Tuesday, June 06, 2017 12:45:24 PM
ASCO'17: “Prelim-Correlative-Analysis of PD-L1 Expr. from SUNRISE (Biomarker#4)” - press release, poster image, abstract detail...
6-5-17/PR: Peregrine Pharmaceuticals Presents Preliminary Correlative Analysis of PD-L1 Expression from SUNRISE Trial at ASCO 2017
-- Negative PD-L1 Expression was Associated with a Significantly Longer Median Overall Survival Compared to Positive PD-L1 Expression in Patients Receiving Docetaxel Plus Bavituximab
-- Presented Results Support Hypothesis that Bavituximab May Demonstrate Greater Effect in "Cold" Tumors Expressing Low to No PD-L1
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1028997
TUSTIN, June 05, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by advancing its proprietary R&D pipeline and manufacturing high quality products for biotechnology and pharmaceutical companies, today announced the presentation of promising new data from its Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC). Presented results demonstrated that patients in the study's bavituximab treatment arm who had low baseline PD-L1 expression levels had a statistically significant improvement in median overall survival (mOS) as compared to patients in the same treatment arm who had higher baseline levels of PD-L1. Data were presented by Peregrine scientists at the Annual Meeting of the American Society of Clinical Oncology (ASCO), being held June 2-6, 2017 in Chicago.
Data presented demonstrated that patients in the study's docetaxel plus bavituximab (D+B) treatment arm with a pre-treatment PD-L1 expression level on tumor cells of < 1% (TC0) had a mOS of 12.1 months compared to a mOS of 6.1 months for patients with PD-L1 expression >=1% (TC1/2/3) (HR = 0.42 p=0.007). There was no difference in mOS based on PD-L1 expression levels observed in the study's docetaxel plus placebo (D+P) control arm (10.7 months for TC0 vs. 11.1 months for TC1/2/3; HR = 0.87; p=0.609).
"We believe that these latest observations from the SUNRISE trial further support the hypothesis that bavituximab, through its immune modulating mechanism, may have more effect on tumors without pre-existent immunity. These ‘cold' tumors suppress normal anti-tumor immune response and are categorized by very low to no PD-L1 expression on tumor cells," said Joseph Shan, VP of Clinical & Regulatory Affairs at Peregrine. "These latest findings, along with other recently announced clinical and preclinical data from our PS-targeting program, inform our clinical development strategy going forward and provide additional rationale for combining bavituximab with checkpoint inhibitors."
As part of the SUNRISE clinical study protocol, researchers requested but did not require that patients provide a tumor tissue sample at the time of diagnosis. In total, tissue samples were collected from 129 of the trial's 597 patients and were assessed retrospectively for baseline PD-L1 expression levels on tumor cells. Of the 129 tissue samples collected, 122 were evaluable for PD-L1 expression on tumor cells (54 in D+B arm and 68 in D+P control arm). Of the evaluable samples in the D+B arm, 69% demonstrated PD-L1 expression levels < 1%, as compared to 59% in the D+P arm.
Bavituximab is an investigational immune-modulatory monoclonal antibody that targets phosphatidylserine (PS). PS inhibits the ability of immune cells to recognize and fight tumors. Bavituximab is believed to reverse PS-mediated immunosuppression by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS-targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes grants awarded by the National Comprehensive Cancer Network (NCCN) to support 3 different clinical trials of bavituximab treatment combinations. These trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center (MSK) with the goal of evaluating combinations of PS targeting antibodies with checkpoint inhibitors and other immune stimulatory agents. Peregrine's intent behind this strategy is to focus its research and development spending to further validate bavituximab's combination potential as the company seeks to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com

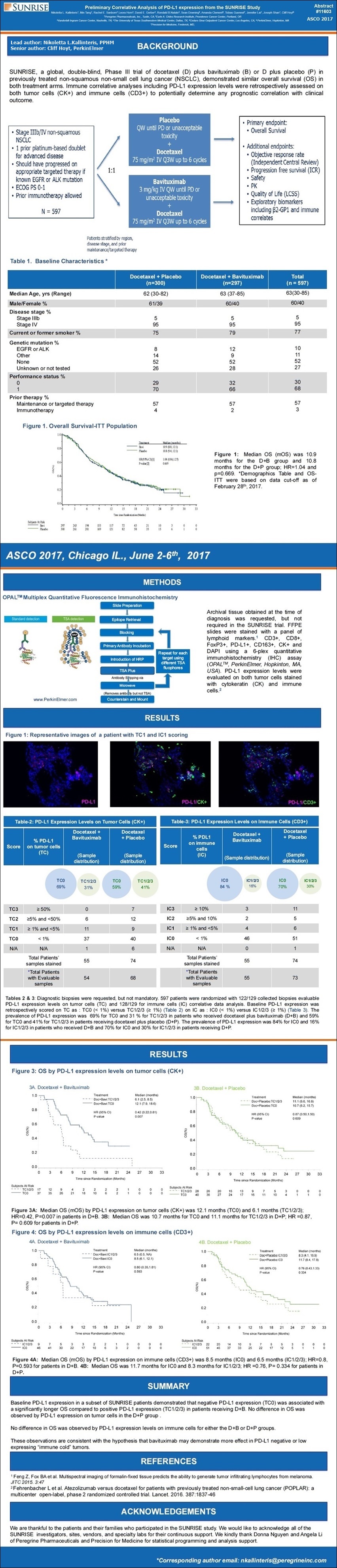
Poster #11603 Image:
http://www.peregrineinc.com/images/stories/pdfs/asco2017kallinteris.pdf
= = = = = = = = = = = = = = = = = = = = = = = = = = = = =
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
Note: this is known Sunrise Biomarker #4 (PD-L1 Expr.)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ORIG. ABSTRACT (superceded by 6-5-17 PPHM PR)...
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab (Opdivo) on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela Dir.Res. (LEAD), Melba Marie Page, Jenny Kreahling ”Nilogen: Bringing the complex tumor microenvironment to life with 3D ex vivo assays”
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org - NOTE: Moffitt’s S.Antonia & S.Altiok are CMO & CSO of Nilogen http://www.nilogen.com/our-team
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. [NOTE: a/o 5-2017, Dr. Antonio is also CMO of Nilogen Oncosystems http://www.nilogen.com ]. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD (also CSO of Nilogen Oncosystems http://www.nilogen.com ) - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic & prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab [BMS’ Opdivo] may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.
Also See: http://tinyurl.com/ydhf6tfx

