Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Dr. Bruce Freimark (Res.Dir./Preclin.Oncology) speaks today: GTC/I-O/SanDiego
Feb6-7 2017: “GTCbio’s 9th Immunotherapeutics & Immunomonitoring Conf.”, SanDiego
“Leading scientific & clinical experts from academia and industry will present the latest findings and developments in the exciting area of immunotherapeutics & immunomonitoring. New immunological concepts with clinically translational implications will be discussed, along with evaluations of novel drug targets & therapeutic approaches, the role of biomarkers in immunomonitoring, and the latest data on combinatorial treatments in the clinic.”
https://www.gtcbio.com/conferences/immunotherapeutics-immunomonitoring-overview
AGENDA: https://www.gtcbio.com/conferences/immunotherapeutics-immunomonitoring-agenda
2-7-17 TRACK: Combination Immunotherapy
2:05pm: “Tipping the Balance: Overriding Phosphatidylserine-mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
Dr. Bruce Freimark (Res.Dir./Preclin.Oncology), Peregrine
Phosphatidylserine (PS) exposure in tumors induces non-inflammatory signals via multiple specific receptors for PS on tumors and immune cells, which contribute to immunosuppression in the tumor microenvironment. Antibody blockade of PS engages with innate immune responses and activates adaptive immune responses by promoting M1 macrophages, maturation of dendritic cells and tumor specific T-cell responses. Combinations of PS with CTLA-4, PD-1 and LAG-3 immune checkpoint antibodies have been shown to induce high levels of tumor regression and long-term anti-tumor immunity in models of breast and melanoma. These immune responses correlate with an overall increase in TILs, including CD8+ T cells with a reduction in suppressive Tregs and MDSCs, and tumor gene expression signatures indicating enhanced antigen presentation. Overall, PS blockade can complement immune checkpoint therapy by promoting a localized anti-tumor response and represents a promising strategy to enhance cancer immunotherapy.
Thx Jbain: 5-24-16/OncoTarget/Brekken/etal: “PS: the Target of Unbiasedly Selected Cancer Specific Peptide-Peptoid Hybrid PPS1”
5-24-16/OncoTarget: “Identification of Lipid-Phosphatidylserine (PS) as the Target of Unbiasedly Selected Cancer Specific Peptide-Peptoid Hybrid PPS1”
Tanvi J. Desai, Jason E. Toombs, John D. Minna, Rolf A. Brekken [PPHM SAB], Damith Gomika Udugamasooriya [MDA, UTSW, Univ. of Houston]
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5058709/
“There is an increasing need for anti-cancer agents that are effective against broad types of cancers, as the efficacy of protein targeted drugs are limited to certain subpopulations of cancer types due to the heterogeneous expressions of those protein drug targets. Phospholipid asymmetry and elevated PS levels is observed in the tumor microenvironments of most cancers analyzed to date. We propose that PPS1D1 may have efficacy in multiple tumor types and also has the potential to safely increase the efficacy of standard cancer therapy.”
**“This manuscript is dedicated to the memory of Dr. Philip E. Thorpe (1951–2013), who passed away in March 2013. Phil made significant contributions to cancer research over the course of his career and was instrumental in identifying PS as the target of PPS1 in this study.”
1-30-17/MolCancer: MDA/Brekken/etal, “PreClin. DNX2401+FhuBAVI Pancreatic”
1-30-17 Mol Cancer Therapy: “Preclinical Evaluation of Sequential Combination of Oncolytic Adenovirus Delta-24-RGD and Phosphatidylserine-targeting Antibody in Pancreatic Ductal Adenocarcinoma”
Dai B 1, Roife D 1, Kang Y 1, Gumin J 2, Rios Perez MV 1, Li X 1, Pratt M 1, Rolf Brekken [PPHM SAB] 3, Fueyo-Margareto J 4, Lang FF 2, Fleming JB 5
1 Dept of Surgical Oncology, Univ. of Texas MDA CC
2 Dept of Neurosurgery, Univ. of Texas MDA CC
3 Hamon Ctr for Therapeutic Oncology Res., Depts Surgery/Pharmacology, UTSW-MC
4 Dept of Neuro-Oncology, Univ. of Texas MDA CC
5 Dept of Surgical Oncology, Univ. of Texas MDA CC
https://www.ncbi.nlm.nih.gov/pubmed/28138026
ABSTRACT
Delta-24-RGD (DNX-2401) [DNXtrix Co.] is a conditional replication-competent oncolytic virus engineered to preferentially replicate in and lyse tumor cells with abnormality of p16/RB/E2F pathway. In a phase 1 clinical trial, Delta-24-RGD has shown favorable safety profile and promising clinical efficacy in brain tumor, which prompted us to evaluate its anticancer activity in pancreatic ductal adenocarcinoma (PDAC), which also has high frequency of homozygous deletion and promoter methylation of CDKN2A encoding the p16 protein. Our results demonstrate that Delta-24-RGD can induce dramatic cytotoxicity in a subset of PDAC cell lines with high Cyclin D1 expression. Induction of autophagy & apoptosis by Delta-24-RGD in sensitive PDAC cells was confirmed with LC3B-GFP autophagy reporter and Acridine Orange staining as well as Western blotting analysis of LC3B-II expression. Notably, we found that Delta-24-RGD induced phosphatidylserine (PS) exposure in infected cells independent of cells' sensitivity to Delta-24-RGD, which renders a rationale for combination of Delta-24-RGD viral therapy and PS targeting antibody for PDAC. In a mouse PDAC model derived from a liver metastatic pancreatic cancer cell line, Delta-24-RGD significantly inhibited tumor growth compared with control (p<0.001), and combination of PS targeting antibody 1N11 [Fully Human Bavituximab] further enhanced its anticancer activity (p<0.01) possibly through inducing synergistic anticancer immune responses. Given that these 2 agents are currently in clinical evaluation, our study warrants further clinical evaluation of this novel combination strategy in pancreatic cancer therapy.
-----------
NOTES:
1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
DNX-2401 (DNAtrix Co. http://www.DNAtrix.com ) is a potent oncolytic adenovirus that targets and kills cancer cells, while leaving normal cells intact. Multiple clinical studies in patients with recurrent glioblastoma and gynecologic cancer have shown that DNX-2401 has a favorable safety profile, strong tumor-killing potential and can trigger an anti tumor immune response. http://www.dnatrix.com/clinical-trials/
= = = = = = = = = = = = = = = =
I. Phase II Bavi+GEM vs. Front-Line Adv. PANCREATIC (randomized, unblinded, n=70)
Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01272791 (15 U.S. + 4 Ukraine = 19 as of 6-7-2012)
…1-22-13: FTM's post of 13 Ph3 Gem+Treatment Pancreatic Trials ('02-'13) - Mean MOS: GEM=6.4mos., GEM+TR=7.3mos. http://tinyurl.com/al99hx9
......Another FTM Pancreatic Phase3 trials table showing HR's, P-Values, and ORR% stats: http://tinyurl.com/btzkw4l
…6-3-13 ASCO’13/final data: n=70, Ctl=>Bavi MOS 5.2=>5.6mos, ORR 13%=>28% HR=.75 http://tinyurl.com/kq3uv4e
......Promising ‘immuno-indicative’ 1yr SURVIVAL results: GEM-Only(n=31): 0%, Bavi+GEM(n=32): 24.5% - see: http://tinyurl.com/lz5yg4f
...2-13-13 Topline Data: “Bavi+Gem resulted in more than a doubling of ORR” http://tinyurl.com/aqny7ny
...6-25-12: Enrollment complete. http://tinyurl.com/72tvnfj
...6-20-12: Early data (cutoff=6/6/12 bavi=15 ctl=17) presented at AACR Pancreatic Conf. http://tinyurl.com/77m9fw2
...1-5-11: U.S. Ph.2 randomized trial initiated http://tinyurl.com/26hnuzv "up to 70 front-line patients at ~10 clinical sites."
“NO INTENTION of conducting further [Exosomes] dev. work beyond the proof-of-concept stage...”
7-14-16-CC/J.Hutchins: “We have no intention of conducting further [Exosomes] development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this pgm in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.” http://tinyurl.com/jg2k8pt
7-14-16-CC/S.King: “Our goal is not to become a diagnostics company, but to put this [Exosomes] in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself.” http://tinyurl.com/jg2k8pt
1-22-17: 1-22-17/Oncotarget: “Detection Of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study” http://tinyurl.com/jg2k8pt
2014-2015 Exosomes Prep. Work by Philip.Thorpe/Alan.Schroit
Filed 2-27-15: Schroit/Thorpe “Isolating Exosomes” Patent App#20150241431
Title: “Methods & Compositions for Isolating Exosomes”
http://tinyurl.com/Schroit-Thorpe20150241431
ABSTRACT:
“Disclosed are surprising new methods & compositions for isolating extracellular microvesicles such as exosomes, particularly disease-related and phosphatidylserine (PS)-positive extracellular microvesicles as exemplified by tumor- and viral-derived exosomes. The methods of the invention are rapid, efficient, cost-effective and, importantly, are suitable for use with large volumes of biological fluids and produce antigenically intact extracellular microvesicles & exosomes. The methods & compositions are based on the surprising use of acetate buffers to isolate large quantities of extracellular microvesicles, particularly tumor-derived exosomes, from solution, without damaging their morphological or functional properties or antigenicity.”
Inventors: Alan J. Schroit; Philip E. Thorpe; Shelley Fussey
Applicant: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
May2014, J Immunol Methods, Thorpe/Schroit/etal, “A Novel ‘Salting-Out’ Procedure for the Isolation of Tumor-Derived Exosomes”
https://www.ncbi.nlm.nih.gov/pubmed/24735771
ABSTRACT
“The last decade has seen an exponential growth in the number of exosome-related publications. Although many of these studies have used exosomes from biological fluids (blood, and ascites or pleural effusions) the vast majority employed vesicles isolated from large volumes of tissue culture supernatants. While several techniques are available for their isolation, all require a significant reduction in volume to obtain sufficient concentrations for study. One approach is to concentrate the medium before proceeding with their isolation, however, these procedures are very time consuming and require specialized laboratory equipment. Here we provide a new & effective method for the isolation of tumor-derived exosomes based on "charge neutralization" with acetate. We show that titration of tissue culture supernatants with 0.1M acetate to pH4.75 results in immediate precipitation of virtually all the exosomes. The precipitated exosomes can be washed to remove residual media and are readily "resolubilized" upon resuspension in acetate-free buffer at neutral pH. This simple cost effective method significantly increases the yield of exosomes from an unlimited quantity of culture supernatants. Exosomes isolated by this technique are indistinguishable from exosomes recovered by direct ultracentrifugation.”
= = = = = = = = = =
PPHM’s EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/jg2k8pt
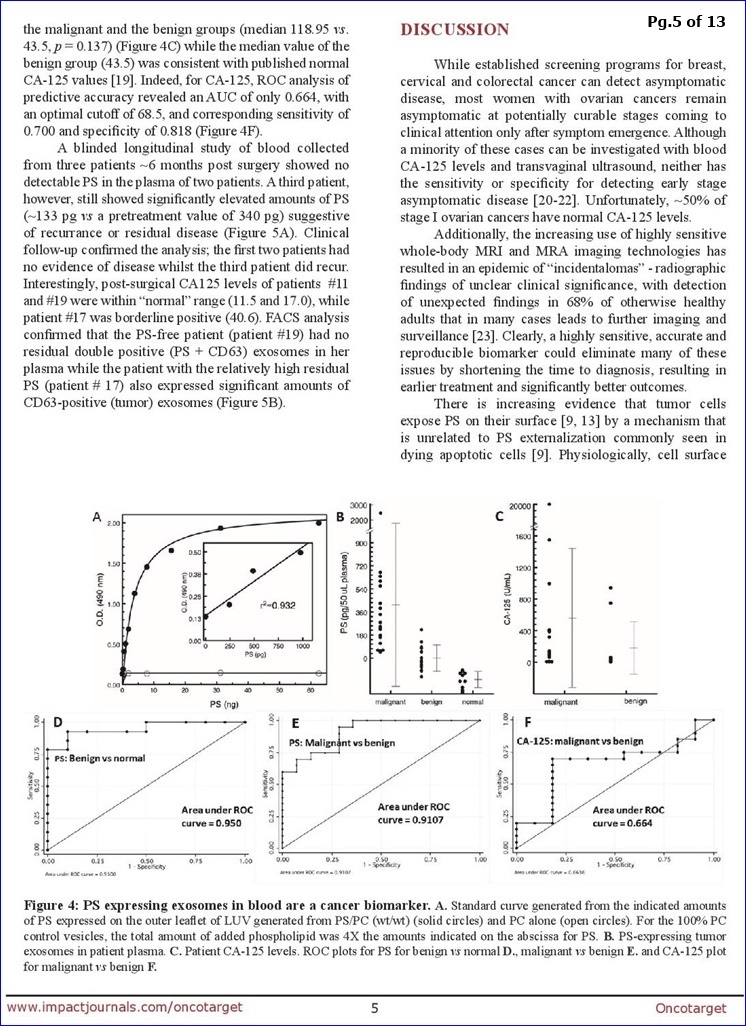
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
1-22-17/OncoTarget/PS-Exosomes: PPHM SAB’r Dr. Alan Schroit: Proof-of-Concept data supports the “high diagnostic power” of PS-Positive Exosomes in Ovarian Malignancies. ...Basically, Dr. Schroit (cell membrane guru) & his UTSW team used a variant of fully-human Bavi (1N11) to detect the PS on the exosomes…
FROM PG.8: “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
**NOTE: “PPHM’s PS+ Exosomes Pgm – known history to date” section added at bottom.
1-22-17 Oncotarget: “Detection Of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study”
Lea J 1, Sharma R 2, Yang F 2, Zhu H 1, Ward ES 3,4, Alan J. Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/jlrkxma ) 1,3
1 Harold Simmons Comprehensive CC, UTSW-MC/Dallas
2 Hamon Center for Therapeutic Oncology Res., UTSW-MC/Dallas
3 Dept of Immunology, UTSW-MC/Dallas
4 Dept of Molecular & Cellular Medicine, Texas A&M Univ. Health Science Ctr
https://www.ncbi.nlm.nih.gov/pubmed/28122335
ABSTRACT
There are no suitable screening modalities for ovarian carcinomas (OC) and repeated imaging and CA-125 levels are often needed to triage equivocal ovarian masses. Definitive diagnosis of malignancy, however, can only be established by histologic confirmation. Thus, the ability to detect OC at early stages is low, and most cases are diagnosed as advanced disease. Since tumor cells expose phosphatidylserine (PS) on their plasma membrane, we predicted that tumors might secrete PS-positive exosomes into the bloodstream that could be a surrogate biomarker for cancer. To address this, we developed a highly stringent ELISA that detects picogram quantities of PS in patient plasma. Blinded plasma from 34 suspect ovarian cancer patients and 10 healthy subjects were analyzed for the presence of PS-expressing vesicles. The nonparametric Wilcoxon rank sum test showed the malignant group had significantly higher PS values than the benign group (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups had significantly higher PS values than the healthy group (median 0.237 vs -0.158, p<0.0001 and -0.027 vs -0.158, p=0.0002, respectively). ROC analysis of the predictive accuracy of PS-expressing exosomes/vesicles in predicting malignant against normal, benign against normal & malignant against benign revealed AUCs of 1.0, 0.95 and 0.911, respectively. This study provides proof-of-concept data that supports the high diagnostic power of PS detection in the blood of women with suspect ovarian malignancies.
