Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Grip,
Thank you for reposting your opinion again.
The company is still very small, but the leadership are formidable and tenacious. The independent peer review took a long time too, but it was successfully completed and published in JAMA Oncology, and it is an asset as we move further into the regulatory process.
The CEO has been quite an effective and successful leader of a small clinical-stage biotech. She is responsible for leading the company to the point where NWBO now has a valuable cell-based platform technology that has completed all three phases of clinical investigation. PD1 combo and platform potential represent additional value beyond the P3...
DCVax-L has been investigated in other clinical trials and is currently being studied in combo with PD1 blockade at UCLA, and this novel technology will be studied in other combos and other cancers too. The CEO put the company in a position where these forthcoming developments are possible. After successful commercialization, we can learn a lot more about this technology as a platform.
The leadership team have positioned the company well globally and they have secured a solid comprehensive worldwide patent portfolio. The results presented in the JAMA publication represent the greatest prospective value that NWBO has ever had since the company was founded.
In my opinion, JAMA Oncology put Northwest Bio on the radar of institutional investors, and MHRA will likely be the next tipping point.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171297915
https://www.lifespan.org/centers-services/lifespan-cancer-institute


https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
brazen22, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for asking!
Here are some progress updates and news headlines that are a bit more recent than the 2018 article that you posted. I’m sure that you will find these recent developments representative of the greatest prospective value that the company has had since its founding:
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.theguardian.com/science/2022/nov/17/im-just-carrying-on-vaccine-gives-brain-cancer-patient-years-of-extra-life
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.nice.org.uk/guidance/indevelopment/gid-ta10143
brazen22, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Your posts represent misinformation in my view.
Thanks for posting an article from 2018, but there have been significant developments since 2018.
Rather than entertaining the noise of baseless distractions, I would suggest researching the evidence supporting the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















brazen22,
PFS is an intermediate endpoint that predicts the hard endpoint of OS.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172692156
aesop1,
Manufacturing capacity is a very relevant topic to discuss at this stage following the successful completion of the P3 and the independent peer review of the P3 clinical data. As a component of regulatory review, it is crucial that a company build and development a manufacturing system that can scale up rapidly to serve a global market. Everyone is eagerly awaiting news of regulatory developments, and manufacturing is a foundational pillar of the path forward alongside regulatory advisory.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171781313
HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt

Thank you for asking!
Yes, the hope is always that PFS aligns with OS and accurately predicts OS. Unfortunately, trial investigators cannot know in advance precisely how well PFS data actually predict OS until OS data are reached.





https://www.fda.gov/media/71195/download

https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(20)30076-6/fulltext
https://www.ncbi.nlm.nih.gov/books/NBK137763/
https://www.eu-openscience.europeanurology.com/article/S2666-1683(22)02684-2/pdf
https://www.fda.gov/media/113423/download
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172671647
Regarding the DCVax-L P3, actual PFS was subsequently enlightened by the OS data, but the predictive PFS data were contaminated and therefore the investigators could not use the predictive PFS data as a reliable surrogate predictor of OS. Instead, they had to wait for actual OS data.
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
The P3 for the DCVax-L cell-based technology spanned many years. If the trial were designed and commenced in 2023, the investigators would appropriately attempt to measure progression and predict survival using the best methods and most current medical knowledge available in 2023, as any cancer researcher would.
Don’t forget to follow the combo trial!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172584227
HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thank you for again highlighting the pembrolizumab combo study that is ongoing at UCLA today! You are 100% correct—the UCLA investigators are measuring PFS and predicting survival. Those PFS data will be available much sooner than the OS data.

