Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
TNXP..20%+ in Pre-Mkt ..Big rally could start today ..stock is to cheap at $13 million valuation
https://stockaholics.net/threads/tnxp-tonix-pharma.9291/
TNXP ..Phase 3 interim results of the posttraumatic stress disorder (PTSD) trial expected during this Quarter .Extremely low valuation of only $13 million this could run towards $10 and higher especially on positive phase 3 outcome .
from Zacks article :
We are looking forward to the results of the interim analysis for the RECOVERY trial in the first quarter of 2020, and pending the outcome of that analysis, topline results in the second quarter of 2020. Based on the results of 2.8 mg TNX-102 SL in the AFFIRM trial, we believe there is a high likelihood of success for 5.6 mg TNX-102 SL in the RELIEF trial, and we anticipate updates regarding an interim analysis for that trial in 2020.
TNXP ..$2++ tomorrow
TNXP=MC $11 M another LIFETIME OPP trading under cash and with major milestones around the corner see chart below ..Could hit $5++ easily ahead of the phase 3 interim results expected this Quarter and if positive this low float gem could run toward $20 .GL
Tonix Pharma (TNXP)
Market Cap: $12 M
Cash: $18 M
Price:$1.40 M
Shares Out: 8.5 M

TNXP=MC $11 M another LIFETIME OPP trading under cash and with major milestones around the corner see chart below ..Could hit $5++ easily ahead of the phase 3 interim results expected this Quarter and if positive this low float gem could run toward $20 .GL
Tonix Pharma (TNXP)
Market Cap: $12 M
Cash: $18 M
Price:$1.40 M
Shares Out: 8.5 M

TNXP=MC $11 M another LIFETIME OPP trading under cash and with major milestones around the corner see chart below ..Could hit $5++ easily ahead of the phase 3 interim results expected this Quarter and if positive this low float gem could run toward $20 .GL
Tonix Pharma (TNXP)
Market Cap: $12 M
Cash: $18 M
Price:$1.40 M
Shares Out: 8.5 M

could run to $2 before the ADCOM meeting next month
IPCIF = UP 100% since yesterday and still terrible underpriced here at market cap of $18 million .ADCOM meeting mid next month
https://www.advfn.com/p.php?pid=staticchart&s=NO%5EIPCIF&t=37&p=0&dm=0&vol=0&width=280&height=200&min_pre=0&min_after=0
strong buy
(SNGX) Mcap $ 27 M /2 Phase 3 (Cancer Drugs) results expected within 5 months --low float stock ..Potential 10 bagger stock
Major Milestones for Soligenix Bring 2019 to a Strong Close..December 11, 2019
https://marketwirenews.com/news-releases/major-milestones-for-soligenix-bring-2019-to-a-stron-6268818629888602.html
(SNGX) Mkt-cap $ 27 M /2 Phase 3 (Cancer Drugs) results expected within 5 months --low float stock ..Potential 10 bagger stock
Major Milestones for Soligenix Bring 2019 to a Strong Close..December 11, 2019
https://marketwirenews.com/news-releases/major-milestones-for-soligenix-bring-2019-to-a-stron-6268818629888602.html

(SNGX) Mkt-cap $ 27 M /2 Phase 3 (Cancer Drugs) results expected within 5 months --low float stock ..Potential 10 bagger stock

Major Milestones for Soligenix Bring 2019 to a Strong Close..December 11, 2019
https://marketwirenews.com/news-releases/major-milestones-for-soligenix-bring-2019-to-a-stron-6268818629888602.html
ASRT =FDA DECISION IN 11 DAYS....Drug targeting billion dollar market
[ASRT] Price $1.33 // Market Cap $103 // Cash $76 Million // FDA DECISION ON OCTOBER 19 = 500% Potential
Cosyntropin (Synthetic ACTH Depot) is currently not available in the U.S., but Synacten® Depot is approved and broadly used around the world for a wide array of diagnostic and therapeutic uses with broadly similar indications to Mallinckrodt’s H.P. Acthar® Gel. Presently the U.S. ACTH depot market is served by H.P. Acthar Gel which generated an estimated $1.2 billion in net sales for the twelve-month period ending June 30th, 2017. Depomed intends to seek approval for cosyntropin (Synthetic ACTH Depot) in the US and subsequent to approval Depomed and Slán will share in the net sales of cosyntropin (Synthetic ACTH Depot) for a 10-year period (after which time the product will revert back to Slán).