FULL ARTICLE (13pgs):
HTML: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=14795&path%5B%5D=47251
PDF: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=download&path%5B%5D=14795&path%5B%5D=47248
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
PDF EXCERPTS 1-22-17/OncoTarget/A.Schroit/PPHM-Exosomes (5 pgs of the 13):




= = = = = = = = = = = = = = = = = = =PPHM’s PS+ Exosomes Pgm – known history to date:
EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
7-14-16/PR: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
7-14-16/CC S.King PREPARED: “...In addition to bavituximab program which continues to have tremendous potential value, the company announced earlier today that it has licensed-in a Novel PS Exosome Technology, with the potential to detect & monitor cancer at an early stage through a simple blood test [ 7-14-16: http://tinyurl.com/zszd4fj ]. I recognize that some of you might think this theme is contrary to controlling spending as we move toward profitability, but in fact we believe that given our already existing knowledge base in Targeting PS and our already available infrastructure for developing & validating tests, that so a very modest capital investment we can quickly reach proof-of-concept with a goal of partnering this technology, which could then bring in addl. revenue, with another potential upside of the technology being that it can possibly be useful in the continued dev. of bavituximab. Jeff will talk more about this during his prepared remarks. Given the strategy of R&D targeted toward early partnering, will allow the company to continue its R&D activities with significant upside coming from partnering as we move to our profitability.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Hutchins PREPARED: “I'm very happy to be able to discuss a number of exciting developments in our preclinical group. First, as Steve discussed, we have executed the licensing agreement with UT Southwestern Medical Center, for novel exosome technology [ 7-14-16: http://tinyurl.com/zszd4fj ] . While many of you are familiar with exosomes, I’ll provide a brief overview for those who are not. Exosomes are cell-secreted vesicles, or mini-cells if you will, that are present in nearly all bodily fluids, including blood. Likewise, tumor-derived exosomes represent small pieces of tumor cells that are released into the blood as tumors grow. As well, these tumor-derived exosomes have Phosphatidylserine or PS on their surface as a marker and can also contain DNA, RNA and proteins as markers of malignant disease. It is believed that even small tumors begin to release PS-positive exosomes, and thus the ability to detect these exosomes in the blood may be an indicator of presence or progression of a tumor. The licensed technology is designed to detect & monitor PS-positive exosomes in a patient's blood sample, providing clinicians with detection & monitoring information regarding the presence & prevalence of cancer. These exosomes have PS flipped to the outside of the surface and demonstrate immunosuppressive activity, just as we find with tumor cells. Preliminary studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS-positive exosomes that are detected in the blood of cancer patients and the severity or extent of their disease burden. Given our in-house expertise in PS-targeting, we believe that we are uniquely qualified to advance this technology. As Steve stated, there are significant opportunities to use this technology as both a complimentary tool in bavituximab's ongoing development, which Joe will address later, as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of our partnering efforts. It is our goal to develop, optimize, and validate a functional detection & monitoring assay capable of detecting PS-positive exosomes from a simple blood sample, and, given the company's extensive experience in developing assays of this type, we do not anticipate the need for added personnel or any specialized equipment for this project. Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.”
...SUMMARY: “And lastly, we've in-licensed a new exosome technology for a minimal cost that leverages our existing in-house expertise and provides us with another opportunity for us to create value to product development. Together, we believe the strategy will provide success, as it will allow us to focus the majority of our resources on achieving our primary corporate goal, future sustainable profitability within 24mos. At same time, we will focus our R&D efforts on small early stage trials and development of the exosome technology in an effort to attract partners. We believe this strategy will allow us to build near-term revenues through Avid, while maintaining the potential for significant addl. value creation associated with our R&D efforts.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Shan PREPARED: “I'd first like to comment on our new exosome program. One of the most exciting aspects of this technology is the potential synergy that it offers with our bavituximab clinical dev. program. Through our ongoing work with bavituximab, we have gained significant understanding of PS-mediated immuno-suppression in cancer. The availability of the PS specific biomarker, which can be implemented in our planned future bavituximab clinical trials, aligns nicely with our refocused bavituximab dev. strategy aimed at generating the most meaningful data possible from small, early stage clinical trials to support partnering efforts. We are very anxious to bring this new technology to Peregrine and we look forward to the value it brings to our bavituximab program.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/Pantginis – SK: “(On the Exosome pgm), …it fits right in; we’re not having to hire addl. people, we’re not having to bring in addl. equipment. This really fits in with everything we are doing on both studying PS, as well as on the assay development side of the business. We think it’s actually going to be complementary. There are other technologies out there looking at exosomes; they're are all taking a very different approach to what we’re doing and we actually think they could be very complementary to each other. We also see a need, even as interest in exosomes begins to pick up, to actually utilize this in conjunction with other things that are in development.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/T.Yip – SK: “(On the Exosome pgm), the next step will be to validate that through patient samples. The beauty of this is that while you do need IRB approval of course, you’re not running really clinical trials, so this can be done in conjunction with either our ongoing trials or partners' trials, or there are many other sources of just receiving these types of blood samples. This gives us the ability to very quickly go through and test hundreds or thousands of patient samples as part of the validation process. At that point, we can zero in on what are the potential applications of the technology, outside of what we might do with our own PS Targeting programs – what would be the potential utility of this for patients. Our goal is not to become a diagnostics company, but to put this in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself. Our benefit at that point would become, hopefully, some residual royalties, milestones and what have you, which feeds back into our revenue goals of becoming profitable. So, we thought that this is a very attractive technology that just fits right in with what we’re doing and requires almost no addl. resources whatsoever…
Q2:/TY: ”when should we expect to see more preclinical data on this front?”… SK: “Our goal is, probably towards for the end of this year, to be in a position to have data that we can present. That will come in a lot of different formats, so it will be in conjunction with other ongoing studies that maybe taking place already to be standalone just on the diagnostic itself. So, you'll be hearing a lot about this, and one of the reasons when to get this news out there is because what you think sooner than later we will build a talk about this technology.” http://tinyurl.com/h8eqtg5
9-8-16/PR: “Peregrine in-licensed a novel exosome technology from UTSW that has potential for cancer detection and monitoring applications. This technology aligns directly with the company's expertise, its proprietary PS-targeting platform and the bavituximab development program. As such, there are opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts.” http://tinyurl.com/jydtkoy
9-8-16/CC J.Hutchins PREPARED: “1st, an an update on our PS Exosome Program. As we announced in July, we executed a licensing agreement with UT Southwestern Medical Center for a novel exosome technology designed to detect PS-positive exosomes in a patient blood sample. It is our belief that this technology can provide clinicians with detection & monitoring information regarding the presence and the prevalence of cancer. Preliminary indep. studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS positive exosomes detected in the blood of cancer patients and the severity and extent of their disease burden. Given our in-house expertise in PS targeting, we believe that we are uniquely qualified to advance this technology. We believe there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of partnering efforts. It is our goal to develop, optimize and validate a functional screening assay capable of detecting PS positive exosomes in a blood sample and to initiate partnering discussions for commercialization of the pgm in 2017. We are on track to achieve this goal and we look forward to providing further updates.” http://tinyurl.com/jydtkoy
10-13-16/ASM REPORT BY ATTENDEE COPPER888: “...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...” http://tinyurl.com/jx7ouay

12-12-16/CC: No mention of Exosomes! http://tinyurl.com/hhn4gga
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/jg2k8pt
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
PPHM’s PS+ Exosomes Pgm – known history to date
EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy")
...Excellent Exosome (aka microparticles, microvesicles) info: http://www.exosome-rna.com
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
7-14-16/CC S.King PREPARED: “...In addition to bavituximab program which continues to have tremendous potential value, the company announced earlier today that it has licensed-in a Novel PS Exosome Technology, with the potential to detect & monitor cancer at an early stage through a simple blood test [ 7-14-16: http://tinyurl.com/zszd4fj ]. I recognize that some of you might think this theme is contrary to controlling spending as we move toward profitability, but in fact we believe that given our already existing knowledge base in Targeting PS and our already available infrastructure for developing & validating tests, that so a very modest capital investment we can quickly reach proof-of-concept with a goal of partnering this technology, which could then bring in addl. revenue, with another potential upside of the technology being that it can possibly be useful in the continued dev. of bavituximab. Jeff will talk more about this during his prepared remarks. Given the strategy of R&D targeted toward early partnering, will allow the company to continue its R&D activities with significant upside coming from partnering as we move to our profitability.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Hutchins PREPARED: “I'm very happy to be able to discuss a number of exciting developments in our preclinical group. First, as Steve discussed, we have executed the licensing agreement with UT Southwestern Medical Center, for novel exosome technology [ 7-14-16: http://tinyurl.com/zszd4fj ] . While many of you are familiar with exosomes, I’ll provide a brief overview for those who are not. Exosomes are cell-secreted vesicles, or mini-cells if you will, that are present in nearly all bodily fluids, including blood. Likewise, tumor-derived exosomes represent small pieces of tumor cells that are released into the blood as tumors grow. As well, these tumor-derived exosomes have Phosphatidylserine or PS on their surface as a marker and can also contain DNA, RNA and proteins as markers of malignant disease. It is believed that even small tumors begin to release PS-positive exosomes, and thus the ability to detect these exosomes in the blood may be an indicator of presence or progression of a tumor. The licensed technology is designed to detect & monitor PS-positive exosomes in a patient's blood sample, providing clinicians with detection & monitoring information regarding the presence & prevalence of cancer. These exosomes have PS flipped to the outside of the surface and demonstrate immunosuppressive activity, just as we find with tumor cells. Preliminary studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS-positive exosomes that are detected in the blood of cancer patients and the severity or extent of their disease burden. Given our in-house expertise in PS-targeting, we believe that we are uniquely qualified to advance this technology. As Steve stated, there are significant opportunities to use this technology as both a complimentary tool in bavituximab's ongoing development, which Joe will address later, as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of our partnering efforts. It is our goal to develop, optimize, and validate a functional detection & monitoring assay capable of detecting PS-positive exosomes from a simple blood sample, and, given the company's extensive experience in developing assays of this type, we do not anticipate the need for added personnel or any specialized equipment for this project. Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical & clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage. Rather, we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we'll have more details to offer in the coming months.”
...SUMMURY: “And lastly, we've in-licensed a new exosome technology for a minimal cost that leverages our existing in-house expertise and provides us with another opportunity for us to create value to product development. Together, we believe the strategy will provide success, as it will allow us to focus the majority of our resources on achieving our primary corporate goal, future sustainable profitability within 24mos. At same time, we will focus our R&D efforts on small early stage trials and development of the exosome technology in an effort to attract partners. We believe this strategy will allow us to build near-term revenues through Avid, while maintaining the potential for significant addl. value creation associated with our R&D efforts.” http://tinyurl.com/h8eqtg5
7-14-16/CC J.Shan PREPARED: “I'd first like to comment on our new exosome program. One of the most exciting aspects of this technology is the potential synergy that it offers with our bavituximab clinical dev. program. Through our ongoing work with bavituximab, we have gained significant understanding of PS-mediated immuno-suppression in cancer. The availability of the PS specific biomarker, which can be implemented in our planned future bavituximab clinical trials, aligns nicely with our refocused bavituximab dev. strategy aimed at generating the most meaningful data possible from small, early stage clinical trials to support partnering efforts. We are very anxious to bring this new technology to Peregrine and we look forward to the value it brings to our bavituximab program.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/Pantginis – SK: “(On the Exosome pgm), …it fits right in; we’re not having to hire addl. people, we’re not having to bring in addl. equipment. This really fits in with everything we are doing on both studying PS, as well as on the assay development side of the business. We think it’s actually going to be complementary. There are other technologies out there looking at exosomes; they're are all taking a very different approach to what we’re doing and we actually think they could be very complementary to each other. We also see a need, even as interest in exosomes begins to pick up, to actually utilize this in conjunction with other things that are in development.” http://tinyurl.com/h8eqtg5
7-14-16/CC Q&A/T.Yip – SK: “(On the Exosome pgm), the next step will be to validate that through patient samples. The beauty of this is that while you do need IRB approval of course, you’re not running really clinical trials, so this can be done in conjunction with either our ongoing trials or partners' trials, or there are many other sources of just receiving these types of blood samples. This gives us the ability to very quickly go through and test hundreds or thousands of patient samples as part of the validation process. At that point, we can zero in on what are the potential applications of the technology, outside of what we might do with our own PS Targeting programs – what would be the potential utility of this for patients. Our goal is not to become a diagnostics company, but to put this in the hands of a good organization that's already established in the diagnostics area and then have them finish up the commercialization and expansion of the utility of the actual assay itself. Our benefit at that point would become, hopefully, some residual royalties, milestones and what have you, which feeds back into our revenue goals of becoming profitable. So, we thought that this is a very attractive technology that just fits right in with what we’re doing and requires almost no addl. resources whatsoever…
Q2:/TY: ”when should we expect to see more preclinical data on this front?”… SK: “Our goal is, probably towards for the end of this year, to be in a position to have data that we can present. That will come in a lot of different formats, so it will be in conjunction with other ongoing studies that maybe taking place already to be standalone just on the diagnostic itself. So, you'll be hearing a lot about this, and one of the reasons when to get this news out there is because what you think sooner than later we will build a talk about this technology.” http://tinyurl.com/h8eqtg5
9-8-16/PR: “Peregrine in-licensed a novel exosome technology from UTSW that has potential for cancer detection and monitoring applications. This technology aligns directly with the company's expertise, its proprietary PS-targeting platform and the bavituximab development program. As such, there are opportunities to use this technology as both a complementary tool in bavituximab's ongoing development, as well as more broadly as the basis for novel cancer detection & monitoring tests that can be the focus of partnering efforts.” http://tinyurl.com/jydtkoy
9-8-16/CC J.Hutchins PREPARED: “1st, an an update on our PS Exosome Program. As we announced in July, we executed a licensing agreement with UT Southwestern Medical Center for a novel exosome technology designed to detect PS-positive exosomes in a patient blood sample. It is our belief that this technology can provide clinicians with detection & monitoring information regarding the presence and the prevalence of cancer. Preliminary indep. studies have demonstrated that the levels of PS-positive exosomes present in the blood of cancer patients are higher than levels found in the blood of healthy volunteers. Furthermore, study findings also suggest that there is a correlation between the level of PS positive exosomes detected in the blood of cancer patients and the severity and extent of their disease burden. Given our in-house expertise in PS targeting, we believe that we are uniquely qualified to advance this technology. We believe there are significant opportunities to use this technology as both a complementary tool in bavituximab's ongoing development as well as more broadly as the basis for a novel cancer detection & monitoring test kit that will be the focus of partnering efforts. It is our goal to develop, optimize and validate a functional screening assay capable of detecting PS positive exosomes in a blood sample and to initiate partnering discussions for commercialization of the pgm in 2017. We are on track to achieve this goal and we look forward to providing further updates.” http://tinyurl.com/jydtkoy
10-13-16/ASM REPORT BY ATTENDEE COPPER888: “...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...” http://tinyurl.com/jx7ouay

12-12-16/CC: No mention of Exosomes!
