Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
aesop1, https://t.co/BYogOUXX8T comments on the 8/9/23 SEC 10-Q for $nwbo. The MD and A Reveal. pic.twitter.com/3wBlUyjjZf This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
I agree with you on one point—there is a lot of positive support for the DCVax-L cell-based platform technology among those who have been following the small pre-commercial company and following the great research that is ongoing at UCLA!
https://twitter.com/mc17595091/status/1666528319209103361?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172579930><i class=
aesop1, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I believe that you are misinformed. Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology was a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















Exactly.
Thanks for the reply!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172456463><i class=
HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
BP is currently running a DCVax-L study at UCLA in combination with one of BP’s blockbuster drugs. Skip down below to review the pembrolizumab study supported by Merck and NIH.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















HyGro,
Thanks for the reply. I have studied the full-context link too—there is so much included on the JAMA Network site! The independent peer review of the DCVax-L cell-based platform technology P3 study is worth exploring, especially in the broader context of the emerging combo data at UCLA!
The Liau podcast interview linked to the publication foreshadows combos and implications for other cancers as Liau repeatedly uses the term “platform.”
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


learningcurve2020,
Wow—I didn’t know that NWBO has the most popular board—interesting! I was only aware that the publication of the independent peer review of the clinical study of the DCVax-L cell-based platform technology has been very popular on the JAMA Network site.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
iclight, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh OCE's Project Orbis provides a framework for concurrent submission and review of oncology products among international partners. Learn more: https://t.co/fiGFjKUo5T#ProjectOrbis
You are misinformed, but thank you for highlighting the topic of the MHRA.
Just like the FDA in the United States, the MHRA is highly significant. The only regulatory agency that anyone could argue is more powerful is the FDA, and the FDA considers the MHRA a partner in advancing public health and innovation in medicine worldwide.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107><i class=



HyGro,
Thanks for posting. Here is the full-context link to the JAMA Oncology independent peer review of the DCVax-L cell-based platform technology P3 study, including the comments that you referred to and additional supplements. The Liau podcast interview is also linked to the publication.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
learningcurve2020 & iclight,
You are misunderstanding the SP. The ‘catalyst’ will be regulatory developments across the globe.
The ‘short & distort’ crowd exploit the price action created by naked shorts or spoofers to tell a negative story. It is one of the strategies that they employ to manufacture disinformation—If the price of a stock looks bad after the spoofers or nakeds put a dent in it, then the short & distort vultures can build a negative story around it.
https://blog.geniusgroup.net/
Fortunately, I believe that the immeasurable value of the murcidencel (DCVax-L) cell-based platform technology will soon outshine and overcome all of the illegal attacks—Its value to medical science and to humanity will render any criminals insignificant.
Avoid online disinformation. Do your own research. Seek trustworthy full-context sources.
Regulatory developments will be the most determinative factor following the independent peer review, and NWBO is currently on the OTC market. PD1 combo data are also immensely significant and developments on that front will also prove consequential going forward.
The value of this cell-based immunotherapy platform technology will be determined by the integrity of the science, and the regulatory process will be the catalyst.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


iclight, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
You repost and repost the same misinformation/disinformation over and over. You need to seek full-context credible sources of independently verifiable information. Please do some research before posting meaningless statements recklessly again and again.
The clinical data in total include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thanks for reposting your opinions once again, but you are still misinformed and need better sources of credible and verifiable information…
The clinical data in total include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.
The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















iclight, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thanks for reposting your opinions again, but I cannot discern any meaning from your recent posts. I believe that you need better sources of credible and verifiable information…
The clinical data in total include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















ilovetech, Immune System Researchers Win Nobel Prize in Medicine https://t.co/SMYpQLLHOK @alphavestcap
Yes! That is why I post the Brown press conference as a signature at the end of almost every response post —so more people might watch, listen, and appreciate the shift toward a new cell-based era of medical technology!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/



https://twitter.com/mc17595091/status/1666528319209103361?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
learningcurve2020,
Thank you for highlighting the safety profile!
Safety is one point that nobody argues about with regard to DCVax-L, not even the critics who have conflicts of interest because they’re involved in competing therapies…
The DCVax-L cell-based technology has rock-solid safety data spanning decades and multiple trials. Additionally, there are compassionate use safety data spanning years. Furthermore, on balance against nGBM and rGBM, no drug could possibly pose a safety risk (see the list of side effects on the Avastin label for example).
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
iclight,
Thanks for the post about Advent and manufacturing!
There have been some recent updates from the FDA, and the new draft guidance released a few days ago is highly relevant and favorable to DCVax-L in terms of manufacturing and approval:


