Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
From the 10-K: ''In 2021, we entered into a Research Collaboration Agreement with CRISPR (as amended, the "CRISPR Agreement") to establish research plans for the purpose of collaboratively designing and advancing up to two allogeneic, gene-edited NK-cell therapies and one allogeneic, gene-edited NK+T-cell therapy for use in the treatment of autoimmune diseases, oncology, or infectious disease up to the filing of an application to a regulatory authority to request the ability to start a clinical trial. The first product candidate being developed in partnership with CRISPR was NKX070, and together with CRISPR, we had planned to advance NKX070 in clinical development for the treatment of solid tumors and blood cancers. The second product candidate being developed in partnership with CRISPR was NK+T. Both the NKX070 and NK+T programs have been deprioritized, however, to allow us to focus our resources on developing NKX019 for the treatment of B-cell mediated autoimmune diseases.''
NKTX.................................https://stockcharts.com/h-sc/ui?s=NKTX&p=W&b=5&g=0&id=p86431144783
Anyone know of a reason for NKTX's decline today?
I assume (rightly or wrongly) that expression of HLA class I and II will be disrupted through genetic ablation of beta 2 microglobulin (B2M) and the class II transactivator (CIITA), respectively, allowing evasion of the patient's adaptive immune system, with the CD38 gene also being ablated as well.
If so, CD38-targeted mAbs, such as daratumumab, are approved for the treatment of multiple myeloma. CD38 has also been shown to be highly expressed on activated immune cells, including CD8+ T-cells, CD4+ T-cells, and NK cells. FATE has incorporated the knock-out of CD38 into some of its iPSC-derived products, which uniquely allows for CD38-null, iPSC-derived cells to be combined with CD38-targeted mAbs and avoid fratricide. In one ASH presentation [1], scientists from the company showed that the administration of daratumumab selectively depleted allogeneic CD38+ NK and T-cells and uniquely enabled CD38-null iNK cells to functionally persist through Day 28 compared to non-edited iNK cells. These preclinical data were supported by translational findings from the PhI trial of FT576, its multiplexed-engineered, BCMA-targeted CAR-NK cell product candidate that incorporates the knock-out of CD38, where combination with daratumumab rapidly and selectively eliminated CD38+ patient immune cells through the first month of therapy. The translational findings suggest that following administration of daratumumab, CD38-null iNK cells may avoid rejection by activated host immune cells without requiring conditioning chemotherapy.
Ref:
1 https://ashpublications.org/blood/article/140/Supplement%201/7388/492103/iPSC-Derived-CD38-Null-NK-Cells-in-Combination
(OT) Last week, Tome Bio disclosed its pipeline for the first time. Its two most advanced programs are a pair of off-the-shelf cell therapies for autoimmune kidney diseases, including lupus nephritis. The engineered NKs are among the most heavily edited, with three genes knocked out and four to five genes added.

(OT) A post on LinkedIn by Max Qian (CEO and Co-Founder at Rui Therapeutics):
''I am so pleased today to share an exciting clinical development with our CAR-NK therapeutic platform on Autoimmune disease space. Attached six slides describe our ongoing FIH clinical trial of KN5501 (CD19 CAR-NK) that is used to treat three severe SLE (systemic lupus erythematosus) patients. We innovatively designed this dose-escalation clinical trial with 1+2+3=6 patients to complete low, medium and high dose.
SLEDAI 2K score (SLE disease activity index 2000), the most commonly used system that evaluates the severity of SLE, is used to determines changes in the disease activity of patients diagnosed with SLE: 1) mild activity (SLEDAI-2K = 6); 2) moderate activity (SLEDAI-2K 7 to 12); 3) severe activity (SLEDAI-2K>12).
· The higher the score, the more significant is the degree of disease activity.
· Scores of 6 and above are considered to be consistent with active disease requiring therapy. However, scores greater than 20 are very rare.
· Modifications of score of 6 (improvement) and of 8 (worsening) are considered clinically relevant.
Consistently, as we observed in r/rDLBCL trials, CAR-NK demonstrated a supper safety profile, i.e., no CRS nor ICANS. Very most exciting is that we observed the efficacy from the 1st three patients on low and medium dose (we even have not completed three doses with medium dose patients yet!). This might be the very 1st FIH data available globally to support CAR-NK therapy in SLE.