1-22-17: OncoTarget article on PS-Exosomes: PPHM SAB’r Dr. Alan Schroit (UTSW): Proof-of-Concept data http://tinyurl.com/hv8dcpd
...Data (blinded plasma from 34 O.C. pts & 10 healthy subjects) supports the “high diagnostic power” of PS+ Exosomes in Ovarian Malignancies. “The data (Fig.4) show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
1-22-17/OncoTarget/PS-Exosomes: PPHM SAB’r Dr. Alan Schroit: Proof-of-Concept data supports the “high diagnostic power” of PS-Positive Exosomes in Ovarian Malignancies. ...Basically, Dr. Schroit (cell membrane guru) & his UTSW team used a variant of fully-human Bavi (1N11) to detect the PS on the exosomes…
FROM PG.8: “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies.”
1-22-17 Oncotarget: “Detection Of Phosphatidylserine-Positive Exosomes as a Diagnostic Marker for Ovarian Malignancies: A Proof of Concept Study”
Lea J 1, Sharma R 2, Yang F 2, Zhu H 1, Ward ES 3,4, Alan J. Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/jlrkxma ) 1,3
1 Harold Simmons Comprehensive CC, UTSW-MC/Dallas
2 Hamon Center for Therapeutic Oncology Res., UTSW-MC/Dallas
3 Dept of Immunology, UTSW-MC/Dallas
4 Dept of Molecular & Cellular Medicine, Texas A&M Univ. Health Science Ctr
https://www.ncbi.nlm.nih.gov/pubmed/28122335
ABSTRACT
There are no suitable screening modalities for ovarian carcinomas (OC) and repeated imaging and CA-125 levels are often needed to triage equivocal ovarian masses. Definitive diagnosis of malignancy, however, can only be established by histologic confirmation. Thus, the ability to detect OC at early stages is low, and most cases are diagnosed as advanced disease. Since tumor cells expose phosphatidylserine (PS) on their plasma membrane, we predicted that tumors might secrete PS-positive exosomes into the bloodstream that could be a surrogate biomarker for cancer. To address this, we developed a highly stringent ELISA that detects picogram quantities of PS in patient plasma. Blinded plasma from 34 suspect ovarian cancer patients and 10 healthy subjects were analyzed for the presence of PS-expressing vesicles. The nonparametric Wilcoxon rank sum test showed the malignant group had significantly higher PS values than the benign group (median 0.237 vs. -0.027, p=0.0001) and the malignant and benign groups had significantly higher PS values than the healthy group (median 0.237 vs -0.158, p<0.0001 and -0.027 vs -0.158, p=0.0002, respectively). ROC analysis of the predictive accuracy of PS-expressing exosomes/vesicles in predicting malignant against normal, benign against normal & malignant against benign revealed AUCs of 1.0, 0.95 and 0.911, respectively. This study provides proof-of-concept data that supports the high diagnostic power of PS detection in the blood of women with suspect ovarian malignancies.
FULL ARTICLE (13pgs):
HTML: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=14795&path%5B%5D=47251
PDF: http://www.impactjournals.com/oncotarget/index.php?journal=oncotarget&page=article&op=download&path%5B%5D=14795&path%5B%5D=47248
EXCERPTS FROM 1-22-17 ONCOTARGET ARTICLE:
**From DISCUSSION (pg. 8): “The data summarized in Fig. 4 show that quantification of PS-exosomes in blood distinguishes, with 100% accuracy, healthy tumor-free individuals from patients with ovarian malignancies… In summary, this study provides proof-of-concept data that supports the high diagnostic power of PS-expressing tumor exosome detection in blood from women with suspect ovarian malignancies. Ultimately, these studies could lead to earlier stage diagnosis, substantial cost savings, reduced patient exposure to radiation and invasive procedures, and improved clinical outcomes. The assay might also find utility in patients with radiographic abnormalities, even before clinical detection. Indeed, an accurate biomarker predicting the likelihood of malignancy would be extremely beneficial to such a population since they often face long periods of anxiety and uncertainty inherent to a “wait & watch” approach. Finally, if PS-exosome diagnostics are confirmed in a large study to be an accurate and reproducible biomarker of ovarian malignancies, the assay could be applied to the early detection of other visceral malignancies."
**From METHODS (pg.9): “Expression of an engineered tetravalent antibody for PS-detection Monoclonal 1N11 is a human IgG1^ that binds PS through the PS-specific plasma protein B2GP1. A tetravalent variant of 1N11 (1N11-T), with 4 binding sites per molecule was designed to generate a high avidity PS binding agent (Fig.1).”
……...[NOTE: 1N11 is Fully-Human Bavituximab (aka PGN635=AT004), B2GPI-dep. Binding]
**From FINANCIAL SUPPORT (pg.11): “Supported by Cancer Prevention & Res. Inst. of TX (CPRIT) Grant #RP110441, and a Simmons CC Support Grant 5P30 CA142543.”
**EDITORIAL NOTE (pg.11): “This paper has been accepted based in part on peer-review conducted by another journal and the authors’ response & revisions as well as expedited peer-review in Oncotarget."
= = = = = = =
EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") - Excellent Exosome info: http://www.exosome-rna.com
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”
10-13-16 ASM REPORT BY ATTENDEE COPPER888: http://tinyurl.com/jx7ouay
“...I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there...”
PDF EXCERPTS 1-22-17/OncoTarget/Schroit/PPHM-Exosomes (5 pgs of the 13):





Loof, Westy got his 47-1 $$$, HL45 his 35-1 $$$ 4wks ago, I got my CU NC’16, so now all attention turns to YOU in ‘17: UL/NC’17 AND PPHM SP turnaround. Those are my wishes for you (and pt.B for All Longs). ...Let Loof’s Pure Kentucky Korn Licker FLOW bigtime in ‘17!
I'm with you, Westy! This little company is going to Win before the final chapter is written. Good luck All Longs!
Pipeline Chart on website upd. 1-17-17: AZN entry changed (since 10-13-16 ASM Pipeline chart) from “Mult. Solid Tumors: Combo with durvalumab(anti-PD-L1) + Chemo” to “Mult. Solid Tumors: Combo with durvalumab(anti-PD-L1)”. Ie, “+ Chemo” dropped.
http://www.peregrineinc.com/pipeline/overview.html

- - - - - - - - -
10-13-16 Peregrine's ASM: ATTENDEE Reports & Link to CEO Steve King's 35min/45slide webcast: http://tinyurl.com/jx7ouay
CEO Steve King speaking 1-18-17 at Immunotherapy World Conf./Miami (replacing Jeff Hutchins)…
Jan17-20 2017: “Phacilitate's Immunotherapy World Conf.”, Miami
”Meet the future of immuno-oncology - Combining science & partnering. Join 850+ senior cell, gene, and immunotherapy executives in Miami for partnering & business opportunities.
http://www.immunotherapyforum.com
Agenda: http://www.immunotherapyforum.com/immunotherapy-world-2017-agenda
Jan18(Day1):
Focus Session2, “IMMUNOTHERAPY: EXTENDING ITS REACH, PREDICTING RESPONSES”
1-18-17 12:30-12:45pm: Stephen King (Pres&CEO, Peregrine), “Turning up the Heat: Phosphatidylserine-Targeting Antibodies Modulate the Tumor Microenvironment & Enhance Checkpoint Blockade”

Known Upcoming Events
Jan17-20: Phacilitate's Immunotherapy World Conf., Miami http://tinyurl.com/zzdbzst
...Jan18 12:30-12:45pm: Steve King (CEO), “Turning up the Heat: PS-Targeting Antibodies Modulate the Tumor Microenvironment & Enhance Checkpoint Blockade”
Jan24-26/Avid/Table#22: WCBP’17: 21st Symposium on Interface of Reg. & Analytical Sciences, WashDC http://www.casss.org/page/WCBP1700 (Theme: “Convergence & Harmonization”)
Feb6-7: GTCbio’s Immunotherapeutics & Immunomonitoring Conf., SanDiego
https://www.gtcbio.com/conferences/immunotherapeutics-immunomonitoring-overview
...AGENDA: http://tinyurl.com/gmrlupq
...Feb7 2:05pm: Dr. Bruce Freimark(Res.Dir./Preclin.Oncology), “Blockade of PS Enhances the Anti-Tumor Activity of Immune Checkpoint Inhibitors by Induction of Adaptive Immune Responses Mechanism”
Feb20-22: CHI’s 5th Translational Models in Oncology & I-O, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Feb21 11:15-11:45am: Michael Gray(Sr.Res.Sci.), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
??Apr1-5: AACR 2017, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins***(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
***Note: 1-4-17: Dr. Hutchins left Peregrine to become CSO of Heat-Biologics.
??Jun2-6: ASCO 2017, Chicago https://am.asco.org
June12-17: CHI’s 2nd Annual Immunotherapy & Combinations Conf., Boston http://www.worldpreclinicalcongress.com/Cancer-Immunotherapy-Combinations
...Dr. Jeff Hutchins***(VP/PreClinRes.): “Expansion & Activation of T-Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
***Note: 1-4-17: Dr. Hutchins left Peregrine to become CSO of Heat-Biologics.
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Stephen Worsley(VP/BusDev) has not “been moved” from Peregrine to Avid. No change in the Mgt.Team listings at http://www.peregrineinc.com/about-us/management-team.html and http://avidbio.com/team . King, Lytle, Worsley, Garnick are listed the same on BOTH the Peregrine & Avid websites, as they have all along. There’s been no change.
CHI/I-O 2-21-17 Talk switched from JH to Dr.Michael.Gray, Senior Research Scientist at Peregrine. I actually noticed this chg. several days ago, didn’t think much of it, and had it queued to update iBox the next time I update.
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & Immuno-Oncology”, SanFran
“Models & Approaches to Bring Combination Therapies to the Clinic”
Part of the 24th Intl. Molecular Medicine Tri-Conference
http://www.triconference.com/Pre-Clinical-Oncology-Models
CHI = Cambridge Healthtech Institute http://www.healthtech.com
Track: TUMOR MODELS FOR CANCER IMMUNOTHERAPY
2-21-17 11:15-11:45am: Dr. Michael Gray (Sr.Res.Scientist), Peregrine Pharmaceuticals - “Case Study: Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
SUMMARY:
Phosphatidylserine (PS) exposure in tumors induces non-inflammatory signals which contribute to an immunosuppressive environment. Antibody blockade of PS activates immune responses by promoting M1 macrophages, maturation of dendritic cells and inducing adaptive T-cell responses. PS targeting antibodies enhance the anti-tumor activity of checkpoint antibodies in preclinical tumor models.
= = = = = = = =Recall:
9-27-16 AACR-CRI/Dr. Michael Gray: Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells)
http://tinyurl.com/zy9yv78
“...the presentation of preclinical study data demonstrating that phosphatidylserine (PS)-targeting antibodies similar to bavituximab are able to enhance the anti-tumor activity of multiple checkpoint targeting agents including anti-PD-1 & anti-LAG3 therapies in a model of triple negative breast cancer (TNBC). Data showed that 8 of the 10 (80%) animals receiving the preclinical bavituximab equivalent (ch1N11) combined with anti-PD-1 and anti-LAG3 therapies ("Triple Combination") experienced complete tumor regressions, whereas there were no animals (0/10) in the anti-PD-1 & anti-LAG3 combination treatment arm that had a complete regression. Addl. data demonstrated that the Triple Combination featuring ch1N11 led to a 99% reduction in total tumor volume at the interim analysis point (Day 25) across all animals as compared to the control arm. In addition, the Triple Combination showed a statistically significant increase in tumor growth inhibition (TGI) as compared to the anti-PD-1 & anti-LAG3 combination treatment (99% vs. 62%; p < 0.05). Peregrine's Michael J. Gray, PhD, the study's lead scientist, presented the study findings at the 2nd CRI-CIMT-EATI-AACR Intl. Cancer Immunotherapy Conf. Sept. 25-28, 2016, in NYC.”
**DR. MICHAEL GRAY: https://www.linkedin.com/in/michael-gray-18447328 “Principal Scientist, joined Peregrine 12-2014, prev=OSI, Educ=MDA"
Dr. Bruce Freimark(PhD) is currently Research Director, Preclinical-Oncology for Peregrine Pharmaceuticals. He joined PPHM in 4-2006.
2-7-17/GTC I-O Conf: “Dr. Bruce Freimark, PhD. is currently Research Director, Preclinical Oncology for Peregrine Pharmaceuticals. He is responsible for preclinical studies focusing on the Peregrine’s lead technology involving immune-oncology therapeutics targeting the membrane lipid phosphatidylserine. Previous to Peregrine has held mgt. positions for the past 25 years in the pharmaceutical & biotechnology industry in the areas of cancer & inflammation at CancerVax, Nexell Therapeutics, Valentis, and Dupont-Merck. Dr. Freimark received his Ph.D. in Pathology at SUNY/Buffalo and post-doctoral training at Scripps Clinic & Research Foundation, La Jolla, CA”
https://www.gtcbio.com/preview/immunotherapeuticssummit/bios/feimark_bruce.pdf
NOTE that he & Dr. Hutchins basically shared Bavi I-O Conference presentation duties…
UPCOMING...
