Tuesday, April 30, 2024 2:37:05 PM
(OT) A post on LinkedIn by Max Qian (CEO and Co-Founder at Rui Therapeutics):
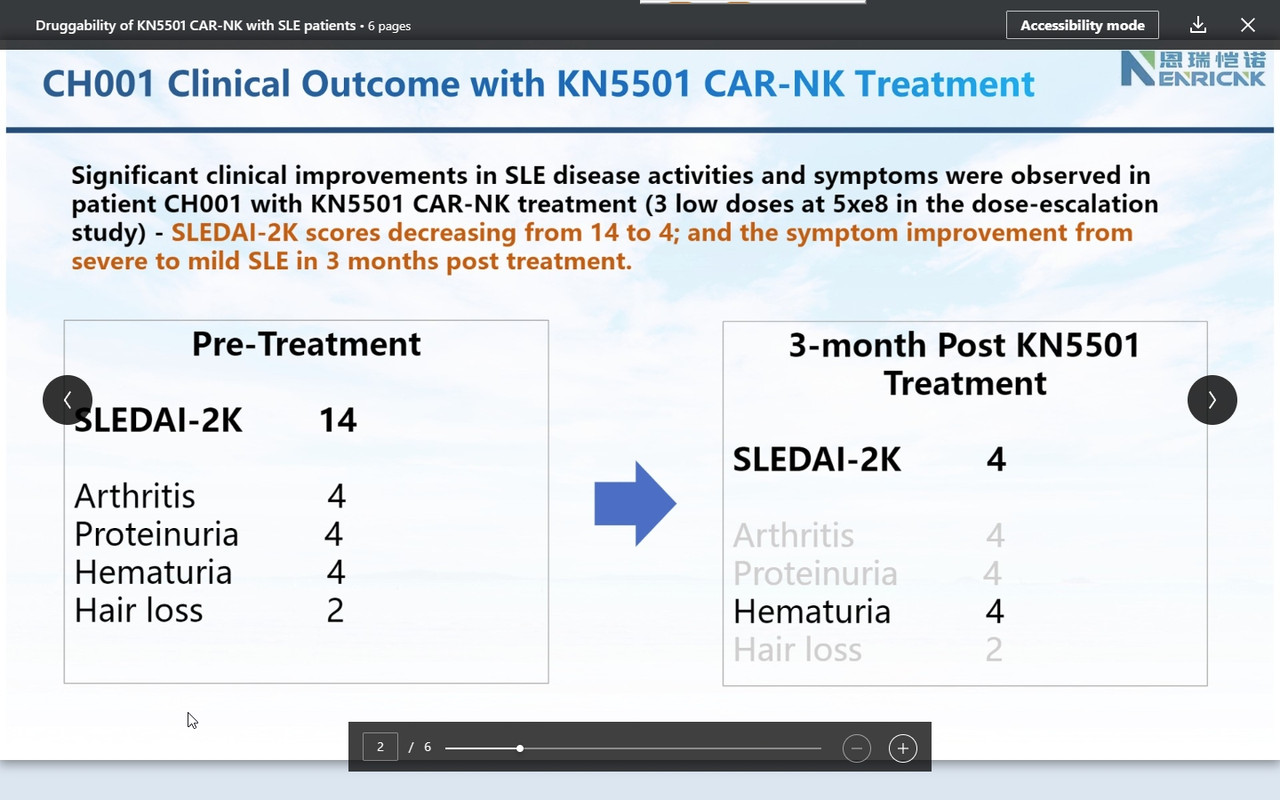
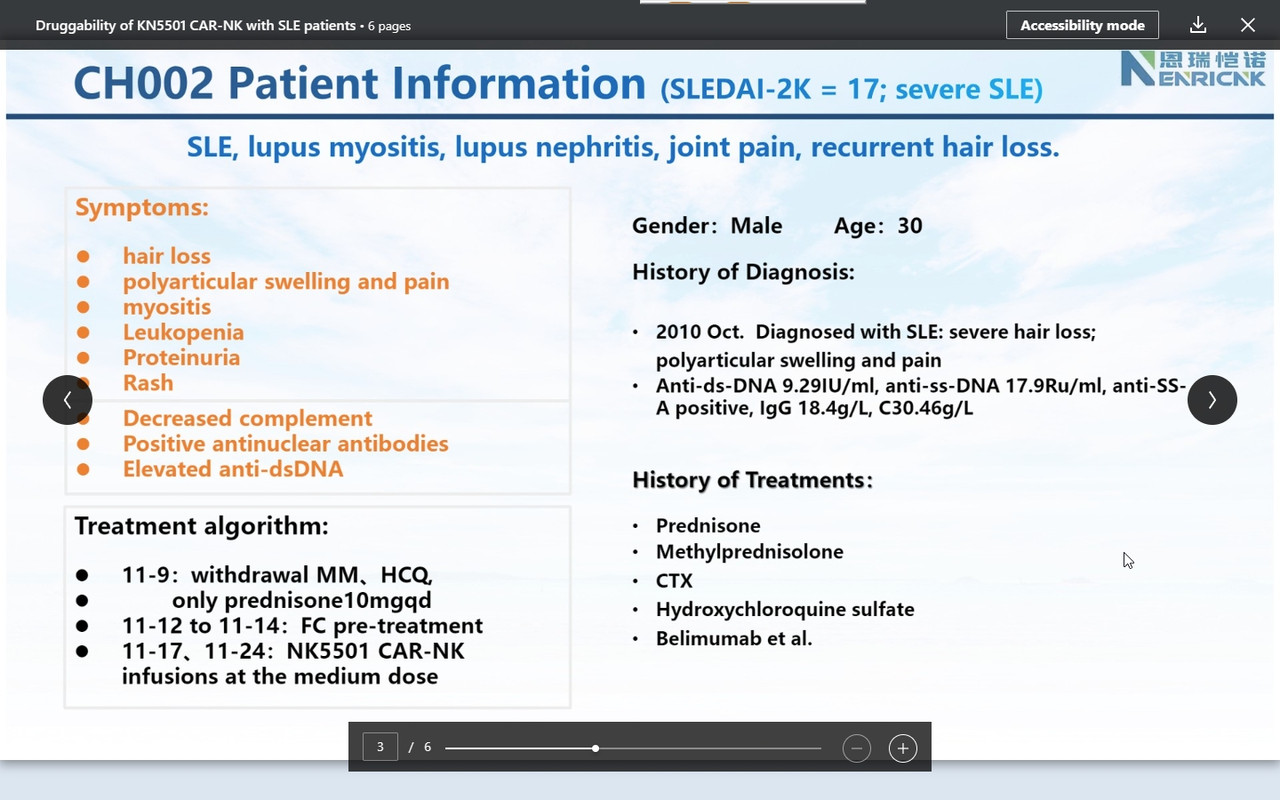
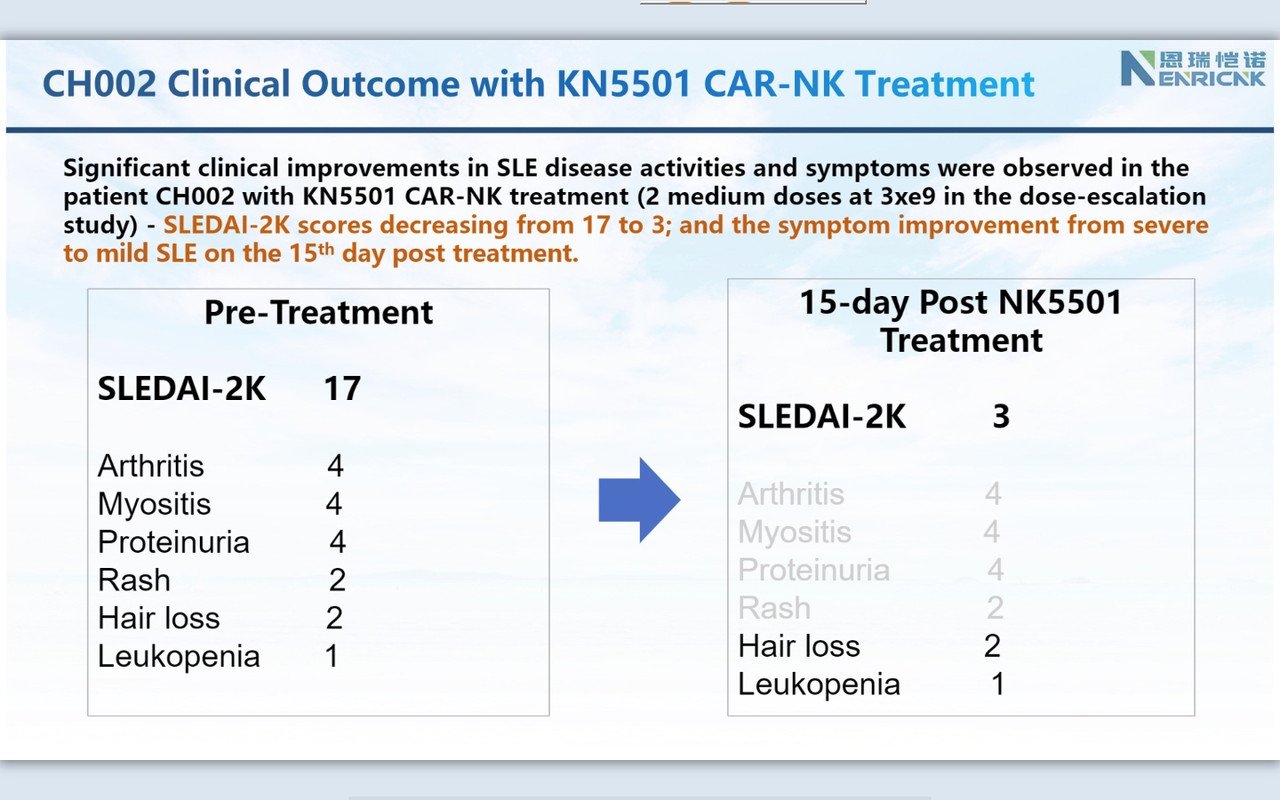
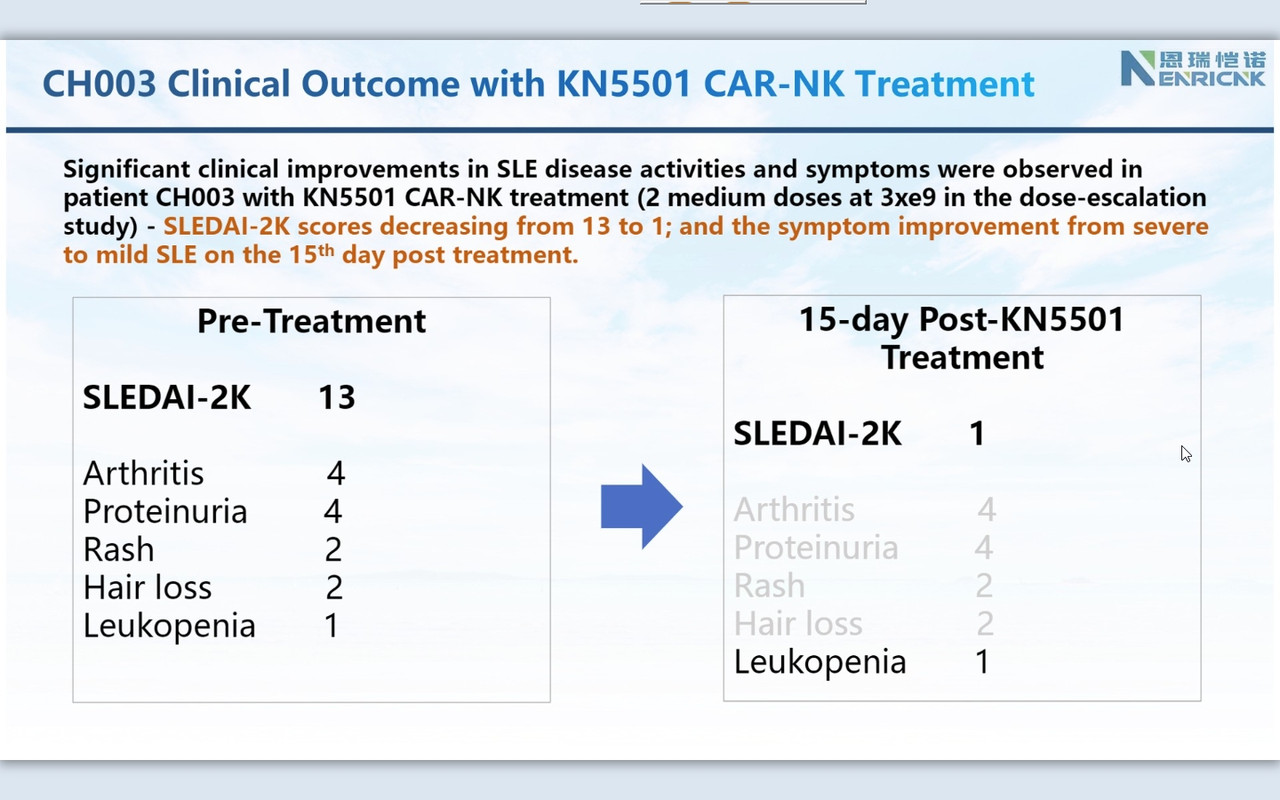
''I am so pleased today to share an exciting clinical development with our CAR-NK therapeutic platform on Autoimmune disease space. Attached six slides describe our ongoing FIH clinical trial of KN5501 (CD19 CAR-NK) that is used to treat three severe SLE (systemic lupus erythematosus) patients. We innovatively designed this