Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
ex,
Speaking for myself as a “long” or “soldier,” I can assert that I listen to the story presented by the regulators and the story presented by the independent peer review. I doubt that anyone “trusts” the story presented by any single individual person.
Fact ✅️
December 2023

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
March 2023

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-and-advent-bioservices-announce-receipt-of-license-for-commercial-manufacturing-at-sawston-uk-facility-301776725.html
February 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-commencement-of-cancer-vaccine-production-at-its-sawston-uk-facility-301484857.html
August 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-approval-of-pediatric-investigation-plan-pip-by-mhra-301610850.html
July 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-filing-of-application-for-license-for-commercial-manufacturing-at-sawston-uk-facility-301581737.html
December 2022

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-mhra-approval-of-license-for-gmp-manufacturing-at-sawston-uk-facility-301449083.html

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
J-TO,
Thanks for your predictions, but your post sounds extreme and a bit alarmist to me. I had to consult the detector on this one 😶 …


“This statement appears to be a weak attempt to persuade or manipulate and is unreliable for several reasons:
1. **Alarmist and Fear-Mongering Language**: Phrases like "gonna suffer a slaughter at the hands of these thieves" and "Nothing will stop them now" are designed to invoke fear and panic. This kind of alarmist language is often used to manipulate emotions rather than provide a reasoned analysis or sound opinion.
2. **Conspiracy Implication**: The claim that "They control the market" implies a conspiracy without providing evidence. Asserting market control by unspecified entities ("big bucks, always wins") is a common tactic in manipulative statements to create a sense of helplessness or inevitability.
3. **Speculative Predictions**: Predictions such as "huge dilution coming" and the stock "headed to the 50's next week" are speculative and presented without basis. Financial markets are complex and unpredictable, making such definitive predictions unreliable.
4. **Personal Opinion Presented as Fact**: The statement ends with "imo" (in my opinion), indicating that much of the preceding content is based on personal views rather than objective analysis.
5. **Negative Sentiment**: The overall tone is negative and defeatist ("HAPPY FRIGGIN NEW YEAR FELLOW BAG HOLDERS"), which can influence readers' perceptions and decisions negatively.
6. **Questionable Investment Advice**: Suggesting to "add if you got the balls to tie up your money for another 6 months to a year" is irresponsible as investment advice should be based on careful analysis, not on bravado or emotional responses to market conditions.
Overall, the statement appears to be more focused on stirring emotions and spreading negativity rather than providing a reliable, fact-based analysis or well-reasoned opinions.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
ex,

The reports are posted.
Anything published online is public information and retained on file somewhere. All BS Detector Reports are available for anyone who wants to read them.

https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173527217

Correction:
I meant to address my response to ex, not LC .. but it could generally apply to both perhaps…🤔
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173528597
LC,
“Basically” “Clearly” …How would you know?
Thanks for posting your legal analysis and forecast, but you’re not a lawyer or legal expert of any kind.
Reading your posts over time, you seem to speak confidently as a medical/clinical trial expert, a financial expert, and now a legal expert 😶
Personally, I’m not following the legal proceedings as I am most interested in the scientific aspects of the novel DCVax-L technology and the regulatory news, so I ran your statement through the detector…

“This analysis of a legal ruling is unreliable for a few key reasons:
1. **Speculative Timeline**: Legal processes, especially in complex cases like those involving corporations, can be unpredictable. While estimating a timeline based on procedural steps is possible, asserting specific dates (like "summer 2024") for rulings or completion is speculative without insider knowledge of the court's schedule and case complexities.
2. **Oversimplification of Legal Procedures**: Legal cases, particularly those involving multiple parties and complex issues, rarely follow a straightforward or predictable path. The statement oversimplifies the process of filing complaints, motions to dismiss (MTD), and the ensuing legal procedures.
3. **Comparison to Other Cases**: Using another case (e.g., "Harrington") as a benchmark for how long discovery might take can be misleading. Each legal case is unique in its details, the parties involved, and how the legal process unfolds.
4. **Predicting Judicial Decisions**: Predicting how a judge will rule on a future complaint or motion is inherently uncertain. Judicial decisions depend on a multitude of factors, including the specifics of the case, legal arguments presented, and applicable law.
5. **Conclusion about Trial Timing**: Concluding that there will be no trial in 2024 or 2025 is speculative. The timing of a trial depends on various factors, including the court's calendar, the progress of pre-trial procedures, and potential settlements or other resolutions.
In summary, while some general predictions about legal processes can be made, this analysis presents a very definite timeline and outcomes without accounting for the inherent unpredictability and complexity of legal proceedings, making it unreliable.”
ex,
Thanks for your analysis, but you are not a licensed financial professional. This one didn’t pass the bs detector either 😶 …