ASRT =FDA DECISION IN 11 DAYS....Drug targeting billion dollar market
[ASRT] Price $1.33 // Market Cap $103 // Cash $76 Million // FDA DECISION ON OCTOBER 19 = 500% Potential
Cosyntropin (Synthetic ACTH Depot) is currently not available in the U.S., but Synacten® Depot is approved and broadly used around the world for a wide array of diagnostic and therapeutic uses with broadly similar indications to Mallinckrodt’s H.P. Acthar® Gel. Presently the U.S. ACTH depot market is served by H.P. Acthar Gel which generated an estimated $1.2 billion in net sales for the twelve-month period ending June 30th, 2017. Depomed intends to seek approval for cosyntropin (Synthetic ACTH Depot) in the US and subsequent to approval Depomed and Slán will share in the net sales of cosyntropin (Synthetic ACTH Depot) for a 10-year period (after which time the product will revert back to Slán).

ASRT =FDA DECISION IN 11 DAYS....Drug targeting billion dollar market
[ASRT] Price $1.33 // Market Cap $103 // Cash $76 Million // FDA DECISION ON OCTOBER 19 = 500% Potential
Cosyntropin (Synthetic ACTH Depot) is currently not available in the U.S., but Synacten® Depot is approved and broadly used around the world for a wide array of diagnostic and therapeutic uses with broadly similar indications to Mallinckrodt’s H.P. Acthar® Gel. Presently the U.S. ACTH depot market is served by H.P. Acthar Gel which generated an estimated $1.2 billion in net sales for the twelve-month period ending June 30th, 2017. Depomed intends to seek approval for cosyntropin (Synthetic ACTH Depot) in the US and subsequent to approval Depomed and Slán will share in the net sales of cosyntropin (Synthetic ACTH Depot) for a 10-year period (after which time the product will revert back to Slán).

Big news imminent like Eskata approval espected within 2 weeks/ Partnerships for the commercial and some pipeline assets and the second phase 3 readout of the A-101 trial expected during this quarter ..Still trading heavily under cash balance market cap of $47 million is a pure gift
ACRS= RALLYYYYYYYYYYYYY
MONSTER OPP here trading MASSIVE UNDER cash balance -Drug Approval imminent +Phase 3 data imminent :
(ACRS)--Market-Cap $42 M --Cash $115 M untill Q3 2021---2 APPROVED Drugs / Drug approval expected before mid October / second Phase 3 readout imminent for a Drug for which there is NO FDA approved rx treatment (first P3 data was strong) ---/New Partner Deal(s) also expected during Q4 = MEGA Rebound Gem with 1000% Upside potential
Actively Seeking a Commercialization Partner for RHOFADE® (oxymetazoline hydrochloride) cream, 1%
Aclaris will also seek strategic partners for ESKATA® (hydrogen peroxide) topical solution, 40% (w/w).
Aclaris is actively seeking a strategic partner to commercialize its drug candidate A-101 45% Topical Solution, an investigational compound being developed as a potential treatment for verruca vulgaris (common warts). The Company’s two ongoing Phase 3 pivotal clinical trials, THWART-1 and THWART-2, in which A-101 45% Topical Solution is being evaluated as a potential treatment for common warts, are progressing as planned. Aclaris has completed enrollment of more than 1,000 patients across these two trials, and data from both trials are expected in the second half of 2019.
Aclaris is actively seeking a development and commercialization partner for its drug candidates, ATI-501 (oral) and ATI-502 (topical), which are investigational Janus Kinase (JAK) 1/3 inhibitor compounds for the potential treatment of alopecia.
