Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
positive phase 3 results out today ,,, next is FDA decision on Sept 12 = $10++ stock here----LOVE IT
https://finance.yahoo.com/news/ardelyx-announces-positive-results-pivotal-120000881.html
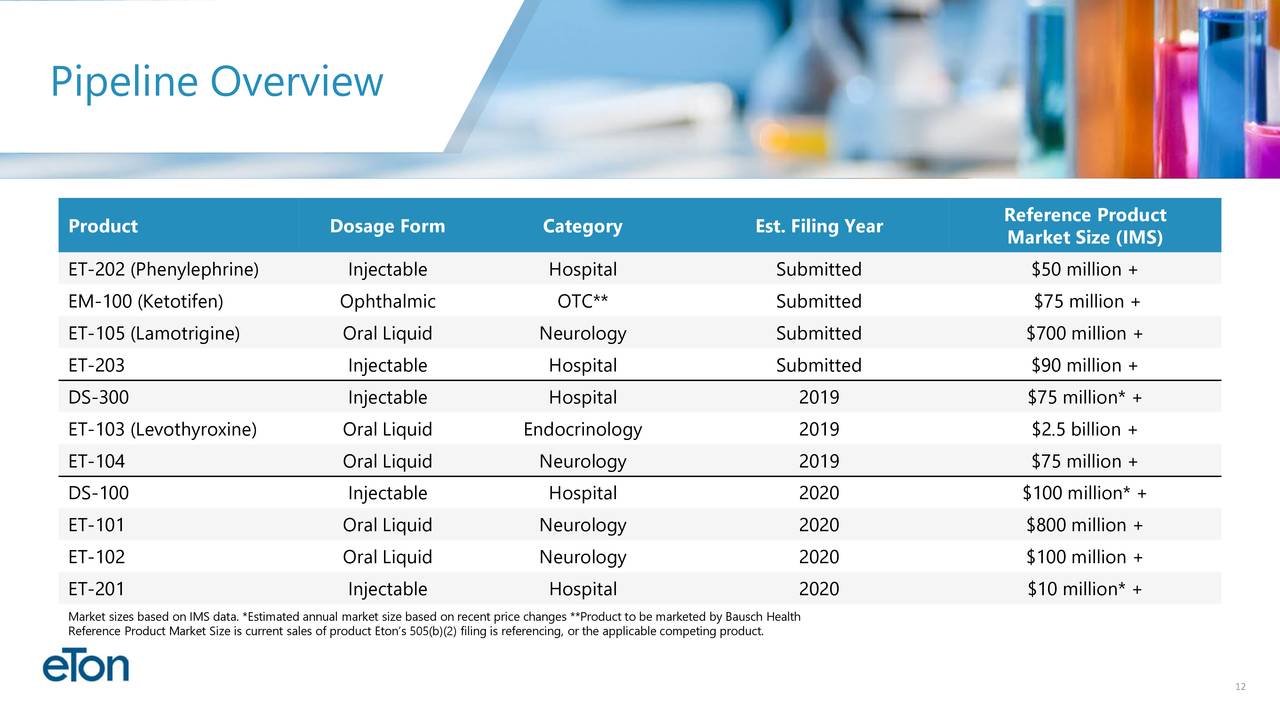
ETON..4 Drugs close to FDA approval..MC $104 M
ETON (Market Cap $104 Mil) 4 Drugs close to FDA approval --first FDA decision on October 21 -- 3 NDA submission expected in Q4 2019 --Low float and undiscovered Stock = Potential 10 Bagger Gem
Potential Upcoming Business Milestones:
ET-103 Clinical Results (Third Quarter 2019)
ET-202 PDUFA Date (October 21, 2019)
Potential ET-103 NDA Submission (Fourth Quarter 2019)
Potential ET-104 NDA Submission (Fourth Quarter 2019)
Potential DS-300 ANDA Submission (Fourth Quarter 2019)
EM-100 Amendment Submission (Fourth Quarter 2019)
ET-105 PDUFA Date (March 17, 2020)
Potential EM-100 FDA Response (First Quarter 2020)
Great move and still undepriced ,,,, FDA Decision for IBS-C drug in 3 Weeks and Phase 3 readout of the hyperphosphatemia trial imminent (this Quarter)
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
$5 coming closer
what a nice jump
ARDX = MC $155 M / Cash $124 M / FDA Decision for potential Blockbuster Drug on September 12 / Second Big Drug with Phase 3 Results also expected this Quarter ($700 M peak sales Potential ) = Potential 10 bagger here guys..RALLYYYYYYYYYYYYYY
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.