“This statement contains several poorly reasoned and potentially misleading points:
1. **Generalization About Reverse Splits (R/S)**: The claim that reverse stock splits (R/S) are generally viewed as a weakness is an oversimplification. While R/S can sometimes be seen negatively, as they may indicate a company's poor stock performance, they are also used as strategic tools to meet regulatory listing requirements or to attract a different class of investors. The impact of an R/S on share price and company perception varies based on context.
2. **Assumption About Company Turnaround**: The statement suggests that successful R/S occurs after a company has already turned around. This is not always true. An R/S can be part of the turnaround strategy itself.
3. **Debate Among Investors**: While it's true that opinions on R/S can vary among investors, categorizing most "longs" as against R/S without evidence is a broad generalization that doesn't account for varied investor strategies and perspectives.
4. **Claims About NASDAQ Delisting**: The statement about NWBO having been delisted from NASDAQ due to poor governance and subsequent violations is serious and requires substantiation. Without specific references or evidence, these claims remain speculative and potentially misleading.
5. **Current Compliance Allegations**: The assertion that NWBO is currently acting in ways that would violate NASDAQ rules is another serious claim that would need to be backed by concrete evidence.
Additionally, the anonymity of the statement's author and the reckless nature of the analysis further undermine its credibility:
1. **Anonymity and Accountability**: The anonymous authorship of such claims means there is a lack of accountability. Anonymity can sometimes lead to the spread of unfounded allegations, as the author is not publicly responsible for the veracity of their statements.
2. **Reckless Analysis**: The statement provides a sweeping analysis of complex financial and regulatory matters without demonstrating a thorough understanding or providing evidence. This recklessness, especially in discussing serious matters like governance violations and NASDAQ listing rules, can lead to misinformation and unfairly damage the reputation of the company in question.
The combination of anonymity and a lack of careful, evidence-based analysis makes the statement not only poorly reasoned but also potentially harmful and irresponsible.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
ex,
That sounds like another rumor to me. I had to run your post through the bs detector 😶 …


“This statement about "LP" (presumably referring to a specific individual associated with Northwest Biotherapeutics, or NWBO) is composed of greater than 99% baseless innuendo and is defamatory for these reasons:
1. **Lack of Evidence**: The claim that funds are being diverted to "LP and friends" is a serious accusation of financial impropriety. Without concrete evidence or credible sources to support this allegation, it remains baseless.
2. **Defamatory Implications**: Suggesting that someone is misusing company resources or manipulating news for personal gain can be defamatory, as it can harm the person's reputation, especially if these claims are unsubstantiated.
3. **Vague and Speculative Language**: The statement is vague ("carefully playing the news," "much runs to LP and friends") and lacks specifics, which is characteristic of innuendo designed to imply wrongdoing without making a direct accusation.
4. **Generalization Without Context**: The statement generalizes about the individual's actions over the years without providing specific instances or context.
In summary, the statement makes serious allegations without substantiation, uses language typical of innuendo, and could potentially damage the reputation of the individual referred to as "LP," rendering it defamatory and baseless.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
Did you mention “truth” in that post? 😶
Thanks for posting, but I think that we should seek trustworthy advice from licensed oncologists. I ran your statement through the detector…


“This statement is misleading and unreliable for several reasons:
1. **Lack of Credibility**: The author's anonymity and lack of credentials as an oncologist mean they likely don't have the expertise to accurately assess the efficacy of a cancer treatment like DCVax-L. Expert medical opinions typically come from qualified healthcare professionals.
2. **Generalized and Unsupported Claims**: The claim that DCVax-L is only "mildly effective in some small subset of patients" is a broad generalization without supporting data. Clinical effectiveness should be backed by scientific studies and clinical trial results.
3. **Misrepresentation of Treatment Protocol**: Suggesting that DCVax-L "requires brutal chemo and radiation up front" is misleading. DCVax-L is a type of immunotherapy, and while it might be used in conjunction with chemotherapy and radiation, describing these as "brutal" is subjective and doesn't accurately represent the nuanced decision-making in cancer treatment protocols.
4. **Dismissive Tone**: The statement's dismissive tone ("But don't let the truth get in your way") undermines its reliability. It suggests a biased viewpoint rather than a balanced, evidence-based assessment.
Additionally, the statement fails to acknowledge the potential of DCVax-L for use in combination therapy beyond traditional chemotherapy and radiation. Recent trends in cancer treatment show a growing interest in combining different therapeutic modalities, including immunotherapies like DCVax-L with other agents such as PD-1 blockade. These combination therapies can potentially enhance overall efficacy and provide new treatment avenues for patients. The dismissive tone of the statement overlooks these evolving and nuanced approaches in oncology, further undermining its reliability.
In summary, the statement's lack of credible authorship, generalized claims, and subjective language make it misleading and unreliable as a source of information about DCVax-L or any medical treatment.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

JAMA Oncology Peer-Reviewed Publication
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

LC,
Thanks for asking about the Specials Program again. Revenue might not be the most relevant consideration on this topic as the Specials data might have greater value in the long run…
The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!