dose-escalation clinical trial with 1+2+3=6 patients to complete low, medium and high dose.
SLEDAI 2K score (SLE disease activity index 2000), the most commonly used system that evaluates the severity of SLE, is used to determines changes in the disease activity of patients diagnosed with SLE: 1) mild activity (SLEDAI-2K = 6); 2) moderate activity (SLEDAI-2K 7 to 12); 3) severe activity (SLEDAI-2K>12).
· The higher the score, the more significant is the degree of disease activity.
· Scores of 6 and above are considered to be consistent with active disease requiring therapy. However, scores greater than 20 are very rare.
· Modifications of score of 6 (improvement) and of 8 (worsening) are considered clinically relevant.
Consistently, as we observed in r/rDLBCL trials, CAR-NK demonstrated a supper safety profile, i.e., no CRS nor ICANS. Very most exciting is that we observed the efficacy from the 1st three patients on low and medium dose (we even have not completed three doses with medium dose patients yet!). This might be the very 1st FIH data available globally to support CAR-NK therapy in SLE.
In earlier this year, we may hear CAR-T on SLE, but latest FDA announcement may give us (patients, hospital and manufacturers) a 2nd thought of the CAR-T approach in autoimmune disease spaces; don’t mention its other safety and economic concerns. Welcome discussions!''


''I am so pleased today to share an exciting clinical development with our CAR-NK therapeutic platform on Autoimmune disease space. Attached six slides describe our ongoing FIH clinical trial of KN5501 (CD19 CAR-NK) that is used to treat three severe SLE (systemic lupus erythematosus) patients. We innovatively designed this dose-escalation clinical trial with 1+2+3=6 patients to complete low, medium and high dose.
