Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
More than 80% of intubated COVID patients die in Brazil
https://medicalxpress.com/news/2021-03-intubated-covid-patients-die-brazil.html
I wonder if Mahboob will return the 200,000 shares he got ?
Friends of Leronlimab.
https://www.getleronlimab.com/support.aspx
https://www.sciencedirect.com/science/article/pii/S2589909021000174?dgcid=rss_sd_all
3. Case DESCRIPTION
The male subject was admitted to a London teaching hospital in the United Kingdom with confirmed nasopharyngeal swabs positive for SARS-CoV-2 infection by reverse transcriptase polymerase chain reaction (RT–PCR). He is of mixed race in his late 50’s with critical COVID-19. Other pertinent characteristics include a BMI of 37 kg/m2, prior smoker, and well-controlled hypertension. Following a positive test for SARS-CoV-2 infection, the subject was admitted to hospital with dyspnoea and pyrexia (Day 0). The subject received several investigational treatments prior to treatment with leronlimab. Upon admission, the subject was treated with dexamethasone for 10 days. Remdesivir was initiated on Day 1; a plasma exchange was administered on Day 4 for 10 days. Other drug interventions included intravenous antibiotics. The subject’s condition continued to deteriorate, and ECMO was initiated on Day 19. Four doses of leronlimab (700 mg), obtained from CytoDyn (CytoDyn inc. WA, USA) through an Emergency Investigational New Drug (EIND) application, were administered on Days 79, 86 and 93 and 100 post diagnosis. The subject responded extremely rapidly and was weaned off ECMO between Days 82 to 84, and he was discharged from the ECMO intensive care unit on Day 91. No adverse safety issues were identified with the administration of leronlimab in this subject. Oxygen therapy and intravenous antibiotics for ventilator-associated pneumonia were administered post weaning off ECMO. At last follow up the subject’s condition continues to improve and he is undergoing rehabilitation.
4. Discussion
Recent data suggest that severely dysregulated host immune responses to SAR-CoV-2, referred to as ‘cytokine storm’, may predominately mediate the morbidity and mortality of severe-to-critical COVID-19[13, 22, 23]. Cytokine storm primarily leads to lung inflammation causing acute respiratory distress syndrome (ARDS), but cytokine storm can also result in severe extrapulmonary manifestations, including thrombotic complications, myocardial dysfunction, and liver and kidney injury[24]. CCR5-expressing proinflammatory immune cells such as activated T-cells and macrophages are thought to play a key role in the cytokine storm response to COVID-19, suggesting that drug interventions that target the CCR5 system may represent a promising approach to treating COVID-19[13].
Leronlimab is a C-C chemokine receptor type 5 (CCR5)-specific humanised IgG4 monoclonal antibody. In a Phase 2, placebo-controlled, randomized clinical trial in subjects with mild-to-moderate COVID-19 leronlimab was shown to provide clinical benefit, primarily in subjects with more severe disease. A number of recent case series have demonstrated that treatment with leronlimab restores immune function and achieves clinical improvement in people with critical COVID-19. Taken together therapeutic interventions that target the chemokine receptor–ligand system may be an effective approach to treating COVID-19 because chemotaxis and trafficking of CCR5 expressing proinflammatory immune cells are thought to play an important role in this process. Importantly the long-term safety of leronlimab is already well established in the treatment of HIV[[17], [18], [19]].
This case is of particular interest because to the best of our knowledge this subject received ECMO for the longest period of any person in the United Kingdom with COVID-19 (66 days). He received his first dose of leronlimab on Days 79 after diagnosis and was successfully weaned off ECMO between Days 82 to 84 and discharged from the ECMO intensive care unit on Day 91. Considering the length of time this subject was on ECMO and the speed of the subject’s response to leronlimab we believe that this case adds critical insight to the growing body of evidence for leronlimab in treatment of critical COVID-19.
Perhaps you may want to read another letter written to Dr.Fauci
https://www.villagevoice.com/2020/05/28/an-open-letter-to-dr-anthony-fauci/
He does not know what he does not know. That was said by a former Chairman of Gilead, who also appeared to have no pharma knowledge.
At a February 12, 2002, news briefing, Secretary of Defense Donald Rumsfeld explained the limitations of intelligence reports: "There are known knowns. There are things we know we know. We also know there are known unknowns. That is to say, we know there are some things we do not know. But there are also unknown unknowns, the ones we don't know we don't know."
A mentor of mine imparted two pearls of wisdom which I have used for over 50 years -
1) Do not attempt to soft peddle the unpalatable.