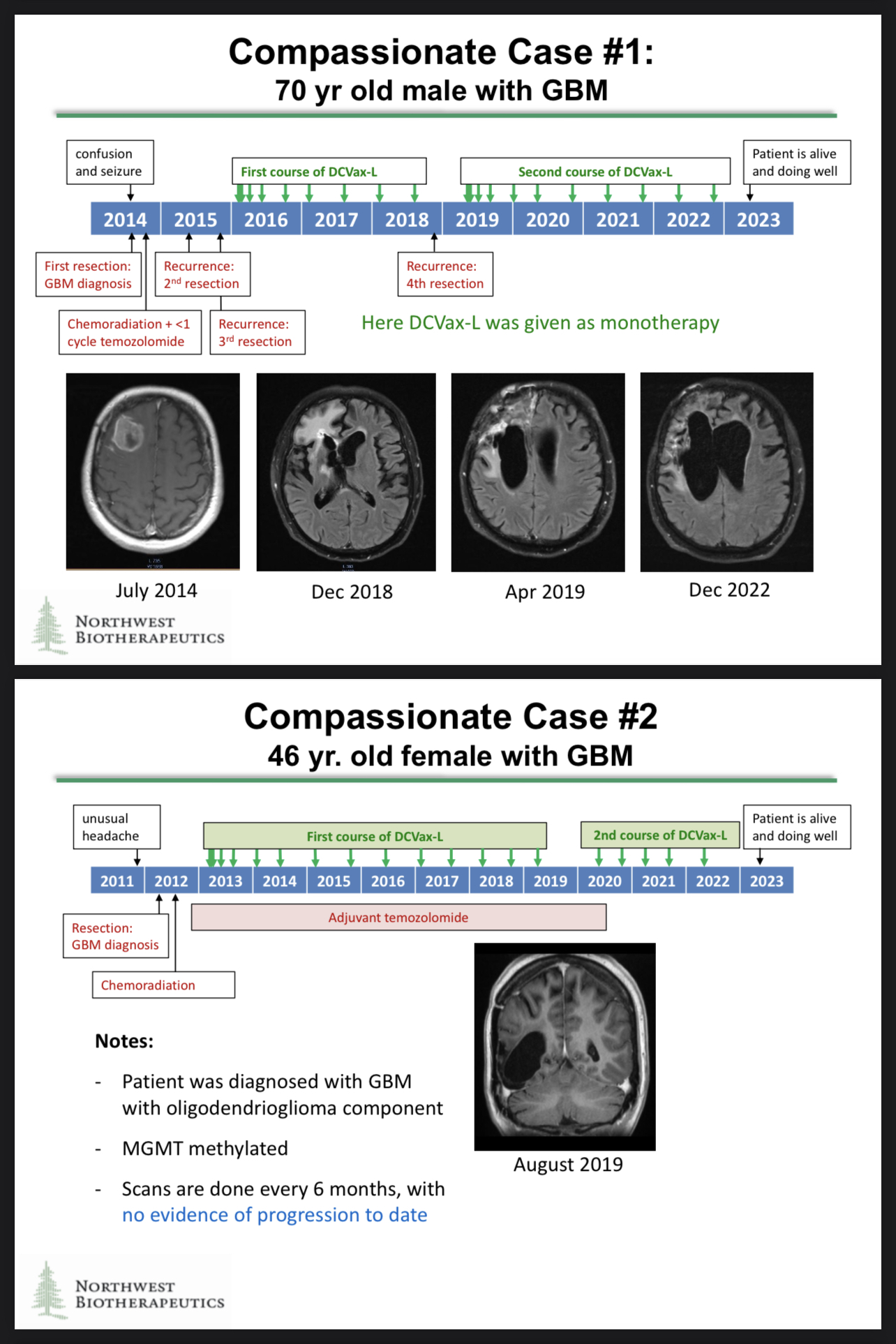

“Compassionate use data, also known as expanded access or named patient programs, can be included in regulatory submissions for drug approval. Compassionate use refers to the use of an investigational drug outside of a clinical trial to treat a patient with a serious or life-threatening illness for whom no other treatment options are available.
Regulatory agencies, such as the U.S. Food and Drug Administration (FDA), recognize that compassionate use programs can provide valuable information about a drug's safety and potential efficacy. However, the inclusion of compassionate use data in regulatory submissions is subject to certain conditions:
1. **Data Quality and Relevance:**
- The compassionate use data must meet the standards of quality, relevance, and reliability expected in clinical trials. Robust data collection and reporting practices are crucial.
2. **Consistency with Regulatory Requirements:**
- The data collected under compassionate use should align with the regulatory requirements for the type of approval sought (e.g., New Drug Application, Biologics License Application).
3. **Ethical Considerations:**
- Ethical considerations surrounding patient safety and informed consent are essential. The compassionate use program should adhere to ethical standards, and patients should be well-informed about the risks and benefits.
4. **Corroboration with Controlled Clinical Trial Data:**
- While compassionate use data can be informative, regulatory agencies typically prioritize data from well-designed, controlled clinical trials. Compassionate use data may complement such trial data but is not a substitute for it.
5. **Regulatory Agency Consultation:**
- It's advisable to consult with the relevant regulatory agency early in the drug development process to discuss the potential inclusion of compassionate use data in regulatory submissions. Agencies may provide guidance on how this data can be integrated appropriately.” —ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://www.theguardian.com/science/2022/nov/17/im-just-carrying-on-vaccine-gives-brain-cancer-patient-years-of-extra-life
https://www.braintumourresearch.org/stories/in-hope/in-hope-stories/kat-charles
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
LC,
Your curiously vague reference to LP and Asia didn’t pass the detector either 😶 …


“The statement is potentially defamatory and appears intended to fuel a rumor for several reasons:
1. **Implied Wrongdoing**: The suggestion that "Powers" (presumably a reference to a specific individual in a position of authority or influence) might exploit her "Asian contacts" implies unethical or underhanded behavior without providing any evidence or context. This insinuation can be damaging to her reputation.
2. **Lack of Specificity and Evidence**: The statement is vague about what "Powers" might "try" with her Asian contacts. This lack of specificity, coupled with no evidence, suggests that the intent is to insinuate wrongdoing or illicit activities without making a direct accusation.
3. **Feeding Speculation**: By mentioning that "this regulator never gets discussed here," the statement seems designed to spark curiosity and speculation, characteristic of rumor-mongering.
4. **Potential for Misinformation**: The statement's speculative nature and lack of factual basis can lead to the spread of misinformation.
In summary, the statement appears to be crafted to cast aspersions and fuel rumors without providing any substantiating information, which can be damaging to the individual's reputation and is characteristic of defamatory intent.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
Thanks for posting, but this sounds like another rumor to me. I ran it through the detector 😶 …

