Thursday, September 15, 2016 8:56:00 AM
Lisa Stepp(PhD,PPHM Sen.Dir/MedicalAffairs) today, then EVENTS Heat UP
Sept14-16 2016: “EXL's 11th Medical Affairs Executive Forum”, SanDiego
http://exlevents.com/medical-affairs-executive-forum-west
“The role of medical affairs teams in the life science industry has seen an expansion of functionality & responsibility, especially with easier access to information through digital platforms. A primary focus for medical affairs leadership is to ensure the compliant separation of clinical & commercial teams, particularly regarding their access to therapeutic area experts. Communication channel mgt. will serve as one of the driving themes for the Medical Affairs Strategic Summit (MASS) West 2016...”
9-15-16 11:15–12pm: “The Effects of Company Size on the Establishment of an Advisory Board Benchmark” - Lisa Stepp, PhD, Sen.Dir./Medical Affairs, Peregrine Pharmaceuticals
- - - -
Lisa Stepp (PhD/MBA) joined Peregrine 3-2015 as “Sr. Dir./Medical Affairs” - formerly with Gilead, Allos, Genentech, and Wyeth.
https://www.linkedin.com/in/lisa-stepp-phd-mba-345942a
= = = = = = = = = = = = = = = = =KNOWN UPCOMING:
Sep15: EXL's 11th Medical Affairs Executive Forum, SanDiego http://exlevents.com/medical-affairs-executive-forum-west
...11:15–12pm: Dr. Lisa Stepp, PPHM's PhD/Sen.Dir./MedAffairs, “<uiEffects of Co. Size on the Est. of an Advisory Board Benchmark”
Sep22: Phacilitate’s "Immunotherapy Europe" (Strategic Partnering Event), Berlin http://tinyurl.com/hwxax88
...9:50-10:20am WEBINAR - CEO Steve King is Panelist w/BMS/Merck/Agenus: “Analyzing The Business & Partnering Model in Industry & Academia for the Future Dev. of I-O Combination Therapies”
Sep25-28: 2nd AACR-CRI Intl. Cancer Immunotherapy Conf.”, NYC http://tinyurl.com/hj3ar5q
...Sep26 5:15-7:45pm: Bruce Freimark(Dir.Res/Preclin.Oncology) poster B019, “LAG3 is an Immunotherapeutic Target in Murine Triple- Breast Cancers, Whose Activity is Significantly Enhanced in Combo with PS-Targeting Antibodies”
...Sep26 5:15-7:45pm: Dr. Raymond Birge**(Rutgers) & PPHM poster B119, ”Characterization of a PS, TAM Receptor (Tyro3, Axl, Mertk), PDL1 Axis in Breast Cancer”
**Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. “The Birge laboratory conducts basic science focused on the eradication of cancer.” http://birgelab.org
Oct4-7/Avid/Booth#716: IBC's BioProcess Intl. Conf., Boston http://www.ibclifesciences.com/BPI/overview.xml & http://tinyurl.com/zyblds7
...10-5-16/8-8:30am: Peter Gagnon(Avid's VP/Process-Svcs), ”Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”
Oct7-11: 41th ESMO European Cancer Congress, Copenhagen, Denmark http://tinyurl.com/j2cng2y (Oral Pres. of Topline Data from the Ph3 SUNRISE trial)
...10-10-16 9:15-9:30am: Dr. David Gerber(UTSW) Proffered Oral Presentation, “Top-line Results from Ph3/SUNRISE..." (incl. “initial results from ongoing biomarker analysis”)
...10-9-16 1:00-2:00pm: Jeff Hutchins(VP/PreClin.Res), “Antibody Mediated Blockade of PS Improves Immune Checkpoint Blockade...”