ACRS..Trading HEAVILY under Cash and with lotsof upcoming news :
ACRS = Market Cap $43 M / Cash $115 M or untill end 2021 / 2 Marketed Products / 1 Phase 3 trial (common warts) with results expected in Q4 2019 and many other phase 2 programs / because of their strategic change they wants to out-license all their Prodcuts which means HUGE upfront payments for the company in short term (more infos below)= CHEAPEST Biotech you can get at this time could triple very quickly
Aclaris Therapeutics (ACRS)
Market Cap: $43 M
Cash: $115 M or enough untill end 2021
Price 1.08
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-details/aclaris-therapeutics-announces-new-strategic-direction
ACRS aquired a attractive drug from Allergan last year paid $65 m upfront alone for this drug which they now looking to outlicense .
https://www.globenewswire.com/news-release/2018/10/15/1621519/0/en/Aclaris-Therapeutics-to-Acquire-Worldwide-Rights-to-RHOFADE-from-Allergan.html
Pipeline
https://www.aclaristx.com/pipeline/
Largest Shareholders:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
ACRS..Trading HEAVILY under Cash and with lotsof upcoming news :
ACRS = Market Cap $43 M / Cash $115 M or untill end 2021 / 2 Marketed Products / 1 Phase 3 trial (common warts) with results expected in Q4 2019 and many other phase 2 programs / because of their strategic change they wants to out-license all their Prodcuts which means HUGE upfront payments for the company in short term (more infos below)= CHEAPEST Biotech you can get at this time could triple very quickly
Aclaris Therapeutics (ACRS)
Market Cap: $43 M
Cash: $115 M or enough untill end 2021
Price 1.08
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-details/aclaris-therapeutics-announces-new-strategic-direction
ACRS aquired a attractive drug from Allergan last year paid $65 m upfront alone for this drug which they now looking to outlicense .
https://www.globenewswire.com/news-release/2018/10/15/1621519/0/en/Aclaris-Therapeutics-to-Acquire-Worldwide-Rights-to-RHOFADE-from-Allergan.html
Pipeline
https://www.aclaristx.com/pipeline/
Largest Shareholders:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
ACRS..Trading HEAVILY under Cash and with lotsof upcoming news :
ACRS = Market Cap $43 M / Cash $115 M or untill end 2021 / 2 Marketed Products / 1 Phase 3 trial (common warts) with results expected in Q4 2019 and many other phase 2 programs / because of their strategic change they wants to out-license all their Prodcuts which means HUGE upfront payments for the company in short term (more infos below)= CHEAPEST Biotech you can get at this time could triple very quickly
Aclaris Therapeutics (ACRS)
Market Cap: $43 M
Cash: $115 M or enough untill end 2021
Price 1.08
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-details/aclaris-therapeutics-announces-new-strategic-direction
ACRS aquired a attractive drug from Allergan last year paid $65 m upfront alone for this drug which they now looking to outlicense .
https://www.globenewswire.com/news-release/2018/10/15/1621519/0/en/Aclaris-Therapeutics-to-Acquire-Worldwide-Rights-to-RHOFADE-from-Allergan.html
Pipeline
https://www.aclaristx.com/pipeline/
Largest Shareholders:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
ACRS..Trading HEAVILY under Cash and with lotsof upcoming news :
ACRS = Market Cap $43 M / Cash $115 M or untill end 2021 / 2 Marketed Products / 1 Phase 3 trial (common warts) with results expected in Q4 2019 and many other phase 2 programs / because of their strategic change they wants to out-license all their Prodcuts which means HUGE upfront payments for the company in short term (more infos below)= CHEAPEST Biotech you can get at this time could triple very quickly
Aclaris Therapeutics (ACRS)
Market Cap: $43 M
Cash: $115 M or enough untill end 2021
Price 1.08
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-details/aclaris-therapeutics-announces-new-strategic-direction
ACRS aquired a attractive drug from Allergan last year paid $65 m upfront alone for this drug which they now looking to outlicense .