5 Biggest New Drug Approvals Potentially on the Way in 2019....August 19, 2019
https://finance.yahoo.com/news/5-biggest-drug-approvals-potentially-100000979.html
5. Tenapanor
Small biotech Ardelyx should be only a few weeks away from some big news. The FDA is scheduled to announce an approval decision for tenapanor in treating irritable bowel syndrome with constipation (IBS-C) by Sept. 12, 2019.
EvaluatePharma expects that the drug could pull in nearly $700 million annually by 2024 if it's approved. Ardelyx hopes that the IBS-C is only the first of two approved indications for tenapanor. The company is also evaluating the drug in a phase 3 clinical study for treating hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis.
Big impacts
FDA approvals for lumateperone and tenapanor would definitely have huge impacts for Intra-Cellular Therapies and Ardelyx. Neither company has an approved product on the market yet.

ARDX = MC $155 M / Cash $124 M / FDA Decision for potential Blockbuster Drug on September 12 / Second Big Drug with Phase 3 Results also expected this Quarter ($700 M peak sales Potential ) = Potential 10 bagger here guys..RALLYYYYYYYYYYYYYY
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.
5 Biggest New Drug Approvals Potentially on the Way in 2019....August 19, 2019
https://finance.yahoo.com/news/5-biggest-drug-approvals-potentially-100000979.html
5. Tenapanor
Small biotech Ardelyx should be only a few weeks away from some big news. The FDA is scheduled to announce an approval decision for tenapanor in treating irritable bowel syndrome with constipation (IBS-C) by Sept. 12, 2019.
EvaluatePharma expects that the drug could pull in nearly $700 million annually by 2024 if it's approved. Ardelyx hopes that the IBS-C is only the first of two approved indications for tenapanor. The company is also evaluating the drug in a phase 3 clinical study for treating hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis.
Big impacts
FDA approvals for lumateperone and tenapanor would definitely have huge impacts for Intra-Cellular Therapies and Ardelyx. Neither company has an approved product on the market yet.

step by step going higher ,fantastic move
$3++ close today possible
Booooooooooooooooooooom and still 3 weeks away from FDA decision could run to $4-5 before the meeting
major breakout on the way stock likely to hit $6+ on FDA approvalby mid next month
5 Biggest New Drug Approvals Potentially on the Way in 2019....August 19, 2019
https://finance.yahoo.com/news/5-biggest-drug-approvals-potentially-100000979.html
5. Tenapanor
Small biotech Ardelyx should be only a few weeks away from some big news. The FDA is scheduled to announce an approval decision for tenapanor in treating irritable bowel syndrome with constipation (IBS-C) by Sept. 12, 2019.
EvaluatePharma expects that the drug could pull in nearly $700 million annually by 2024 if it's approved. Ardelyx hopes that the IBS-C is only the first of two approved indications for tenapanor. The company is also evaluating the drug in a phase 3 clinical study for treating hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis.
Big impacts
FDA approvals for lumateperone and tenapanor would definitely have huge impacts for Intra-Cellular Therapies and Ardelyx. Neither company has an approved product on the market yet.
and phase 3 readout for another indication also expected this quarter
fda decision next month
low float stock
great move
ARDX..FDA Decision on Sept 12...Trading near cash level
ARDX = MC $132 M / Cash $124 M / FDA Decision for Blockbuster Drug on September 12 / Second Big Drug with P3 Results also this Quarter ($700 M peak sales Potential ) =MONSTER OPPORTUNITY potential 10 bagger here guys .
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.

ARDX = MC $132 M / Cash $124 M / FDA Decision for Blockbuster Drug on September 12 / Second Big Drug with P3 Results also this Quarter ($700 M peak sales Potential ) =MONSTER OPPORTUNITY potential 10 bagger here guys
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.

ARDX = MC $132 M / Cash $124 M / FDA Decision for Blockbuster Drug on September 12 / Second Big Drug with P3 Results also this Quarter ($700 M peak sales Potential ) =MONSTER OPPORTUNITY potential 10 bagger here guys
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.

ARDX = MC $132 M / Cash $124 M / FDA Decision for Blockbuster Drug on September 12 / Second Big Drug with P3 Results also this Quarter ($700 M peak sales Potential ) =MONSTER OPPORTUNITY potential 10 bagger here guys
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.