Oct13/10am: Annual SHM, Avenue of the Arts Hotel, Costa Mesa – Final Proxy: http://tinyurl.com/gsrmgs2
Nov9-13: (SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD http://www.sitcancer.org/2016
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
~Dec8: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & I-O”, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Bruce Freimark(Dir.Res/Preclin.Oncology), ”Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
- - - - - - - - - -
9-8-16 CC/Steve King: “Today, we are very pleased to announce that the topline data from our Phase III SUNRISE trial have been accepted for a late-breaking oral presentation at the upcoming European Society of Medical Oncology (ESMO) Congress to be held in Copenhagen in early October [Oct7-11]. ESMO is the premier European oncology meeting, attended by thousands of oncologists. The presentation at ESMO will be a great opportunity to share clinical data from the study in conjunction with initial results from ongoing biomarker analysis, which are already highly encouraging. Biomarker analysis was built into the SUNRISE trial from the beginning, including the collection of thousands of patient samples that could be analyzed once of the trial was unblinded. The primary goal the biomarker analysis is to identify a biomarker pattern, present in patients that receive the most benefit from a bavituximab containing therapeutic regimen, and we look forward to sharing the results of the ongoing analysis, with more data expected later in the year. The impact of the effective biomarker identification is already quite evident in oncology drug development, with the latest evidence being PD1/PD-L1 in the development of Keytruda. These types of biomarkers can only be identified through analysis of larger patient populations, such as in the SUNRISE trial. The results of identifying effective biomarkers can potentially have a huge impact on clinical development, potentially reducing the size of future trials, including making possible biomarker-driven clinical designs which could provide even more rapid readouts. Taken together, these developments are setting the stage for new data throughout the rest of 2016 and into 2017.” http://tinyurl.com/jydtkoy
9-8-16 CC/Joe Shan: “QWe are very excited to have the opportunity for a late-breaking oral presentation on the Phase III SUNRISE trial at the ESMO Congress about a month from now [Oct7-11 http://www.esmo.org/Conferences/ESMO-2016-Congress ]. Even as the trial continues to wind down, treatment, follow-up, and data collection are still ongoing for a number of patients, with some continuing to receive bavituximab over a year. At ESMO, we will be presenting both topline clinical data from the trial as well as initial results from our ongoing biomarker analysis, the results of which have been promising to-date. Because these data have been accepted for presentation at ESMO, they are under embargo until the actual presentation, but we will provide addl. details on the ESMO presentation and our findings as soon as we can. As a reminder, the biomarker program was prospectively built into the SUNRISE protocol, and is designed to help us better understand how bavituximab works and which patients may benefit most. This is a massive effort, and though there are still much ongoing sample testing and data analysis, our goal is to share these results as they become available throughout the rest of this year and into 2017.” http://tinyurl.com/jydtkoy
9-8-16 CC/Rob Garnick: “I’d like to emphasize the strategic importance of the biomarker study, which, as Joe said, was built into the SUNRISE trial. The identification of a positive biomarker or relevant biomarker pattern with clinical efficacy is an important facet of clinical trial design that can be used to inform addl. new studies that in turn might be used to select patients who would best benefit from bavituximab therapy. For example, the identification of the HER2 biomarker was critical in the development of Herceptin. Its importance was only critically established based on the results of a Phase II clinical trial. As Steve previously described, the critical role of the PD-L1 biomarker is emerging as a major factor in the selection of patients for the clinical use of Opdivo & Keytruda. In the case of bavituximab, once a promising biomarker or biomarker pattern is identified, we strategically plan to include these results into potential new clinical trials.” http://tinyurl.com/jydtkoy
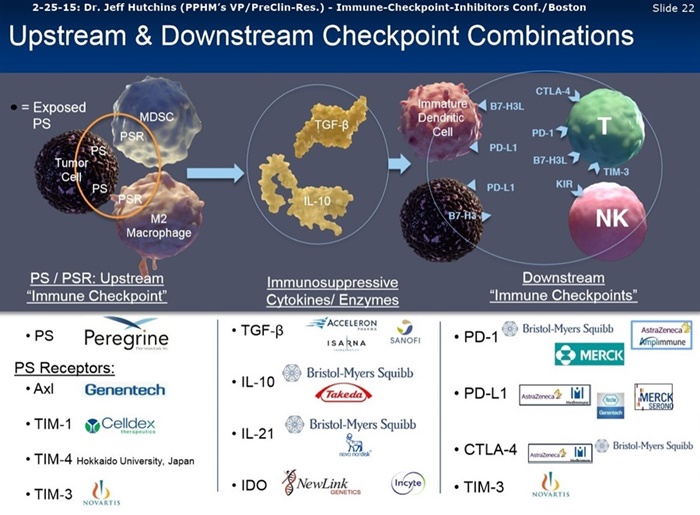
9-8-16 CC/Jeff Hutchins: “I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering CC. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the CRI/AACR Immunotherapy Meeting in NY later this month and at ESMO in early October. We expect the first results from our collaboration with the Wolchok Lab investigators to be presented at SITC in November and we will provide more detailed information as that presentation becomes available.” http://tinyurl.com/jydtkoy
Sept14-16 2016: “EXL's 11th Medical Affairs Executive Forum”, SanDiego
http://exlevents.com/medical-affairs-executive-forum-west
“The role of medical affairs teams in the life science industry has seen an expansion of functionality & responsibility, especially with easier access to information through digital platforms. A primary focus for medical affairs leadership is to ensure the compliant separation of clinical & commercial teams, particularly regarding their access to therapeutic area experts. Communication channel mgt. will serve as one of the driving themes for the Medical Affairs Strategic Summit (MASS) West 2016...”