https://www.globenewswire.com/news-release/2018/10/15/1621519/0/en/Aclaris-Therapeutics-to-Acquire-Worldwide-Rights-to-RHOFADE-from-Allergan.html
Pipeline
https://www.aclaristx.com/pipeline/
Largest Shareholders:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
ACRS..BRUTAL Hot Rebound Play which is massive underpriced here trading wayyy below cash balance
ACRS = Market Cap $43 M / Cash $115 M or untill end 2021 / 1 Marketed Product / 1 Phase 3 trial (common warts) with results expected in Q4 2019 and many other phase 2 programs / because of their strategic change they wants to out-license all their Prodcuts which means HUGE upfront payments forthe company (more infos below)= CHEAPEST Biotech you can get at this time
Aclaris Therapeutics (ACRS)
Market Cap: $43 M
Cash: $115 M or enough untill end 2021
Aclaris Therapeutics Announces New Strategic Direction
https://investor.aclaristx.com/news-releases/news-release-details/aclaris-therapeutics-announces-new-strategic-direction
Pipeline
https://www.aclaristx.com/pipeline/
Largest Shareholders:
Deerfield Management Company LP...5 893 416
Franklin Advisers, Inc....3 953 203
BlackRock Fund Advisors ...2 359 223
Broadfin Capital LLC...2 280 260
D. E. Shaw & Co. LP....2 049 462
Sofinnova Ventures, Inc.....1 911 573
Vivo Capital LLC ....1 647 214
Fidelity Management & Research Co...1 562 190
Polar Capital LLP...1 443 920
The Vanguard Group, Inc...1 142 022
Market cap $35 M /2 Approvals expected next month
Cipher pharma (CPH.TO / CPHRF) (Market cap $35 M) Cash $10 M /Shares Out 26M / Profitable Pharma / 2 Drug Approvals expected next month /undiscovered Low Float stock / =Potential 10 Bagger here guys .GLTA
Presentation
https://filecache.investorroom.com/mr5ircnw_cipher/217/IR%20Deck%20May%202019%20Final.pdf
Cipher Pharma anticipates several key milestones in 2019 that will continue the growth of its Canadian commercial platform, including:
Regulatory approval for plecanatide from Health Canada in Q4 2019 ($200+ Million Market)
Regulatory approval for A-101 from Health Canada in Q4 2019 ($100+ Million Market)
Commercial launch of XYDALBA (dalbavancin) ($60 Million Market)
North American top line results for MOB-015 in Q4 2019
Market cap $35 M /2 Approvals expected next month
CPHRF (Market cap $35 M) Cash $10 M /Shares Out 26M / Profitable Pharma / 2 Drug Approvals expected next month /undiscovered Low Float stock / =Potential 10 Bagger here guys .GLTA
Presentation
https://filecache.investorroom.com/mr5ircnw_cipher/217/IR%20Deck%20May%202019%20Final.pdf
Cipher Pharma anticipates several key milestones in 2019 that will continue the growth of its Canadian commercial platform, including:
Regulatory approval for plecanatide from Health Canada in Q4 2019 ($200+ Million Market)
Regulatory approval for A-101 from Health Canada in Q4 2019 ($100+ Million Market)
Commercial launch of XYDALBA (dalbavancin) ($60 Million Market)
North American top line results for MOB-015 in Q4 2019
clinical results should be released any day now
clinical results of ET 103 trial imminent and next FDA decision in 60 days ..
Presentation
https://seekingalpha.com/article/4282869-eton-pharmaceuticals-eton-investor-presentation-slideshow#
4 Drugs nearing FDA approval first one in October .....
(ETON) Market Cap $99 Million / Cash $16 Million enough until late 2020 / 4 Drugs awaiting FDA approval/ first FDA decision on October 21 / 3 NDA submission expected in Q4 2019 (more infos below) / Low float and undiscovered Stock with massive upside potential .
