Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
July 2023




https://www.bnosconference.co.uk/bnos-conference/
September 2023






https://www.cns.org/annualmeeting
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
HyGro,
Thanks for the response. Just to clarify: The MIA is a prerequisite of the MAA for DCVax-L.
The Specials License is referred to as “MS,” not MIA. The MIA is a commercial license that will allow DCVax-L to be manufactured commercially once it has its own approval. The MIA will apply to the DCVax-L product manufactured at Advent following the approval of DCVax-L.

https://nwbio.com/northwest-biotherapeutics-and-advent-bioservices-announce-receipt-of-license-for-commercial-manufacturing-at-sawston-u-k-facility/
https://mhrainspectorate.blog.gov.uk/2019/10/04/mhra-process-licensing-useful-information/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
HyGro,
Thank you for highlighting the topic of the MHRA! I would like to add a reminder to this conversation about the highly significant recent developments:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
HyGro,
Thanks for reosting your perspective again about the FDA and MHRA.
The DCVax-L cell-based technology has been under clinical investigation for a significant period of time, and the overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from trials with external controls and trials with placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The NIH is the most significant player in this game—If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












HyGro,
“Purgery” can only be found in a sugar factory.
Thank you for reposting that again, but the document you are referring to and your comments are outdated and out of context.
The NIH is the most significant player in this game if you are looking for “proof” or some kind of assurance. If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












Foogie88,
Congratulations on your long position! We are always happy to welcome a long investor who recognizes the value to humanity of the DCVax-L cell-based platform technology!
You are 100% correct—NWBO has committed a lot of diligent work and resources to build a hefty BLA that has been under construction for a significant period of time and likely includes a voluminous catalogue of clinical data.
Just like the JAMA Oncology independent peer review, the regulatory filings will be executed with precision to ensure a favorable outcome.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
BTW, have you met Poor Man? Any relation?
HyGro,
Thanks for reposting to ask about that again!
From my perspective, the NIH is the most significant player in this game if you are looking for “proof” or some kind of assurance as the NIH shares a common mission with the FDA. If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












aesop1,
Yes, NWBO is the “tortoise.”
The independent peer review took a long time too, but it was successfully completed and published in JAMA Oncology, and it is a solid asset as we move further into the regulatory process.
Slow and steady wins the race!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Grip,
Good point! There always are two sides and opposing opinions…
Everyone must do their own research to determine for themselves which direction we’re likely going. Seek trustworthy full-context sources, and verify information found posted online.




https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers


Exactly.

https://www.cancerresearchuk.org/about-cancer/find-a-clinical-trial/how-clinical-trials-are-planned-and-organised/how-long-it-takes-for-a-new-drug-to-go-through-clinical-trials
https://www.drugs.com/fda-approval-process.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
iclight,
That does not make sense. The Expanded Access Program is specifically for drugs that have not yet been evaluated for approval.
Additionally, the FDA approved the PD1 combo study to commence, and that study is ongoing at UCLA today.
https://www.fda.gov/news-events/public-health-focus/expanded-access
https://www.uclahealth.org/departments/neurosurgery/expanded-access-protocol-treatment-glioblastoma-multiforme
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Grip,
The company is still very small, but the leadership are formidable and tenacious. The independent peer review took a long time too, but it was successfully completed and published in JAMA Oncology, and it is an asset as we move further into the regulatory process.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
iclight,
Excellent point! The fact that he is alive to have additional resections is significant. Patients who enjoy extended survival have the chance to try any number of additional medical treatments or wellness therapies.
I agree with you about NWBO only offering compassionate care in the U.K. Specials Program. It needs to be more available globally! Stay tuned!


