Friday, April 19, 2019 4:39:07 PM
How to Vote your ATOS Proxy IMO.
I received my proxy today. The following is why and how my vote differed from Board recommendations.
Proposal No. 1 – Election of Directors
Mr. Quay should not be both the CEO and Chairman of the Board. Unfortunately, it does not appear that shareholders have a say in this matter. The election of Mr. Quay and Mr. Weaver looks to be pretty much a done deal. However, I withheld my vote for Mr. Quay as a protest in that he has too much control over the Company.
I voted FOR Mr. Weaver and WITHHOLD on Mr. Quay
Proposal No. 3 – To Approve the 2019 Amendments to the Atossa Genetics Inc 2010 Stock Option and Incentive Plan and to Increase The Number of Shares Authorized for Issuance Under The Plan By 3,600,000
This is one of the most egregious requests I have ever seen by a public company and my vote here is a no-brainer. A 26% dilution to existing shareholders that is almost exclusively for the benefit of the CEO and CFO as a reward for what? Management’s performance since Atossa became a public company has been atrocious. On a split adjusted basis, the stock once traded at over $1,500/share:
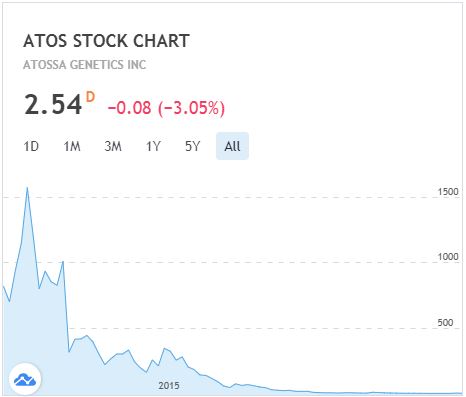
Note at the IPO in 2012, Mr. Quay owned over 5,000,000 shares (37% post IPO). However, through two reverse splits (and probably some sales along the way), his beneficial share interest has dropped to 82,667. Would have been great for investor confidence if he bought 200,000 shares at $1.36 instead of taking 2.3M no-risk options.
Lastly, the wording of Proposal No. 3 is very deceptive for what it doesn’t say. View prior post here.
I voted AGAINST Proposal No. 3
Proposal No. 4 - Advisory Vote on Executive Compensation
While I agree with the stated philosophy of “tying compensation to increases in stockholder value,” the number of option shares granted to Mr. Quay (2,300,000) and Mr. Guse (800,000) is over the top. Fortunately shareholders can stop this by voting Against Proposal No. 3. A similar vote is required here to express shareholder dissatisfaction with the overall compensation package.
What is the right number of options? Given an exercise price of $1.36/share, I could support 230,000 shares for Mr. Quay and 80,000 shares for My Guse. If Atossa hits on its Oral Endoxifen, the Company could easily run to a $500M market cap. With 9,124,447 common shares outstanding as of the record date, that equals a per share price of $54.80. In other words, $12.3M for Mr. Quay (as opposed to $123M) and $4.3M for Mr. Guse (as opposed to $43M). Most CEO's of Fortune 500 companies don't earn $123M in stock compensation. $166M for the CEO/CFO of a micro cap, outrageous.
I voted AGAINST Proposal No. 4
Thanks for taking the time to read my post. Now go vote your Proxy.
I received my proxy today. The following is why and how my vote differed from Board recommendations.
Proposal No. 1 – Election of Directors
Mr. Quay should not be both the CEO and Chairman of the Board. Unfortunately, it does not appear that shareholders have a say in this matter. The election of Mr. Quay and Mr. Weaver looks to be pretty much a done deal. However, I withheld my vote for Mr. Quay as a protest in that he has too much control over the Company.
I voted FOR Mr. Weaver and WITHHOLD on Mr. Quay
Proposal No. 3 – To Approve the 2019 Amendments to the Atossa Genetics Inc 2010 Stock Option and Incentive Plan and to Increase The Number of Shares Authorized for Issuance Under The Plan By 3,600,000
This is one of the most egregious requests I have ever seen by a public company and my vote here is a no-brainer. A 26% dilution to existing shareholders that is almost exclusively for the benefit of the CEO and CFO as a reward for what? Management’s performance since Atossa became a public company has been atrocious. On a split adjusted basis, the stock once traded at over $1,500/share:
Note at the IPO in 2012, Mr. Quay owned over 5,000,000 shares (37% post IPO). However, through two reverse splits (and probably some sales along the way), his beneficial share interest has dropped to 82,667. Would have been great for investor confidence if he bought 200,000 shares at $1.36 instead of taking 2.3M no-risk options.