Jan24-26/Avid/Table#22: WCBP’17: 21st Symposium on Interface of Reg. & Analytical Sciences, WashDC http://www.casss.org/page/WCBP1700 (Theme: “Convergence & Harmonization”)
Jan17-20: Phacilitate's Immunotherapy World Conf., Miami http://www.immunotherapyforum.com
...Jan18 12:25-12:40pm: Dr. Jeff Hutchins (VP/PreClinRes), “Combination Immunotherapies - Opening the Gate: Increasing Tumor Infiltrating Activated T-Cells to Optimize & Expand the Benefits of Immune Checkpoint Therapies”
Feb6-7: GTCbio’s Immunotherapeutics & Immunomonitoring Conf., SanDiego
https://www.gtcbio.com/conferences/immunotherapeutics-immunomonitoring-overview
...AGENDA: http://tinyurl.com/gmrlupq
...Feb7 2:05pm: Dr. Bruce Freimark(Res.Dir./Preclin.Oncology), “Blockade of PS Enhances the Anti-Tumor Activity of Immune Checkpoint Inhibitors by Induction of Adaptive Immune Responses Mechanism”
Feb20-22: CHI’s 5th Translational Models in Oncology & I-O, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
??Apr1-5: AACR 2017, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
??Jun2-6: ASCO 2017, Chicago https://am.asco.org
June12-17: CHI’s 2nd Annual Immunotherapy & Combinations Conf., Boston http://www.worldpreclinicalcongress.com/Cancer-Immunotherapy-Combinations
...Dr. Jeff Hutchins(VP/PreClinRes.): “Expansion & Activation of T-Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
Jeff Hutchins(VP/PreClinRES) leaves Peregrine for CSO@Heat-Biologics
1-4-16: ...“Most recently, Dr. Jeff Hutchins served as VP of Preclinical Research for Peregrine Pharmaceuticals, Inc., a biopharmaceutical company developing therapeutics to fight cancer and infectious diseases. Dr. Hutchins was responsible for building out the research program for Peregrine’s lead product candidate, bavituximab, a chimeric monoclonal antibody designed to target phosphatidylserine. Currently in clinical development, bavituximab is being evaluated in combination through Peregrine-funded trials and strategic collaborations, including AstraZeneca and the National Comprehensive Cancer Network (NCCN).”
http://www.heatbio.com/newsroom/press-releases/detail/544/heat-biologics-appoints-jeff-hutchins-ph-d-as-chief
Known Upcoming Events
Jan24-26/Avid/Table#22: WCBP’17: 21st Symposium on Interface of Reg. & Analytical Sciences, WashDC http://www.casss.org/page/WCBP1700 (Theme: “Convergence & Harmonization”)
Jan17-20: Phacilitate's Immunotherapy World Conf., Miami http://www.immunotherapyforum.com
...Jan18 12:25-12:40pm: Dr. Jeff Hutchins (VP/PreClinRes), “Combination Immunotherapies - Opening the Gate: Increasing Tumor Infiltrating Activated T-Cells to Optimize & Expand the Benefits of Immune Checkpoint Therapies”
Feb6-7: GTCbio’s Immunotherapeutics & Immunomonitoring Conf., SanDiego https://www.gtcbio.com/conferences/immunotherapeutics-immunomonitoring-overview
...AGENDA: http://tinyurl.com/gmrlupq
...Feb7 2:05pm: Dr. Bruce Freimark(Res.Dir./Preclin.Oncology), “Blockade of PS Enhances the Anti-Tumor Activity of Immune Checkpoint Inhibitors by Induction of Adaptive Immune Responses Mechanism”
Feb20-22: CHI’s 5th Translational Models in Oncology & I-O, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
~Mar8: FY'17Q3 (qe 1-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
??Apr1-5: AACR 2017, WashDC http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=105
Apr10-12: Immune Profiling World Congress 2017, WashDC (part of “World Vaccine Congress 2017”) http://tinyurl.com/zlr5cyj
...4-11-17 12:10pm: Dr. Jeff Hutchins(VP/PreClinRes.), “Increasing the Benefits of Immune Checkpoint Therapies Thru Increased Tumor Infiltrating Activated T-Cells”
??Jun2-6: ASCO 2017, Chicago https://am.asco.org
June12-17: CHI’s 2nd Annual Immunotherapy & Combinations Conf., Boston http://www.worldpreclinicalcongress.com/Cancer-Immunotherapy-Combinations
...Dr. Jeff Hutchins(VP/PreClinRes.): “Expansion & Activation of T-Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
H.N.Y. to you, AndyC (all All longs) - I still have on my bucket list buying the first 2 rounds for you and our close friends in Celebration at a time & place to be determined!!
Duke has been working w/Peregrine on Anti-Cancer since at least 2015. As of 10-24-16, Peregrine “plans to continue to work with our collaborators at Duke University to further study the therapeutic potential of PS-targeting agents in combo with checkpoint inhibitors like anti-PD-L1 & conventional therapies that augment immunotherapy mechanisms."
= = = = = = = = = = = = =
10-24-16: Duke’s Herbert.Lyerly presents AntiPS/TNBC data at AACR’s “Tumor Immunology & Immunotherapy Conference” in Boston. We first learned of the DUKE/PPHM Cancer Collab. in Bavi/TNBC back in Nov. 2015 at SITC'15 (See 11-9-15: Duke’s Dr. Herbert K. Lyerly http://tinyurl.com/pbof95w )...
10-24-16: Preclinical Research Demonstrates Peregrine Pharmaceuticals' PS-Targeting Antibodies Enhance the Anti-Tumor Activity of PD-L1 Checkpoint Inhibitors in Model of Triple Negative Breast Cancer (TNBC)
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=994994
TUSTIN, Oct. 24, 2016: Peregrine… presentation of preclinical study data demonstrating that PS-targeting antibodies similar to bavituximab are able to enhance the anti-tumor activity of anti-PD-L1 therapy in a model of triple negative breast cancer (TNBC). Data showed that a combination of anti-PS and anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations with paclitaxel, in the well-characterized E0771 murine model of TNBC. Study results were presented by researchers from Duke University Medical Center at the AACR’s Tumor Immunology & Immunotherapy Conference held Oct. 20-23, 2016 in Boston. In addition to evaluating the anti-tumor activity of the various treatment combinations, researchers also examined the impact of various traditional cancer therapies on PS expression in cancer cells. Study results confirmed that levels of PS expression were upregulated in E0771 and 4T1 TNBC cells following treatment with chemotherapy, radiation or photodynamic therapy. Photodynamic therapy also was shown to increase PS expression on tumor cells.
"These study results provide the latest support for the belief that PS-targeting therapies can enhance the anti-tumor activity of checkpoint inhibitors such as anti-PD-L1 therapy in the treatment of TNBC. Just last month, we announced results from another preclinical study [9-27-16/AACR-CRI: http://tinyurl.com/zy9yv78 ] in TNBC demonstrating that 80% of animals receiving the triple combination of anti-PS, anti-PD-1 and anti-LAG3 therapies experienced complete tumor regressions, whereas there were no animals in the anti-PD-1 and anti-LAG3 combination treatment arm that had a complete regression," said Jeff T. Hutchins, Ph.D., Peregrine's VP, Preclinical Research. "Additionally, these latest study findings related to increased PS expression on the surface of tumor cells following traditional cancer treatments demonstrate important activity within the tumor microenvironment that offers rationale for the potential of anti-PS agents in combatting cancer. We plan to continue to work with our collaborators at Duke University Medical Center to further study the therapeutic potential of PS-targeting agents in combination with checkpoint inhibitors like anti-PD-L1 and conventional therapies that augment immunotherapy mechanisms."
**snip**
- - - - - - - - - - - -
[ http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=92
10-22-16 POSTER SESSION B:
#B36 “Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
Kensuke Kaneko 1, Takuya Osada 1, Bruce D. Freimark 2, Herbert Kim Lyerly ** (Duke Univ.) 1
1=DUKE UNIVERSITY, DURHAM, NC
2=Peregrine Pharmaceuticals, Inc.
**Dr. Herbert Kim Lyerly: https://immunology.duke.edu/people/herbert-kim-lyerly-md (George Barth Geller Professor, Duke Univ. MC)
= = = = = = = = = = =
BAVI MOA 11-14-16: SITC’16: Joint Memorial Sloan Kettering (Wolchok Lab) & PPHM poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma http://tinyurl.com/js3fca4
...Dr. Jedd Wolchok states, “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/hlwu72q
...”Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray: Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
–----
11-9-15: BREAST CANCER - Researchers from Duke University and Peregrine evaluated the combination of ch1N11 (preclinical bavituximab equivalent) and anti-PD-1 therapy versus anti-PD-1 stand-alone therapy in well-characterized murine breast cancers, including the triple negative breast cancer (TNBC) model E0771. Study data showed that the combination therapy significantly enhanced overall survival (p=0.0016) and was capable of mediating complete tumor regressions in a greater number of subjects compared to single agent treatments (60% vs. 20%). Data also demonstrated that animals receiving combination treatment had significant increases in tumor associated indicators of immune system activation, including CD45+, CD8+ and CD3+ T-cells. Importantly, the combination treatment led to a prolonged anti-tumor immune response which protected the animals against a re-challenge with the same tumor. This sustained anti-tumor response suggests the potential of the combination therapy to trigger immune system memory and support adaptive immune responses against reemerging disease in breast cancers. All study animals experienced no signs of adverse effects following repeated doses of all therapeutic agents.
BAVI MOA 8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow http://tinyurl.com/qz64pzg
…“Expansion & Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
• PGN632 (B2GPI-indep.) is the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p
Wernaaa, I missed the Sunrise-Update end-of-year promise. Where did you see that?
Shareholder Attendee Reports (10-13-16 ASM) – as more come in, I will delete, update, and re-post. THANKS to all attendees for sharing. This update adds FU’s by Eb0783.
WEBCAST Replay: http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
By: Eb0783 10-13-16 #275973
ASM attendees - roughly 40. 20 of us being shareholders.
FU/275979(re: Garnick?): He was a very valuable asset in the meeting.
FU/276020: From the ASM: King said (in answer to a question about B2GP1 stability, a la GJH) that B2GP1 is “relatively stable” among the patients and, although not as much is known about it compared to other proteins, there probably is no more than a +/-10% variation in a given patient.
Dr Garnick said that B2GP1 is only one (and maybe not the most important) of a number of biomarkers and they will have a number of them creating a profile. Lytle said the $$ spend on Sunrise will be finished by around end of calendar year. My take on these two statements is that they will have the analysis done and the biomarker profile will be complete. imo
Garnick shared a story about an early biomarker at Genentech with us that very few people know. Herceptin failed its first trials but one scientist identified the HER2 mutation and saw how many with HER2 lived longer. So they ran another trial which also failed. Garnick and his peers didn't believe in it but one other guy, identified a biomarker that corrolated with the ones who lived, and talked them into spending another $60mm on one more trial. Since then Herceptin has save 1000's of lives and earned $Billions of dollars. He believes in the value of biomarkers and he has been driving it for us.
As to my question of filing the BLA with what we have, [sorry CP, I did agree with your view) Dr Garnick point blank said our data is “not fileable.” We have now proof of concept and need now another trial, even a small phase II, to validate. Keep in mind however that means a smaller trial and with biomarkers involved it could be shorter than we imagine. imo
On a sidebar with just Shelley and I, Shelly Fussey said they have filed a detailed provisional patent on the Biomarkers which sets the filed date, keep it from being published/shared, and allows inexpensive additions/updates for a number of months (forget if it was 6, 9, or 12). Basically, we the public, and their competitors, will not be able to access it for 18mos. It also adds another year to the patent protection that way.
Edit: Avid III has not actually begun construction as the lease is still being negotiated. However the design, equipment models, etc is ready to begin. Some others will probably add some color because they gave 11 of us a tour of the Myford facility. I call that "shareholder friendly."
FU/276047(To HD): King told us before that ps targeting will be an astronomical value and your verbose presence here continues to confirm that for me. Thank you again for your support.
FU/276054(re: Expenses?): You are right. In fact Lytle said they have already reduced expenses by 38% and will be completely finished with Sunrise expense by end of year. Don't believe those who try to say they have not reduced.
FU/276076(Future?): LOL Loofman, You are still here for the same reason the rest of us "believers" are: The potential rewards are Astronomical (even hornet knows it as he is trying to talk us out of our PEARL every day) but it is only taking a little longer.
FU/276334: I wanted to add one more item from the Avid tour. In past Q&A during one of the latest CCs, King talked about how profitable “fill & finish” is and how he wanted to get into that part in the future. Well, part of John Haney’s presentation was a discussion on what “Fill & Finish” is and why we would want to do it (profitable due to difficulties in keeping the product pure). From his explanation, description, and excitement, my take-away is that he has the design, equipment layout, and process already to fit into the next space.
FU/276490(to ExW re: Herceptin data mining analogy): Missing the ASM made you miss this. Herceptin was NOT originally developed for HER2: data mining pointed them there. Garnick said they did a phase III after screening for HER2 and it also failed! Then one more round of "data mining" gave one scientist enough leverage to convince them to do one more. At that time they had nothing else in the pipeline. Sound familiar? With that addl. biomarker screening they ran a successful phase III and the rest is history. That is how I remember it. Some here seem to denigrate and demonize "data mining" but I see the benefit.
FU/276505(to ExW re: Herceptin analogy): I’m just telling you what I think I heard from Dr. Garnick who said, “very few people know this.” That implies to me that it is not included in any public documents. On top of that, one of the most discussed “problems” still today [there is even talk of legislation to correct it] in getting FDA approvals is that BPs often do not include “all” the information (like trial failures) in the BLAs that they file. I’m not making something up, just bringing into question rational reasons why this FDA document may not tell us about failed trials. Why would it? I may be incorrect that it was a phase III as it could have been a phase II but he said they spent $30?mm on the failed trial and this one guy convinced them (even though their committee didn't believe in it) to spend another $60mm on another trial, Phase III. ASM attendees please correct me.
FU/276539(re: Planning future trials): Yes, this is exactly what King said. The biomarker (profile) will let them filter out those who do not benefit and that will be entrance criteria for the next trial which only needs to be about 30% of the Sunrise 600 and that 30% will provide stat.sig. performance (74%?) on OS or other chosen end points. Addl. biomarkers found that can add to the profile will make it even more precise and possibly less than 200 pts could be used.
FU/276543: Brandon Cox was talking to Jeff Hutchins & Steve Worsley before the meeting started and much of that was about the aPS + aPD1 + aLAG3 data. I don’t think MSK was mentioned in that discussion (I was listening to some of it). I know he was asking about next steps for that data. I will let Brandon share that with you, if he will.
FU/277081: Jake, At the ASM Q&A King was asked is we were talking with Merck and he succinctly answered, "Yes."
FU/277180(Avid Expansion): Jake, I wish you had been there for the Avid tour so you could hear & see what is happening there. You would have gotten a great deal from it. I spent a number years doing project eng. & mgt. work myself, so I think I can see where some items can be checked off. Peregrine has been a neighbor to the tenants & landlords around them for a few decades. They are well enough known to them, have relationships with them, and have expanded a number of times in that locale over the years. I don’t think the landlords around them have any issues about their credit worthiness. They have surveyed the available sites, have leased some already for various purposes, and they know the advantages/disadvantages of each of them for the purposes they will use them for. The design for the new facility is a smaller but cookie cutter version on Avid II. John Haney also mentioned that the parking used by PPHM at Myford is considerably less than an office tenant would need and he spoke as if that was a plus for them at that location. The only hold-up (time-wise) I see right now is whether they can get the lease signed in the most desirable site or if they will relent and use one that may take a little more infrastructure build-out. It is their timeline and they will decide how much negotiation time is too much (it could be done as we type this). I might also add, imo, that if the demand for a product/service is great enough, there are a myriad of ways to get it financed, including various levels of customer input/$$. I experienced that in another industry.
By: Robert C Jonson 10-13-15 #276028
Eb, I thought I heard Rob Garrick say our trial didn’t give proof of concept, just a correlation with the Bavi + 200-240 group, and another proof of concept trial would have to be done. I asked Steve King about betabodies and he said they're still in the picture and should help out with B2GP1 problem. I also asked about earthquake provisions and he said the production labs have been designed to weather earthquakes, but the ultimate provisions are to expand our mfg. facilities to other locales, possibly to include international sites. I asked him lastly about timing to implement the RS and he said as late as possible to give other upcoming events as much time as possible to raise our pps back to the minimum required (or higher). This ASM was as good as one could be after a company has failed a phase III trial, and the NCCN preliminary data on the triple combination of Bavi, PD-L1, and LAG-3 is particularly exciting. It was particularly fun to socialize with PPHM shareholders, most of whom I knew, but some whom I met for the first time. RCJ
FU/276161: Eb, or North, or someone else who attended the ASM yesterday, please correct or expand me, but in response to a question about advancing our finding of the biomarker, Steve said he'd have to be 99% certain of the correlation first. Brandon asked if that meant a p value of .01 instead of .05 and Steve answered "yes." He then said further analysis of Sunrise data might provide addl. biomarker identification that could bring the confidence level up to 99%.