ARDX = MC $132 M / Cash $124 M / FDA Decision for Blockbuster Drug on September 12 / Second Big Drug with P3 Results also this Quarter ($700 M peak sales Potential ) =MONSTER OPPORTUNITY
Remaining Expected 2019 Milestones
The company's New Drug Application for U.S. marketing authorization of tenapanor for patients with IBS-C has a target action date under the Prescription Drug User Fee Act (PDUFA) of September 12, 2019.
Results from the AMPLIFY clinical trial, the company's Phase 3 clinical trial evaluating tenapanor's efficacy in combination with phosphate binders, are currently expected to be announced in the third quarter of 2019.
Results from the PHREEDOM clinical trial, the company's second Phase 3 clinical trial evaluating tenapanor as a monotherapy treatment for hyperphosphatemia in patients with end-stage renal disease (ESRD) who are on dialysis, are currently expected to be announced in the fourth quarter of 2019.
The company's most promising late-stage product candidate is called Tenapanor. Thanks to a unique mechanism of action, this molecule demonstrates potential to treat two separate conditions: irritable bowel syndrome with constipation (IBS-C) and hyperphosphatemia in end-stage renal disease (ESRD).
IBS-C: An estimated one million Americans currently are taking or seeking prescription medication for IBS-C, while millions more suffer from IBS-C but go undiagnosed. Unfortunately, just 40% of patients in need of medication are sufficiently treated today because existing treatment options work only in a minority of patients. Tenapanor offers a novel mechanism of treatment for IBS-C in patients who do not respond to available drugs. Additionally, clinical trials have shown that Tenapanor's efficacy and safety profile is comparable to and in some respects better than that of the leading IBS-C drug, which is called Linzess and which generates $1 billion in sales annually.
Hyperphosphatemia: Tenapanor's potential in hyperphosphatemia is even greater than that in IBS-C. Every year, 90,000 ESRD patients die in the United States alone, and elevated phosphate levels are a major cause of ESRD-related deaths. Despite a mortality rate of greater than 20%, almost one-half of all ESRD patients are not compliant with their prescribed pill regimen. To a large extent, this noncompliance is a result of the overwhelming pill burden: ESRD patients, many of whom are limited to just 32 ounces of water intake per day, swallow roughly 20 pills daily with almost one-half of these pills being hyperphosphatemia drugs. Tenapanor could resolve this problem by providing the same level of efficacy and tolerability with only two pills per day.
https://www.forbes.com/sites/adamstrauss/2018/12/31/happy-new-year-here-is-a-2019-stock-idea-for-your-investment-consideration/#6492d88f75d0

Super hot stock from australia called Antisense Therapeutics (ANP.AX /OTC: ATHJF) has low Market cap of $15 Million and a very attractive product pipeline targeting blockbuster markets like DMD and Multiple Scleroses . Phase 2 Results of the DMD Trial expected next Quarter / Former Chairman of Sarepta and inventors of Eteplirsen are on board of this small company , Sarepta (SRPT) went from $8 to over $150 per share = Next Potential 5000%+ Play with this unknown Gem . There is zero interest on the OTC but good volume in australia (ASX) .Its also a super attractive takeover candidate .check it out guys before she gets discovered .GL
insert-text-here
Antisense Therapeutics (ANP.AX / OTC: ATHJF) australian company
Market Cap $15 Million
Cash $3 Million ( enough untill mid 2020)
"Former SRPT Chairman, William (Bill) Goolsbee, is a director on ANP’s Board. Goolsbee is also the Chairman of the company’s scientific advisory board which includes the two Australian scientists (Profs Steve Wilton and Sue Fletcher) who are the inventors of Sarepta’s drug, eteplirsen. Bill has extensive experience and networks in the field of DMD drug development and commercialisation which he is imparting on ANP in their development of ATL1102."
Could Antisense be the next multi-billion Duchenne Muscular Dystrophy biotech?
insert-text-here
“I joined the board of Sarepta in 2007. We were in a very similar circumstance as Antisense,” he told Stockhead.
“We had a low market cap, we had low cash balance, we were fighting to get to the point where we could prove out our drug. Now look at what’s changed in the last 10 years. That’s what we’re trying to achieve at Antisense.”
Company Presentation
insert-text-here
Live Presentation
insert-text-here
Phase II clinical trial in DMD on track for dosing completion
insert-text-here
ASX Junior’s Flagship Immunomodulatory Drug Could Turn the Tide in Duchenne Muscular Dystrophy Treatment
insert-text-here
ANP: Why partnering with big biotech has its benefits insert-text-here
One such disease for which ASX junior Antisense Therapeutics (ASX:ANP) could deliver much improved outcomes is Duchenne Muscular Dystrophy (DMD).