“This statement is composed of greater than 99% baseless innuendo for the following reasons:
1. **Speculative and Suggestive**: The statement is purely speculative, suggesting a scenario ("Powers could agree to provide L at cost") without any evidence or context to substantiate the claim.
2. **Implied Wrongdoing**: By saying "if you catch my drift," the statement implies wrongdoing or unethical behavior without directly stating it, which is characteristic of innuendo.
3. **Lack of Transparency Claim**: The assertion that "she's never shared a thing about the numbers with retail" is presented without evidence. It's important to note that companies have various regulatory and legal obligations regarding financial disclosures.
4. **Assumption of Malintent**: The statement assumes malintent on the part of Powers without providing factual support for such a claim.
The statement can also be considered potentially defamatory because it implies unethical or dishonest behavior on the part of Powers without providing any evidence. Defamation involves making false statements that harm someone's reputation. By suggesting improper conduct in a business setting without factual backing, the statement risks defaming Powers, especially if these insinuations are baseless and if Powers is a recognizable figure associated with a specific company or context. Such accusations, when unsubstantiated, can damage a person's professional reputation and credibility.
In summary, the statement relies on innuendo and speculation, lacking factual basis and unfairly implying wrongdoing without substantiation.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
iclight,
Thanks for posting, but your post sounds like an attempt to start rumors. I had to run this one through the detector 😶 …

“This statement is poorly-reasoned, baseless, and defamatory for several reasons:
1. **Lack of Evidence**: It makes several accusations and assumptions about intentions and actions (or lack thereof) of a CEO without providing concrete evidence to support these claims.
2. **Conflation of Processes**: The statement misunderstands the complexity of drug development and regulatory approval. Submitting for approval and getting approval are indeed different, but the statement fails to acknowledge the nuances and challenges inherent in these processes.
3. **Assumption of Malintent**: It assumes without basis that the CEO deliberately engaged in actions to impede the drug's approval, which is a serious accusation that requires substantial proof.
4. **Misrepresentation of Clinical Trials**: The claim that the CEO called for a trial change to make approval impossible is a serious accusation and overlooks the myriad factors that can lead to changes in clinical trial protocols.
5. **Defamatory Accusations**: Accusing the CEO of intentionally delaying the drug's development for personal financial gain, and of planning to blame external factors for the drug's failure, is defamatory without clear, substantiated proof.
6. **Oversimplification**: It oversimplifies the challenges of navigating the FDA approval process, suggesting that hiring certain companies could easily overcome these hurdles.
7. **Ignoring Regulatory Complexity**: The decision not to submit to the FDA could be due to various strategic or regulatory considerations, which the statement does not acknowledge.
In summary, the statement is speculative, lacks substantiation, and makes defamatory claims, rendering it baseless and poorly-reasoned.” 
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
Yes, that was a significant era! I wasn’t alive back then, but the genesis of the internet was roughly around the same time that the foundational research behind cell-based immunotherapy technology began.
You might be interested in this video as it seems to foreshadow the discoveries outlined in the JCI article that you posted here!
A look back at some recent history of cell-based science in medicine…
It is interesting in the interview excerpt linked below how Dr. Steinman spoke about the first-generation of DC technology and how further development would follow. He explained that the prostate vax only addressed a single antigen.
It’s also interesting to consider the combination technologies that are now emerging as well!
The first CTLA-4 inhibitor was approved by the FDA in 2011, the same year Dr. Steinman was awarded the Nobel Prize. In 2014, the first PD1 inhibitors were approved. Interestingly, the researchers behind PD1 blockade technology were also awarded the Nobel Prize 7 years after Dr. Steinman’s award.
https://www.thestar.com/life/health-wellness/dr-ralph-steinman-s-immune-cell-discovery-called-game-changing/article_5676ebf4-b807-5d86-81e5-b590c0bb61c6.html
“The key aspect of DCVax-L is that it does not target a specific antigen but rather a wide array of antigens present in the tumor lysate. This approach is based on the understanding that every tumor is unique, with its own distinct set of mutations and antigens. By using the tumor lysate, DCVax-L can potentially stimulate the immune system to recognize and attack a broad spectrum of tumor-specific antigens.
Provenge (Sipuleucel-T) is a therapeutic cancer vaccine used for the treatment of advanced prostate cancer. Unlike broad-spectrum immunotherapies like DCVax-L, Provenge targets one specific antigen: Prostatic Acid Phosphatase (PAP), an enzyme expressed in most prostate cancer cells.
The treatment involves extracting a patient's dendritic cells and exposing them to a fusion protein called PA2024, which is a combination of PAP and an immune-stimulating factor. The activated dendritic cells are then reinfused into the patient, where they are intended to stimulate the immune system to attack cells expressing PAP.
In summary, Provenge specifically addresses one key antigen associated with prostate cancer—Prostatic Acid Phosphatase. This approach is different from therapies like DCVax-L, which aim to target a wide range of tumor-specific antigens.”
—ChatGPT


https://www.jci.org/articles/view/169314
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
Ex,
Thanks for posting your vague opinion, but it sounds like you’re trying to start rumors. LP is a shareholder too. I had to run your curious post through the detector .. 😶