FU/276173: One person who really impressed me yesterday was John Haney, who I call Peregrine's/Avid's logistician. He conducted the first half of our tour of Avid's facilities and told us that Avid was NOT growing: it is EXPLODING! He said a major reason for this is the tremendous increase in efficiency of the manufacturing process. In the past it was only possible to obtain a few mg's of product from a batch of mix. Now, from the same amount of mix, they are obtaining 2-4 grams of product.
FU/276212: Right, JJ, another surprise for me was that we found out Avid has 10+ customers now vs the 1-2 we had heard previously!
FU/276465: Data mining may not be a preferred means of getting results, but it can be good. Rob Garnick told us at the ASM that without data mining there would be no Herceptin today, a drug that's sold $8-10bb.
FU/276485(to IFU): Rob Garnick brought up Herceptin as an analogy to how/why PPHM is analyzing the Sunrise data.
By North40000 10-14-16 #276066
The attendees at the ASM did not care how timely the meeting started---why should you? Attendees, including us, were quite satisfied talking with management and BOD members pre ASM. The meeting started at 10 or shortly thereafter.
FU/276071: Reverting to an earlier exwannabe post prior to start of ASM, ex is correct no BLA will or would be filed today, per Dr. Garnick's opinion expressed post-close of original ASM presentations...it would be "laughed out of Washington" he said. I rejoined that a lot of funny things happen in Washington these days. Dr. Garnick responded with reliance on his Herceptin experience with Genentech[DNA], and the need--in his opinion--for completion of analysis of Sunrise trial to determine existence of further biomarker data or other data that would provide concrete evidence of validity of the proof of concept data now extant today.
More on this issue when I return to East Coast next week, with access to transcripts or audio record, if any, on what was said.
FU/276075: SK: the RS split will not be immediate, answering your speculation about today or tomorrow. Rather, he said, any RS will be delayed as long as possible. Others who attended likely have already commented to same effect.
FU/276077(Worsley?): Worsely was there. I had no discussions with him, and have no idea what others may have discussed with him before or after ASM.
FU/276086(To Eb0783): We enjoyed visiting with you again, as well as the Avid facilities tour.
FU/276088: As further proxies were collected from attendees at ASM, and were yet to be counted, the actual vote count on proposal 1 was not announced during the meeting itself, only the result. Results themselves were obviously apparent from data and votes on hand during the ASM.
FU/276204: I noted that PPHM had obtained a new stock transfer agent some time ago, and asked whether PPHM had monitored who, or what entity, was buying, selling or accumulating shares of PPHM; I further noted that share price had been cabined in a lid of some sort day to day for some time. PL, CFO, responded to my question and comment. As CP has noted previously on other occasions, and consistent with PL's response, the stock transfer agent is not necessarily aware of certain trades itself known by a certain acronym. As to trades known by the agent, PPHM is aware of them as well as any accumulation > 5%. He reminded us of shareholder protection plan in place that enables certain activities by the BOD. I will not detail those here - they can be posted by others as necessary or desirable.
FU/276323(re: AZN?): The analysis of the Sunrise data in regards to biomarkers is critical to the design of all future trials. Peregrine knows it and so does AZN. AZN is not gone. The future of biomarkers is the ability to zero in on target patients to greatly improve the odds of success while helping patients survive. Any person who attended the ASM would have heard that message from King, Garnick, Shan and others. They have valuable data that could lead to a lighted pathway. According to King, that analysis should be completed by yr-end. The Herceptin story was used as an example by Garnick to illustrate a drug developed from data after failing 2 clinical trials. Garnick also commented that the MOA of Herceptin is still unknown, but as science advances we gain the ability to identify and target specific biomarkers that improve patient outcomes.
FU/276472(re: Avid II & III in Calif): Training purposes for later duplicate facilities to built elsewhere: Dr Garnick's words, not mine.
FU/276598: Dr. Garnick spoke with at least some PPHM ASM attendees, including my wife and me, after the meeting was adjourned: 1) Herceptin experience as he[Dr. Garnick] related it...DNA had only prelim. data that targeting Her2 worked or gave good results in some patients... [DNA] needed further addl. data from another, prospective trial---that trial needed to include the pre-identified Her2 patients. Only a very few, including the original project leader and perhaps Garnick himself, thought THAT addl. trial would produce info that would be useful or that Her2 was in fact useful as a target; top mgt did not. But DNA had nothing more in its pipeline at the time. Finally, or ultimately, top mgt of DNA was convinced to invest what Garnick said was $60mm for that addl. trial. 2) As a further side-note, CJ told me directly that, following the ASM, he and the BOD would convene an extended meeting until what he expected to be early evening hours to discuss various matters/issues....what those might be he did not say, nor did I ask.
FU/276514: "BJ", one of our guides on the tour of the Avid facilities on the afternoon of 10/13/2016, was a neuro-scientist himself with PPHM before switching over to Avid, for how long I did not ask. He originally was from the bay area of SanFran. John Haney, who began our tour, is a mechanical engineer with a great deal of experience in property location & acquisition for purposes intended. Like SK, John had a great many slides shown to us that he employs to demonstrate Avid facilities to customers & visitors. I have not looked on Avid web-site to see what slide decks may be there.
By Djohn 10-13-16 #276058
Expanding head count? Of course. Avid is expanding like wildfire. FDA, biosimilars. 12 customers, $20mm in revenue turned down in 2017 because of capacity constraints.
FU/276225: I would say The GM of biotech in 10 years. From what I saw yesterday PPHM has perfected the manufacturing assemble line of biotech. It will be replicated all over the world IMO. Nice meeting all of you at the ASM and Avid tour!!
By JJ1223 10-14-16 #276197
John Haney was well versed and impressive with his presentation on future Avid growth. I was surprised that Peregrine had to turn down $20MM of business in the past year due to capacity. He made it easy to see Avid's vision of growth and why SK see's total corporate profitability within the next 21mos. Also impressed by his comment of 1-2 new customer visits per month, and their enthusiasm to "get" started. No one I spoke with was happy with the Sunrise results, but the Biomarker data could surprise. We will know by year-end. I am not happy with delay, but I walked away with a clear picture and understanding that my long term investment is probably more secure than at any time in the past. For those who believe Steve King was not positive, I would have to disagree.
FU/276237(to HD): Not true at all. Fact is they tried to lease addl. in the Myford building, but the landlord had made improvements and wanted an unreasonable price. The addl. space in that building remains vacant today. Avid leased across the street at a much better price/sf and a more accommodating landlord. BTW, 6 new clients have already booked for the new facility expected to be operational mid 2017. The search for addl. space is underway at present, with a vision of major expansion planned. "Explosion of business", and "perfect storm" in terms of the business environment were the themes we heard during the Avid tour. Avid now employs 220 people (growing 6 just this week) and the facilities are massive and very impressive. If anyone gets the opportunity to hear John Harney speak about Avid, or tour the facility, you should do so. Sounds like Biosimiliars will play a big role in the future growth of the business at Avid. IMO, Pete Gagnon plays a very important role here.
FU/276487(to IFU re: Herceptin data mining analogy) Perhaps you missed Garnick's point since you did not hear it directly from him. He stressed the importance of analyzing the data by saying that Herceptin failed a PI and PII trial dramatically. No one at Genetech wanted to proceed any further. It was dead until one scientist noticed something in the data and convinced others to take a look. The comparison to Herceptin is not the point. In a Biomarker world, it is critical to understanding.
By Hawkfan1 10-15-16 #276290
I also think that John Haney (Avid Sr.Proj.Mgr, prev: Genentech, Pfizer) is a very impressive gentleman, and his enthusiasm is contagious. It's too bad that he couldn't finish the rest of the Avid tour with us. The tour that Paul and I got with him last year was much better than this year's. I haven't seen anyone else post about this, so I thought I'd add a few details about Avid's current state and future plans. Anyone who was at the ASM, please feel free to make additions or correct any errors. Avid has leased 2 buildings since last year. The one that they are calling the Michelle building (it's a half a block northeast on Michelle Drive) is all office space. They have also leased another building on the other side of Franklin Ave. (I think they called it Franklin II, if I'm not mistaken). They leased this one to be the home of Avid III, or a water purification plant when they couldn't work out a deal with landlord of the Myford building. He had decided to improve his building in the hopes of enticing a Google type business at three times the rate that we are paying to lease Avid II. He added all kinds of amenities (including an outdoor seating area with a fire pit!) to attract creative programmer types, but a warehouse district is not the most desirable location for this type of business, and it has been vacant for the past year. John has been negotiating with the owner, and while he stopped short of saying that he is becoming more reasonable, he noted that he has investors too, and John is hopeful that they can come to some kind of suitable arrangement. He would like to lease the other half of the Myford building to build a water purification plant to support Avid. Currently, Avid buys the water that they use in the cell growing process in barrels. I've forgotten how many gallons John said that they use, but each run costs about $150,000.00 just for the water that they buy. If he is successful in negotiating a lease of the Myford building, then the Franklin II building would be used for warehouse space. The Myford building has a limited amount, but not enough. John also talked a little about some pf the challenges of building the water purification plant. Interestingly, in it's finished state, the water is so pure that it can leach the ions out of the stainless steel pipes that carry it, and they must keep the water circulating to prevent this. Before John had to leave, he also made the comment that he is already looking for his next building, which I assume would be the home of Avid IV, since he has already made other plans for the other 2 buildings.
FU/276506(to ExW re: Herceptin data-mining analogy): My purely uneducated guess as to why we didn't data mine the phII is that there were insufficient numbers in that study to achieve statsig. in any subgroups that they might have found, so it wouldn't have meant much. Just a guess… Regarding the Herceptin trial, I have in my ASM notes that the PhI showed safety, but the phII didn't show much, if any, benefit at all. Most of the powers that be at Genentech (including Garnick) were for dropping the drug, but as eb points out, they had nothing else in the pipeline. Then Dr. Cowen re-analyzed the data, and found that a subgroup of patients that over-expressed Her2 had benefited. He developed a test for Her2 expression, and convinced Genentech to run a $60mm PhIII that pre-selected for the over-expression of Her2, and the rest is history. Hope that clarifies some of the confusion.
By Copper888 10-18-16 #276580
Thanks to all attendees of the ASM for your great recaps and it was a great pleasure to meet you and spend some time together! Everyone did a great job of recapping the discussion portion of the meeting, so I don't have much to add except for a few observations and verifications.
1. B2GP1 discussion - I wrote in my notes several times the phrase "layering of biomarker data" and "layering selection criteria" They have the B2GP1 data but are looking for all the data to come in to further "explain the effects" of the stat sig population. New Data is coming in "every day" and should be complete in the next 3 months. They stated that they want to be 99% sure before moving forward with any confirmatory trials, whether that is a registrational PHII or Phase 3 trial.
2. Avid - John Haney indeed described the business as exploding and attributed the growth to what he called the "perfect storm" of market factors aiding growth including:
i) FDA now has a clear path for approval of biosimilars
ii) Production Tech has now improved to the point that, in his words, "little Avid" can produce a worldwide supply of a given drug and compete with the largest producers of biologics.
He also mentioned that the speed to enter into production agreements used to be a year long courtship. Now companies are begging to get on the schedule and using early test runs just to get a relationship with Avid and hopefully try to jump the line.
They expressed their interest in building new facilities - mentioning places like Europe, Singapore, and other parts of the US taking advantage of local tax incentives to build and to decentralize for risk mitigation (earthquakes and such).
Now comes my personal gut feeling about what I heard...let the chastising begin :)
I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there. Well, that is it...I tried to furiously write direct quotes as much as I could. For those in attendance, Please feel free to correct or add to anything written here… Hope that this is of some value!
