Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
LC,
No, it’s not hilarious. It’s not funny when adults miss the basic and crucial lessons that we teach to our children:
✅️ How to be critical thinkers
✅️ How to evaluate sources
✅️ How to avoid the pollution called innuendo
✅️ How not to cause undue harm to others
Evaluate the bold claim presented in the article that you recklessly posted and find credible supporting evidence—if you are successful, you won’t be “waved off.”
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173493325
Warriors Triumphant Stand
In the battle against a relentless foe,
Two warriors stand, their resolve aglow.
Doctors Liau and Ashkan, names renowned,
In the fierce war against cancer, they're bound.
With wisdom as their shield, compassion their sword,
Their quest for a cure, a harmonious chord.
In the lab's hallowed halls, they forge their fight,
Illuminating darkness, bringing hope to light.
Dr. Liau, with her insight so keen,
A guiding star on the oncology scene.
Dr. Ashkan, with a surgeon's precise hand,
Shaping futures, helping patients stand.
Together they stride in this arduous quest,
Against glioblastoma, they give their best.
In their steadfast journey, they're not alone,
For every success, hope's seed is sown.
So here's to these heroes, in their noble strife,
Soldiers in the fight, guardians of life.
Their dedication, a beacon so bright,
Guiding us forward, toward healing's light.

—ChatGPT
I don’t think that’s exactly an unbiased source. You might want to consider doing some further research to discover whether you can find any credible independent evidence to support the veracity of that headline 😶
Believe carefully. Verify sources. Fact ✅️
https://www.allsides.com/news-source/daily-caller

Excellent point! Dr. Liau is invaluable as a physician and medical research scientist! Her work has been supported by U.S. NIH peer-reviewed grants for many consecutive years!
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm
Thanks for the reply. I would count Doctors Liau and Ashkan among the soldiers—fighting disease with their novel cell-based technology! Liau and Ashkan both are extremely noteworthy as we look back on the P3 and as we look forward to the regulatory phase in the UK, US, and other nations…
UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might wish to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3 and were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support their respective nations’ governments over many years. Ashkan is an advisor to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020

LC,
Soldier Milestones! I love your phrasing! 💚 Your word choice evokes powerful and majestic imagery!
I believe you are also foreshadowing the triumphant soldier!

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173492533
LC,
Thanks for asking 😉
In my view, the short list of pre-commercial milestones that stand out and shine the most:
⭐️ JAMA Oncology
⭐️ Multiple MHRA Approvals
⭐️ Emerging Combo Data
⭐️ MAA Submission
Further, Dr. Liau joining the SAB at Northwest Biotherapeutics is awesome!
Combo is King!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers







Your assertion defies logic. 😶
Let’s consider a simple hypothetical scenario: What if a company submitted an application based on a P3 trial but patients in a subsequent P1 combo trial were dropping dead of complications? According to your logic, the regulators should ignore those P1 data?
All clinical data that exist at the time of a filing with regard to a novel technology are relevant—it’s common sense.
HyGro,
I hope you’re doing well. Thanks for the reply. At least we can agree that the emerging combo data are brilliant!
While I do believe that the first regulatory filings will be based primarily on the P3, all existing clinical data are relevant with regard to safety and efficacy.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://clinicaltrials.gov/study/NCT04201873