In earlier this year, we may hear CAR-T on SLE, but latest FDA announcement may give us (patients, hospital and manufacturers) a 2nd thought of the CAR-T approach in autoimmune disease spaces; don’t mention its other safety and economic concerns. Welcome discussions!''

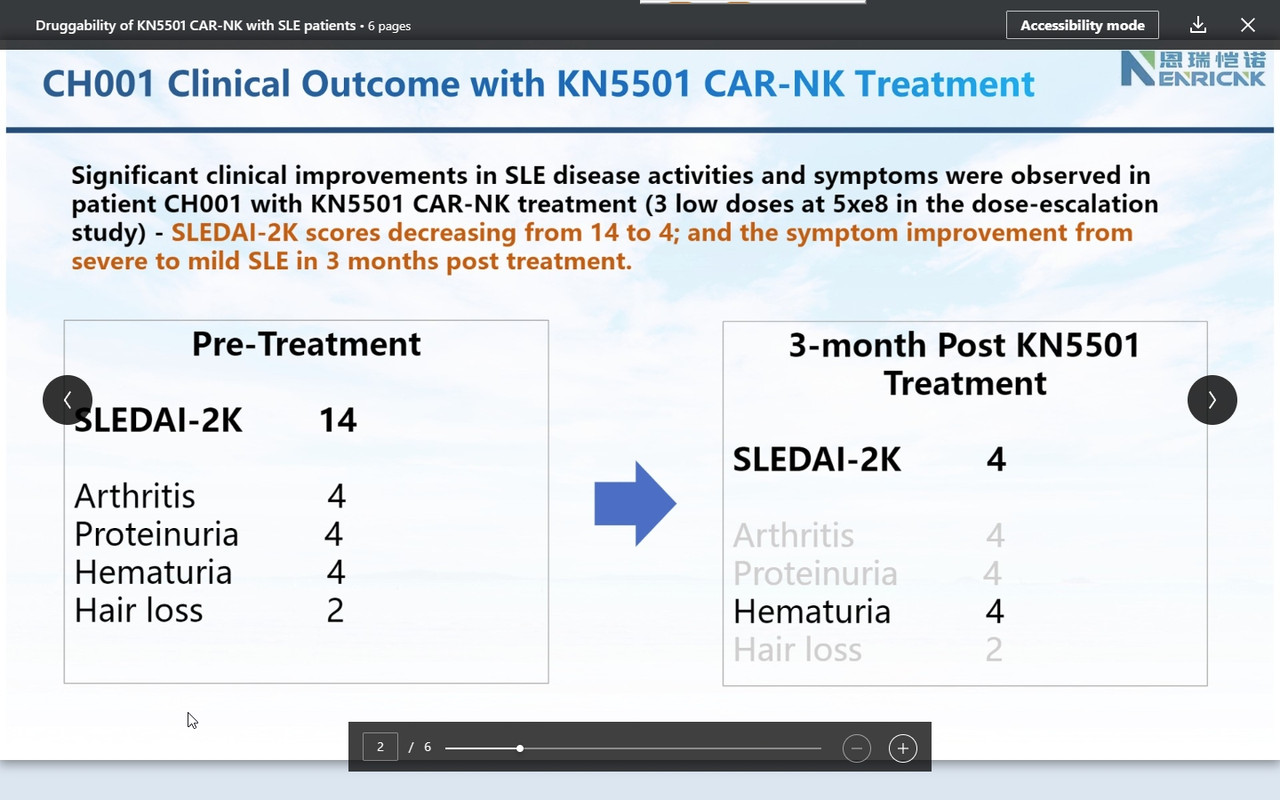
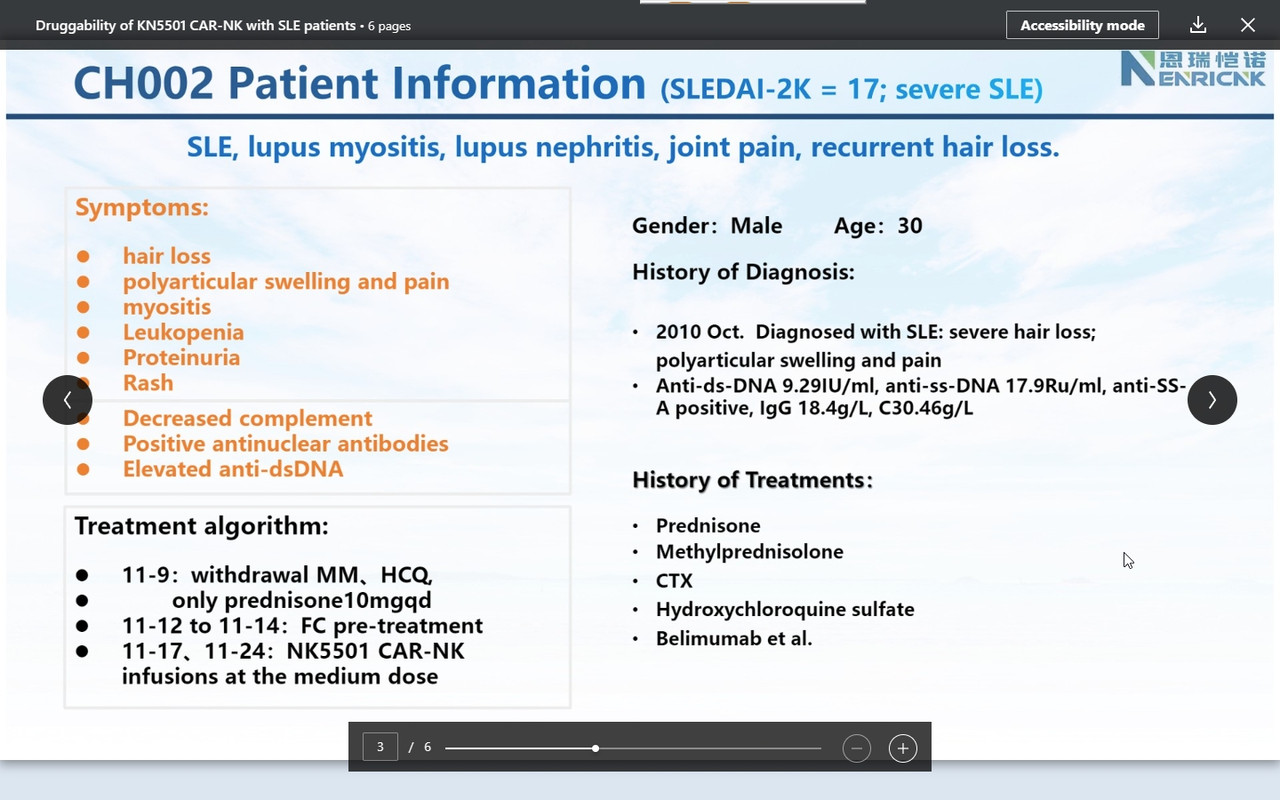
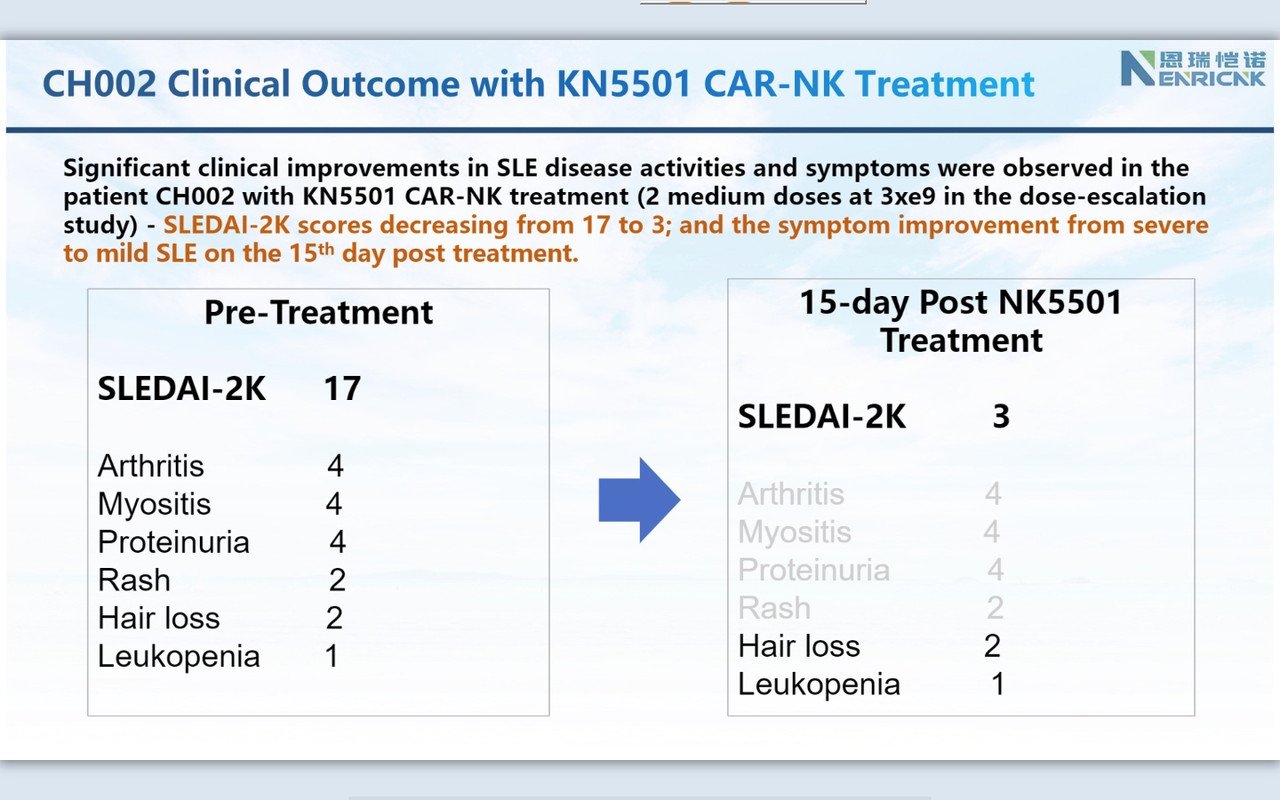

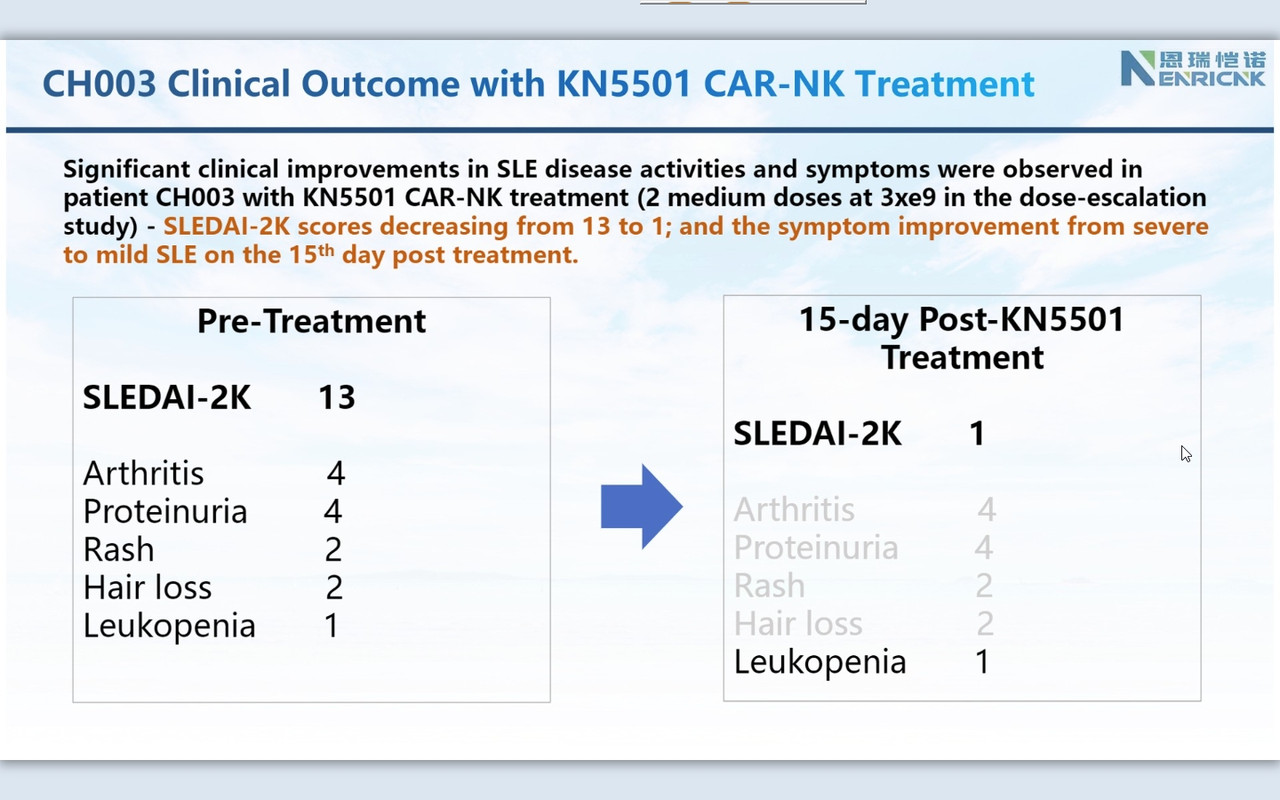
Companies usually raise cash after positive (trial) results. This secondary was on the promise in autoimmune https://www.globenewswire.com/news-release/2024/03/25/2851470/0/en/Nkarta-Announces-Pricing-of-240-Million-Underwritten-Offering.html
AACR abstracts
3603 / 9 - NKX101, an allogeneic off-the-shelf CAR NK cell therapy targeting NKG2D-Ls, has potent anti-leukemic activity alone or in combination with Ara-C https://www.abstractsonline.com/pp8/#!/20272/presentation/7138
3604 / 10 - NKX101, an allogeneic off-the-shelf NKG2D CAR-NK cell therapy, has potent in vitro cytotoxicity against patient-derived AML leukemic stem cells and non-leukemic stem cell blasts https://www.abstractsonline.com/pp8/#!/20272/presentation/7139