Potential Upcoming Business Milestones:
ET-104 Clinical Results (Third Quarter 2019)
ET-103 Clinical Results (Third Quarter 2019)
ET-202 PDUFA Date (October 21, 2019)
Potential ET-103 NDA Submission (Fourth Quarter 2019)
Potential ET-104 NDA Submission (Fourth Quarter 2019)
Potential DS-300 ANDA Submission (Fourth Quarter 2019)
EM-100 Amendment Submission (Fourth Quarter 2019)
ET-105 PDUFA Date (March 17, 2020)
Potential EM-100 FDA Response (First Quarter 2020)
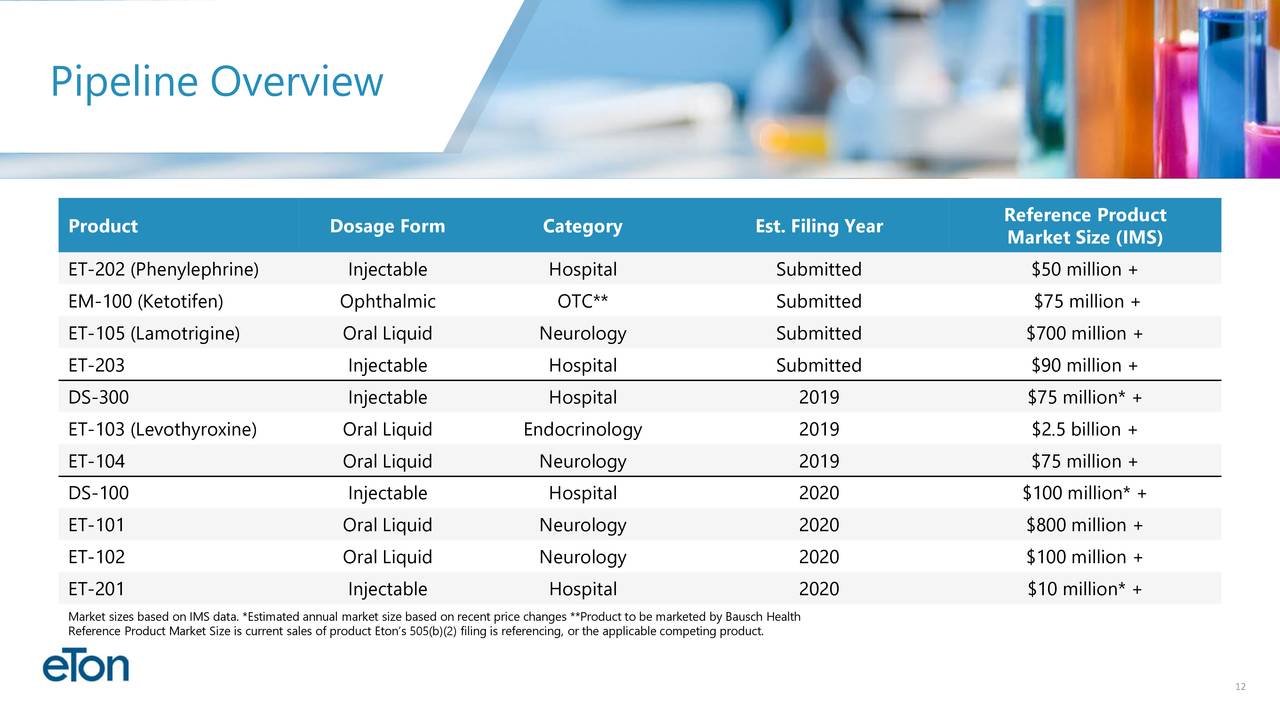
4 Drugs nearing FDA approval first one in October .....
(ETON) Market Cap $99 Million / Cash $16 Million enough until late 2020 / 4 Drugs awaiting FDA approval/ first FDA decision on October 21 / 3 NDA submission expected in Q4 2019 (more infos below) / Low float and undiscovered Stock with massive upside potential .
Potential Upcoming Business Milestones:
ET-104 Clinical Results (Third Quarter 2019)
ET-103 Clinical Results (Third Quarter 2019)
ET-202 PDUFA Date (October 21, 2019)
Potential ET-103 NDA Submission (Fourth Quarter 2019)
Potential ET-104 NDA Submission (Fourth Quarter 2019)
Potential DS-300 ANDA Submission (Fourth Quarter 2019)
EM-100 Amendment Submission (Fourth Quarter 2019)
ET-105 PDUFA Date (March 17, 2020)
Potential EM-100 FDA Response (First Quarter 2020)

insider buying ,, time to load up ahead to the clinical results expected this quarter and the FDA decision in october ...
https://fintel.io/n/us/eton
Massive opportunity to add or for new entry , lots of big news on the way ....