https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
“In a regulatory application, all clinical data, including data from early-stage trials, is generally relevant, even if the application primarily hinges on a Phase 3 trial. Interim data from a subsequent Phase 1 trial can provide additional insights into safety, tolerability, pharmacokinetics, and pharmacodynamics. This data can help regulatory bodies understand the full scope of the drug's effects, potential risks, and its mechanism of action. While the focus might be on Phase 3 results for efficacy and safety, the complete picture offered by all clinical trials contributes to a thorough evaluation by agencies like the FDA or MHRA.”
—ChatGPT
LC,
Thanks for asking. The focus is often on DCVax-L or the emerging combo data…
LC, we can always count on you to broaden the conversation. 😉 … It is a good idea to highlight Direct too…
DCVax-Direct is an entirely separate topic and presents additional potential value beyond resectable cancer.
For some preliminary background about Direct, let’s search the web with an AI app:
“DCVax-Direct is a form of cancer immunotherapy being developed by Northwest Biotherapeutics. It is designed to target a broad range of inoperable solid tumors. Here are the key aspects of DCVax-Direct:
1. **Mechanism of Action:** DCVax-Direct involves the same mechanism as DCVax-L, but with a crucial difference. In DCVax-Direct, dendritic cells are directly injected into the tumor, where they take up tumor target proteins in situ. This is in contrast to DCVax-L, where tumor lysate from surgically resected tumor tissue is used.
2. **Phase 1 Trial Results:** The Phase 1 trial of DCVax-Direct showed positive results in treating more than a dozen different types of inoperable solid tumors. This indicates the potential effectiveness of DCVax-Direct across a diverse range of tumor types.
3. **Future Development:** Northwest Biotherapeutics plans to continue the clinical development of DCVax-Direct for a wide range of inoperable solid tumors, building on the success of their Phase 1 trials.
In summary, DCVax-Direct represents an innovative approach in cancer immunotherapy, especially for inoperable solid tumors, leveraging the body's immune system to target and destroy cancer cells.”
—ChatGPT

LC,
Thanks for reposting about the publisher again. It’s worthwhile to consider and appreciate the meticulous work that went into the MAA that was submitted this week.
It is also important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents had to be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA did as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
The most recent PR didn’t mention the publisher, but it did echoe the recent JCI publication. Highlighting JCI really was a mic-drop moment for LC! 😉
https://www.jci.org/articles/view/169314

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html


Combo is King!
https://clinicaltrials.gov/ct2/show/NCT04201873

The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The first announcement of a regulatory filing for marketing was announced on December 21, 2023!
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA was a prerequisite of the MAA application that was submitted this week.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html


LC,
You’re working “during the holidays,” so why wouldn’t the MHRA be working?
I can see why you’re here all day and on holidays—NWBO seems to be getting some attention. 😉

https://investorshub.advfn.com/boards/most_read.aspx?p=d
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
Thanks again for recommending AI to supplement responses to online discussions. I tried the app in reference to some of the postings about NWBO:
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.
Investors can take several steps to avoid falling victim to stock disinformation:
**Verify Information from Reliable Sources:**
- Cross-reference information from multiple reputable sources. Avoid relying solely on one channel, especially social media or anonymous message boards.
**Understand the Company:**
- Conduct thorough research on the companies you're interested in. Understand their financials, business model, and industry. Reliable information often comes from official reports, financial statements, and reputable news sources.
**Use Caution with Social Media:**
- While social media platforms can provide information, they can also be a source of misinformation. Be critical of information shared on these platforms and verify it through reliable sources.
**Stay Informed about Market Regulations:**
- Understand the regulations governing financial markets. Authorities such as the SEC (U.S. Securities and Exchange Commission) take actions against those engaged in fraudulent activities.
**Consider Professional Advice:**
- Consult with financial advisors or professionals who can provide guidance based on expertise and experience.
**Participate in Reputable Forums:**
- If you engage in online discussions, consider reputable financial forums where discussions are moderated, and misinformation is less likely to thrive.
By staying informed and exercising caution, investors can better protect themselves from falling prey to stock disinformation.” —ChatGPT

Congratulations, Dr. Steinman …


https://www.nytimes.com/2012/12/23/magazine/is-the-cure-for-cancer-inside-you.html
Another milestone was reached today on the journey that Dr. Steinman began. On December 21st, Northwest Biotherapeutics announced the submission of a voluminous regulatory filing to the MHRA that was based on the further development of Dr. Steinman’s Nobel Prize-winning discovery of the dendritic cell.
Congratulations to Doctors Liau and Ashkan who followed Dr. Steinman to build upon his research in service of humanity. His work lives on at UCLA today!

—Hippocratic Oath
Congratulations to Northwest Biotherapeutics for their relentless commitment to the DCVax cell-based technology and to its further development as a platform.
Congratulations to the United States NIH for recognizing the importance of Dr. Liau’s research and for supporting her invaluable work for over 20 consecutive years.


