Saturday, January 26, 2019 4:22:53 PM
Reformulated Patent Pending SequestOx™ is the SAFEST IR Opioid in the World.
It will easily receive Oral (chewing) Abuse Deterrent Labeling like no other IR ADF Opioid has.
Here’s how it works:
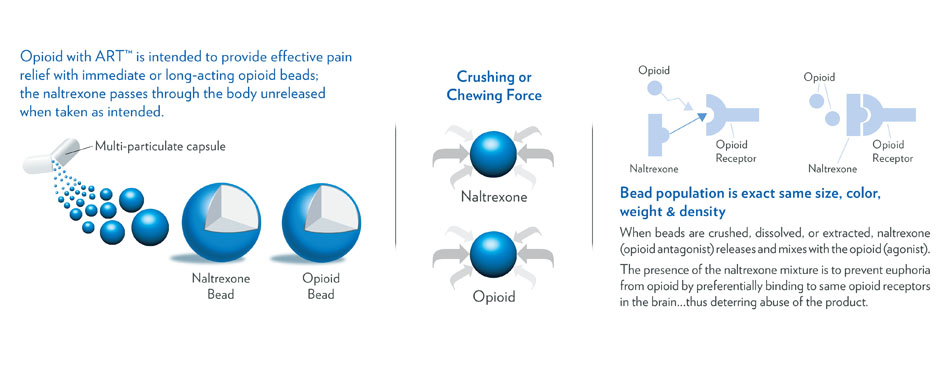
FDA validated the entire ER ADF line of opioids
The FedTmax is not an issue with ER Opioids
Elite's tech can be used on all ER opioids
FDA did not find one issue with Elite 2 bead ADF except for fed Tmax which only applies to IR opioids.
Not one issue with HAL studies.
Quote:
"the FDA made no comment about Elite’s technology because it works and it’s solid. They made no comment on the human abuse liability studies because again they were solid and they work; or with the withdrawal study or the efficacy study – the Bunionectomy. This is great news for Elite’s ADT platform. There is no effect on Elite’s ER formulations"
Recent Pilot study for Re-formulated SequestOx validated entire lineup of IR ADFs Now Elite's tech can be used on all IR opioids

Pilot Study: shows a positive FedTmax for SequestOx which was the ONLY ISSUE with the IR ADF lineup.
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0
ELITE PHARMA
ELTP
rma.com/wp-content/uploads/2014/12/How_It_Works.jpg[/img]
Q The only issue with SequestOx™ was the FedTmax.
That was fixed months ago the PR was early this year.
See below for supporting link and exceptional results.
Quote:
“We resolved that issue and our patent attorney, Dr. Smith determined that this is something patentable and we will be filing a patent sometime next week.”
Pilot Study:
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0

IR SequestOx™ will establish a New Standard of Care 2018-2019.
The most frequently abused opioids are currently immediate-release (IR) opioids.

In 2016 93% of new opiod RXs were for IR(Immediate Release) opiods.
Reformulated SequestOx™ with lowered fed Tmax is the SAFEST Immediate Release opiod in the world, and the only one using a Pharmalogical Approach to abuse deterrence.
According to data from “Do abuse deterrent opioid formulations work?” Richard C. Dart, MD, PhD; Janetta L. Iwanicki, MD; Nabarun Dasgupta, PhD; Theodore J. Cicero, PhD; Sidney H. Schnoll, MD, PhD, in the Journal of Opioid Management 13:6, November/December 2017, reported that Immediate Release (IR) opioids are 4.6 times more likely to be intentionally abused and 6.1 times more likely to be diverted than Extended Release (ER) formulations. Not surprisingly, cited in the aforementioned work is a real-world study of 300 opioid abusers entering treatment for substance abuse who indicated they prefer abusable IR opioid products because they provide faster onset of the rewarding effects.
SequestOx™ had impeccable HAL studies.
The only issue with SequestOx was the FedTmax.
That was fixed months ago the PR was early this year.
See below for supporting link. Glad to Help.
LAST box to be checked off: Reformulated SequestOx THE Holy Grail of IR opioids
996. Bioequivalent ---------------[X]
997. HAL study -------------------[X]
998. Cmax ———————— [X]
999. Fasting Tmax. -———— [X]
1000. Safe FedTmax. -——— [_]
Expedited Review
Pilot Study:
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0