2) Do not do business with a crook, you will get burned.
Good luck to us.
Or BP can do it for them. How Pfizer tried to bully Argentina and Brazil in exchange for vaccines.
https://www.wionews.com/world/how-pfizer-tried-to-bully-argentina-and-brazil-in-exchange-for-vaccines-366037
What is the source for those amounts ??
How is SNCGF related to MNXXF ??
What I found most informative, from the last 10Q,
On October 30, 2020, the Company signed a term sheet with Alpha Holdings, Inc (Alpha) whereby Alpha, subject to a definitive agreement agreeable to both parties will invest US $ 5,000,000 in the Company. The term sheet outlines the terms of the agreement with a projected closure by December 31, 2020 for an initial investment of $2,500,000 and an additional investment of $2,500,000 on about April 30, 2021. The investment will be in the Company’s common stock to be priced at a low of nine cents per share and a high of 10 cents per share subject to a 90 day weighted average pricing before closing. Upon completion of the $5,000,000 investment Alpha may elect a majority of the board of directors of the Company. The Company has determined that Alpha was treating this as an acquisition and not investment requiring control of the board of directors. the Company determined in the best interest of all shareholders to terminate it discussions with Alpha Holdings, Inc.
They moved on from CYDY, but got rebuffed by NBIO.
Meanwhile BLACK HART over on YMB said -
Verano trading at 28.50 times 290m shares equal 8.265b market cap. Sol has 9.3 percent which is 768.645m of current value. Divided by approx. 66m shares equates to 11.64/share plus 1.34 from Bluma value as per previous posts by calculations from Jtottham, is 12.98/share Cdn. Call other holdings and debt a wash and you can see how undervalued this is. Once the initial momentum from today is over this stock will return to closer to what it's value is actually calculated is. (just my opinion , but in the long run the numbers are usually right, or close to it !) GLTA
CAN $12.98 = US$10.22
https://seekingalpha.com/article/4395356-solcf-finding-value-in-weeds
That article mentioned an SOl price of $5.50, based on a VRNO IPO at US$10.00. However, VRNO is today currently trading around US$23.50, so perhaps we should be looking at $9.00 on SOLCF.
https://thedeepdive.ca/verano-holdings-to-commence-trading-this-morning/
Coming soon, another SOL investment being bought - BLUMA Wellness.
https://seekingalpha.com/article/4400961-bluma-wellness-built-in-profits-downside-protection-from-cresco-labs-acquisition
Chicago pot giants GTI, Verano quietly team up in joint venture
https://chicago.suntimes.com/cannabis/2021/2/9/22275233/marijuana-cannabis-illinois-dispensary-gti-verano-ildisp-nh-medicinal-dispensaries
OGI seems to be where the action is. Opened Monday @ $2.54, is now $7.00 after hours. Sold 55% and 58% of float Tue & Wed. Their sweet spot is the deal with Hyasynth, which had/has backing from Bill Gates. https://hyasynthbio.com
From cbdstockwatcher on the MGCLF board.
- MGC Pharmaceuticals (MXC) has appointed Nicole Godresse as Global Chief Sales Officer, effective immediately
- Nicole has more than 20 years experience in the pharmaceutical and healthcare industries, having worked with the likes of Johnson & Johnson and Eli Lilly
- Her most recent role at Tilray saw her play a part in launching the first medical cannabis brand legally in Australia and New Zealand
- She also helped launch one of the first medical cannabis clinics in the country and deliver some of Australia's first government-funded medical cannabis trials
https://themarketherald.com.au/mgc-pharmaceuticals-asxmxc-appoints-nicole-godresse-as-global-chief-sales-officer-2021-01-13/
Nicole's target (KPI) is for MGC Pharmaceuticals to reach $1million in sales per month.
https://www.proactiveinvestors.com.au/companies/news/939936/mgc-pharma-s-new-head-of-global-sales-has-big-ambitions-and-says-they-re--ready-to-go--939936.html
They do that all the time, report something, only to drop it as the night progresses. This time they are not even showing volume ?
OGI opened on 1/4/21 @ $1.36 closed today $2.41
Looks like the Cannabis sector is on the move.
https://www.booker.senate.gov/news/press/booker-wyden-schumer-joint-statement-on-cannabis-reform-legislation
Thats right, a JOINT statement. lol.
https://www.booker.senate.gov/news/press/booker-appointed-to-senate-agriculture-committee
ALEAF was 0.51 on 2/1/21. Closed today 0.698
SOLCF was up .24 today, based on one of its holdings VERANO Holdings doing an IPO next week. Should be good for another .50 hopefully.