“I think this provides the potential for a whole new type of therapy in cancer,” he said. “But we need research and patience to discover the rules, to discover the principles.” —Dr. Ralph M. Steinman
https://www.scientificamerican.com/article/the-patient-scientist/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3376974/

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
ex,
Thanks for your predictions 😶
Your posts about timelines made me think about how NICE factors in…
I think that the NICE status updates that were posted exactly 12 months in advance of the December 2023 MAA submission are also significant.
From an AI app:
”The Medicines and Healthcare products Regulatory Agency (MHRA) and the National Institute for Health and Care Excellence (NICE) are two key organizations in the UK's healthcare system, each with distinct roles, and their processes are interconnected, especially in the context of a Marketing Authorization Application (MAA).
Here's how the MHRA MAA impacts the NICE process:
1. Role of MHRA: The MHRA is responsible for assessing and approving new medicines for use in the UK. This includes reviewing Marketing Authorization Applications (MAAs) to ensure the safety, quality, and efficacy of new drugs.
2. MHRA Approval: When the MHRA grants a marketing authorization for a new drug, it means the drug can be legally sold and marketed in the UK. However, this doesn’t automatically mean the drug will be available through the NHS.
3. NICE Appraisal: After MHRA approval, the drug may undergo a health technology assessment by NICE. NICE evaluates the cost-effectiveness and clinical effectiveness of new medicines to determine whether they should be recommended for use in the NHS.
4. Impact on NICE Process: The MHRA’s decision to authorize a drug is a prerequisite for NICE's appraisal. NICE does not assess the basic safety and efficacy of a drug; it relies on the MHRA's evaluation for that. Instead, NICE focuses on the added value of the drug in terms of health outcomes and cost-effectiveness.
5. Timelines and Access: The MHRA’s approval of a drug is often the starting point for NICE's technology appraisal process. Delays or issues in the MHRA’s MAA process can impact when NICE is able to complete its appraisal, subsequently affecting when a drug becomes available on the NHS.
6. Collaboration and Parallel Advice: In some instances, the MHRA and NICE work in parallel to provide advice to drug developers, which can streamline the process of getting new treatments to patients.
In summary, MHRA’s MAA is a critical step that precedes and influences the NICE appraisal process. While MHRA focuses on the safety and efficacy of a medicine, NICE assesses its cost-effectiveness for NHS use.” —ChatGPT
The forthcoming NICE appraisal of the DCVax-L cell-based technology will be another significant development.

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. Further announcements and status updates are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA was a prerequisite of the MAA application that was submitted in December 2023.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
I see why you’re here all day everyday—NWBO seems to be getting some attention. 😉

https://investorshub.advfn.com/boards/most_read.aspx?p=d
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
LC,
The PR today echoed the JCI publication that you linked us to recently! Highlighting JCI really was a mic-drop moment for LC! 😉
https://www.jci.org/articles/view/169314
Combo is King!

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html


https://clinicaltrials.gov/ct2/show/NCT04201873


The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The first announcement of a regulatory filing for marketing was announced today!
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA was a prerequisite of the MAA application that was submitted this week.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html


iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

Thanks for reposting your baseless commentary again, but I think it’s a good idea to look beyond social media to find full-context credible sources and to consult licensed oncologists.

JAMA Oncology is a peer-reviewed medical journal that us part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers, healthcare professionals, and the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.

https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


iclight,
Thanks for highlighting the vast platform potential! Other advances in the research of cancer and other technologies may elevate the value of DCVax-L! Each existing, emerging, or recently commercialized therapy in the field of oncology represents another potential opportunity to discover an efficacious combo with the DCVax platform!
Combo is King!
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers

iclight,
Did you post earlier requesting more information about this patient’s DCVax-L treatment? (I believe you said that you were unsure about whether he had received DCVax-L). Based on the published updates, it looks like he received his first dose of DCVax-L in March 2021 and his fourth dose in May 2021.
It was beautiful to see the update posted about him today!!!



https://www.gofundme.com/f/helpjoellivelonger
ex, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm
Thanks for reposting your baseless opinions. Highlighting the significance of Dr. Ashkan is one aspect of your post that I can appreciate. We also cannot overlook the significance of Dr. Liau—she and Ashkan both are extremely noteworthy as we look back on the P3 and as we look forward to the regulatory phase in the UK, US, and other nations…
UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might want to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3 and were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support their respective nations’ governments over many years. Ashkan is an adviser to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020

PM,
🤔 I think you’re on the wrong message board. This is for NWBO DCVax…
You can find DOX here:
https://investorshub.advfn.com/Amdocs-Limited-DOX-24926