ANP is one of only a few companies working in the DMD space targeting the largely unmet need of treating the inflammation associated with the disease.
This is a market that is significantly underserved, meaning there is substantial value creation potential as evidenced by comparable DMD therapeutics company Sarepta Therapeutics (NASDAQ:SRPT).
ANP anticipate that its drug could be complementary to other DMD programs as treatment development is across all DMD intervention points, as seen below.

CHEK = MC $17 Million / Cash $18 Million / Shares Out 8.2 Million / ultra Low Float stock with MASSIVE upside potential . $20++ stock trading at ridiculous $2 .Super Strong Buy before stock gets discovered .GL
Colorectal Cancer Screening Pill to Be in US Pilot Study
https://www.mddionline.com/colorectal-cancer-screening-pill-be-us-pilot-study
New Presentation
http://ir.check-cap.com/download/CHECK+Corp+Presentation_January+2019.pdf
Check-Cap: This Overlooked Medical Device Company Is Extremely Undervalued
As a result of the much more established market and reimbursement paradigm for capsule endoscopy, in conjunction with C-Scan’s unique ability to circumvent the bowel preparation requirement and detect precancerous polyps, it is conceivable that Check-Cap could scale more than twice as quickly as Given Imaging did since it launched its first product. This implies potential sales for Check-Cap of more than $60M in 2020. Check-Cap believes that the global market opportunity for C-Scan in the US is $5.4B annually (See slide 10 in this presentation), comprised of a global population at age 50 and above of 710 million, an average procedure cost of $600, and a screening frequency of once every 10 years. Hence, $60M of sales contemplates only minimal penetration relative to the aggregate market opportunity. To demonstrate the potential revenue ramp of C-Scan, investors could also study the sales trajectory of Exact Sciences’ (NASDAQ:EXAS) Cologuard. Cologuard generated $26.5M in sales during the first four quarters since the product launched, and within 8 quarters, it exceeded a $100M annualized run-rate.
In my valuation, I was conservative and used only $50M of sales that are only reached in 2022. I used an EV/Sales multiple range of between 5 to 8 (given that the company will still be growing extremely fast). I then conservatively assumed that on top of all warrants being exercised there would be another 100% dilution, so a total of 17M shares. That leads me to a price target in the range of $15.8 and $24.6 at 2022, or 4.5X to 7X current prices. I then assign a 50% chance of success which brings me to a probability weighted price target of $8 to $12.3 or an IRR of 18% to 29%.
However, if you are optimistic and believe the $60M sales in 2020, then $100M of sales is plausible by 2022, and using an 8X multiple, this could potentially be a $800M EV by 2022 or ~$50/share using 15M shares (assuming 75% dilution on top of all warrants being exercised). Obviously, this is pretty much a best case scenario.
CHEK = MC $17 Million / Cash $18 Million / Shares Out 8.2 Million / ultra Low Float stock with MASSIVE upside potential . $20++ stock trading at ridiculous $2 .Super Strong Buy before stock gets discovered .GL
Colorectal Cancer Screening Pill to Be in US Pilot Study
https://www.mddionline.com/colorectal-cancer-screening-pill-be-us-pilot-study
New Presentation
http://ir.check-cap.com/download/CHECK+Corp+Presentation_January+2019.pdf
Check-Cap: This Overlooked Medical Device Company Is Extremely Undervalued
As a result of the much more established market and reimbursement paradigm for capsule endoscopy, in conjunction with C-Scan’s unique ability to circumvent the bowel preparation requirement and detect precancerous polyps, it is conceivable that Check-Cap could scale more than twice as quickly as Given Imaging did since it launched its first product. This implies potential sales for Check-Cap of more than $60M in 2020. Check-Cap believes that the global market opportunity for C-Scan in the US is $5.4B annually (See slide 10 in this presentation), comprised of a global population at age 50 and above of 710 million, an average procedure cost of $600, and a screening frequency of once every 10 years. Hence, $60M of sales contemplates only minimal penetration relative to the aggregate market opportunity. To demonstrate the potential revenue ramp of C-Scan, investors could also study the sales trajectory of Exact Sciences’ (NASDAQ:EXAS) Cologuard. Cologuard generated $26.5M in sales during the first four quarters since the product launched, and within 8 quarters, it exceeded a $100M annualized run-rate.