AFMD has been hit with the CEO going, and CMO acting as the interim, both the CSO and CFO resigning, and R/S. Let's hope the data is positive as times remain tough in the NK field. If so, investors should probably expect a cash raise.
As for NKTX, hope they start selecting optimal donors and testing ML NKs, especially for solid tumours https://aacrjournals.org/clincancerres/article/27/17/4859/671641/Memory-like-Differentiation-Enhances-NK-Cell https://aacrjournals.org/cancerres/article/83/7_Supplement/893/719928/Abstract-893-Memory-like-differentiation-enhances
I’m at $7.10 just sold half at 10 in less than 20 minutes
MF SZ CTCL must be the best friend for bios. TRIL, AFMD, ... all had good response with their CD47, CD30 ..
Yuppers trust the Nickle
Thanks for heads up this is going nuts lol
Nkarta's Stock Climbs 28% After FDA Approves Drug Application
Nkarta's shares rallied after the company said its investigational new drug application for NKX019, a treatment for lupus nephritis, was approved by the U.S. Food and Drug Administration.
The stock climbed 28% to $1.89 in late morning trading Tuesday. The stock hit a 52-week low of $1.28 earlier this month, and the shares are down 69% this year.
The company said its multi-center, open label, dose escalation clinical trial will assess the safety and clinical activity of NKX019 in patients with refractory lupus nephritis.
Nkarta, a biopharmaceutical company based in South San Francisco, Calif., said it is a developer of engineered natural killer cell therapies.
Lupus nephritis is a form of systemic lupus erythematosus, and is developed by about 40% of the estimated 200,000 people in the U.S. diagnosed with the disease. Of that group, 30% will develop end stage kidney disease, which can be fatal unless they receive dialysis or a kidney transplant.
https://ih.advfn.com/stock-market/NASDAQ/nkarta-NKTX/stock-news/92295683/nkartas-stock-climbs-28-after-fda-approves-drug-a
$NKTX