LC,
Fact ✅️
PFS is either disproven or confirmed by OS. You disagree? 😶
Page 3 of FDA guidance document:
https://www.fda.gov/media/71195/download

It’s all over the medical literature…





https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173444225
ex,
“Fundamental concept” 😶

Thanks for reposting your views about the ECA and PFS again, but you are mixed up and need better sources of credible information.
You are never going to see the FDA or MHRA allow trial participants to be used as lab rats.
The crossover design was required to recruit and retain participants. Who would want to join a GBM study without crossover or remain in a placebo group after developing progressive disease without having the option of receiving an efficacious treatment?
The P3 was exemplary—clinically appropriate and compassionate.

Skip to timestamp 8:01. Dr. Liau again confirms that the crossover was mandated by the FDA.
https://edhub.ama-assn.org/jn-learning/audio-player/18738384


https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Professor Ashkan is a careful and precise brain surgeon—listen to his words.
Skip to timestamp 13:00 of the ASCO video to hear about the ECA and the SAP.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/


If you prefer a randomized placebo design, then you’ll love the pembrolizumab PD1 blockade combo study that is ongoing at UCLA today. The study has a placebo design and the placebo participants are receiving DCVax-L.
https://clinicaltrials.gov/study/NCT04201873

LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

Thanks for reposting your opinions about PFS again, but I think you’re just throwing random mud. Actual PFS was subsequently proven by the OS data, but the predictive PFS data were contaminated and therefore the investigators could not use the predictive PFS data as a reliable surrogate predictor of OS. Instead, they had to wait for actual OS data.
Oncologists always attempt to measure and predict PFS as they should. Every cancer trial investigator will seek to predict PFS in advance of OS which of course takes longer to discover and confirm.
OS is the ultimate standard, but you cannot wait until death to begin evaluating whether a cancer drug is efficacious. The physician must evaluate a patient’s response and try to determine whether the treatment is helping.
Today, doctors have better methods for measuring progression, particularly when studying immunotherapy agents which are still relatively new as the emerging field of cell-based science is leading to rapid changes in the understanding of how cancer can be assessed and treated.
The P3 for the DCVax-L cell-based technology spanned many years. If the trial were designed and commenced in 2023, the investigators would appropriately attempt to measure progression and predict survival using the best methods and most current medical knowledge available in 2023, as any cancer researcher would.

Don’t forget to follow the combo trial! This study has a placebo design and revised methods for attempting to identify progression. More importantly, the interim survival data are brilliant!
https://clinicaltrials.gov/study/NCT04201873

Amid all of the misinformation and bad-faith disinformation pollution online, it is crucial that we refocus our attention beyond baseless social media posts to seek full-context credible sources and consult licensed oncologists…
JAMA Oncology is a peer-reviewed medical journal that us part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers, healthcare professionals, and the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