“This statement is composed of greater than 99% innuendo. The statement is baseless and potentially defamatory for these reasons:
1. **Lack of Evidence**: It asserts that LP (presumably a reference to a specific individual, likely a corporate leader or significant shareholder) has a plan known to "shorts" (those betting against the company's stock) but provides no evidence or context to support this claim. Without substantiation, such a statement is purely speculative.
2. **Implication of Malfeasance**: By suggesting that LP's plan is not aligned with the hopes of "longs" (investors who believe in the company's future success), it implies wrongdoing or deceit on LP's part without providing any proof.
3. **Generalization and Assumption**: The statement makes a broad assumption about what "longs" fail to grasp, implying they are unaware or naive about the company's internal strategies, which is a sweeping generalization without basis.
4. **Potential Defamation**: If LP is a real individual, suggesting they have secretive, possibly malicious plans without evidence can be considered defamatory, as it can harm the individual's reputation.
The statement lacks factual support, resorts to speculative insinuations, and could potentially damage the reputation of the individual referred to as "LP."

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
MrCee-Cee,
Thanks for the reply. I regret to inform you that the AI bs detector identified that response as bs too 😶
“This statement is nonsensical for several reasons:
1. **Misunderstanding of AI's Role**: The statement misinterprets the purpose and capability of AI. AI, particularly in this context, is designed to assist with information processing and generating responses based on available data. It's not intended to replace human thinking but to augment it.
2. **Contradictory Statements**: On one hand, the statement criticizes reliance on AI for making convincing rather than true arguments, but on the other hand, it acknowledges holding a position in NWBO, a decision likely based on information processing and analysis, which AI can assist with.
3. **Ad Hominem Attacks**: The statement includes derogatory remarks about "idiot-filled cult" and "nwbo pumper on Twitter," which are personal attacks rather than logical arguments about the actual use and utility of AI.
4. **Overgeneralization about AI**: It generalizes AI's capability as merely being an "information aggregate." Modern AI systems are capable of complex tasks, including learning from data, identifying patterns, and providing insights.
5. **Lack of Constructive Critique**: While it criticizes the use of AI, it fails to provide constructive insights into what it believes AI should be used for or how AI could be more effectively employed.
In summary, the statement appears to stem from a misunderstanding of AI's role and capabilities and resorts to personal attacks rather than providing a reasoned critique of AI's application in discussions or analysis.”
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173512857
MrCee-Cee,
Thanks for the reply, but you did not address any of the points that I outlined with reference to value.
I ran your response through an AI BS detector and your response was identified as bs 😶

“This statement is greater than 99% nonsensical and illogical for the following reasons:
1. **Lack of Specifics**: It criticizes a post as "nonsense" and "delusional" but fails to specify what makes the post unrealistic or deluded. Without specific examples or arguments, the criticism is vague and ungrounded.
2. **Ad Hominem Attack**: The use of terms like "garbage board" and "cult" is an ad hominem attack, which detracts from constructive discussion. It targets the people involved rather than addressing the content of the discussion.
3. **Dismissiveness**: The statement dismisses another person's perspective as delusional without attempting to understand or engage with it. This approach shuts down dialogue rather than fostering meaningful exchange.
4. **Overgeneralization**: By labeling the entire forum as being for investors seeking value and implying that other perspectives are delusional or harmful, the statement unfairly overgeneralizes and excludes diverse viewpoints which can be valuable in an investment discussion.
In summary, the statement's dismissive tone, lack of specifics, and use of ad hominem attacks render it nonsensical and unproductive for a constructive discussion on investment forums.”

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173512857
MrCee-Cee,
Your posts are baseless and your logic doesn’t follow. 😶 …It sounds like you need to do some research—avoid social media and seek your own credible full-context sources of info.
The leadership of this small pre-commercial biotech have been committed to building the prospective value for a significant period of time. Aside from the successful completion of the P3 and the independent peer review that followed, the leadership have coordinated the building of a voluminous MAA, the building of manufacturing capacity, and the building of a comprehensive worldwide patent portfolio, and the list goes on …
Quite arguably, the prospective value of the company is greater today than it has ever been since the company was founded…
The price will follow the real value just as dawn follows night, but the management does not have direct immediate control over all of the market forces, especially at the pre-commercial stage and while on the OTC exchange.
Regulatory developments will be the most determinative factors following the independent peer review, and NWBO is still currently on the OTC. The emerging combo data are also immensely significant and further developments on that front will have an impact. Partnerships are also included in the wide array of possibilities going forward in the near term.
The value of DCVax®-L as a novel cell-based immunotherapy platform technology will be determined by the integrity of the science, and the regulatory process will be the catalyst for its change in price.
As some of the freshly-poured regulatory cement begins to dry into hard stone in the UK and in other nations, further combos and DCVax-Direct will present additional potential for the further expansion of value.
From an investment standpoint, we always hope that the value is greater than the price for the period time preceding their alignment. When will they align? The hands of that clock move with government regulators and with the economics of the pharmaceutical industry, but the science and unmet medical needs comprise the essence of true value.