No. The ticker for the IPO has not yet been announced. I tagged $SOL because they own 12.6% of Verano and it is a way of indirectly investing in Verano ahead of the IPO for those who feel inclined to do so 😊 pic.twitter.com/9jVrswtCeO
— jungle.java (@junglejava1) January 22, 2021
Took a minute position in AMYZF - small cash balance in an account.
Giddy up
Looks like the GME action may be trailing off today.
Meanwhile CYDY is up $1.00+ and is trading over 9.2MM.
BANG !
The BANG stocks (Blackberry (NYSE: BB), AMC Entertainment (NYSE: AMC), Nokia Inc. (NYSE: NOK), GameStop Corp (NYSE: GME) are experiencing wild price swings in recent days. GameStop is on a meteoric run up in price fueled by what CNBC called a “Reddit Rebellion.”
https://insiderfinancial.com/reddit-rebellion-targeting-the-short-mafia-next/180939/
What interesting times.
I see over on Reddit, they are saying they are not the cause of any perceived silver short, it is just naturally organic.
Am waiting to see how they will proceed against Zuckerberg and his need to feed off of peoples data.
I think it will come to pass tomorrow, Monday. A few minutes ago I was seeing B/A $29/$30, now it's down to $28/$29. Spread has gone from $0.10 to $1.00 in my neck of the woods. I also just looked up Reddit users, and Google says 330 million monthly active users, of which USA is under 25 million.
https://www.kitco.com/news/video/show/Market-Analysis/3193/2021-01-31/Silver-price-moves-6-on-short-squeeze-pressure-is-that-it-Phil-Streible#_48_INSTANCE_puYLh9Vd66QY_=https%3A%2F%2Fwww.kitco.com%2Fnews%2Fvideo%2Flatest%3Fshow%3DMarket-Analysis
The times are about to get more interesting.
https://www.reddit.com/r/Silverbugs/comments/l6fnto/rwallstreetbets_looks_like_it_might_start_leaning/
WSJ interviewed a guy about GME, he said -
“Please tell the wolf of
Wall Street that the pigeon of
San Francisco is gonna eat your
lunch,”
Seen on another board....
Reported on another board that the Schall Attorney's office had to take their call center offline due to being flooded with angry phone calls. LOL. I guess they didn't know they were messing with Cytonuts!
Ain't that something. A message board recommends buying a stock, and hedge funds are annoyed by that.
I would have thought that funds were selling to get cash to cover their underwater shorts, however volume does not support that. Will have to wait for tomorrow to see.
Had to share this Mathematics:
This comes from 2 math teachers with a combined total of 70 yrs. experience.
It has an indisputable mathematical logic.
This is a strictly ..... mathematical viewpoint.... and it goes like this:
What Makes 100%?
What does it mean to give MORE than 100%?
Ever wonder about those people who say they are giving more than 100%? We have all been to those meetings where someone wants you to give over 100%.
How about achieving 103%?
What makes up 100% in life?
Here's a little mathematical formula that might help you answer these questions:
If:
A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
Is represented as:
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26..
Then:
H-A-R-D-W-O-R-K
8+1+18+4+23+15+18+11 = 98%
And
K-N-O-W-L-E-D-G-E
11+14+15+23+12+5+4+7+5 = 96%
But ,
A-T-T-I-T-U-D-E
1+20+20+9+20+21+4+5 = 100%
And,
B-U-L-L-S-H-I-T
2+21+12+12+19+8+9+20 = 103%
AND, look how far ass kissing will take you.
A-S-S-K-I-S-S-I-N-G
1+19+19+11+9+19+19+9+14+7 = 118%
So, one can conclude with mathematical certainty, that while Hard work and Knowledge will get you close, and Attitude will get you there.
Its the Bullshit and Ass Kissing that will put you over the top.
Now you know why some people are where they are!
Australian daily volume can be seen on Yahoo Finance, under MXC.AX
https://finance.yahoo.com/quote/MXC.AX/history?p=MXC.AX
During the quarter, MGC Pharma completed the 100% acquisition of the operating telehealth clinic-based assets, data and intellectual property of Medicinal Cannabis Clinics (MCC), a wholly owned subsidiary of Cannvalate Pty Ltd, with over 300 patient consults conducted since completion on the 23rd of November. This following the signing of a binding term sheet in July 2020. Consideration of $1m MGC Pharma Ordinary Shares (2/3 subject to trading restrictions) and $400,000 in cash has been paid for the acquisition.