FDA looking for an IR ADF opiod with Oral labeling
INSIDERS ACCUMULATED SHARES !!! Insiders refused to sell at $0.97.
IR SequestOx™ The New Standard of Care 2018-2019
the most frequently abused opioids are immediate-release (IR)
Elite's 2 bead ER and IR ADF opiods
will be the first to obtain complete ORAL, Nasal and IV ADF labeling
SequestOx will make ALL other IR opiods obsolete.
Inspirion's RoxyBond lost big by not getting oral AD labeling
Reformulated SequestOx™ with lowered fed Tmax is the SAFEST Immediate Release opiod in the world, and the only one using a Pharmalogical Approach to abuse deterrence.
SequestOx™ had impeccable HAL studies.
SequestOx II™ Elite's market disruptive ADF labeled Immediate Release opiod
can not be abused by:
NASAL:
Insuffalation/snorting all particle sizes
IV:
injecting/IV/poor syringe ability
Smoking:
ORAL:
chewing
crushing/mechanical manipulation
grating
milling
sucking
splitting
breaking
grinding
use with alcohol
sandpapering
blended
melting
microwave
baking
parachuting
freezing
extracting
centrifuging
bead separation
Cant be defeated with common solvents
It will easily receive Oral (chewing) Abuse Deterrent Labeling like no other IR ADF Opioid has.
Here’s how it works:

FDA validated the entire ER ADF line of opioids
The FedTmax is not an issue with ER Opioids
Elite's tech can be used on all ER opioids
FDA did not find one issue with Elite 2 bead ADF except for fed Tmax which only applies to IR opioids.
Not one issue with HAL studies.
Quote:
"the FDA made no comment about Elite’s technology because it works and it’s solid. They made no comment on the human abuse liability studies because again they were solid and they work; or with the withdrawal study or the efficacy study – the Bunionectomy. This is great news for Elite’s ADT platform. There is no effect on Elite’s ER formulations"
Recent Pilot study for Re-formulated SequestOx validated entire lineup of IR ADFs Now Elite's tech can be used on all IR opioids

Pilot Study: shows a positive FedTmax for SequestOx which was the ONLY ISSUE with the IR ADF lineup.
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0
ELITE PHARMA
ELTP
rma.com/wp-content/uploads/2014/12/How_It_Works.jpg[/img]
Q The only issue with SequestOx™ was the FedTmax.
That was fixed months ago the PR was early this year.
See below for supporting link and exceptional results.
Quote:
“We resolved that issue and our patent attorney, Dr. Smith determined that this is something patentable and we will be filing a patent sometime next week.”
Pilot Study:
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0

IR SequestOx™ will establish a New Standard of Care 2018-2019.
The most frequently abused opioids are currently immediate-release (IR) opioids.
In 2016 93% of new opiod RXs were for IR(Immediate Release) opiods.
Reformulated SequestOx™ with lowered fed Tmax is the SAFEST Immediate Release opiod in the world, and the only one using a Pharmalogical Approach to abuse deterrence.
According to data from “Do abuse deterrent opioid formulations work?” Richard C. Dart, MD, PhD; Janetta L. Iwanicki, MD; Nabarun Dasgupta, PhD; Theodore J. Cicero, PhD; Sidney H. Schnoll, MD, PhD, in the Journal of Opioid Management 13:6, November/December 2017, reported that Immediate Release (IR) opioids are 4.6 times more likely to be intentionally abused and 6.1 times more likely to be diverted than Extended Release (ER) formulations. Not surprisingly, cited in the aforementioned work is a real-world study of 300 opioid abusers entering treatment for substance abuse who indicated they prefer abusable IR opioid products because they provide faster onset of the rewarding effects.
SequestOx™ had impeccable HAL studies.
The only issue with SequestOx was the FedTmax.
That was fixed months ago the PR was early this year.
See below for supporting link. Glad to Help.
LAST box to be checked off: Reformulated SequestOx THE Holy Grail of IR opioids
996. Bioequivalent ---------------[X]
997. HAL study -------------------[X]
998. Cmax ———————— [X]
999. Fasting Tmax. -———— [X]
1000. Safe FedTmax. -——— [_]
Expedited Review
Pilot Study:
http://elite.irpass.com/profiles/investor/ResLibraryView.asp?ResLibraryID=86991&BzID=2258&g=939&Nav=0&LangID=1&s=0