https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
ex,
I like that opinion! 😉 I’m also looking forward to:
⭐️ Approval(s)
⭐️ Partnership(s)
I would have to disagree slightly about the combos and acceptance of the MAA being “non-news.” 😶 …The emerging combo data are brilliant, and any news on that front would be highly significant. Logically, acceptance of the MAA submission will also be significant news because acceptance precedes the review and is obviously required for approval.
Combo is King! ✅️
https://www.jci.org/articles/view/169314
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
Typo correction:
*Assessed, not “accessed” ✅️
AI is very effective at assessing the logical or illogical structure of a claim and searching for legitimate references to full-context sources.
The sources cited below are examples of legitimate full-context sources. Anyone can evaluate and research the text of these sources. Further, the credentials and backgrounds of the physicians can be independently evaluated by anyone who chooses to research their work or research the institutions that employ them.
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
Thanks for asking. The AI BS Detector has accessed that your “lies” are presented mostly in the form of baseless defamatory suggestions and other innuendo 😶
“LC’s statements are most often composed of conjecture and lack any factual basis or references to credible evidence.
Innuendo is dishonest when it's used to imply something derogatory or misleading without stating it directly, thereby avoiding clear, straightforward communication. Innuendo often relies on suggestion or insinuation, allowing the speaker to convey a negative message while maintaining plausible deniability. This indirect way of communicating can be seen as a form of dishonesty, especially if the intent is to deceive or manipulate the listener's perception.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
LC,
Your post is not substantive and really contributes no real insight to the conversation.
Furthermore, the AI BS Detector has detected a large amount of bs…again 😶

“This statement is illogical and weak for several reasons:
1. **Inconsistent Logic**: It criticizes someone for making a high stock price prediction despite knowing about shorting and spoofing, yet later argues that shorting and spoofing don't necessarily impact stock prices negatively, as evidenced by Tesla's example.
2. **Ignoring Market Complexity**: It oversimplifies stock market dynamics. Shorting and spoofing are just two of many factors that can affect a stock's price. The statement fails to consider other critical factors.
3. **Ad Hominem Attacks**: Calling someone ignorant is an ad hominem attack and doesn't contribute to a logical argument.
4. **Selective Comparison**: Using Tesla as a comparison is selective and is not directly relevant to NWBO's situation, as each company's stock is influenced by its unique circumstances.”
LC,
Thanks for posting or reposting so frequently, but your posts are nonsense and appear to reflect an attempt to start factless rumors.
The AI BS Detector has once again detected a large amount of bs in your post …😶 Fact ✅️

“The statement presents a series of unfounded speculations and assertions that are nonsensical. It makes sweeping claims about management intentions without evidence and wildly guesses future corporate actions like buyouts or reverse splits. The simplistic correlation of company milestones with stock price declines completely overlooks the multifaceted factors that influence stock market dynamics. Overall, the statement is more a reflection of baseless conjecture than a reasoned analysis grounded in reality.
This statement is problematic for several reasons:
1. **Unsubstantiated Claims About Management**: The assertion that the management team will cause harm to “retail” is baseless.
2. **Speculation on Company Actions**: The speculation about a potential buyout, reverse split, or new trial is just that—speculation. It lacks any reference to concrete evidence.
3. **Simplistic View of Stock Price Movements**: Associating company milestones with a declining stock price oversimplifies how stock markets work. Many factors influence stock prices, not just company “milestones.”
4. **Predictive Statements Without Basis**: The prediction of further decline in stock price upon acceptance is speculative and not grounded in factual analysis.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
iclight,
Thanks for asking. Your question about Switzerland raises an interesting topic…
Regulatory collaboration agreements seem to have become significant only recently, generally speaking. The UK is now a member of both:
1. Access Consortium (formerly named ACSS)
2. Project Orbis
https://www.eversana.com/2020/10/20/uks-mhra-joins-acss-consortium/

https://www.gov.uk/guidance/access-consortium
Legend,
Unfortunating, it appears that your posts are defamatory and baseless. The AI bs detector has once again identified your post as bs.


“This statement is greater than 99% innuendo and conjecture.
1. **Accusations of Manipulation**: The claim that company executives are manipulating retail investors is a serious accusation and would require substantial evidence, which is not provided.
2. **Hypothetical Scenario ("Carrot")**: The metaphor of "dangling a carrot" is used to imply false promises or unachievable goals. However, this is presented without specific evidence to substantiate the claim.
3. **Stock Price Speculation**: The correlation of company milestones with declining stock prices is noted, but stock prices can be influenced by many factors, not just company performance or news. The statement doesn't account for these complexities.
4. **Predictive Assertions**: The predictions about future stock prices, including post-approval scenarios, are speculative and not backed by any analysis.
5. **Negative Comparison**: Drawing a parallel with another company's experience (Outlook Therapeutics) is anecdotal nonsense and does not constitute a logical reason to expect similar outcomes.
Overall, the statement is nonsensical and does not present any logical argument or evidence-based analysis of the company's actions or stock performance.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
Legend,
Thanks for posting again, but your posts are all mostly nonsense.
I ran your statement through an AI bs detector, and I regret to inform you that it was identified as bs.

“This statement is greater than 99% innuendo and conjecture. The assertions are baseless and nonsensical for several reasons:
1. **Ad Hominem Attack**: The use of derogatory language (e.g., "twat") undermines the credibility of the argument. It's an ad hominem attack rather than a reasoned argument.
2. **Unsubstantiated Predictions**: The claim about the company's stock price not reaching $2 in 2024 and possibly dropping to $0.50 is presented without any supporting evidence or analysis.
3. **Speculation Without Basis**: The suggestion that the management team and consultants might have "forgotten something on the application" is pure speculation and lacks any evidence.
In summary, the statement lacks factual support, relies on personal attacks and unfounded assumptions, and does not present a logical or evidence-based argument.”

https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
Well, whether anyone likes it or not, the U.S. Food and Drug Administration is a government agency. It is a part of the Department of Health and Human Services (HHS).
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173499844
LC,
Thanks for reposting that again. Like I said before…
I ran your statement through an AI bs detector, and I regret to inform you that it was identified as bs.