WEBCAST Replay (10-13-16 ASM): http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
Slideshow PDF (45 Slides): http://files.shareholder.com/downloads/PPHM/2879585795x0x911689/C8068896-AF8B-4CAA-A08C-FB483775B2AD/20161013-_PPHM_Corporate_Overview_-_ASM_FINAL.pdf
EXCERPTS:

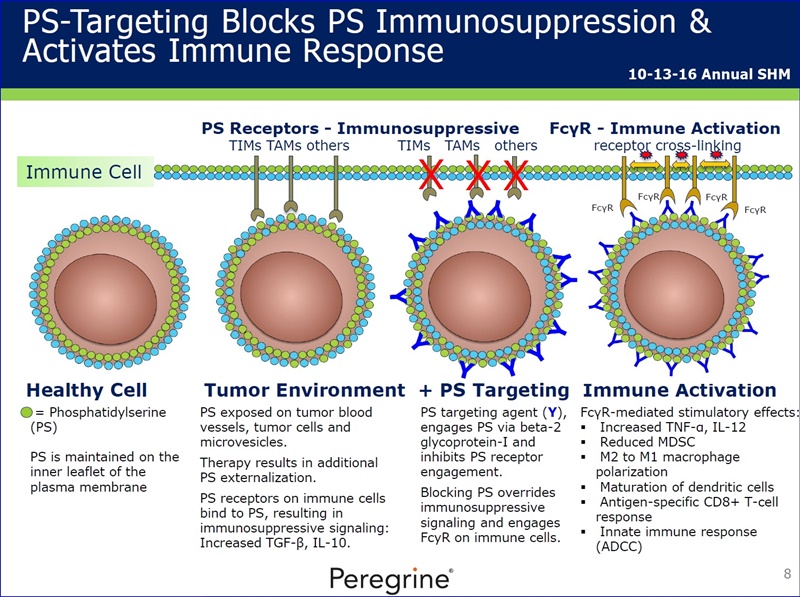


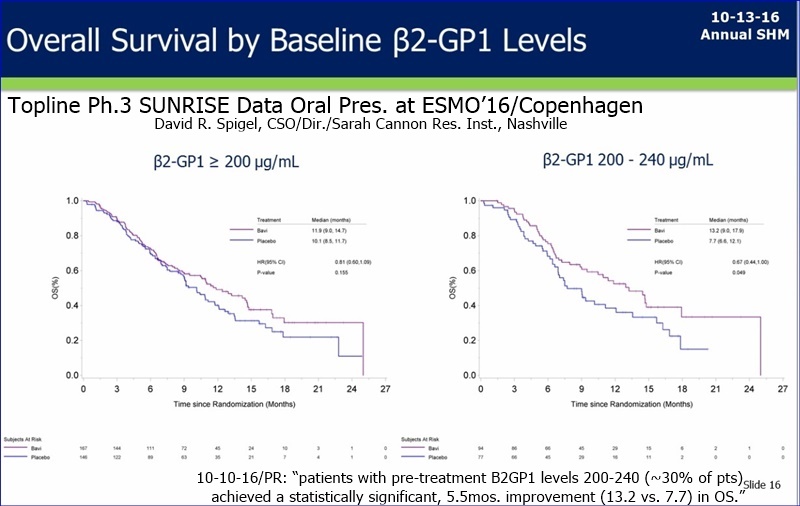
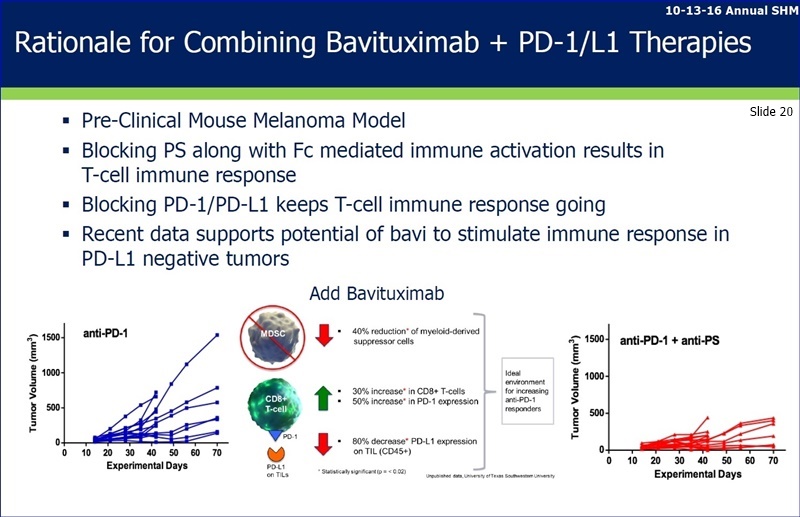

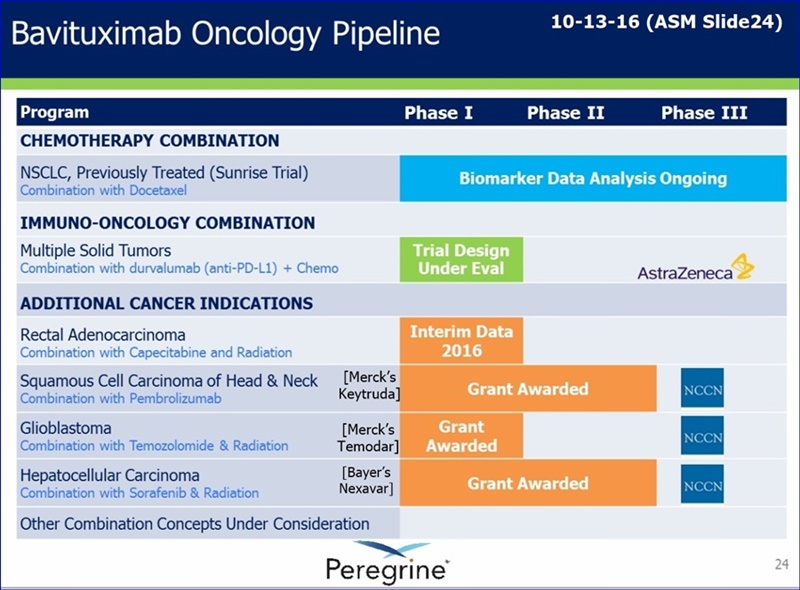

----------
BAVI MOA 10-24-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
Nov9-13 2016: (SITC’16) Society for Immunotherapy of Cancer 31st Annual Meeting, Natl-Harbor MD http://tinyurl.com/j4tw5p9
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
...I. “PS Targeting Antibody in Combo w/Checkpoint Blockade & Tumor Rad. Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC
...II. “Antibody Targeting of PS Enhances the Anti-Tumor Responses of Ibrutinib & anti-Pd-1 Therapy in a Mouse Triple Neg. Breast Tumor Model” - Jian Gong, PhD, PPHM
...III. Monoclonal Antibodies Targeting PS Enhance Combinational Activity of the the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD, PPHM
...Dr. Jedd Wolchok spoke about Bavi in the 11-14-16/PR (SITC’16, Joint MSKCC & PPHM Poster, “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”)…
11-14-16: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model," stated Dr. Jedd Wolchok. "It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact." http://tinyurl.com/js3fca4
11-14-16: "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model," said Dr. Taha Merghoub, PhD, co-director of the Ludwig Collaborative Laboratory at MSK. "We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers." http://tinyurl.com/js3fca4
Note: FULL SITC’16 ABSTRACT #199: http://bit.ly/2dHTEVn
CONCLUSION SECTION FROM THE JOINT MSKCC/PPHM SITC’16 POSTER:


Per Stephanie Diaz (PPHM IR), Dr. Gerber’s 12-7-16 IASLC’16 New Sunrise Biomarker #2 Poster was not presented since the Analysis on the final ~400 pts was not completed in time for inclusion as intended in the final poster. Recall, the orig/prelim. Abstract said superb Biomarker data for Pts 1-200, N=50, MOS 5.9=>12.5mos; Pts 300-600 data to be presented at the Conference)...
12-20-16/S.Diaz(per Cheynew post #282068): “There was no poster in Vienna. The team was working on a very tight timeframe and, despite their best efforts, couldn’t complete the data analysis in time. Given the crunch, we knew there was a possibility that we wouldn’t meet the timing, so we never issued a PR announcing that we would present. Hence no press release announcing that we did not present.”
= = = = = = = = =
12-7-16/IASLC/WCLC: UTSW’s David.Gerber to Present addl. Biomarker Data from Ph3.SUNRISE Trial. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
=>11/30/16 update: ABSTRACT published – see below.
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...”
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
http://wclc2016.iaslc.org
IASLC = Intl. Association for the Study of Lung Cancer - http://iaslc.org
WCLC = World Conference on Lung Cancer (IASLC)
Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
ABSTRACT Book PDF: http://wclc2016.iaslc.org/wp-content/uploads/2016/12/WCLC2016-Abstract-Book_vF-WEB_revDec12.pdf
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with high complement activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [median OS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded independent validation.
= = = = = = = = = =
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ] “Founded in 2005, Biodesix discovers & commercializes cancer tests (diagnostics) that help patients & their doctors make more informed decisions about treatment based on a patient’s unique molecular profile.”
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
= = = = = = = = = =BIOMARKER #1 (B2GPI):
10-10-16/PR: ESMO’16 Topline/SUNRISE; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.
10-10-16: “Peregrine Pharmaceuticals Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at European Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Additional Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Additional patient sample testing and analysis is ongoing and may result in other biomarkers of importance.
** Data presented at ESMO demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approximately 30% of randomized patients) achieved a statistically significant, 5.5-month improvement (13.2 months vs. 7.7 months) in median overall survival (OS) as compared to patients in the control group with the same range of B2GP1 levels [p = 0.049; hazard ratio (HR) = 0.67].
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP of Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific and medical conferences as the more results become available. It is not uncommon in the cancer field for therapeutic candidates to suffer clinical trial setbacks as researchers continue to learn more about the most appropriate patient populations for those drugs. In this landscape, biomarkers play an increasingly important role in helping identify specific patient characteristics that may impact responses to a treatment. This has been seen historically with targeted cancer treatments, as well as more recently with checkpoint inhibitors including PD-1 inhibitors. We look forward to identifying the equivalent markers for bavituximab that will help guide its clinical development.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
- - - - - - - - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
10-10-16 ESMO’16 Topline/Ph3.SUNRISE Data: Biomarker B2GPI/200-240(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
12-12-16 Qtly CC-Transcript, PR(Financials Q2FY17/qe10-31-16), updated Avid Revenues History Table By Quarter…
=> Total Revs May06-Oct16: $202.5mm/Avid + $24.1mm/Govt + $2.4mm/Lic. = $229.2mm.
Cash at 10-31-16: $49.1mm (Op. Cash Burn for q/e 10-31-16 was $2.6mm – see below).
As of Dec. 8, 2016, there were 257,141,534 shares outstanding.
This large post has 3 sections:
I. 12-12-16 Q2/FY17 Qtly. Earnings Conf. Call TRANSCRIPT (q/e 10-31-16)
II. 12-12-16 PPHM Press Release: Q2/FY17 Earnings & Developments
III. Updated Table of Avid Revenues By Quarter (May’06-Current)
…Recall: Peregrine’s FY runs May-Apr, so FY’17 = May’16-Apr’17.
((( Orig. transcript from SeekingAlpha.com [ http://tinyurl.com/z9p7pf4 ], with numerous corrections made. )))
Link to webcast replay: http://ir.peregrineinc.com/events.cfm => http://edge.media-server.com/m/p/x8b7exde
FULL TRANSCRIPT…
12-12-2016 FY’17/Q2 Earnings Conf. Call (q/e 10-31-16) (King/Shan/Hutchins/Lytle)
WELCOME & FWD-LOOKING STATEMENTS: Tim Brons, Vida Strategic Partners (IR) http://www.peregrineinc.com
CEO STEVE KING – OPENING COMMENTS:
Thanks, Tim, and thanks to all of you who have dialed in and to all of you who are participating via webcast today. I like to begin by just reminding everyone that we operate 2 separate, but linked, businesses under the Peregrine umbrella. These businesses represent our R&D efforts and our contract mfg. business, Avid, respectively. Taken together, our goal is to bring the overall company to profitability within the next 18mos. by controlling costs and growing revenues. Equally important is to make sure that our market cap adequately reflects the value of each of these business units, which currently does not seem to be the case. Given our revenue history, current year revenue projections at $50-55mm, and backlog of future business at over $70mm, we believe just the value of Avid Bioservices is far greater than our current market cap and is only growing in value. The Avid business is on track to continue its revenue growth this FY. Actually, our 2 facilities have the potential to generate in excess of $80mm in revenue, leaving addl. capacity for revenue growth beyond the current FY and into next year. Even with this available capacity, we are moving forward with our plans to construct a 3rd mfg. facility in order to meet the anticipated commercial needs of our clients, while continuing to grow the business by bringing in new projects. As we continue planning the new facility, we're keeping a close eye on efficiencies that will reduce the overall cost of construction, and once built, the cost of operations. While this may delay the new facility launch until later in CY2017, we currently have adequate existing capacity to continue meeting the needs of our current clients while also bringing in new customers. So, we do not expect any delay in construction to impact our near term or long term ability to grow top line revenue as originally planned. Business at Avid is brisk, with several process validation campaigns either underway or completed in our Myford [“Avid II”] facility, continuing commercial & clinical production at our Franklin [Avid I”] facility, and new projects being initiated. Taken together, this gives us great confidence in the future of the business. In addition, another milestone was achieved during the quarter as we successfully completed a pre-approval inspection with Health Canada for a client product that is currently under regulatory review in that country. This adds the Avid’s strong regulatory track record with multiple successful inspections from regulatory agencies in the US, EU, Brazil, and Canada. Our regulatory success, our unique ability to provide a wide range of services from cell line development to commercial mfg., and our reputation for delivering quality products continues to drive demand for Avid’s capacity. We are also continuing to evaluate other growth opportunities for the business. This includes the evaluation of downstream, high margin services, which will allow us to increase our footprint with clients.
On the R&D side of the business, we announced a series of important findings in recent months, all of which will contribute to our future development of bavituximab. Our ongoing analysis of the Phase III SUNRISE data has revealed a promising biomarker that may give us insight into key patient populations. We are currently evaluating addl. biomarkers that we hope will allow us to identify a profile for patients who will receive therapeutic benefits from a bavituximab-containing treatment regimen. Concurrent with our internal clinical work, our collaborators at NCCN are in the process of initiating trials for 3 new bavituximab combination treatments, which we expect to begin enrolling patients in the coming months. What is exciting is that the NCCN studies will help build on developments that we are seeing from our internal scientists, as well as our collaborators at Duke, Rutgers, and Memorial Sloan Kettering Cancer Center. Together, we presented compelling data supporting our long-standing belief that bavituximab significantly impacts the tumor microenvironment, creating a more immune -active environment in which other therapies, including checkpoint inhibitors, are able to have a greater anti-tumor effect. These findings are highly validating and we look forward to continuing our work with these world-class institutions to help guide clinical development. We believe the data from the new clinical studies being initiated, other clinical opportunities in development, and the continuing mechanistic and immune-oncology combination insight provided by our scientists & collaborators can add significant value to our R&D business, including creating partnering opportunities, even with a tight control of expenditures that will allow us to reach our profitability goal. I'll now turn the call over to the other members of our team who will give a detailed overview of our clinical, pre-clinical, and corporate activities, as well as our Avid Bioservices contract mfg. business. We will begin with Joe Shan, VP/Clinical & Regulatory Affairs.
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
The SUNRISE Phase 3 trial was discontinued earlier this year. Patient treatment and follow-up on the study were allowed to continue, and currently a number of patients are still receiving bavituximab maintenance. As we previously reported, the study protocol pre-specified the collection of thousands of patient samples for exploratory analyzes over a wide range of potential biomarkers. This analysis has been taking place as patient follow up has continued. Through this biomarker analysis, our team has identified a promising correlation between OS and pre-treatment levels of beta-2 glycoprotein-1 (B2GP1), which was presented at ESMO in October [10-10-16: http://tinyurl.com/hp73njt ]. These results were based on a data cut-off after 70% of the targeted overall survival events in SUNRISE had been reached. Interim analysis demonstrated that patients with pretreatment beta-2 levels between 200 and 240 µg/mL, representing approx. 30% of randomized patients, achieved a statistically significant 5.5mos. improvement in MOS from 7.7mos. in the control group to 13.2mos. in the patients receiving bavituximab & docetaxel. A similar trend was observed with pretreatment B2GP1 levels greater than 200 micrograms per mL, representing approx. 50% of randomized patients. We believe that these observations strongly suggest that B2GP1 levels may be useful for identifying patients who are more likely to benefit from a bavituximab containing therapeutic regimen. We plan to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials, and the company has filed a patent application directed to the use of this initial biomarker discovery. While we are disappointed SUNRISE did not meet the pre-specified clinical endpoints, the data from SUNRISE will be critical in guiding the future clinical dev. of bavituximab. In a rapidly changing oncology treatment landscape, biomarkers are playing an increasingly important role in helping identify specific patient subgroup characteristics that may predict response to a treatment. This has been seen historically with targeted therapies, as well as more recently with checkpoint inhibitors, including PD-1 inhibitors. Numerous addl. biomarkers are currently being evaluated with the goal of developing a multi-marker signature that can potentially identify patients that are likely to receive clinical benefit from bavituximab. Ideally, these biomarkers can be applied not only to chemotherapy combination approaches, but also to I-O combinations, including the checkpoint inhibitors. As we'll hear from Jeff in a minute, we and our research collaborators are generating very exciting data to support the advancement of clinical dev. of bavituximab with I_O approaches, and we plan to incorporate potential biomarkers identified from SUNRISE into future clinical trials.