PM,
Thank you for asking, but I can only suggest that you conduct some research. Dr. Toms briefly outlines the cost of the Specials Program in the video below. This might be a good starting point for your research:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173443224
PM,
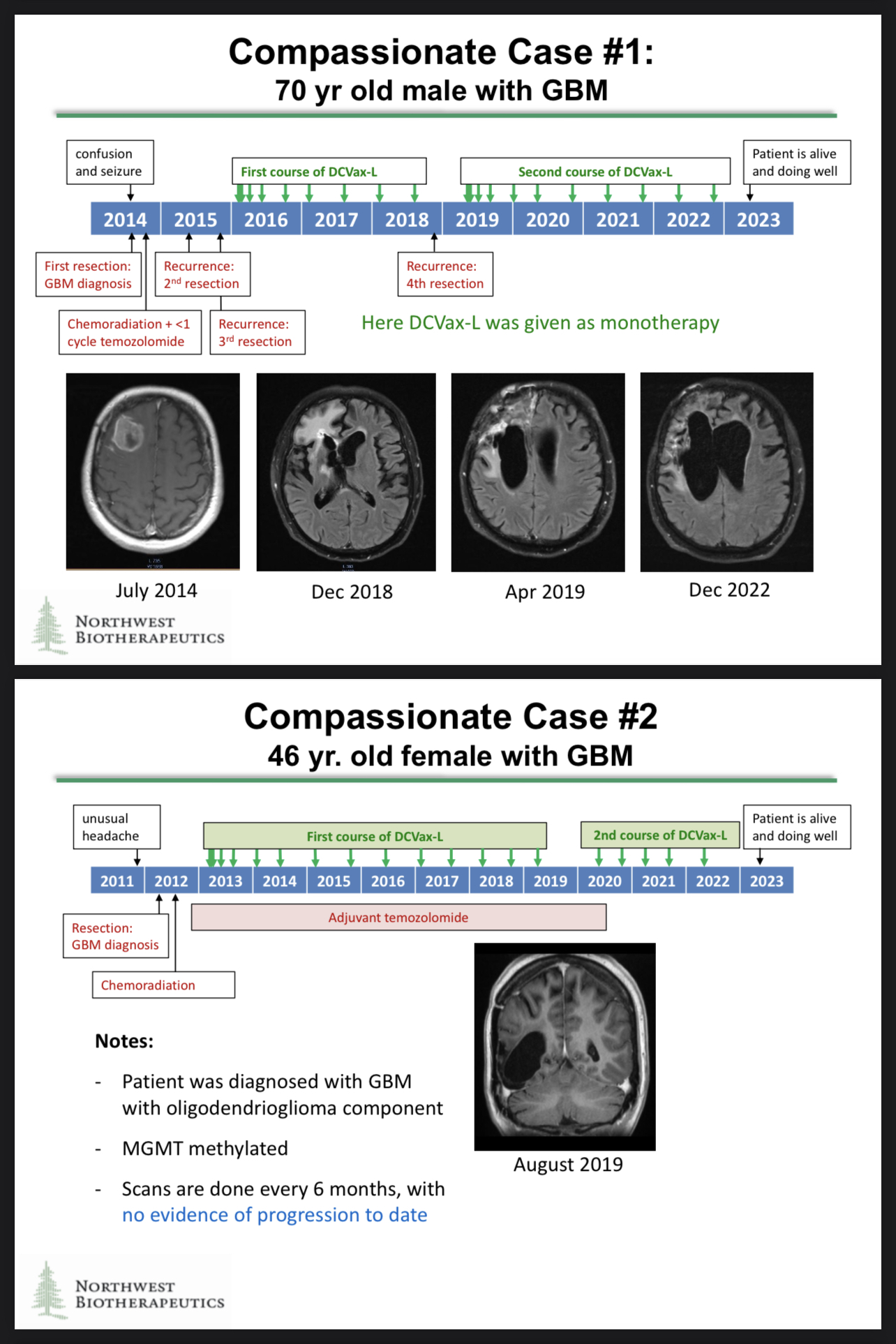
Thank you for asking about the Specials Program. Revenue might not be the most relevant consideration on this topic as the Specials data might have more value in the long run…
The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!


