xuWD.png
https://scienceofparkinsons.com/2019/02/28/february-2019/#more-52900
Results Imminent
Quote:
STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia on May 7, 2019.
https://scienceofparkinsons.com/2019/02/28/february-2019/#more-52900
There are +1,000,000 Parkinson’s patients and another 1,000,000 hypertensives with a family history of PD
TWO
Pharmaceuticals to supply:
Watson and Elite .
$500per year per patient
Split $1 Billion between Watson Elite and the Pharmacies
Watch what happens to the SP
Watch for leaks
—————————————————————————
Scientists believe isradipine works to prevent the death of dopamine-producing cells and therefore may slow the progression of PD.
Isradipine can affect the function of specialized channels that are present in the types of brain cells that are affected in PD patient. These cells are usually responsible for making dopamine, which is depleted in patients with PD. Isradipine may block the damage caused by the flow of certain chemicals through these channels. Laboratory data has showed that Isradipine may prevent the development of Parkinson-like symptoms in animals.
5 mg of isradipine taken twice daily for 36 months.
https://clinicaltrials.gov/ct2/show/NCT02168842
Elite’s e’s Isradipine is a calcium channel blocker approved for the treatment of high blood pressure and was recently tested as a disease-modifying drug for Parkinson’s Disease.
There is solid scientific basis as well as preclinical and epidemiological data that Elite’s isradipine has the potential to prevent development of parkinsonian signs.
Positive results from a Phase II study have provided support for a Phase III trial, STEADY-PD III, a major step toward a novel treatment to slow the progression of PD.
A lack of healthy dopamine neurons in the brain is the major cause of the motor symptoms of Parkinson disease (PD).
Dopaminergic nerve cells are unique in the brain because they have their own pacemaking rhythms.
These special rhythm changes are being driven by the chemical channels that are either sodium or calcium.
Calcium is more energy demanding and with age, these neurons increasingly rely on calcium channels.
What is the science behind it? The neuron cells’ reliance on calcium to drive this activity creates a lot of ‘wear and tear’ on dopamine neurons-aging them more rapidly than other neurons and making them vulnerable.
Elite’s Calcium channel blockers (class of drugs frequently used to treat high blood pressure), have possible neuroprotective effects that can slow or stop Parkinson’s progression.
Population studies have shown that current long-term use of calcium channel blockers has been associated with a significantly reduced risk of Parkinson disease.
STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia May 7, 2019.

STEADY-PD III (isradipine) clinical trial results will be released at the 2019 American Academy of Neurology meeting in Philadephia on the 4th-10th May, 2019.
https://investorshub.advfn.com/uimage/uploads/2019/4/2/eifnxGg4xuWD.png
https://scienceofparkinsons.com/2019/02/28/february-2019/#more-52900
Everyone you meet is fighting a battle you know nothing about BE KIND
Recent ELTP News
- Form 8-K - Current report • Edgar (US Regulatory) • 04/02/2026 12:05:23 PM
- Elite Pharmaceuticals Announces Commercial Launch of Methadone Hydrochloride Tablets • Newsfile • 04/02/2026 12:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/17/2026 10:10:32 PM
- Elite Pharmaceuticals, Inc. Reports Financial Results for the Third Quarter of Fiscal Year 2026 (three months ended December 31, 2025) and Provides Conference Call Information • Newsfile • 02/17/2026 09:19:00 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 02/17/2026 09:06:15 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/11/2026 09:35:26 PM
- Elite Pharmaceuticals, Inc. to Host Conference Call to Provide Corporate Update and Discuss Third Quarter 2026 Financial Results on February 18, 2026 • Newsfile • 02/11/2026 09:30:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/13/2026 09:35:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/14/2025 10:10:23 PM
- Elite Pharmaceuticals, Inc. Reports Financial Results for the Second Quarter of Fiscal Year 2026 Ended September 30, 2025 and Provides Conference Call Information • Newsfile • 11/14/2025 09:19:00 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:11:38 PM
- Form 8-K/A - Current report: [Amend] • Edgar (US Regulatory) • 11/12/2025 10:19:32 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/12/2025 12:15:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/10/2025 12:15:46 PM
- Elite Pharmaceuticals, Inc. to Host Conference Call to Provide Corporate Update and Discuss Second Quarter 2026 Financial Results on November 17, 2025 • Newsfile • 11/10/2025 12:00:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 08/26/2025 09:17:39 PM
- Form 144 - Report of proposed sale of securities • Edgar (US Regulatory) • 08/22/2025 01:39:01 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/14/2025 08:37:09 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/08/2025 08:35:45 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/25/2025 10:05:07 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/10/2025 08:17:22 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/08/2025 08:15:11 PM
- Form 144 - Report of proposed sale of securities • Edgar (US Regulatory) • 07/08/2025 02:58:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 06/30/2025 08:27:51 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 06/30/2025 08:05:59 PM