With all the edits, CAR NK is more like ADC, not NK cells that have also been described as ‘immune-regulatory’ because of their ability to produce an array of cytokines and chemokines, through which they help shape B cell and T cell responses, and impact the function of DC, macrophages and neutrophils
More from
$NKTX hopes to avoid the fate of $FATE, via @evaluatevantage -> https://t.co/1SOK7gOAbS
— Jacob Plieth (@JacobPlieth) June 27, 2023
The update will be today at 8:00 a.m. ET https://nkx101-clinical-update-conference-call.open-exchange.net/registration
PR https://www.globenewswire.com/news-release/2023/04/17/2648511/0/en/Nkarta-Presents-Preclinical-Data-Exploring-Gene-Knockout-Strategies-and-Combination-Agents-with-NK-Cell-Based-Therapies-at-the-2023-AACR-Annual-Meeting.html
Posters https://www.nkartatx.com/file.cfm/75/docs/nkarta_aacr2023_poster%20890_adam17%20ko%20of%20nk%20cells.pdf https://www.nkartatx.com/file.cfm/75/docs/nkarta_aacr2023_poster%203183_nkx101%20and%20cetuximab%20combo.pdf
Yes, it could have been primary resistance due to low MICA/B (or other NKG2DLs) expression, but without any translational data from the company there is no way of knowing.