Meanwhile, as Steve mentioned, we're pleased by the NCCN’s award of 3 grants to investigators for clinical trials of bavituximab in combination with other therapeutics for the treatment of Glioblastoma, Head & Neck Cancer and Hepatocellular Carcinoma [9-6-16: http://tinyurl.com/gutgwb5 ]. These grants were awarded to researchers at the Moffitt Cancer Center, Mass General Hospital Cancer Center, and The Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins. These NCCN studies which we expect to be initiated in the coming months align with our dev. strategy for bavituximab, which is currently focused on small early stage clinical trials evaluating the drug in combination with other cancer treatments, including chemotherapy, radiation, and checkpoint inhibitors. Collaborators such as NCCN play a central role in this strategy and we look forward to integrating the valuable clinical data generated by these investigators to expand our knowledge regarding bavituximab-focused cancer treatment combinations. That concludes my comments today. I’ll now turn the call over to Jeff Hutchins, VP/Pre-Clinical Research.
JEFF HUTCHINS (VP/PreClinical Res.)
As Joe's team mentioned and the important clinical progress they've made during the qtr, the pre-clinical group has also reported findings that further illuminate bavituximab’s role in treating cancer. In recent months, we have announced results from studies conducted with collaborators at Memorial Sloan Kettering Cancer Center, Duke University, and Rutgers University College of Medicine, as well as our own internally conducted pre-clin. research. The presented results of this work reinforced our belief that bavituximab plays an important role in transforming what we call “cold” tumor microenvironments, which are characterized by immunosuppression and an inability to generate an effective attack on tumors into “hot” tumor microenvironments that exhibit high immune activity and are pronged with specific cells to fight tumors. During the past qtr, we have presented data from several studies showing that meaningful tumor microenvironment shifts occurred when a bavituximab like antibody was administered as part of a combination treatment regime with checkpoint inhibitors such as anti-PD-1, anti-PD-L1, and anti-LAG3, as well as with radiation and/or chemotherapy. This shift in tumor microenvironment from cold to hot as was evidenced by greater increases in the activity of several critical immune activating pathways, including presentation and processing of antigens and signaling and activation of T cells. We believe that these immune activating mechanisms can be an important component of combination treatment approaches to cancer. Currently it is estimated that 40-70% of cancer patients don't respond to available anti-cancer therapies due primarily to the immunosuppression impacting their tumors. Our pre-clinical work suggests that bavituximab can help reverse this immunosuppression creating an immune active environment that can potentially convert these non-responders to responders. Importantly, we think that this priming of the tumor marker environment is not only relevant for enhancing response to immunotherapies, but also for more traditional cancer treatments such as radiation & chemotherapy. While we are excited by the data we've generated, it is important that we also demonstrate the practical impact that this activity can have on tumors. To this end, we have presented promising data from a study in a Triple Negative Breast Cancer model at the 2nd Intl. Cancer Immunotherapy Conference in September [9-27-16/AACR-CRI: http://tinyurl.com/zy9yv78 ]. These findings showed that 80% of animals receiving a pre-clinical bavituximab equivalent combined with anti-PD1 & anti-LAG3 therapies experienced complete tumor regression. This is in contrast to findings that showed no complete regression among the animals in the treatment group that received a combination of anti-PD1 & anti-LAG3 alone, without the equivalent of the bavituximab.
In a follow-up presentation from the same study at the SITC Annual Meeting in November [11-14-16/SITC’16: http://tinyurl.com/js3fca4 ], we reported these complete regressions for long, durable, and suggestive of immune system memory and adaptive immune responses. This is just one example of the type of preclin. work that has generated the excitement at Peregrine about the potential of bavituximab in combination cancer therapy. But we recognize that this work needs independent outside validation from the academic community. To this end, we are thrilled to highlight our ongoing collaboration with scientists at Memorial Sloan Kettering, which is being led by doctors Taha Merghoub & Jedd Wolchok, two of the world's most foremost experts in the field of cancer immunotherapy. Their work in a mouse model of B16 melanoma, also presented at the SITC meeting [11-14-16: http://tinyurl.com/js3fca4 ], showed that PS targeting antibodies synergized with both anti-PD1 & radiation therapy to improve anti-cancer activity for tumor burden & survival. As well, this combination uniquely led to a change in the tumor microenvironment, shifting it from immunosuppressive, in which tumors which are protected, to immune active, in which tumors are more susceptible to immune related treatments. We recognize that these research results while exciting are in preclini. cancer models and must be translated into human studies. This is a key objective of the 3 NCCN clinical trials that Joe just mentioned that are expected to be initiated in the coming months. We look forward to continuing our research activities on bavituximab treatment combinations with our collaborators and present new findings as they are available. That concludes my comments today. I'll turn over the call to Paul Lytle, CFO…
PAUL LYTLE (CFO):
Before I begin, I’d like to reiterate our financial goal of becoming profitable on an overall basis 18mos. from this past qtr-end. Our strategy for achieving this goal is built on growing revenue from our contract mfg. business, Avid Bioservices, while reducing our overall spending on R&D. We have made great strides over the past 2 qtrs in both growing revenue and reducing our R&D spend, thereby reinforcing our commitment to this goal. I’ll first address our contract mfg. revenue. During Q2/FY’17, we achieved all-time high revenue of $23.4mm, and while we expected to have a very strong qtr as some revenue shifted from Q1 to Q2 due to an outside testing delay which was discussed last qtr, I am pleased to say that those delays have been resolved and we have recognized to-date $29mm in revenue over the past 6mos. This represents revenue growth of 53% compared to last FY. Given the performance, we are able to reaffirm our full FY2017 revenue guidance of $50-55mm. We also have a strong revenue backlog of $73mm under signed contracts that supports this revenue growth. This backlog mostly covers services to be completed during the remainder of FY2017 & FY2018. I’ll address the 2nd part of our strategy, which is to reduce our overall spending on R&D. As we announced last June, Peregrine has made a decision to focus our internal drug dev. efforts on small, cost effective, early-stage clinical trials designed to attract potential partners to further advance our products. We believe the strategy will not only help us achieve profitability sooner, but it could also create significant potential upside for our shareholders. As we execute on the strategy, our R&D expenses decreased 51% this qtr compared to the same prior year qtr, and for the current 6mo. period our R&D expenses declined 45% over the same period last year. The result of the strategy has translated into a reduction in our net loss by 69% for the qtr and 44% for the 6mo. period vs. the same prior year periods. And, if you deduct non-cash expenses from our net loss this qtr, we saw our net cash burn rate for the qtr decline 78% to $2.6mm compared to $11.7mm for the same qtr last year. As I mentioned before, we're making great progress towards reaching our goal of profitability. A more detailed analysis of our statement of operations is included in our Form 10-Q that will be filed shortly [ http://tinyurl.com/j2u2bjk ]. This concludes my financial overview. I will now open the call up for questions.
Q&A: [beg. 22:10]
1. Thomas Yip (FBR & Co.): http://www.fbr.com
TY: ”Can you just remind us what was the magnitude of Avid’s delayed revenue recognition between Q1 & Q2?”
Paul Lytle: The overall delay is that last qtr we mentioned that we had an outside testing lab that is responsible for performing certain tests, and that testing lab was backlogged with certain activities and therefore we couldn’t recognize a certain amount of revenue last quarter that was shifted from Q1 into Q2. Well, their delays have now been resolved and we’re back on track and recognizing revenue as we routinely do as we release lots. So, everything has been resolved.
TY: ”Approx. how much was that amount?”
Paul Lytle: If you straight line our revenues, our projections are about $50-55mm over the FY. So you figure at $12.5mm/qtr, you could assume that last qtr was about $5.5mm in total revenue and it could have been on a straight line basis of about $12mm.
TY: ”If I recall correctly the cost of your new [Avid II “Myford”] facility is also included in cost of contract mfg. as well. So how much of the new facility cost is included in Q2/FY17’s contract mfg costs?”
Steve King: If we're talking about the Myford facility which we commissioned in January of this year, that cost would be included in those numbers. Obviously we're just kind of moving toward a 3rd facility that would primarily be used for clinical production, at least initially, could eventually be commercial as well, but it’s really designed to be a clinical production facility. So those numbers other than where we spent on planning up to this point are not reflected as in the current numbers.
TY: “Re: Bavi, now that we’ve seen some positive pre-clin. data in combination with checkpoint inhibitors and as you outlined earlier, there are a number of collaboration of academic partners. Can you tell us what else is needed to move any potential combination to the clinic and would any of these upcoming trials involve existing academic collaborators?”
Steve King: One of the nice things about the NCCN collaboration, which just to remind everybody was based on a $2mm grant from Peregrine, a lot of which has already been put into the program, but basically the NCCN will oversee all of those 3 clinical studies that will be completed. One of those is in an I-O combination, so it gives us one opportunity already to start looking at kind of an extension if you will of the work we're doing with our collaborators into the clinic and so we can start to validate some of that. In addition, we have interest from other academic collaborators in I-O combinations, so we're currently evaluating those and how those would move forward. So, we're hopeful to have some news in the not too distant future on some of those developments as well. Beyond that, we're really in the process of just looking through the SUNRISE data, identifying what's the best way to identify patients. In addition, some of the other data we can learn from SUNRISE are changes on therapy, the primary hypothesis being that we can take, if you will, immunologically cold tumors and make them hot tumors by breaking down the immunosuppression - that's some of the things that we have an option to learn from the SUNRISE study. Overall, the data from SUNRISE, taken together with the work at Sloan Kettering and our other collaborators, all really build the story very nicely to eventually potentially starting more company-sponsored studies, but that's where the balance of our R&D spend vs. our revenue comes into play. So, whatever studies we start we anticipate would be smaller studies, but ones that will be rich in biomarker-type analysis, which we think will be the key to #1 identifying those successful combinations, but secondly to eventual partnering where we can actually help some of the other drugs that are in development to do better.
TY: ”Thanks again for taking my questions. Looking forward to learn more about these trials in coming months.”
MR. KING’S CLOSING COMMENTS:
I'd like to thank all of you again for participating in today’s phone call. As always, I want to thank our stockholders for their continued support and I would like to especially thank our patients, their families, and the investigators that are participating in our bavituximab clinical trials. Looking ahead, we are confident in our ability to achieve profitability through cost reduction and revenue growth, while continuing to work with our collaborators to generate the data required to drive partnering and build shareholder value. With that we will now conclude the call. Thank you again.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = == = = =
12-12-15 PR: Peregrine Pharmaceuticals Reports Financial Results for Second Quarter of FY 2017 and Recent Developments
*Avid Posts Record Revenue of $23.4 Million During Second Quarter FY 2017 with Contracted Backlog of Future Business Currently at $73 Million
*Beta-2 Glycoprotein-1 (B2GP1) Identified as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone from the Phase III SUNRISE Trial
*Multiple Preclinical Presentations Collectively Point to Bavituximab's Ability to Enhance the Efficacy of Checkpoint Inhibitors by Triggering Immune Active Tumor Environment
TUSTIN, Dec. 12, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced financial results for the second quarter of FY (FY) 2017 ended October 31, 2016, and provided an update on its contract manufacturing business, clinical pipeline and other corporate developments.
Highlights Since July 31, 2016
"The Avid business is on track to continue its revenue growth this FY as we move toward overall profitability within the next 18 months. Our two facilities have the potential to generate in excess of $80 million in revenue, leaving additional capacity for revenue growth beyond FY 2017 revenue guidance," stated Steven W. King, President and CEO of Peregrine. "We are moving forward with our plans to construct a third manufacturing facility, with an eye toward efficiencies that will reduce the overall cost of construction and operation. While this may delay the new facility launch until later in calendar year 2017, we currently have adequate existing capacity to continue meeting the needs of our current clients while also bringing in new customers so we do not expect it to impact our near-term ability to grow top-line revenue as originally planned. Independently, Avid is a successful and growing CDMO business generating significant revenue and one of our key goals going forward is to help ensure that its value is appropriately represented in the market cap of our overall business." Mr. King continued, "During, and subsequent to, the second quarter, we announced a series of important findings, all of which will contribute to our future development of bavituximab. Our ongoing analysis of the Phase III SUNRISE data has revealed a promising biomarker that may give us insight into key patient populations. We are actively evaluating additional potential biomarkers and we hope to identify a profile for patients who will receive therapeutic benefit from treatment with bavituximab. Concurrent with our internal clinical work, our collaborators at NCCN are in the process of initiating trials for three new bavituximab combination treatments, which we expect to begin enrolling patients in the coming months. What is exciting is that the NCCN studies will help build on developments we are seeing from our internal scientists, as well as our collaborators at Duke, Rutgers, and Memorial Sloan Kettering Cancer Center. Together, we presented compelling data supporting our long-standing belief that bavituximab significantly impacts the tumor microenvironment, creating a more immune active environment in which other therapies, including checkpoint inhibitors, are able to have a greater anti-tumor effect. These findings are highly validating and we look forward to continuing our work with these world-class institutions to help guide clinical development."
Avid Bioservices Highlights
"Growing top-line revenue is a key focus and we are pleased to report a 53% improvement in contract manufacturing revenue for the current six-month period compared to the same period last FY. In addition, our revenue guidance for the second quarter was targeted to exceed $20 million and we achieved $23.4 million in contract manufacturing revenue as we worked closely with the third-party testing laboratory to resolve the unexpected delays in testing we encountered during the first quarter. As a result, we reaffirm our manufacturing revenue guidance of between $50 and $55 million for the full FY," stated Paul Lytle, CFO of Peregrine. "We also continued to advance the validation of three separate manufacturing processes related to third-party customer products that could lead to future commercial manufacturing for these products. While these activities generally have a higher cost of manufacturing, which impacted our gross margin during the second quarter, we believe our investment in these products will provide us future revenue opportunities once these products are approved."
The company reaffirms its manufacturing revenue guidance for the full FY 2017 of $50 - $55 million.
Avid's current manufacturing revenue backlog is $73 million, representing estimated future manufacturing revenue to be recognized under committed contracts. This backlog mostly covers revenue to be recognized during the remainder of FY 2017 and FY 2018.
Clinical Development Highlights
Through the ongoing analysis of the Phase III SUNRISE data, Peregrine scientists identified a correlation between overall survival and pre-treatment levels of the biomarker, beta-2 glycoprotein-1 (B2GP1), which we presented at ESMO in October [10-10-16: http://tinyurl.com/hp73njt ]. Data demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 µg/mL - representing approximately 30% of randomized patients - achieved a statistically significant, 5.5-month improvement, from 7.7 months to 13.2 months, in median overall survival as compared to patients in the control group with the same range of B2GP1 levels.
Peregrine's research collaboration with NCCN is advancing as planned, with grants awarded to three investigators [9-6-16: http://tinyurl.com/gutgwb5 ] to support research of bavituximab in combination with other therapeutics for the following studies:
1. Phase I Trial of Sorafenib and Bavituximab Plus Stereotactic Body Radiation Therapy (SBRT) for Unresectable Hepatitis C Associated Hepatocellular Carcinoma
2. Phase I/II Clinical Trial of Bavituximab with Radiation and Temozolomide for Patients with Newly Diagnosed Glioblastoma
3. Phase II Study of Pembrolizumab and Bavituximab for Progressive Recurrent/Metastatic Squamous Cell Carcinoma of the Head and Neck
The company expects these trials to begin over the coming months.