Alongside revenue generated from consults, this acquisition provides MGC Pharma with an operating platform with both import and export capacity that will significantly expand market access and provide control of the supply chain from manufacturing through to patients.
The acquisition also allows the Company to continue providing its high-quality GMP certified medications to patients in Australia and further improves profit margins while keeping product at the current competitive prices.
The acquisition of the MCC Assets is the next step in building on-the-ground distribution assets allowing the Company to wholesale and distribute directly to other clinics and pharmacies to reduce storage and distribution costs.
Launch of CannEpil® App
As part of its ongoing work with the Royal Melbourne Institute of Technology (RMIT), MGC Pharma launched the CannEpil® App, and is providing medical access to the International Library of Cannabinoids (ILC).
The App is a cross platform application available to download from both the Apple App Store and Google Play Store and is designed to be used by patients (or the patient’s guardian) taking CannEpil® as a prescription treatment. The App will record patient responses to medical questionnaires as part of their treatment plan and the treating practitioner will be able to view the responses in real-time.
The ILC is a world first centralised platform compiling the diverse range of existing data on the therapeutic benefits of cannabinoids. The ILC database has been designed to collect comprehensive information about clinical trials, including details of diseases and follow up treatments, as well as product identifiers, including genetics, grow conditions and chemical profile to provide doctors with an encyclopaedia of exhaustive information on the best treatment for patients using cannabinoids.
First shipment of MP Line products directly to patients in Brazil
MGC Pharma’s first batch of MP Line products were shipped directly to patients in Brazil in October 2020, through its binding supply and distribution agreement with Brazil-based ONIX Empreendimentos e Participações (‘ONIX’). MGC Pharma is the first company globally to ship high THC formulations directly to a patient’s door in Brazil, without the need to visit a pharmacy.
The shipment was completed under Brazil’s Compassionate Use Program following the receipt of patients’ prescriptions provided by an ONIX referring doctor. ONIX currently has more than 100 referring doctors in Brazil able to prescribe cannabinoid products under the Compassionate Use Program and is targeting to have over 1,000 referring doctors by mid-2021.
Research and Development
Completion of Phase II clinical trial on COVID-19 patients
MGC Pharma’s Phase II double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of anti-inflammatory treatment, ArtemiCTM, on 50 patients diagnosed with COVID-19 has completed.
The trial included 50 patients of which 33 were in the treatment group and 17 in the placebo group and took place across three independent hospital sites across Israel and India.
The full results have demonstrated to improve the health status of COVID-19 patients delivering a NEWS score of less than or equal to 2. None of the patients in the treatment group required additional oxygen, mechanical ventilation or admission to intensive care where all of these events were reported in the placebo group. The average NEWS score of patients in the placebo group was 2.25 statistically significantly higher (p<0.04) than in the treatment group – 0/5.
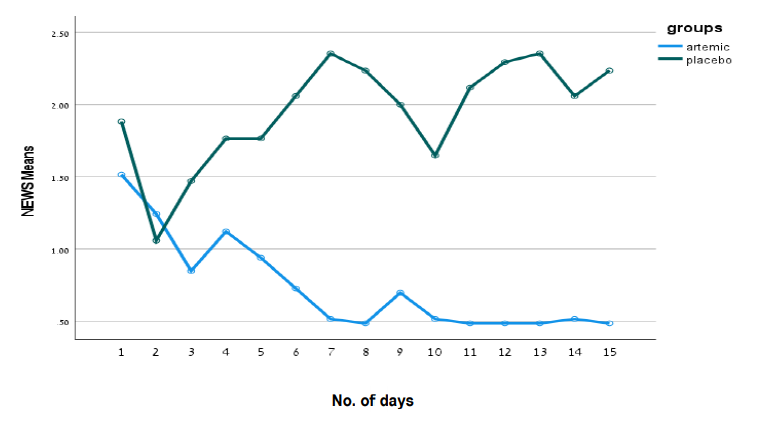
The Trial met all the FDA requirements for a COVID-19 study including population diversity (age, medical history, and genetic diversity) and demonstrated a full safety profile with no drug related adverse events. This resulted due to ArtemiCTM and the trial being focused on the immunomodulation specific for the prevention of cytokines storm, as opposed to other immunomodulators.