9-15-16 11:15–12pm: “The Effects of Company Size on the Establishment of an Advisory Board Benchmark” - Lisa Stepp, PhD, Sen.Dir./Medical Affairs, Peregrine Pharmaceuticals
- - - -
Lisa Stepp (PhD/MBA) joined Peregrine 3-2015 as “Sr. Dir./Medical Affairs” - formerly with Gilead, Allos, Genentech, and Wyeth.
https://www.linkedin.com/in/lisa-stepp-phd-mba-345942a
= = = = = = = = = = = = = = = = =KNOWN UPCOMING:
Sep15: EXL's 11th Medical Affairs Executive Forum, SanDiego http://exlevents.com/medical-affairs-executive-forum-west
...11:15–12pm: Dr. Lisa Stepp, PPHM's PhD/Sen.Dir./MedAffairs, “<uiEffects of Co. Size on the Est. of an Advisory Board Benchmark”
Sep22: Phacilitate’s "Immunotherapy Europe" (Strategic Partnering Event), Berlin http://tinyurl.com/hwxax88
...9:50-10:20am WEBINAR - CEO Steve King is Panelist w/BMS/Merck/Agenus: “Analyzing The Business & Partnering Model in Industry & Academia for the Future Dev. of I-O Combination Therapies”
Sep25-28: 2nd AACR-CRI Intl. Cancer Immunotherapy Conf.”, NYC http://tinyurl.com/hj3ar5q
...Sep26 5:15-7:45pm: Bruce Freimark(Dir.Res/Preclin.Oncology) poster B019, “LAG3 is an Immunotherapeutic Target in Murine Triple- Breast Cancers, Whose Activity is Significantly Enhanced in Combo with PS-Targeting Antibodies”
...Sep26 5:15-7:45pm: Dr. Raymond Birge**(Rutgers) & PPHM poster B119, ”Characterization of a PS, TAM Receptor (Tyro3, Axl, Mertk), PDL1 Axis in Breast Cancer”
**Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. “The Birge laboratory conducts basic science focused on the eradication of cancer.” http://birgelab.org
Oct4-7/Avid/Booth#716: IBC's BioProcess Intl. Conf., Boston http://www.ibclifesciences.com/BPI/overview.xml & http://tinyurl.com/zyblds7
...10-5-16/8-8:30am: Peter Gagnon(Avid's VP/Process-Svcs), ”Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”
Oct7-11: 41th ESMO European Cancer Congress, Copenhagen, Denmark http://tinyurl.com/j2cng2y (Oral Pres. of Topline Data from the Ph3 SUNRISE trial)
...10-10-16 9:15-9:30am: Dr. David Gerber(UTSW) Proffered Oral Presentation, “Top-line Results from Ph3/SUNRISE..." (incl. “initial results from ongoing biomarker analysis”)
...10-9-16 1:00-2:00pm: Jeff Hutchins(VP/PreClin.Res), “Antibody Mediated Blockade of PS Improves Immune Checkpoint Blockade...”