In my valuation, I was conservative and used only $50M of sales that are only reached in 2022. I used an EV/Sales multiple range of between 5 to 8 (given that the company will still be growing extremely fast). I then conservatively assumed that on top of all warrants being exercised there would be another 100% dilution, so a total of 17M shares. That leads me to a price target in the range of $15.8 and $24.6 at 2022, or 4.5X to 7X current prices. I then assign a 50% chance of success which brings me to a probability weighted price target of $8 to $12.3 or an IRR of 18% to 29%.
However, if you are optimistic and believe the $60M sales in 2020, then $100M of sales is plausible by 2022, and using an 8X multiple, this could potentially be a $800M EV by 2022 or ~$50/share using 15M shares (assuming 75% dilution on top of all warrants being exercised). Obviously, this is pretty much a best case scenario.
MONSTER OPPORTUNITY here on this one which trading heavily under cash and is close to FDA approval
CLSD = Market cap $39 Mil // Cash $65 Mil //NDA acceptance IMMINENT & FDA DECISION by MID YEAR for potential Blockbuster // Shares Out 32 Mil = 15 BAGGER GEM...UNDISCOVERED Stock trading almost 40% below Cash.
Clearside BioMedical (CLSD)
Market Cap: $40 Million
Cash: $65 Million
Price: 1.24
Shares Out: 32 Million
New Presentation
http://ir.clearsidebio.com/static-files/7edc337d-df24-4e66-b1f8-97ff01b3c9a0
Clearside Biomedical Submits New Drug Application for XIPERE™ for the Treatment of Macular Edema Associated with Uveitis
https://www.barrons.com/articles/PR-CO-20181219-911604
Uveitis is a set of ocular inflammatory conditions and is one of the leading causes of vision loss, affecting about 350,000 patients in the U.S. and more than one million worldwide.
If approved by the FDA, XIPERE would be the first therapy for macular edema associated with uveitis. The uveitis market is expected to grow to nearly $550-million in the U.S. and more than $1-billion globally by 2024.
(CLSD) Trading way below cash..check it out guys
CLSD = Market cap $39 Mil // CASH $65 Mil) NDA acceptance IMMINENT & FDA DECISION this year for potential BLOCKBUSTER = 15 Bagger GEem here...Undiscovered Stock trading almost 40% below Cash.
New Presentation
http://ir.clearsidebio.com/static-files/7edc337d-df24-4e66-b1f8-97ff01b3c9a0
Clearside Biomedical Submits New Drug Application for XIPERE™ for the Treatment of Macular Edema Associated with Uveitis
https://www.barrons.com/articles/PR-CO-20181219-911604
Uveitis is a set of ocular inflammatory conditions and is one of the leading causes of vision loss, affecting about 350,000 patients in the U.S. and more than one million worldwide.
If approved by the FDA, XIPERE would be the first therapy for macular edema associated with uveitis. The uveitis market is expected to grow to nearly $550-million in the U.S. and more than $1-billion globally by 2024.
the cheapest FDA Gem out there
going up day after day but its still under radar once it get discovered then $3+ will come quickly
canopy partner delivra skyrocketing but still deeply undervalued at 17 million valuation .check it out guys before she runs higher .
Delivra Corp – The Biotech-Cannabis Company You’ve Never Heard Of
http://thedeepdive.ca/delivra-corp-biotech-cannabis-company-youve-never-heard/
Presentation
http://www.delivracorp.com/wp-content/uploads/2018/11/DVA-Delivra-Corp.-Deck-December-2018.pdf
DVA.V and DLRVF both ticker are from Delivra ..do your own dd and then decide
in usa the currenncy is $ and in canada its C$ which has lower value that US$ .
only listing is on TSXV ticker is DVA
Marktcap $16 million apure joke for this company but it wont stay for long at this price
Partner Deal(s) news is imminent which means upfront payments for DVA .Market Cap of C$ 15 million is a PURE GIFT !
Delivra Receiving Interest for its Expertise and Technology for Use in the Cannabis and Pharma Industries
https://finance.yahoo.com/news/delivra-receiving-interest-expertise-technology-110000081.html
TORONTO , Oct. 1, 2018 /CNW/ - Delivra Corp. (TSXV: DVA - "Delivra" or the "Company"), a scientific and biotechnology company, today announced that it has recently received inquiries from a number of interested parties in relation to the Company's proprietary transdermal delivery system platform, including its proprietary applications for the cannabis and pharma industries.
looking great so far