FDA looking for an IR ADF opiod with Oral labeling
INSIDERS ACCUMULATED SHARES !!! Insiders refused to sell at $0.97.
IR SequestOx™ The New Standard of Care 2018-2019
the most frequently abused opioids are immediate-release (IR)
Elite's 2 bead ER and IR ADF opiods
will be the first to obtain complete ORAL, Nasal and IV ADF labeling
SequestOx will make ALL other IR opiods obsolete.
Inspirion's RoxyBond lost big by not getting oral AD labeling
Reformulated SequestOx™ with lowered fed Tmax is the SAFEST Immediate Release opiod in the world, and the only one using a Pharmalogical Approach to abuse deterrence.
SequestOx™ had impeccable HAL studies.
SequestOx II™ Elite's market disruptive ADF labeled Immediate Release opiod
can not be abused by:
NASAL:
Insuffalation/snorting all particle sizes
IV:
injecting/IV/poor syringe ability
Smoking:
ORAL:
chewing
crushing/mechanical manipulation
grating
milling
sucking
splitting
breaking
grinding
use with alcohol
sandpapering
blended
melting
microwave
baking
parachuting
freezing
extracting
centrifuging
bead separation
Cant be defeated with common solvents
Everyone you meet is fighting a battle you know nothing about BE KIND
Recent ELTP News
- Form 8-K - Current report • Edgar (US Regulatory) • 04/02/2026 12:05:23 PM
- Elite Pharmaceuticals Announces Commercial Launch of Methadone Hydrochloride Tablets • Newsfile • 04/02/2026 12:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/17/2026 10:10:32 PM
- Elite Pharmaceuticals, Inc. Reports Financial Results for the Third Quarter of Fiscal Year 2026 (three months ended December 31, 2025) and Provides Conference Call Information • Newsfile • 02/17/2026 09:19:00 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 02/17/2026 09:06:15 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/11/2026 09:35:26 PM
- Elite Pharmaceuticals, Inc. to Host Conference Call to Provide Corporate Update and Discuss Third Quarter 2026 Financial Results on February 18, 2026 • Newsfile • 02/11/2026 09:30:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/13/2026 09:35:20 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/14/2025 10:10:23 PM
- Elite Pharmaceuticals, Inc. Reports Financial Results for the Second Quarter of Fiscal Year 2026 Ended September 30, 2025 and Provides Conference Call Information • Newsfile • 11/14/2025 09:19:00 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/14/2025 09:11:38 PM
- Form 8-K/A - Current report: [Amend] • Edgar (US Regulatory) • 11/12/2025 10:19:32 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/12/2025 12:15:37 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/10/2025 12:15:46 PM
- Elite Pharmaceuticals, Inc. to Host Conference Call to Provide Corporate Update and Discuss Second Quarter 2026 Financial Results on November 17, 2025 • Newsfile • 11/10/2025 12:00:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 08/26/2025 09:17:39 PM
- Form 144 - Report of proposed sale of securities • Edgar (US Regulatory) • 08/22/2025 01:39:01 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/14/2025 08:37:09 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 08/08/2025 08:35:45 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/25/2025 10:05:07 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/10/2025 08:17:22 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 07/08/2025 08:15:11 PM
- Form 144 - Report of proposed sale of securities • Edgar (US Regulatory) • 07/08/2025 02:58:10 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 06/30/2025 08:27:51 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 06/30/2025 08:05:59 PM









