Tuesday, November 08, 2016 11:18:51 AM
SITC’16 MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #P194: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #P205: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #P208: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
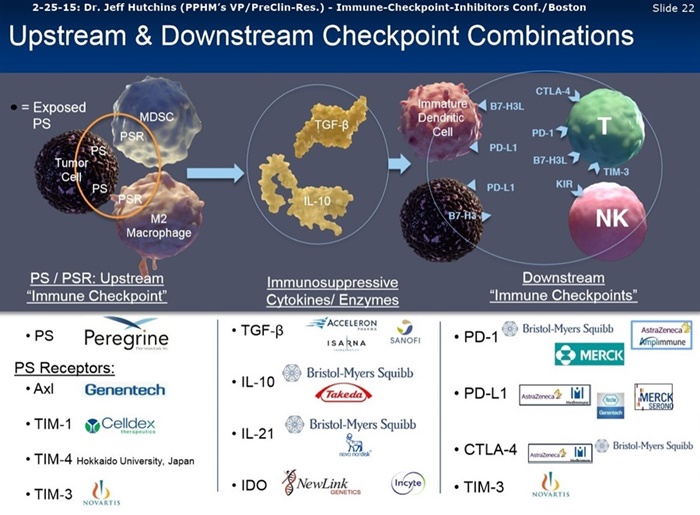
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
----------------
II. P205/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
----------------
III. P208/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #P194: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #P205: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #P208: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
----------------
II. P205/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
----------------
III. P208/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."