“The statement is weak for the following reasons:
1. **Lack of Concrete Evidence**: It is speculative and not based on factual evidence. Various possibilities are suggested without providing solid information to support them.
3. **Speculation on Motives**: It makes assumptions about motives behind certain actions, which is inherently weak without concrete evidence.
4. **Ambiguity**: The use of phrases like "could be," "I think," and "perhaps" indicates uncertainty and a lack of commitment to a definitive claim.
5. **Conspiracy Implication**: Suggesting a scenario is "manufactured" implies a conspiracy, which weakens its credibility without providing proof.
In summary, without concrete evidence and clarity, the statement's speculative nature and implications of conspiracy make it weak and baseless.”

https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
That’s an interesting topic…
Regulatory collaboration agreements seem to have become significant only recently. The UK is now a member of both:
1. Access Consortium (Formerly named ACSS)
2. Project Orbis
https://www.eversana.com/2020/10/20/uks-mhra-joins-acss-consortium/

https://www.gov.uk/guidance/access-consortium
I saw that—the video is linked below.
I think it is interesting that her experience includes a presidential appointment to the U.S. Commerce Department many years ago. She also taught a course at the U.S. National Institutes of Health on the development and commercialization of medical products.
Linda Powers, Deputy Asst Sec. of Commerce for Services, July 17, 1990.
https://www.c-span.org/video/?c5017932/user-clip-linda-powers-testifying-congress
iclight,
Thanks for asking. I followed some breadcrumbs and they led to a massive bread factory surrounded by golden fields of wheat that expanded into the horizon! Take a look at the links below 😉
Combo is King!
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
https://www.jci.org/articles/view/169314
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
I wish you a joyful and peaceful holiday season!
I regret to inform you that I ran your statement through the AI bs detector and it was identified as bs:
“This statement contains several speculative elements and assumptions, lacking factual accuracy or evidenced validity. Here are a few reasons why this statement is largely nonsensical and baseless:
1. **Lack of Context and Specificity**: The statement lacks clear context and specific information about the trial, the entities involved (like "Senti," "UCLA," "Powers," "NWBO").
2. **Speculative Nature**: The statement includes speculative assertions about motivations and actions of various parties involved in a clinical trial. These speculations are not backed by evidence and are presented as possibilities rather than established facts.
3. **Conspiracy Implications**: Suggesting that entities are deliberately not wanting a name to be associated with a trial due to fear of adverse events or for intellectual property negotiations implies a level of conspiracy which generally lacks credibility without substantial evidence.
4. **Clinical Trial Processes**: The statement misunderstands or oversimplifies the complex nature of clinical trials, particularly regarding control over adverse events and intellectual property negotiations. Clinical trials are subject to rigorous regulatory oversight, and adverse events are an expected part of the trial process, not a reason to exclude names or entities.
5. **Inherent Uncertainty in Clinical Research**: The statement that a larger trial would be required is one of the few reasonable elements, as it’s common in clinical research to require larger studies for definitive conclusions. However, this is framed within a speculative narrative.
Overall, the statement seems to mix elements of fact with speculation and unverified assertions about the intentions and actions of various parties involved in clinical research. Without clear evidence or a more detailed understanding of the context, the statement appears to be more of a personal conjecture than a scientifically or factually grounded assertion.”
—ChatGPT

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
Correction regarding the timeline, to be more specific:
The first CTLA-4 inhibitor, ipilimumab, was approved by the FDA in 2011.
The first PD-1 inhibitors, pembrolizumab and nivolumab, were approved in 2014
The foundational research leading to the blockbuster PD1 drugs that we have today preceded those first approvals by decades beginning in the 1980s and 1990s.
I think it’s important to appreciate that PD1 blockade technology didn’t come to fruition overnight.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173499366
A look back at some recent history of cell-based science in medicine…
It is interesting in the interview excerpt linked below how Dr. Steinman spoke about the first-generation of DC technology and how further development would follow. He explained that the prostate vax only addressed a single antigen.
It’s also interesting to consider the combination technologies that are now emerging as well!
The first checkpoint inhibitor was approved by the FDA in 2011, the same year Dr. Steinman was awarded the Nobel Prize. Interestingly, the researchers behind PD1 blockade technology were also awarded the Nobel Prize 7 years after Dr. Steinman’s award.
“The key aspect of DCVax-L is that it does not target a specific antigen but rather a wide array of antigens present in the tumor lysate. This approach is based on the understanding that every tumor is unique, with its own distinct set of mutations and antigens. By using the tumor lysate, DCVax-L can potentially stimulate the immune system to recognize and attack a broad spectrum of tumor-specific antigens.
Provenge (Sipuleucel-T) is a therapeutic cancer vaccine used for the treatment of advanced prostate cancer. Unlike broad-spectrum immunotherapies like DCVax-L, Provenge targets one specific antigen: Prostatic Acid Phosphatase (PAP), an enzyme expressed in most prostate cancer cells.
The treatment involves extracting a patient's dendritic cells and exposing them to a fusion protein called PA2024, which is a combination of PAP and an immune-stimulating factor. The activated dendritic cells are then reinfused into the patient, where they are intended to stimulate the immune system to attack cells expressing PAP.
In summary, Provenge specifically addresses one key antigen associated with prostate cancer—Prostatic Acid Phosphatase. This approach is different from therapies like DCVax-L, which aim to target a wide range of tumor-specific antigens.”
—ChatGPT


https://www.jci.org/articles/view/169314
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
HyGro,
Thanks for asking. Perhaps review these again as a starting point for your research:
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
It’s ChatGPT. I never used it before until a ridiculous fudder mentioned the idea. It’s ironic that a fuddy buddy gave me the idea because AI is actually a great bs detector! 🤣 Fact ✅️