The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
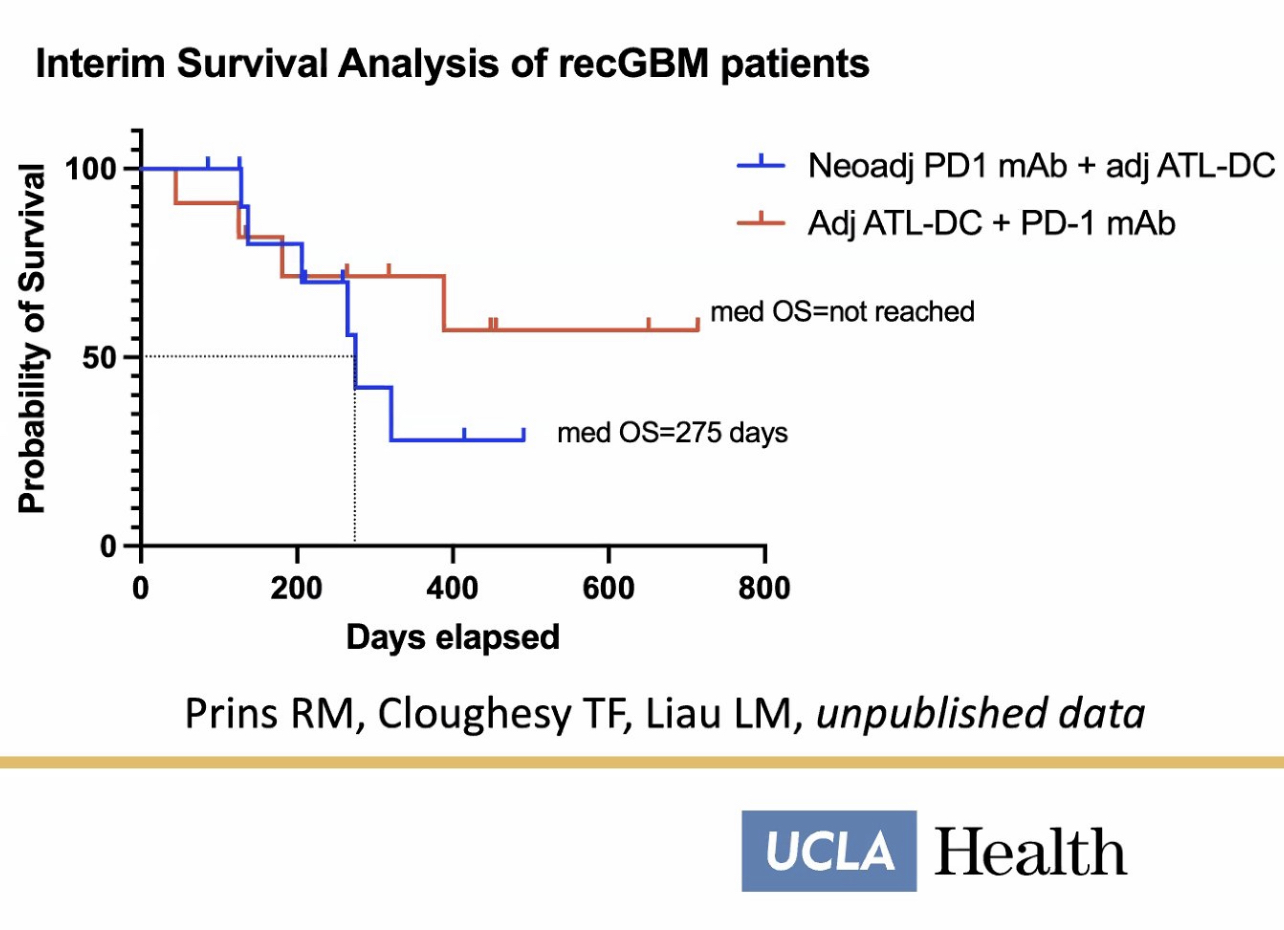
https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.


Exactly. Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy https://t.co/BnJHM3myP9
That’s precisely what I am trying to say — I see the same reposts over and over. I have not read much meaningful discussion…
I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology. Stay tuned!

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172639755%5B/color%5D
Grip, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thanks for reposting your opinion once again, but I would again argue that you’ve drifted off track. Perhaps refocus back on what matters. I believe that you are distracted from the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















Yes, MHRA is one of the most significant, and we are all eagerly awaiting regulatory advisory in several nations.
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
Exactly. This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for the reply!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Maverick0408,
Thanks for the reply and congratulations on coming from this world! My only point was that there is a connection between NIH and the FDA, and there is a connection between NIH drug development and FDA drug approvals.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172652917
Maverick0408,
If you do some research into NIH and the FDA, you will discover that your assertion is not exactly accurate. The NCI division of NIH develops oncology drugs, and FDA approves drugs. There is some connection…
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020

https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter




https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
DavidW2, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thank you for your comments. I can help you understand why I respond to those who post their views—I respond because I choose to participate in the discussion on this discussion board as an investor who is interested in health and wealth! I believe that this technology represents immense prospective value, and I also hope to have access to efficacious immunotherapy technologies if I ever need them.
I am focused on the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















aesop1 Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy https://t.co/BnJHM3myP9
I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology. Stay tuned!