These results also follow safety and toxicity testing completed on mice and in line with FDA requirements for product registration requiring two types of rodents in pre-clinical trials. MGC Pharma completed an in vivo safety and toxicity pre-clinical study, including histology testing, on 24 rats. This included four groups with three study drug dosages being 48ug, 96ug and 196ug per kg rat and a control group.
The rats were observed and tested for clinical changes over seven days. This study included pathological examination of the organs: liver, heart, brain, spleen, spinal cord, sciatic nerve, kidney (L+R), lungs and tongue.
Further successful results from pre-clinical glioblastoma research
Results from the ongoing pre-clinical research program focused on evaluating cannabinoid formulations in the development of a treatment of the most aggressive and therapeutically resistant brain tumour, glioblastoma, have shown further successful results.
The pre-clinical in-vitro research program is being conducted in collaboration with the National Institute of Biology (‘NIB’) and the Neurosurgery Department at the University Medical Centre in Ljubljana, Slovenia.
The results from 18 patient tumour samples show for the first time that the Company’s proprietary formulation, CBG, exerts a superior effect in impairing the major hallmarks of glioblastoma progression, i.e. fast proliferation and invasion, and particularly enhancing glioblastoma cell death. Moreover, CBG can destroy therapy-resistant glioblastoma stem cells, which are the root of cancer development and extremely resistant to various treatments of this lethal cancer. CBG should present a new yet unexplored modality of glioblastoma therapy that could replace Tetrahydrocannabinol (THC) as a more acceptable add-on or adjuvant treatment strategy.
Financial and Corporate
Completion of unmarketable share parcel
The Company completed the sale of 50,696,634 fully paid ordinary shares pursuant to the Company’s Unmarketable Parcel Sale Facility (UMP Facility). MGC Pharma confirms its shareholder base has now been reduced by 5,067 shareholders. This will significantly reduce the Company’s administrative and corporate costs moving forward.
Progress towards completion of MGC Nutraceuticals sale to Onassis
As announced on 2nd December 2020, under the terms of the sale and purchase agreement for the 100% sale of the Company’s subsidiary MGC Nutraceuticals, MGC Pharma will receive shares equating to a value of US$6 million in Onassis Holdings Corp and the Company has secured an exclusive supply agreement for the provision of its CBD, raw materials and proprietary production intellectual property (IP). This follows the signing of a binding acquisition and exclusive CBD supply agreement as announced on 18 June 2020.
During the quarter MGC Pharma provided the 30 June 2020 audited financial statements for MGC Nutraceuticals to Onassis and full settlement of the acquisition is expected to complete over the next quarter. During the December quarter Onassis has commenced the process of finalising the offering submissions for the capital raising with the US SEC. The process from submission to completion is currently expected to complete in H1 2021. Once the offering submission has been approved, Onassis will complete the capital raising which will enable the full and complete settlement of the MGC Nutraceuticals acquisition with MGC Pharma.
Long terms benefits from UN vote to reschedule cannabis
In early December 2020, the United Nations (UN) voted in favour of the removal of cannabis and its derivatives from schedule IV in recommendations from the World Health Organisation (WHO). Cannabis and its derivativities are now contained under Schedule I of the 1961 UN Single Convention on Narcotic Drugs.
This creates a significant opportunity for MGC Pharma by removing red-tape that creates logistical limitations of the movement of products and creates an open pathway for easier and cheaper global distribution. This will also enable significantly more commercial opportunities for MGC Pharma by allowing it to deliver its Mercury Pharma product line to new markets going forward.
Appendix 4C
The Company had $1.57m cash at the end of the December 2020 quarter, with access to $9.25m undrawn from its $15m financing facility with Mercer Street Opportunity Fund LLC (as announced to the ASX on 10 September 2020). In accordance with Section 6 of the attached Appendix 4C, the Company confirms the total $480k was for executive director fees, non-executive director fees and corporate costs during the quarter.
As detailed in the Appendix 4C, expenditure for the quarter has been spent on $1.484m for research and development, $1.363m for manufacturing and operating costs, $152k for advertising and marketing, $258k staffing costs and $852k for administration and corporate costs.
https://stocknessmonster.com/announcements/mxc.asx-6A1016539/
Gives a link to a December quarterly report.
Seems like they are doing a Lazarus comeback.
With Shares Outstanding of 1,788,130,339 then 10MM could be a forgotten account. If the volume picks up over the next 2 weeks, we'll know for sure - something's up. Even today is about 1/2 MM over average. They do seem opportune for a 1 :100 Reverse split.