ETON ( $99 M) 4 Drugs awaiting FDA approval/ first FDA decision on October 21 / 3 NDA submission in Q4 2019 (more infos below) / Low float and undiscovered Stock .....
Potential Upcoming Business Milestones:
ET-104 Clinical Results (Third Quarter 2019)
ET-103 Clinical Results (Third Quarter 2019)
ET-202 PDUFA Date (October 21, 2019)
Potential ET-103 NDA Submission (Fourth Quarter 2019)
Potential ET-104 NDA Submission (Fourth Quarter 2019)
Potential DS-300 ANDA Submission (Fourth Quarter 2019)
EM-100 Amendment Submission (Fourth Quarter 2019)
ET-105 PDUFA Date (March 17, 2020)
Potential EM-100 FDA Response (First Quarter 2020)

next sarepta here is possible
phase 2 results of the DMD trial in September or october
huge upside potential here
great buying opportunity indeed , lots of big news around the corner . And EM-100 still likely to be approved in Q1-Q2 2020 its a light crl .
ET-202. Eton has initiated launch preparations for ET-202, Eton’s ready-to-use injectable formulation of phenylephrine. If approved on its PDUFA date of October 21, 2019, Eton anticipates launching the product in the fourth quarter of 2019. Eton believes the addressable phenylephrine market for ET-202 is more than 10 million units annually.
ET-103. The bioequivalence study for ET-103, a liquid formulation of levothyroxine, is ongoing. Eton expects study results in September and, if successful, plans to submit the NDA in the fourth quarter of 2019.
ET-203. The NDA for ET-203, a ready-to-use formulation of a high-volume injectable product, is expected to be submitted by Eton’s partner by the end of the third quarter of 2019.
ET-104. The bioequivalence study for ET-104, a patent-pending oral suspension pursuing a neurological indication, is ongoing. Eton expects study results in September and, if successful, plans to submit the NDA in the fourth quarter of 2019.
Up 10% looks like good news from FDA coming
RALLYYYYYYYYYYYYYYYY ...FDA Decision tomorrow
looks like the fda did already inspect their manufacturing site if there were an issue the company would get a CRL not a new pdufa date of july 11 we will see what happens tomorrow .
this new pipeline update is from last month
https://ir.etonpharma.com/news-releases/news-release-details/eton-pharmaceuticals-announces-licensing-lamotrigine-new-drug
EM-100. EM-100, Eton’s preservative-free ophthalmic solution for allergic conjunctivitis has been assigned a target action date of July 11, 2019. Bausch Health will be responsible for all remaining regulatory and commercial activities surrounding the product. Eton is entitled to a milestone payment upon product launch and a royalty on commercial sales.
this is from early may
https://ir.etonpharma.com/news-releases/news-release-details/eton-pharmaceuticals-reports-first-quarter-2019-financial
EM-100. EM-100 is Eton’s preservative-free ophthalmic solution for the treatment of allergic conjunctivitis. The product is currently under review with the FDA and has been assigned a target action date of May 2019 or July 2019 depending on whether the FDA chooses to inspect EM-100’s manufacturing site. Bausch Health is responsible for all commercialization activities for the product.
Opaleye bought 5% stake or 881K shares of Eton .Now over 8 million shares of the 17.6 Million O/S held by insiders and institutional alone ..With potential FDA approval tomorrow this low float stock could fly like a bullet
http://www.insidercow.com/institution/subject.jsp;jsessionid=B54DEE86A6DD2C22783F1ABDA5F8B1A6?subject=0001710340&company=ETON
B 2019-05-24 17:20:10.0 Eton Pharmaceuticals ETON Opaleye Management Inc 5.0%