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172639755%5B/color%5D
Grip,
The CEO has been quite an effective and successful leader of a small clinical-stage biotech. She is responsible for leading the company to the point where NWBO now has a valuable cell-based platform technology that has completed all three phases of clinical investigation. PD1 combo and platform potential represent additional value beyond the P3...
DCVax-L has been investigated in other clinical trials and is currently being studied in combo with PD1 blockade at UCLA, and this novel technology will be studied in other combos and other cancers too. The CEO put the company in a position where these forthcoming developments are possible. After successful commercialization, we can learn a lot more about this technology as a platform.
The leadership team have positioned the company well globally and they have secured a solid comprehensive worldwide patent portfolio. The results presented in the JAMA publication represent the greatest prospective value that NWBO has ever had since the company was founded.
In my opinion, JAMA Oncology put Northwest Bio on the radar of institutional investors, and MHRA will likely be the next tipping point.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171542173
HyGro,
Thanks for reply, but I think you’re mixed up. Every cancer trial attempts to measure and predict survival.
PFS is a surrogate predictor of OS. The PFS data will be available sooner. The trial will also collect OS data of course, but OS data are not available as early as PFS data are.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172643007
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172642900
This statement is illogical. PFS does predict OS—that’s its purpose. Cancer trials collect data on both, but one data set is available much sooner than the other. Once OS is finally reached, OS either confirms or invalidates the earlier PFS data. PFS is not a hard endpoint—PFS predicts the hard endpoint of OS.

HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thank you for again highlighting the pembrolizumab combo study that is ongoing at UCLA today! You are 100% correct—the UCLA investigators are measuring PFS and predicting survival. Those PFS data will be available much sooner than the OS data.

The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.


(The P3 linked below was already completed and is separate from the ongoing study linked above.)
HyGro,
Thanks for reposting your opinion again, but you are misinformed.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172639742
I think you are distracted from the significance of the DCVax-L platform technology. Doctors at the Lifespan hospital were on the news again earlier this month highlighting the unmet medical need of GBM and the need for more approved treatment options. They again referred to DCVax-L as a medical “breakthrough” just as they did at the press conference following the publication of the independent peer review.
One of the greatest aspects of immunotherapy such as DCVax-L is that it may be efficacious in combination with a number of other therapies. The pembrolizumab combo is the most exciting and noteworthy combination therapy in my opinion, and I hope that we will see more PD1 combo studies with the DCVax-L platform for cancers beyond nGBM and rGBM.
https://www.lifespan.org/centers-services/brain-and-spine-tumor-center

https://www.wpri.com/health/brain-breakthrough-ri-researchers-working-to-make-progress-against-glioblastoma/
Refocus from distractions and consider the integrity of the science.




https://www.fda.gov/drugs/development-approval-process-drugs

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
I doubt it. All of us who show up and post here everyday seem pretty committed to our respective positions.
Time will tell … Exciting times!
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Happy,
That seems illogical. For any stock or other investment, you want the prospective value to be greater than the current price, especially if you’re making a purchase. But I take your point—if you’ve already made your purchase, you want the prospective value now.
I want everything now too.

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Happy,
Thanks for the reply. I don’t know anything about 2016, but I would argue now that the “governance” has led the company to the greatest prospective value that NWBO has ever had since its founding—following the P3, peer review, ongoing combo study, manufacturing capacity development, manufacturing license, and a comprehensive worldwide patent portfolio.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/

aesop1,
Exactly.
You are 100% correct! SCIENCE trumps all!
The recent scientific developments at UCLA are brilliant and build upon years of science and Nobel Prize-winning science!
https://x.com/mc17595091/status/1666528319209103361?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

https://newsroom.ucla.edu/dept/faculty/grant-awarded-to-researchers-to-develop-immune-based-therapies-for-deadly-brain-tumors
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
aesop1, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thank you for posting an article from 2016. Here are some progress updates and news headlines that are a bit more recent than 2016:
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.theguardian.com/science/2022/nov/17/im-just-carrying-on-vaccine-gives-brain-cancer-patient-years-of-extra-life
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.nice.org.uk/guidance/indevelopment/gid-ta10143
Exactly. Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy https://t.co/BnJHM3myP9
Thanks for the reply!
I recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology. Stay tuned!