Disfunctional NKs have a few more mutations than dysfunctional NKs.
My point was MDS patients has disfunctional NK cells which cause the cancer to slow down on MICA expression. With lowered MICA expression, NKTX cells don't work. I doubt MDS TME is more suppressive than AML.
In patient's there is immune cell dysfunction, but using off-the-shelf should be able to overcome some of that. As for primary, adaptive and/or acquired resistance, only translational data will be able to give insights as to what strategies could combat that.
Yes, just a shame about the target.
It will be interesting to see how this DT HER2 x NKG2D x CD16A engager works since it won't be impacted by the # of ligands on cancer cells.
Cancer is adapting. Why bother expressing ligands if the NK cells are not working anyway.
NKG2D surface down-modulation in freshly isolated NK cells from MDS patients may be a clinically useful “biomarker” of suppressed NK function.
Without any translational data it is hard to say why. I know in one paper the authors reported that just 30% of MDS blasts expressed the NKG2D ligands MICA/B https://ashpublications.org/blood/article/109/11/4816/23111/Reduced-natural-killer-NK-function-associated-with
That was confirmed by another group, showing a low expression of NKG2D ligands https://www.nature.com/articles/leu2010149
There are plenty of other mechanisms, including Tregs, either via cell-to-cell interactions of soluble factors, such as TGF-B https://rupress.org/jem/article-lookup/doi/10.1084/jem.20051511
This one is puzzling. Four patients with MDS were treated, with no response observed.
The update is expected to include longer-term follow up from patients who were in response as of the previous April '22 data cut-off; clinical data from at least ten new R/R AML patients treated at 1.5B (x3); clinical data from patients who received one or more additional three-dose cycles; and clinical data from a new cohort of patients who received lymphodepletion with cytarabine and fludarabine.
Speaking of expansion. From this: ''We apply a human B-lymphoblastoid cell-line 721.221 (hereinafter, 221)-based artificial feeder cell system with membrane-bound interleukin 21 (mIL 21) to propagate NK and CAR-NK cells. The expansion capability, purity, and cytotoxicity of NK cells expanded with 221-mIL-21 feeder cells are superior to that of conventional K562-mIL-21 feeder cells. RNA sequencing (RNA-seq) data show that 221-mIL-21 feeder cell-expanded NK cells display a less differentiated, non-exhausted, limited fratricidal, memory-like phenotype correlated with enriched metabolic pathways, which explains underlying mechanisms.'' https://www.cell.com/molecular-therapy-family/methods/fulltext/S2329-0501(20)30138-8
Also, additional ways that could improve it https://ashpublications.org/blood/article/118/21/4035/68863/Nicotinamide-a-Form-of-Vitamin-B3-Promotes https://aacrjournals.org/cancerres/article/77/20/5664/623001/GSK3-Inhibition-Drives-Maturation-of-NK-Cells-and https://www.cell.com/cell-metabolism/fulltext/S1550-4131(21)00130-3
Some more info. Pre-activation is IL-12/15/18 and expansion with uAPCs (engineered K562 feeder cells expressing mbIL-21 and 4-1BBL) plus IL-2, transduction with a retroviral vector, before another round of expansion. From two cord blood units, 250 doses have been manufactured with a (CAR) transduction efficiency of 60%. Six dose levels will be tested, with DL1 completed. Also, over 90 cryopreservation protocols were tested, with MDACC outperforming standard frozen.
|
Followers
|
9
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
159
|
|
Created
|
08/27/20
|
Type
|
Free
|
| Moderators | |||
| Volume: | 1,155,170 |
| Day Range: | 1.59 - 1.70 |
| Last Trade Time: | 7:02:13 PM EDT |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