https://www.fda.gov/regulatory-information/search-fda-guidance-documents/manufacturing-changes-and-comparability-human-cellular-and-gene-therapy-products
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172029305
iclight,
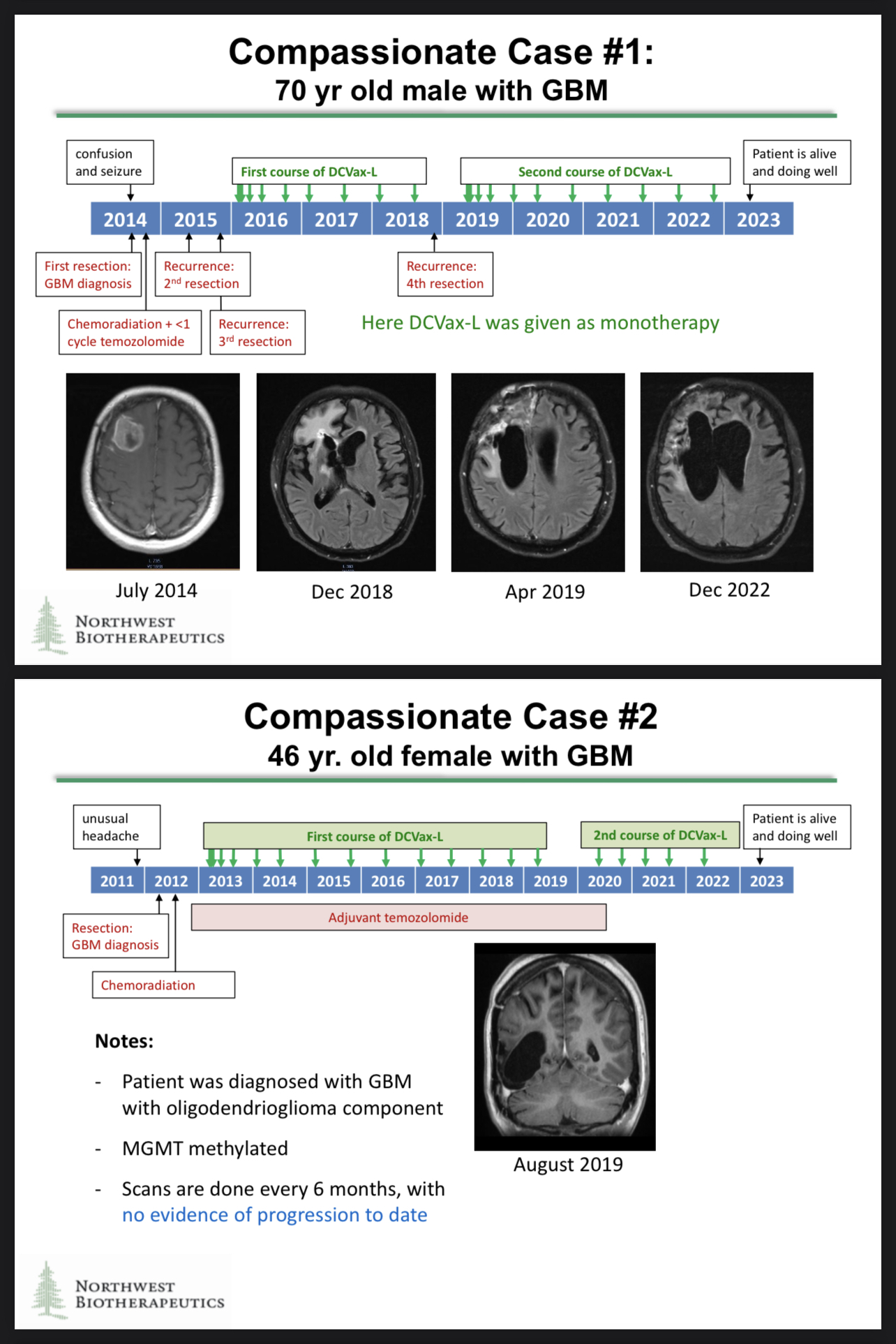
Excellent point! The fact that those patients were alive to have additional resections is significant. Patients who enjoy extended survival have the chance to try any number of additional medical treatments or wellness therapies.
It is time to expand beyond the success of the U.K. Specials Program to make this cell-based platform technology available globally! Stay tuned!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
HyGro,
You are suggesting that we and the FDA should care about comments on the publication, but we should not care about the independent peer review of the clinical study?? That seems “dubious” to me.
This also seems dubious:
Before November 22, we saw a lot of posts about the great value of independent peer review—posts such as these:





After November, we saw a lot of posts about peer reviewers being deceived, or posts that imply that the peer reviewers weren’t part of the process at all:

iclight,
Thank you for reposting again about compassionate use! The Specials Program can help ensure approval so that everyone has access—data gathered from compassionate use can be submitted as supportive extra data on any regulatory application across the globe. Compassionate use is one of the many many reasons that NWBO has built a voluminous initial regulatory application that includes 1.7 million pages.


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
iclight,
Thanks for reposting that once again about ASCO! It was a brilliant presentation in the Industry Expert Theatre! They had a spectacular booth too!
I also agree that combo therapy is the centerpiece of DCVax-L in terms of enhanced efficacy and a broader range of indications beyond GBM!