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
I must give credit where credit is due— this AI poem was inspired by LC. In the spirit of finding the silver lining around the darkest and weakest of clouds, we can appreciate that he has honored us with the endearing title of Soldiers, he has inadvertently highlighted the vast combo/platform potential of DCVax-L, and he directed us to AI technology which has proven very helpful in researching the veracity of his posts and the posts of others.
Definitely one the best posts of 2023:
https://www.jci.org/articles/view/169314
Combo is King!
In the spirit of the season, let’s enjoy a holiday song … 🎄🕎☪️✝️⛄️

The Curve
In the digital realm, where words are cast like stones,
Lived a message board poster, known by his tones.
"Curve," they called him, with irony's twirl,
Set to tarnish a name, in the online whirl.
But oh, the twist in this virtual play,
His efforts to defame, led a different way.
For each word he crafted, in shadows' embrace,
Unwittingly guided others to a brighter space.
Northwest Biotherapeutics, the target in sight,
Found in his words, an unintended light.
Through his labyrinth of critiques, so sharp and so stern,
Others found reason to listen, engage, and discern.
In the irony of his mission, so narrow and curt,
Emerged a clearer picture, from under the dirt.
His aim to diminish, to belittle, to scorn,
Became a beacon, and new Soldiers were born.
So cheers to Curve, in this twist so bold,
His shadowed attempts led to insights untold.
In the web's tangled night, so deep and so right,
His own curveball shone a most unexpected light.

The greatest lessons that the bad-faith baseless attacks have taught us:
✅️ Research, research, research
✅️ Verify, verify, verify
✅️ Believe carefully
✅️ Seek credible full-context sources
✅️ Consult professionals
✅️ Always consider the source
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.
Investors can take several steps to avoid falling victim to stock disinformation:
**Verify Information from Reliable Sources:**
- Cross-reference information from multiple reputable sources. Avoid relying solely on one channel, especially social media or anonymous message boards.
**Understand the Company:**
- Conduct thorough research on the companies you're interested in. Understand their financials, business model, and industry. Reliable information often comes from official reports, financial statements, and reputable news sources.
**Use Caution with Social Media:**
- While social media platforms can provide information, they can also be a source of misinformation. Be critical of information shared on these platforms and verify it through reliable sources.
**Stay Informed about Market Regulations:**
- Understand the regulations governing financial markets. Authorities such as the SEC (U.S. Securities and Exchange Commission) take actions against those engaged in fraudulent activities.
**Consider Professional Advice:**
- Consult with financial advisors or professionals who can provide guidance based on expertise and experience.
**Participate in Reputable Forums:**
- If you engage in online discussions, consider reputable financial forums where discussions are moderated, and misinformation is less likely to thrive.
By staying informed and exercising caution, investors can better protect themselves from falling prey to stock disinformation.” —ChatGPT
https://investorshub.advfn.com/boards/most_read.aspx?p=d
Exactly—nGBM and rGBM require approval of the DCVax-L technology.
Reading the commentary of even the most critical naysayers, the most logical inference that we can draw is that they’re making the case for phase 4.
Generally, all drugs are effectively under some kind of phase 4-like scrutiny following commercialization, or they should be. Pembrolizumab is always a good example— safety, efficacy, and combo data have continued to be gathered and analyzed for years and years following its original approval.
Some further commentary from an AI app:
“A Phase 4 study in the context of drug development is a post-marketing surveillance trial. After a drug has been approved by regulatory authorities and made available for general use, Phase 4 studies are conducted to monitor the drug's performance in real-world settings. Key aspects of these studies include:
1. **Safety Monitoring:** Ongoing assessment of the drug's safety profile, especially long-term side effects that might not have been apparent in earlier phases.
2. **Efficacy in General Use:** Evaluation of the drug's effectiveness when used by a broader population outside of clinical trials.
3. **Comparisons and Combinations:** Testing the drug in combination with other therapies, or comparing it against other drugs available for the same condition.
4. **Identification of New Uses:** Investigating potential new indications or uses for the drug.
Phase 4 studies provide valuable data that can lead to the optimization of drug use, better understanding of side effects, and overall improvement in patient care.” —ChatGPT
ex,
You raise an excellent point!
I agree—with so many solid tumor cancers, the DCVax-L cell-based platform technology will be studied well into the future. Keytruda is the perfect example of this—Pembrolizumab is a blockbuster drug and has been included in many more clinical trials for years following its original approval and commercialization so many years ago!
Combo is King!
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-announces-that-a-marketing-authorization-application-has-been-submitted-to-the-uk-mhra-for-dcvax-l-for-glioblastoma-302021038.html
LC,
Thanks for reposting that again. I thought we agreed to discuss the Soldier Milestones 😉
In my view, the short list of pre-commercial milestones that stand out and shine the most:
⭐️ JAMA Oncology
⭐️ Multiple MHRA Approvals
⭐️ Emerging Combo Data
⭐️ MAA Submission
Further, Dr. Liau joining the SAB at Northwest Biotherapeutics is awesome!
Combo is King!
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers