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172639755%5B/color%5D
Grip, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
Thank you for posting your comments, but I think you’ve drifted off track. Perhaps refocus back on what matters. I believe that you are distracted from the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for reposting your opinions again, but I think that you are mixed up. Actual PFS was subsequently proven by the OS data, but the predictive PFS data were contaminated and therefore the investigators could not use the predictive PFS data as a reliable surrogate predictor of OS. Instead, they had to wait for actual OS data.
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
The P3 for the DCVax-L cell-based technology spanned many years. If the trial were designed and commenced in 2023, the investigators would appropriately attempt to measure progression and predict survival using the best methods and most current medical knowledge available in 2023, as any cancer researcher would.
Don’t forget to follow the combo trial!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172584227
HyGro,
You are correct! The UCLA pembrolizumab combo study includes a PFS measure.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Your assertions are verifiably false. PFS did not not fail as subsequently proven by the OS data. The PFS data were contaminated and therefore the investigators could not use the inaccurate PFS data as a reliable surrogate predictor of OS. Instead, they had to wait for actual OS data.
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
The P3 for the DCVax-L cell-based technology spanned many years. If the trial were designed and commenced in 2023, the investigators would appropriately attempt to measure progression and predict survival using the best methods and most current medical knowledge available in 2023, as any cancer researcher would.
Thank you for mentioning the combo trial!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172584227>
HyGro,
You are 100% correct again! The UCLA pembrolizumab combo trial does not use an ECA! The combo trial is a placebo-controlled study!
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
— JAMA Oncology (@JAMAOnc) February 15, 2023
HyGro,
Thanks for the reply! NIH develops drugs and FDA approves drugs— I agree!

First, it’s important to recognize the independent peer review of the landmark P3 study that was published in JAMA Oncology.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847



https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020

https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter




September 2023






https://www.cns.org/annualmeeting
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172585824><i class=
HyGro,
Thanks for reposting that again, but you are misinformed. I believe that you need to consider better sources.
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048;
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756;

Exactly. This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for posting!
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
exwannabe, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh
Thanks for posting your thoughts about the 2008 data! Dr. Toms commented on the data collected from the completed P3, and he also briefly mentioned the combo data from the pembrolizumab combo trial that is ongoing at UCLA today. Here is the press conference in case you missed it:
Don’t forget to check out the forthcoming NICE appraisal report on the DCVax-L cell-based technology!

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
Grip, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I believe that you are distracted from the vast significance of Liau’s work and the further development of this technology in combination with other technologies. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold, and the SP will follow the regulatory developments worldwide.
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology is a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















aesop1, https://t.co/BYogOUXX8T comments on the 8/9/23 SEC 10-Q for $nwbo. The MD and A Reveal. pic.twitter.com/3wBlUyjjZf This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
I agree with you on one point—there is a lot of positive support for the DCVax-L cell-based platform technology among those who have been following the small pre-commercial company and following the great research that is ongoing at UCLA!
https://twitter.com/mc17595091/status/1666528319209103361?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172579930><i class=
aesop1, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I believe that you are misinformed. Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer
The P3 study of the DCVax-L cell-based platform technology was a landmark trial.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology. You might consider researching credible full-context sources to gather reliable information.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/















Exactly.
Thanks for the reply!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172456463><i class=
HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
BP is currently running a DCVax-L study at UCLA in combination with one of BP’s blockbuster drugs. Skip down below to review the pembrolizumab study supported by Merck and NIH.
The P3 data were peer reviewed by independent physicians and qualified statisticians—please refer to JAMA Oncology.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873
https://www.fda.gov/media/120721/download
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers



https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/