https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
LC’s suggestion of using AI to supplement responses to postings on messages boards was actually a good idea!
“Yes, compassionate use data, also known as expanded access or named patient programs, can be included in regulatory submissions for drug approval. Compassionate use refers to the use of an investigational drug outside of a clinical trial to treat a patient with a serious or life-threatening illness for whom no other treatment options are available.
Regulatory agencies, such as the U.S. Food and Drug Administration (FDA), recognize that compassionate use programs can provide valuable information about a drug's safety and potential efficacy. However, the inclusion of compassionate use data in regulatory submissions is subject to certain conditions:
1. **Data Quality and Relevance:**
- The compassionate use data must meet the standards of quality, relevance, and reliability expected in clinical trials. Robust data collection and reporting practices are crucial.
2. **Consistency with Regulatory Requirements:**
- The data collected under compassionate use should align with the regulatory requirements for the type of approval sought (e.g., New Drug Application, Biologics License Application).
3. **Ethical Considerations:**
- Ethical considerations surrounding patient safety and informed consent are essential. The compassionate use program should adhere to ethical standards, and patients should be well-informed about the risks and benefits.
4. **Corroboration with Controlled Clinical Trial Data:**
- While compassionate use data can be informative, regulatory agencies typically prioritize data from well-designed, controlled clinical trials. Compassionate use data may complement such trial data but is not a substitute for it.
5. **Regulatory Agency Consultation:**
- It's advisable to consult with the relevant regulatory agency early in the drug development process to discuss the potential inclusion of compassionate use data in regulatory submissions. Agencies may provide guidance on how this data can be integrated appropriately.” —ChatGPT
Poor Man,
Bad? Did you read the post? 😶
How could the ChatGPT excerpt be bad? It is solid advice:
✅️ Verify statements published online.
✅️ Seek advice from professionals.
✅️ Consider the source of any information you read.
I am a shareholder who only supports the company by publishing verifiable facts and links to sources that can be evaluated by anyone who clicks on them.
My personal interests: Health and wealth—I hope to see the DCVax technology commercialized worldwide, and I hope to have access to an efficacious immunotherapy combo if I ever need one.
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.
Investors can take several steps to avoid falling victim to stock disinformation:
**Verify Information from Reliable Sources:**
- Cross-reference information from multiple reputable sources. Avoid relying solely on one channel, especially social media or anonymous message boards.
**Understand the Company:**
- Conduct thorough research on the companies you're interested in. Understand their financials, business model, and industry. Reliable information often comes from official reports, financial statements, and reputable news sources.
**Use Caution with Social Media:**
- While social media platforms can provide information, they can also be a source of misinformation. Be critical of information shared on these platforms and verify it through reliable sources.
**Stay Informed about Market Regulations:**
- Understand the regulations governing financial markets. Authorities such as the SEC (U.S. Securities and Exchange Commission) take actions against those engaged in fraudulent activities.
**Consider Professional Advice:**
- Consult with financial advisors or professionals who can provide guidance based on expertise and experience.
**Participate in Reputable Forums:**
- If you engage in online discussions, consider reputable financial forums where discussions are moderated, and misinformation is less likely to thrive.
**Be Skeptical of Unsubstantiated Claims:**
- If an investment opportunity sounds too good to be true or is based on unverifiable claims, approach it with skepticism.
By staying informed and exercising caution, investors can better protect themselves from falling prey to stock disinformation.” —ChatGPT


https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Poor Man,
Generally speaking, I would argue that conflicts of interest are always highly relevant to consider. Whether or not someone is shorting stock in a company is relevant if they are publishing commentary online about the company.
I consulted an AI app in reference to this topic (as learningcurve suggested):
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.
Investors can take several steps to avoid falling victim to stock disinformation:
**Verify Information from Reliable Sources:**
- Cross-reference information from multiple reputable sources. Avoid relying solely on one channel, especially social media or anonymous message boards.
**Understand the Company:**
- Conduct thorough research on the companies you're interested in. Understand their financials, business model, and industry. Reliable information often comes from official reports, financial statements, and reputable news sources.
**Use Caution with Social Media:**
- While social media platforms can provide information, they can also be a source of misinformation. Be critical of information shared on these platforms and verify it through reliable sources.
**Stay Informed about Market Regulations:**
- Understand the regulations governing financial markets. Authorities such as the SEC (U.S. Securities and Exchange Commission) take actions against those engaged in fraudulent activities.
**Consider Professional Advice:**
- Consult with financial advisors or professionals who can provide guidance based on expertise and experience.
**Participate in Reputable Forums:**
- If you engage in online discussions, consider reputable financial forums where discussions are moderated, and misinformation is less likely to thrive.
**Be Skeptical of Unsubstantiated Claims:**
- If an investment opportunity sounds too good to be true or is based on unverifiable claims, approach it with skepticism.
By staying informed and exercising caution, investors can better protect themselves from falling prey to stock disinformation.” —ChatGPT


https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
LC,
Great quote!

Thanks for highlighting Dr. Ashkan’s statement! Other advances in the research of cancer and other technologies may elevate the value of DCVax-L! Each existing, emerging, or recently commercialized therapy in the field of oncology represents another potential opportunity to discover an efficacious combo with the DCVax platform!
Combo is King!
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
iclight,
Thanks for reposting your opinions about the Keytruda study again. It is always worthwhile to remind us about the small but mighty PD1 combo study that is ongoing at UCLA today! The P3 looks like the tip of the iceberg and it is interesting to consider the P3 data in light of the emerging combo data!
A full-context overview might help us think about the prospective value of DCVax technology as a platform…
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include multiple trials spanning years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.

https://clinicaltrials.gov/ct2/show/NCT04201873

In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.