SLEDAI 2K score (SLE disease activity index 2000), the most commonly used system that evaluates the severity of SLE, is used to determines changes in the disease activity of patients diagnosed with SLE: 1) mild activity (SLEDAI-2K = 6); 2) moderate activity (SLEDAI-2K 7 to 12); 3) severe activity (SLEDAI-2K>12).
· The higher the score, the more significant is the degree of disease activity.
· Scores of 6 and above are considered to be consistent with active disease requiring therapy. However, scores greater than 20 are very rare.
· Modifications of score of 6 (improvement) and of 8 (worsening) are considered clinically relevant.
Consistently, as we observed in r/rDLBCL trials, CAR-NK demonstrated a supper safety profile, i.e., no CRS nor ICANS. Very most exciting is that we observed the efficacy from the 1st three patients on low and medium dose (we even have not completed three doses with medium dose patients yet!). This might be the very 1st FIH data available globally to support CAR-NK therapy in SLE.
In earlier this year, we may hear CAR-T on SLE, but latest FDA announcement may give us (patients, hospital and manufacturers) a 2nd thought of the CAR-T approach in autoimmune disease spaces; don’t mention its other safety and economic concerns. Welcome discussions!''






Recent NKTX News
- Three TriKE Candidates In Clinic As NK Cell Engager Field Reorganizes • PR Newswire (Canada) • 05/19/2026 01:00:00 PM
- Three TriKE Candidates In Clinic As NK Cell Engager Field Reorganizes • PR Newswire (US) • 05/19/2026 01:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 05/12/2026 08:04:35 PM
- Nkarta Reports First Quarter 2026 Financial Results and Corporate Highlights • GlobeNewswire Inc. • 05/12/2026 08:01:00 PM
- Form ARS - Annual Report to Security Holders • Edgar (US Regulatory) • 04/23/2026 08:42:24 PM
- Form DEFA14A - Additional definitive proxy soliciting materials and Rule 14(a)(12) material • Edgar (US Regulatory) • 04/23/2026 08:41:50 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 04/23/2026 08:41:05 PM
- Nkarta Announces FDA Agreement on Outpatient Dosing of NKX019 in Autoimmune Disease, Expanding Access to Community Rheumatology Centers • GlobeNewswire Inc. • 04/15/2026 08:01:00 PM
- Nkarta to Participate in Needham Virtual Healthcare Conference • GlobeNewswire Inc. • 04/09/2026 11:00:00 AM
- Form 424B5 - Prospectus [Rule 424(b)(5)] • Edgar (US Regulatory) • 04/03/2026 09:29:06 PM
- Form S-3 - Registration statement under Securities Act of 1933 • Edgar (US Regulatory) • 03/25/2026 09:23:28 PM
- Form 10-K - Annual report [Section 13 and 15(d), not S-K Item 405] • Edgar (US Regulatory) • 03/25/2026 08:07:22 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 03/25/2026 08:04:17 PM
- Nkarta Reports Fourth Quarter and Full Year 2025 Financial Results and Corporate Highlights • GlobeNewswire Inc. • 03/25/2026 08:01:00 PM
- Nkarta to Participate in March Investor Conferences • GlobeNewswire Inc. • 02/27/2026 01:00:00 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/20/2026 10:38:03 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/08/2026 10:50:36 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 01/08/2026 10:49:10 PM
- Form S-8 - Securities to be offered to employees in employee benefit plans • Edgar (US Regulatory) • 01/05/2026 09:43:55 PM
- Nkarta to Participate in Evercore Healthcare Conference • GlobeNewswire Inc. • 12/02/2025 12:09:32 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/10/2025 09:18:53 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/10/2025 09:13:36 PM
- Nkarta Reports Third Quarter 2025 Financial Results and Corporate Highlights • GlobeNewswire Inc. • 11/10/2025 09:01:00 PM