Oct13/10am: Annual SHM, Avenue of the Arts Hotel, Costa Mesa – Final Proxy: http://tinyurl.com/gsrmgs2
Nov9-13: (SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD http://www.sitcancer.org/2016
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
~Dec8: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & I-O”, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Bruce Freimark(Dir.Res/Preclin.Oncology), ”Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
- - - - - - - - - -
9-8-16 CC/Steve King: “Today, we are very pleased to announce that the topline data from our Phase III SUNRISE trial have been accepted for a late-breaking oral presentation at the upcoming European Society of Medical Oncology (ESMO) Congress to be held in Copenhagen in early October [Oct7-11]. ESMO is the premier European oncology meeting, attended by thousands of oncologists. The presentation at ESMO will be a great opportunity to share clinical data from the study in conjunction with initial results from ongoing biomarker analysis, which are already highly encouraging. Biomarker analysis was built into the SUNRISE trial from the beginning, including the collection of thousands of patient samples that could be analyzed once of the trial was unblinded. The primary goal the biomarker analysis is to identify a biomarker pattern, present in patients that receive the most benefit from a bavituximab containing therapeutic regimen, and we look forward to sharing the results of the ongoing analysis, with more data expected later in the year. The impact of the effective biomarker identification is already quite evident in oncology drug development, with the latest evidence being PD1/PD-L1 in the development of Keytruda. These types of biomarkers can only be identified through analysis of larger patient populations, such as in the SUNRISE trial. The results of identifying effective biomarkers can potentially have a huge impact on clinical development, potentially reducing the size of future trials, including making possible biomarker-driven clinical designs which could provide even more rapid readouts. Taken together, these developments are setting the stage for new data throughout the rest of 2016 and into 2017.” http://tinyurl.com/jydtkoy
9-8-16 CC/Joe Shan: “QWe are very excited to have the opportunity for a late-breaking oral presentation on the Phase III SUNRISE trial at the ESMO Congress about a month from now [Oct7-11 http://www.esmo.org/Conferences/ESMO-2016-Congress ]. Even as the trial continues to wind down, treatment, follow-up, and data collection are still ongoing for a number of patients, with some continuing to receive bavituximab over a year. At ESMO, we will be presenting both topline clinical data from the trial as well as initial results from our ongoing biomarker analysis, the results of which have been promising to-date. Because these data have been accepted for presentation at ESMO, they are under embargo until the actual presentation, but we will provide addl. details on the ESMO presentation and our findings as soon as we can. As a reminder, the biomarker program was prospectively built into the SUNRISE protocol, and is designed to help us better understand how bavituximab works and which patients may benefit most. This is a massive effort, and though there are still much ongoing sample testing and data analysis, our goal is to share these results as they become available throughout the rest of this year and into 2017.” http://tinyurl.com/jydtkoy
9-8-16 CC/Rob Garnick: “I’d like to emphasize the strategic importance of the biomarker study, which, as Joe said, was built into the SUNRISE trial. The identification of a positive biomarker or relevant biomarker pattern with clinical efficacy is an important facet of clinical trial design that can be used to inform addl. new studies that in turn might be used to select patients who would best benefit from bavituximab therapy. For example, the identification of the HER2 biomarker was critical in the development of Herceptin. Its importance was only critically established based on the results of a Phase II clinical trial. As Steve previously described, the critical role of the PD-L1 biomarker is emerging as a major factor in the selection of patients for the clinical use of Opdivo & Keytruda. In the case of bavituximab, once a promising biomarker or biomarker pattern is identified, we strategically plan to include these results into potential new clinical trials.” http://tinyurl.com/jydtkoy
9-8-16 CC/Jeff Hutchins: “I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering CC. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the CRI/AACR Immunotherapy Meeting in NY later this month and at ESMO in early October. We expect the first results from our collaboration with the Wolchok Lab investigators to be presented at SITC in November and we will provide more detailed information as that presentation becomes available.” http://tinyurl.com/jydtkoy