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171909324
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/



Don’t miss the next presentation in September 2023!






https://www.cns.org/annualmeeting
HyGro,
Thanks for reposting that again, but your information is still all mixed up. How is it possible that HyGro would have access to more information about the study than the JAMA reviewers did?


https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172163592;
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171927087;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172163592;
iclight, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thanks for reposting that again, but I believe that you are misinformed.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















iclight,
Placebo? Are you referring to the Keytruda combo study at UCLA ?

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/


















HyGro,
Thanks for reposting that again, but you are misinformed. I believe that you need to consider better sources.
While glancing over several of your recent reposts today, this came to mind:
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171927087;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172163592;


HyGro,
Are you referring to the independent peer review of the landmark clinical study of the DCVax-L cell-based platform technology that was published in JAMA Oncology?





https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172163592;
kund,
It sounds like you need some better sources of information—I believe you’re misinformed.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873;
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873;
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












HyGro,
Exactly!
I agree 100% that the peer-reviewed landmark P3 will draw the majority of the focus duing regulatory review and advisory. The other clinical data simply provide extra confirmation of safety and efficacy and provide additional evidence of a broader range of use.

The data were peer reviewed by independent physicians and qualified statisticians (JAMA Oncology).
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












exwannabe,
Thanks for requesting more information.
For information about the ECA/crossover design, endpoints, and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
Skip to timestamp 13:00 of the ASCO video to hear about the ECA and the SAP.

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756
iclight,
It is not accurate to suggest that there have not been any submissions to regulators. Most notably, the MIA was approved and is evidence of a regulatory filing plan that has begun to unfold. Further applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
March 2023

https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
HyGro,
Thanks for reposting that again, but your information is outdated and out of context. Furthermore, the P3 is not the only DCVax-L trial/trial design relevant to the DCVax-L cell-based technology.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172268944
BNOS 2023 Begins! Official welcome from our President @BrainTumourSurg to #BNOS2023 pic.twitter.com/4IT0frA4NY
https://www.bnosconference.co.uk/bnos-conference/programme/
iclight,
Thanks for the reply, but you are mixed up.
If you don’t like the ECA or other aspects of the landmark P3, then you have other DCVax-L clinical trials to consider.
All clinical data are relevant to the FDA whenever they must make a decision about the safety and efficacy of any drug.
DCVax-L has been studied in placebo trials too if you like internally controlled trials. DCVax-L is currently being studied in combo with Keytruda at UCLA in an internally controlled study supported by Merck and the NIH (NCI).
Don’t miss the two conferences where Northwest Biotherapeutics is scheduled to participate—BNOS in July (the conference starts today!) and CNS in September!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172204891
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172268944
HyGro,
Thanks for reposting your opinion again, but you are misinformed about the P3 and the FDA.
The data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












HyGro,
Are you referring to the independent peer review of the landmark P3 study that was published in JAMA Oncology?
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847






https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
HyGro
Thank you for posting about the MHRA. The forthcoming regulatory developments will be quite significant. Commercialization and value measured in dollars requires RA approval.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171785690
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://www.nice.org.uk/guidance/indevelopment/gid-ta10143
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
HyGro,
Thanks for asking about the landmark P3! The data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. Always seek credible full-context sources.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












iclight,
I think your posts are mixed up and inaccurate. Seek credible full-context sources. Perhaps consider the independent peer review that was published in JAMA Oncology.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171785690
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
iclight,
It is not accurate to suggest that there have not been any submissions to regulators. Most notably, the MIA was approved and is evidence of a regulatory filing plan that has begun to unfold. Further applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
March 2023

https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
July 2023




https://www.bnosconference.co.uk/bnos-conference/
September 2023






https://www.cns.org/annualmeeting
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
iclight,
The data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. I believe that your post is baseless and you might consider researching credible sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












JTORENCE,
There are two upcoming conferences where Northwest Biotherapeutics is scheduled to participate—BNOS in July and CNS in September.
Conferences convened by world-renowned organizations such as ASCO, SNO, BNOS, NYAS, or CNS are never “nothing burgers” and conferences are never about any single company or any single presentation.
These events are global snapshots in time of the state of cancer care and new discovery. Medical conferences provide a glimpse, in momentary still frame, of the ever-evolving science toward better technologies and the future of oncology.
NWBO fits in the picture frame of world-renowned oncology conferences beautifully.
These events overall give some context to the emerging field of cell-based science and the future of cancer care. FDA officials are embracing the swift evolution of the science toward cell-based therapies and FDA officials are usually in attendance at global conferences.
Northwest Biotherapeutics is a small company on the cutting edge of science and innovation. The DCVax-L cell-based technology is on par with the biggest and best global blockbuster drugs. Consider the interim data of the pembrolizumab combo study at UCLA to illustrate why Northwest Biotherapeutics appears at worldwide conferences alongside companies like Merck and Bristol Myers Squibb.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172204891
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172194377