https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
iclight,
Great point! Thank you for reminding us about the brilliant compassionate use results. The compassionate use cases generated some astonishing data, and those data could be included in the regulatory filings!
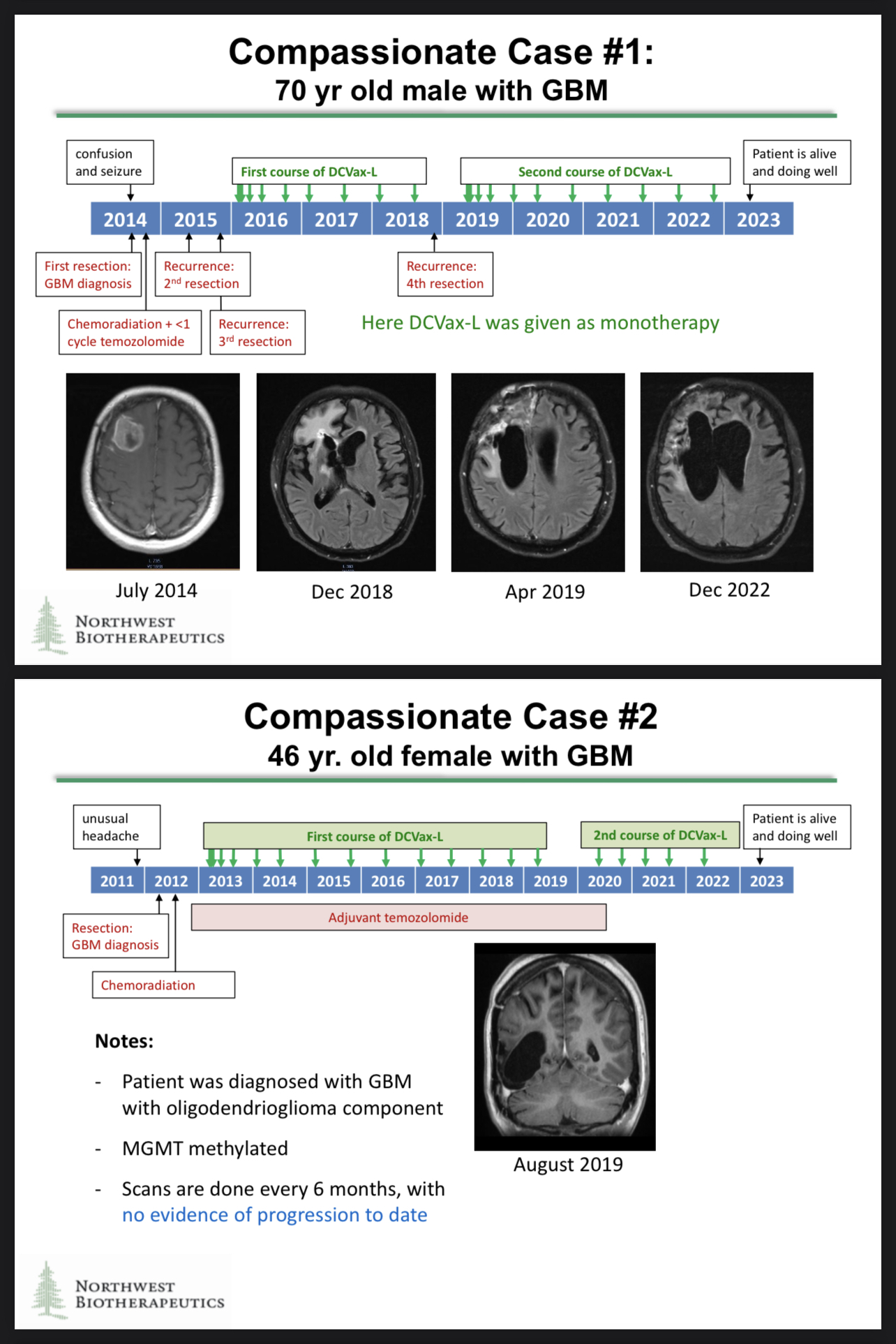
https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
HyGro,
Thanks for the reply and for reposting your perspective again. A little bit of research into the full picture can help to clear up the confusion. Here’s a quick review of some of the most relevant facts:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172139882
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172145669
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171927087
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171781313
exwannabe,
You raise a great point! When you’re looking at ECA data, you might wonder whether there would be proof of efficacy in a placebo study—you’re in luck! The DCVax-L cell-based technology has been under clinical investigation for a significant period of time using internal controls too! The overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from both external controls and placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
https://www.fda.gov/media/120721/download

https://clinicaltrials.gov/ct2/show/NCT04201873
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not proven its efficacy.