6-5-17/PR: Peregrine Pharmaceuticals Presents Preliminary Correlative Analysis of PD-L1 Expression from SUNRISE Trial at ASCO 2017
-- Negative PD-L1 Expression was Associated with a Significantly Longer Median Overall Survival Compared to Positive PD-L1 Expression in Patients Receiving Docetaxel Plus Bavituximab
-- Presented Results Support Hypothesis that Bavituximab May Demonstrate Greater Effect in "Cold" Tumors Expressing Low to No PD-L1
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1028997
TUSTIN, June 05, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by advancing its proprietary R&D pipeline and manufacturing high quality products for biotechnology and pharmaceutical companies, today announced the presentation of promising new data from its Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC). Presented results demonstrated that patients in the study's bavituximab treatment arm who had low baseline PD-L1 expression levels had a statistically significant improvement in median overall survival (mOS) as compared to patients in the same treatment arm who had higher baseline levels of PD-L1. Data were presented by Peregrine scientists at the Annual Meeting of the American Society of Clinical Oncology (ASCO), being held June 2-6, 2017 in Chicago.
Data presented demonstrated that patients in the study's docetaxel plus bavituximab (D+B) treatment arm with a pre-treatment PD-L1 expression level on tumor cells of < 1% (TC0) had a mOS of 12.1 months compared to a mOS of 6.1 months for patients with PD-L1 expression >=1% (TC1/2/3) (HR = 0.42 p=0.007). There was no difference in mOS based on PD-L1 expression levels observed in the study's docetaxel plus placebo (D+P) control arm (10.7 months for TC0 vs. 11.1 months for TC1/2/3; HR = 0.87; p=0.609).
"We believe that these latest observations from the SUNRISE trial further support the hypothesis that bavituximab, through its immune modulating mechanism, may have more effect on tumors without pre-existent immunity. These ‘cold' tumors suppress normal anti-tumor immune response and are categorized by very low to no PD-L1 expression on tumor cells," said Joseph Shan, VP of Clinical & Regulatory Affairs at Peregrine. "These latest findings, along with other recently announced clinical and preclinical data from our PS-targeting program, inform our clinical development strategy going forward and provide additional rationale for combining bavituximab with checkpoint inhibitors."
As part of the SUNRISE clinical study protocol, researchers requested but did not require that patients provide a tumor tissue sample at the time of diagnosis. In total, tissue samples were collected from 129 of the trial's 597 patients and were assessed retrospectively for baseline PD-L1 expression levels on tumor cells. Of the 129 tissue samples collected, 122 were evaluable for PD-L1 expression on tumor cells (54 in D+B arm and 68 in D+P control arm). Of the evaluable samples in the D+B arm, 69% demonstrated PD-L1 expression levels < 1%, as compared to 59% in the D+P arm.
Bavituximab is an investigational immune-modulatory monoclonal antibody that targets phosphatidylserine (PS). PS inhibits the ability of immune cells to recognize and fight tumors. Bavituximab is believed to reverse PS-mediated immunosuppression by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS-targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes grants awarded by the National Comprehensive Cancer Network (NCCN) to support 3 different clinical trials of bavituximab treatment combinations. These trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center (MSK) with the goal of evaluating combinations of PS targeting antibodies with checkpoint inhibitors and other immune stimulatory agents. Peregrine's intent behind this strategy is to focus its research and development spending to further validate bavituximab's combination potential as the company seeks to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com

Poster #11603 Image:
http://www.peregrineinc.com/images/stories/pdfs/asco2017kallinteris.pdf

= = = = = = = = = = = = = = = = = = = = = = = = = = = = =
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
Note: this is known Sunrise Biomarker #4 (PD-L1 Expr.)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ORIG. ABSTRACT (superceded by 6-5-17 PPHM PR)...
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab (Opdivo) on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela Dir.Res. (LEAD), Melba Marie Page, Jenny Kreahling ”Nilogen: Bringing the complex tumor microenvironment to life with 3D ex vivo assays”
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org - NOTE: Moffitt’s S.Antonia & S.Altiok are CMO & CSO of Nilogen http://www.nilogen.com/our-team
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. [NOTE: a/o 5-2017, Dr. Antonio is also CMO of Nilogen Oncosystems http://www.nilogen.com ]. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD (also CSO of Nilogen Oncosystems http://www.nilogen.com ) - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic & prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab [BMS’ Opdivo] may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.
Also See: http://tinyurl.com/ydhf6tfx