Lastly, the wording of Proposal No. 3 is very deceptive for what it doesn’t say. View prior post here.
I voted AGAINST Proposal No. 3
Proposal No. 4 - Advisory Vote on Executive Compensation
While I agree with the stated philosophy of “tying compensation to increases in stockholder value,” the number of option shares granted to Mr. Quay (2,300,000) and Mr. Guse (800,000) is over the top. Fortunately shareholders can stop this by voting Against Proposal No. 3. A similar vote is required here to express shareholder dissatisfaction with the overall compensation package.
What is the right number of options? Given an exercise price of $1.36/share, I could support 230,000 shares for Mr. Quay and 80,000 shares for My Guse. If Atossa hits on its Oral Endoxifen, the Company could easily run to a $500M market cap. With 9,124,447 common shares outstanding as of the record date, that equals a per share price of $54.80. In other words, $12.3M for Mr. Quay (as opposed to $123M) and $4.3M for Mr. Guse (as opposed to $43M). Most CEO's of Fortune 500 companies don't earn $123M in stock compensation. $166M for the CEO/CFO of a micro cap, outrageous.
I voted AGAINST Proposal No. 4
Thanks for taking the time to read my post. Now go vote your Proxy.
Recent ATOS News
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 03/30/2026 11:22:50 PM
- Form ARS - Annual Report to Security Holders • Edgar (US Regulatory) • 03/30/2026 09:33:51 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 03/30/2026 09:30:46 PM
- Atossa Therapeutics Reports Fourth Quarter and Year-End 2025 Financial Results and Provides a Corporate Update • PR Newswire (US) • 03/25/2026 09:30:00 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 03/25/2026 09:21:23 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 03/25/2026 09:20:21 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 03/20/2026 08:52:24 PM
- The Patent Cliff is Coming, Driving Smart Money Towards Precision Oncology • GlobeNewswire Inc. • 03/19/2026 03:00:00 PM
- Atossa Therapeutics Strengthens Clinical Leadership Team with the Addition of Two Experienced Biopharma Executives • PR Newswire (US) • 03/19/2026 12:00:00 PM
- Atossa Therapeutics Presents Clinical Trial Update Highlighting (Z)-Endoxifen Research at the 2026 MDA Clinical & Scientific Conference • PR Newswire (US) • 03/12/2026 12:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/20/2026 09:22:08 PM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 02/20/2026 09:16:05 PM
- Form 424B3 - Prospectus [Rule 424(b)(3)] • Edgar (US Regulatory) • 02/20/2026 09:07:14 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/17/2026 09:30:28 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/11/2026 01:45:24 PM
- Atossa Therapeutics Issues Letter to Shareholders Highlighting 2025 Accomplishments and 2026 Outlook • PR Newswire (US) • 02/11/2026 01:30:00 PM
- Atossa Therapeutics Maintains Strong Market Position for (Z)-Endoxifen for Duchenne Muscular Dystrophy as Congress Reauthorizes Priority Review Voucher Program • PR Newswire (US) • 02/05/2026 01:30:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/26/2026 01:00:26 PM
- Atossa Therapeutics Receives FDA Orphan Drug Designation for (Z)-Endoxifen for the Treatment of Duchenne Muscular Dystrophy • PR Newswire (US) • 01/16/2026 09:40:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/09/2026 01:15:27 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 01/06/2026 01:30:24 PM
- Atossa Therapeutics Receives FDA "Study May Proceed" Letter for (Z)-Endoxifen Investigational New Drug Application for Metastatic Breast Cancer • PR Newswire (US) • 01/06/2026 01:15:00 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 12/22/2025 01:30:27 PM
- Atossa Therapeutics Wins 2025 Clinical Trials Arena Research and Development Excellence Award in Precision Endocrine Therapy Category • PR Newswire (US) • 12/17/2025 01:15:00 PM
- Atossa Therapeutics Presents Four Clinical Trial Updates Highlighting (Z)-Endoxifen Research at the 2025 San Antonio Breast Cancer Symposium • PR Newswire (US) • 12/15/2025 01:00:00 PM