The P3 was brilliantly executed and is more than adequate all by itself, but the application(s) for approval will require lots of paper clips and plenty of staples for all the extra pages that will be included—there is a voluminous catalog of supportive data that has been collected, analyzed, and validated.
All clinical data are relevant to a regulatory application including those data gathered from other studies, interim analyses, and compassionate use. All the DCVax-L trials have produced substantial safety and efficacy data, and two of the three clinical trials ran for over a decade with survivors alive today.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
HyGro,
Thanks for reposting that again, but you are mixed up. Your information is out of context and outdated.
For information about the ECA/crossover design, endpoints, and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756
HyGro,
I forgot about that! That approval is quite significant for anyone who doubted the P3 design. Yes, the P3 methodology was approved for a future clinical trial!
“The primary endpoint for each of the 2 pediatric trials will be overall survival, determined by comparing the survival of DCVax-L treated patients to matched contemporaneous external controls. The external controls will be identified using the same methodology as was used to pre-specify the external controls in the Statistical Analysis Plan for the Company's Phase III trial in adult patients.”
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
Don’t forget that the MIA was also granted!


https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
aesop1,
I think you’re mixed up. If I recall, Provenge was approved but the company did not have the capacity to produce the product. NWBO does not have that problem—NWBO has planned well to expand and scale manufacturing capacity to serve a global market.
Further studies can and should be required after commercialization. For example, Keytruda was commercialized years ago, but clinical studies involving Keytruda have continued ever since and will continue into the future. Safety and efficacy data should always be gathered and new combinations should be tested and investigated.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172029305
HyGro,
Thanks for asking! From my perspective, the NIH is the most significant player in this game if you are looking for “proof” or some kind of assurance. If they had any doubt about the P3, the NIH would not continue their support and the FDA would not have permitted the combo trial to commence or continue.
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












aesop1,
Thanks for the post—manufacturing capacity and RA approval for manufacturing are crucial. NWBO is on top of it!
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171907111
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
jondoeuk,
Your assertion is not from a reliable or verifiable source. The posting from the other message board is not credible and cannot be verified by looking at it.
Here is crystal clear proof directly from UCLA…
The article below was published just yesterday by UCLA where Dr. Prins is employed. At least examine the portion that I have excerpted above the full-context link — it clearly verifies that DCVax-L is same vaccine that was published in JAMA Oncology and it is the same vaccine that is being studied in combination with PD-1 blockade (pembrolizumab). Read the clear print of the article for yourself.
The UCLA article also embeds the link—click on the PD-1 portion inside the article and it directs you to: https://clinicaltrials.gov/ct2/show/NCT04201873

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
HyGro,
Your post does not align with reality.
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756
jondoeuk,
Thanks for the reply. Your post is a distortion—it is missing crucial context and is misleading. The DCVax-L cell-based platform technology was analyzed by an independent peer review that was published in JAMA Oncology. Comments about the publication are not peer reviewed and are not part of the medical literature.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/
iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171785690
June 12, 2023
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
aesop1,
While you’re waiting, don’t forget about the significant regulatory approval that was recently achieved:
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038107
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172038243
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
aesop1,
Perhaps you will see the application submission(s) that you’ve been asking for much sooner than you seem to expect. The JAMA Oncology independent peer review took a long time too, but it was successfully completed and published.
June 12, 2023
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
jondoeuk,
Your information is incorrect about the DCVax-L cell-based platform technology. You need to seek better sources.
While I support the DCVax-L technology as an investor, I also hope for the success of NVCR’s technology, PD1 blockade, and various combo therapies.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171959537
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172099186


iclight,
You are wrong. I recommend doing some research.
Both Liau and Ashkan have been deeply involved in the evaluation of methods of practice of medicine and the analysis of SOC treatments approved by FDA and MHRA. Ashkan is an advisor to the UK government.
No single physician is solely responsible for any drug approval. Liau and Ashkan both have contributed to the peer review process, they both have led clinical studies and led clinical study units at their respective world-renowned institutions. Both have taught other physicians and supervised other physicians who have undertaken clinical investigations as part of their careers or in pursuit of licensing or board certification. Both have authored cutting-edge medical literature at length, and Liau was the editor-in-chief of a peer-reviewed oncology journal for a decade.
Not only have they submitted their own data, they have evaluated data collected and submitted by others at all stages of clinical investigations.
Arguably, both Liau and Ashkan are a part of the system that develops treatments, evaluates treatments, and approves treatments. Furthermore, both Liau and Ashkan have taught other doctors how to develop, evaluate, and approve treatments through their leadership, professorship, authorship, or by serving as advisors to others serving in leadership roles.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172110386
HyGro,
The P3 was led by clinical trial experts.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171927087
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171959537
jammy32,
Thanks for asking.
I think we are above 70, but that is not important. The forthcoming regulatory developments will move the needle. Commercialization and value measured in billions of dollars requires RA approval. The recent MIA signaled that the regulatory filing process has begun and has already resulted in a favorable outcome that foreshadows further developments.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171785690
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=172099186
iclight,
Congratulations on your profitable trades of NWBO!
I would argue that holding is a much smarter strategy. The CEO is very wise and wealthy and she is not a young or new business strategist. She is holding her roughly 130 million shares for commercialization. Day trading seems risky and short-sighted as we near further regulatory developments.
It’s not only more profitable to hold, it is much more fulfilling in my view. I am proud to be a small supportive part of this landmark cell-based platform technology that has already saved lives and will save countless more worldwide. Someday, this technology may play a part in saving my own life. Personally, I’m an investor in this technology for health and wealth.
Remember to always avoid online disinformation. Do your own research. Seek trustworthy full-context sources.
Regulatory developments will be the most determinative factor following the independent peer review, and NWBO is currently on the OTC market. PD1 combo data are also immensely significant and developments on that front will also prove consequential going forward.
The value of this cell-based immunotherapy platform technology will be determined by the integrity of the science, and the regulatory process will be the catalyst.
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
March 2023 Manufacturing News:

https://cms.mhra.gov.uk/mhra/mia/uk-mia-54923
The MIA is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://www.nice.org.uk/guidance/indevelopment/gid-ta10143
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/

HyGro,
You are mixed up. For information about the ECA/crossover design, endpoints, and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756
Dr. Bosch’s June 2023 presentation is now available online. It is another awesome source of information about the landmark P3 study of the DCVax-L cell-based platform technology.
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Always do your own careful research to verify online postings.
aesop,
That’s a great point! Just like Keytruda, DCVax-L will continue to be studied for other indications and in other combinations. Keytruda was approved and commercialized many years ago, but clinical studies involving Keytruda continue today and will continue into the future.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
iclight, #DCVax $nwbo #GBM #nwbo Immune System Researchers Win Nobel Prize in Medicine https://t.co/YW8un0Kujq #CancerResearch #Cancers #cancermoonshot #GBM #Immunotherapy #oncology $nwbo #murcidencel #DCVax #CellTherapy
Dr. Bosch’s presentation is now available online. It is an awesome source of information about the landmark P3 study of the DCVax-L cell-based platform technology.
https://twitter.com/mc17595091/status/1666528319209103361?s=46&t=BbaVVAHSNyBEm-BUTWiU6w
Dr. Steinman’s Nobel Prize-winning work lives on at UCLA!
“I will respect the hard-won scientific gains of those physicians in whose steps I walk, and gladly share such knowledge as is mine with those who are to follow.” pic.twitter.com/WLQ5KpdQqd
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Tell Jeannine about your opinion. This video is many years old. Today, she is celebrating 24 years of life since her GBM diagnosis!
https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
HyGro,
If you don’t like how the ECA was adapted into the trial, I recommend focusing on the DCVax-L trials that have a placebo design.
All of the most relevant context is missing from your curiously composed post, including the fact that the DCVax-L cell-based technology has been under clinical investigation for a significant period of time using internal controls too. The overall clinical data in total include three trials spanning many years. Safety and efficacy data have been gathered from both external controls and placebo controls. The data include DCVax-L as a monotherapy and in combination with other agents.
All data are relevant…
The reason clinical trials must be approved to commence and must be registered on the NIH registry is because all clinical data are relevant and no data pertaining to safety and efficacy can ever be unpublished or deleted from public view. We always must consider all of the data. For example, if a drug were found to be toxic in a certain patient population or in a certain combination with other agents or foods, those data could not be ignored just because they were collected in a separate study. Those data would have bearing on a regulatory application as well.
The PD1 combo trial is currently small but mighty. It is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding. It also follows the P3 in sequence which in itself is a fact that validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L if the preceding P3 had not proven its efficacy.
Regarding the ECA/crossover design and the SAP, please review the video recording of Dr. Ashkan at ASCO 2022 and the JAMA Oncology podcast recording of Dr. Liau. Both recordings are included at the links below.
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254048
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=171254756
Always do your own careful research to verify online postings.
iclight,
The recent presentation by Dr. Bosch is now available online if you are looking for an overview of the DCVax-L cell-based platform technology.

https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/

HyGro,
Your post is very misleading. The link below provides an overview of the landmark P3 results. There is also some information mentioned about the ongoing pembrolizumab combo study at UCLA.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