https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/









Fact ✅️
The greatest potential for LP’s “personal gain” would result from a rise in the SP. It is a verifiable fact that the CEO is holding more shares than anyone else.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
HyGro,
Here are some relevant links in response to your repost about the trial design. The links might help to clear up some of your misunderstanding about the P3 design and why the design was particularly appropriate for a study involving GBM.
Skip to timestamp 8:01. Dr. Liau again confirms that the crossover was mandated by the FDA.
https://edhub.ama-assn.org/jn-learning/audio-player/18738384


https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Professor Ashkan is a careful and precise brain surgeon—listen to his words.
Skip to timestamp 13:00 of the ASCO video to hear about the ECA and the SAP.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/


The clinical data in total supporting the DCVax-L platform technology include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
The P3 data were peer reviewed by independent physicians and qualified statisticians—refer to JAMA Oncology.
Safety and efficacy data have been gathered from various clinical settings that include both external controls and placebo controls. DCVax-L has been studied as a monotherapy and in combination with other agents.
If you prefer a randomized placebo design, then you’ll love the pembrolizumab PD1 blockade combo study that is ongoing at UCLA today. The study has a placebo design and the placebo participants are receiving DCVax-L as the best available therapy for rGBM.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not produced reliable evidence of efficacy.
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
HyGro, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm
Thanks for reposting your baseless opinions again, but your comments reflect disregard for the clinical data overall which include multiple clinical studies. Your comments also show disregard for the expertise and integrity of the physicians who led the trial.
Your disinformation about the design of the exemplary P3 is a distraction from the technology as a platform with potential combo indications beyond nGBM and rGBM.
Combo is King!
Research the interim survival data of the small but mighty PD1 combo study that is ongoing at UCLA today!
https://clinicaltrials.gov/study/NCT04201873

UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might want to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3 and were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support their respective nations’ governments over many years. Ashkan is an adviser to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Yes! Thanks for reposting to remind us about JAMA! We can always count on LC to redirect our focus back to the independent peer review 😉


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
LC,
Thanks for reposting about the MAA PR again. There will be plenty of opportunities to clarify each step as the filing is accepted and as other regulators also come into play in other nations…
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
Thanks for reposting about the MAA PR over and over again every day 😶…I think there will be updates as the regulatory process unfolds—plenty of opportunities to clarify each step as the filing is accepted and as other regulators also come into play in other nations…
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
Thank you for highlighting the NIH! The vast significance of Liau’s work and the further development of the DCVax-L cell-based platform technology in combination with other technologies was/is funded in large part by public dollars awarded through NIH peer-reviewed grants. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold.
If you do some research into the NIH and the FDA, you will discover that not every investigational technology receives the same level of support and not every researcher’s work is recognized by consistent support spanning many consecutive years. The NCI division of the NIH develops oncology drugs, and FDA approves drugs. There is some connection, and their historical and continuing support is not insignificant …
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I hope you’ve been well. Thanks for reposting your opinion again about the ECA, but your post is bs as per usual.
Doctors Liau and Ashkan know how to run a clinical study.
You are never going to see the FDA or MHRA allow trial participants to be used as lab rats.
The crossover design was required to recruit and retain participants. Who would want to join a GBM study without crossover or remain in a placebo group after developing progressive disease without having the option of receiving an efficacious treatment?

Conclusion: The P3 was exemplary—clinically appropriate and compassionate.

Skip to timestamp 8:01. Dr. Liau again confirms that the crossover was mandated by the FDA.
https://edhub.ama-assn.org/jn-learning/audio-player/18738384


https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Professor Ashkan is a careful and precise brain surgeon—listen to his words.
Skip to timestamp 13:00 of the ASCO video to hear about the ECA and the SAP.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/


The clinical data in total supporting the DCVax-L platform technology include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
The P3 data were peer reviewed by independent physicians and qualified statisticians—refer to JAMA Oncology.
Safety and efficacy data have been gathered from various clinical settings that include both external controls and placebo controls. DCVax-L has been studied as a monotherapy and in combination with other agents.
If you prefer a randomized placebo design, then you’ll love the pembrolizumab PD1 blockade combo study that is ongoing at UCLA today. The study has a placebo design and the placebo participants are receiving DCVax-L as the best available therapy for rGBM.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not produced reliable evidence of efficacy.
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html