Research Highlights
Peregrine scientists and collaborators from Duke University Medical Center, Rutgers University College of Medicine, and Memorial Sloan Kettering Cancer Center each presented compelling data demonstrating that shifts in the tumor microenvironment from immune suppressed to immune active occurred when a bavituximab equivalent antibody was administered as part of a combination treatment regimen. Presentations addressed multiple phosphatidylserine (PS)-targeting combinations, including those with checkpoint inhibitors such as anti-PD-1, anti-PD-L1 and anti-LAG3, as well as with radiation or chemotherapy. These data suggest that the addition of PS-targeting reverses an immunosuppressive tumor environment, creating an immune active tumor microenvironment that can potentially convert patients that generally do not respond to immuno-oncology (I-O) therapies into responders. Key presentations were made at the Second International Cancer Immunotherapy Conference in September, the American Association for Cancer Research's Tumor Immunology and Immunotherapy Conference in October, the Society for Immunotherapy of Cancer (SITC) in November, and the San Antonio Breast Cancer Symposium in December.
Financial Highlights and Results
Peregrine continues to execute its previously-announced strategy to reach sustained profitability by increasing contract manufacturing revenue while decreasing research and development expenses, with the goal of reaching profitability 18 months from now. During the first six months of FY 2017, the company made significant progress toward this goal with contract manufacturing revenues increasing 53% compared to the first six months of FY 2016 and research and development expenses decreasing by 45% compared to the first six months of FY 2016.
Contract manufacturing revenue from Avid's clinical and commercial biomanufacturing services provided to its third-party customers increased to $23,370,000 for the second quarter of FY 2017 compared to $9,523,000 for the second quarter of FY 2016. In addition, as previously-announced, a backlog at a third-party testing lab, unrelated to product quality, required that the recognition of some revenue be shifted from the first quarter to the second quarter of FY 2017.
Total costs and expenses for the second quarter of FY 2017 were $27,447,000, compared to $23,347,000 for the second quarter of FY 2016. For the second quarter of FY 2017, research and development expenses decreased 51% to $7,022,000, compared to $14,190,000 for the second quarter of FY 2016. Cost of contract manufacturing increased to $15,441,000 in the second quarter of FY 2017 compared to $4,741,000 for the second quarter of FY 2016, primarily due to an increase in the cost of contract manufacturing associated with higher reported revenue. Also contributing to this increase and impacting gross margins for the period is the higher cost of operating the new Myford facility as well as the higher cost associated with performing process validation runs during the quarter. For the second quarter of FY 2017, selling, general and administrative expenses increased to $4,984,000 compared to $4,416,000 for the second quarter of FY 2016 primarily due to the company's growing manufacturing business.
Peregrine's consolidated net loss attributable to common stockholders was $5,498,000 or $0.02 per share, for the second quarter of FY 2017, compared to a net loss attributable to common stockholders of $14,578,000, or $0.07 per share, for the same prior year quarter. [NOTE: Oper. Cash Burn for Q2(q/e 10-31-16): $2.57mm see below.]
Peregrine reported $49,055,000 in cash and cash equivalents as of October 31, 2016, compared to $61,412,000 at FY ended April 30, 2016.
More detailed financial information and analysis may be found in Peregrine's Quarterly Report on Form 10-Q, which will be filed with the Securities and Exchange Commission today.
Conference Call
Peregrine will host a conference call and webcast this afternoon, December 12, 2016, at 4:30PM ET (1:30PM PT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.peregrineinc.com/events.cfm .
About Peregrine Pharmaceuticals, Inc.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
About Avid Bioservices
Avid Bioservices provides a comprehensive range of process development, high quality cGMP clinical and commercial manufacturing services for the biotechnology and biopharmaceutical industries. With over 15 years of experience producing monoclonal antibodies and recombinant proteins in batch, fed-batch and perfusion modes, Avid's services include cGMP clinical and commercial product manufacturing, purification, bulk packaging, stability testing and regulatory strategy, submission and support. The company also provides a variety of process development activities, including cell line development and optimization, cell culture and feed optimization, analytical methods development and product characterization. For more information about Avid, please visit http://www.avidbio.com .
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS (UNAUDITED)
THREE MONTHS ENDED
OCTOBER 31, SIX MONTHS ENDED
OCTOBER 31 ,
2016 2015 2016 2015
REVENUES:
Contract manufacturing revenue $ 23,370,000 $ 9,523,000 $ 28,979,000 $ 18,902,000
License revenue — — — 292,000
Total revenues 23,370,000 9,523,000 28,979,000 19,194,000
COSTS AND EXPENSES:
Cost of contract manufacturing 15,441,000 4,741,000 18,503,000 9,349,000
Research and development 7,022,000 14,190,000 15,591,000 28,108,000
Selling, general and administrative 4,984,000 4,416,000 10,044,000 9,315,000
Total costs and expenses 27,447,000 23,347,000 44,138,000 46,772,000
LOSS FROM OPERATIONS (4,077,000 ) (13,824,000 ) (15,159,000 ) (27,578,000 )
Interest and other income 21,000 626,000 46,000 657,000
NET LOSS
$ (4,056,000 ) $ (13,198,000) $ (15,113,000) $ (26,921,000)
COMPREHENSIVE LOSS $ (4,056,000 ) $ (13,198,000 ) $ (15,113,000 ) $ (26,921,000 )
Series E preferred stock accumulated dividends (1,442,000 ) (1,380,000 ) (2,477,000 ) (2,413,000 )
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS
$ (5,498,000) $ (14,578,000) $ (17,590,000) $ (29,334,000)
WEIGHTED AVERAGE COMMON SHARES OUTSTANDING:
Basic and Diluted 244,815,767 203,942,411 242,205,428 200,629,892
BASIC AND DILUTED LOSS PER COMMON SHARE $ (0.02 ) $ (0.07 ) $ (0.07 ) $ (0.15 )
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
OCTOBER 31,
2016 APRIL 30, 2016 Unaudited
ASSETS
CURRENT ASSETS:
Cash and cash equivalents $ 49,055,000 $ 61,412,000
Trade and other receivables 6,066,000 2,859,000
Inventories 25,924,000 16,186,000
Prepaid expenses and other current assets 1,711,000 1,351,000
Total current assets 82,756,000 81,808,000
Property and equipment, net 23,957,000 24,302,000
Restricted cash 600,000 600,000
Other assets 2,624,000 2,333,000
TOTAL ASSETS $ 109,937,000 $ 109,043,000
LIABILITIES AND STOCKHOLDERS' EQUITY
CURRENT LIABILITIES:
Accounts payable $ 11,572,000 $ 8,429,000
Accrued clinical trial and related fees 3,639,000 7,594,000
Accrued payroll and related costs 5,280,000 5,821,000
Deferred revenue 17,980,000 10,030,000
Customer deposits 26,928,000 24,212,000
Other current liabilities 1,012,000 1,488,000
Total current liabilities 66,411,000 57,574,000
Deferred rent, less current portion 1,347,000 1,395,000
Commitments and contingencies
STOCKHOLDERS' EQUITY:
Preferred stock - $0.001 par value; authorized 5,000,000 shares; 1,647,760 and 1,577,440 issued and outstanding at October 31, 2016 and April 30, 2016, respectively 2,000 2,000
Common stock-$0.001 par value; authorized 500,000,000 shares; 251,765,279 and 236,930,485 issued and outstanding at October 31, 2016 and April 30, 2016, respectively 252,000 237,000
Additional paid-in capital 566,314,000 559,111,000
Accumulated deficit (524,389,000 ) (509,276,000 )
Total stockholders' equity 42,179,000 50,074,000
TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY $ 109,937,000 $ 109,043,000
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - - -
[From 10-Q header: “As of Dec. 8 2016, there were 257,141,534 shares outstanding.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-16 iss. 7-14-16 PR: http://tinyurl.com/h8eqtg5 (Cash 4-30-16=$61.4mm)
Latest 10Q 10-31-16 iss. 12-12-16 http://tinyurl.com/j2u2bjk PR: http://tinyurl.com/hhn4gga (Cash 10-31-16=$49.1mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q2(qe 10-7-31-16), per the 10-31-16 10-Q ( http://tinyurl.com/j2u2bjk ) issued 12-12-16.
• Total Revs since May’06: ($202.5mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $229.2mm
• 12-12-16: FY'17 (May'16-Apr'17) Avid revs guidance $50-55mm (Committed B/L=$73mm).
• Deferred-Revs at 10-31-16 total $18.0mm, DOWN from $21.5mm at 7-31-16.
• Cust.Deposits at 10-31-16 total $26.9mm, UP from $21.7mm at 7-31-16.
• Inventories at 10-31-16 total $25.9mm, UP from $25.3mm at 7-31-16.
• Avid’s Gross-Profit over last 4 qtrs: $22.3mm on revs of $54.4mm (GP%=41%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16 TOTAL: 44,357,000 22,966,000 21,391,000 48%*
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
FY16Q2 10-31-15 9523 0 0 9523 9688 0 12554
FY16Q3 1-31-16 6672 0 37 6709 15418 0 15189
FY16Q4 4-30-16 18783 0 0 18783 15418 0 15189
FY17Q1 7-31-16 5609 0 0 5609 21531 0 25274
FY17Q2 10-31-16 23370 0 0 23370 21531 0 25274
Totals: 202548 24149 2453 229150 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
FY16 4-30-16 44,686 …Avid(CMO)= 44,357
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
FY16Q2 10-31-15 13,198,000
FY16Q3 1-31-16 16,847,000
FY16Q4 4-30-16 11,884,000
FY17Q1 7-31-16 11,057,000
FY17Q2 10-31-16 4,056,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.25)
FY16Q2 10-31-15 11,701,000 (Q1+Q2: 24,007,000 10Q pg.26)
FY16Q3 1-31-16 15,086,000 (Q1+Q2+Q3: 39,093,000 10Q pg.27)
FY16Q4 4-30-16 10,112,000 (FY'16: 49,205,000 10K pg.39)
FY17Q1 7-31-16 9,607,000 (from 10Q pg.22)
FY17Q2 10-31-16 2,565,000 (Q1+Q2: 12,172,000 10Q pg.24)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
FY’16 total Op-Burn: $49,205,000
Period Halozyme Cust-A Other-Custs
FYE 4-30-14 91% 1% 8%
FYE 4-30-15 79% 12% 9%
FYE 4-30-16 69% 26% 5%
Q/E 7-31-16 65% 29% 6%
Q/E 10-31-16 77% 10% 13%
O/S Shares & ATM Sales History (’06–curr.)
PPHM - O/S Shares History (’06–curr.)
4-30-06 35,876,438
1-31-07 39,222,440
4-30-07 39,222,440
7-6-07 45,233,123
7-31-07 45,242,123
10-31-07 45,242,123
1-31-08 45,242,123
4-30-08 45,242,123
7-31-08 45,242,123
10-31-08 45,242,123
1-31-09 45,242,123
4-30-09 45,537,711
7-10-09 47,392,883
7-31-09 47,393,783
10-31-09 48,869,563 +1,475,780
1-31-10 50,903,404 +2,033,841
4-30-10 53,094,894 +2,191,490
6-21-10 54,388,917 +1,294,023 (6-22-10 ATM/mlv Form424)
7-9-10 55,069,449 +475,987 (4-30-10 10K iss. 7-14-10)
7-31-10 55,784,955 +715,506
10-31-10 59,220,742 +3,435,787
11-30-10 63,932,353 +4,711,611 (10-31-10 10Q iss. 12-9-10)
12-15-10 64,404,097 +471,744 (12-17-10 S-3: $75M Shelf Reg.)
1-31-11 66,813,419 +2,409,322
2-28-11 67,885,811 +1,072,392 (1-31-11 10Q iss. 3-11-11)
4-30-11 69,837,142 +1,951,331
7-8-11 71,069,858 +1,232,716 (4-30-11 10K iss. 7-14-11)
8-22-11 72,704,647 +1,634,789 (Proxy iss. 8-26-11)
8-31-11 73,284,016 +579,369 (424B5 iss. 9-2-11)
9-8-11 79,536,268 +6,252,252 (Roth Sale to 3 Inst’s @ $1.11/sh.)
10-31-11 82,638,201 +3,101,933
12-9-11 86,788,817 +4,150,616 (10-31-11 10Q iss. 12-12-11)
1-31-12 93,146,226 +6,357,409
2-29-12 98,873,172 +5,726,946 (1-31-12 10Q iss. 3-9-12)
4-30-12 101,421,365 +2,548,193
7-13-12 104,174,056 +2,752,691 (4-30-12 10K iss. 7-16-12)
7-31-12 104,178,431 +4,375 (7-31-12 10Q iss. 9-10-12)
8-16-12 104,191,176 +12,745 (prelim. proxy 14A http://tinyurl.com/c48bvof )
9-7-12 104,191,176 nochg (7-31-12 10Q iss. 9-10-12)
10-31-12 123,310,188 +19,119,012
12-7-12 132,539,783 +9,229,595 (10-31-12 10Q iss. 12-10-12)
1-31-13 133,770,614 +1,230,831
3-12-13 137,110,758 +3,340,144 (1-31-13 10Q iss. 3-12-13)
4-30-13 143,768,946 +6,658,188 (4-30-13 10K iss. 7-11-13)
7-5-13 151,602,765 +7,833,819 (4-30-13 10K iss. 7-11-13)
7-31-13 153,506,811 +1,904,046
9-5-13 156,461,114 +2,954,303 (7-31-13 10K iss. 9-9-13)
10-31-13 160,248,742 +3,781,628
12-6-13 160,325,891 +77,149 (10-31-13 10K iss. 12-10-13)
1-31-14 176,453,261 +16,127,370
3-3-14 176,481,054 +27,793 (1-31-14 10Q iss. 3-7-14)
4-30-14: 178,871,164 +2,390,110
7-7-14: 179,209,458 +338,294 (4-30-14 10-K cover page, iss. 7-14-14)
7-31-14: 179,216,032 +6,574 (7-31-14 10Q iss. 9-9-14)
8-22-14: 179,226,424 +10,392 (8-28-14 Proxy/Def14A)
9-5-14: 179,505,424 +279,000 (7-31-14 10Q iss. 9-9-14)
10-31-14: 182,000,583 +2,495,159 (10-31-14 10Q iss. 12-10-14)
12-5-14: 182,081,234 +80,651 (10-31-14 10Q cover pg., iss. 12-10-14)
12-19-14: 182,081,234 -0- (12-23-14 S-3)
1-31-15: 184,244,698 +2,163,464 (1-31-15 10Q iss. 3-12-15)
3-12-15: 188,332,872 +4,088,174 (“ “ “)
4-30-15: 193,346,627 +5,013,755 (4-30-15 10-K iss. 7-14-15)
7-10-15: 199,934,918 +6,588,291 (4-30-15 10-K/cover-pg, iss. 7-14-15)
7-31-15: 200,983,948 +1,049,030 (7-31-15 10Q iss. 9-9-15)
9-4-15: 202,124,031 +1,140,083 (“ “ “)
10-31-15: 225,824,551 +23,700,520 (10-31-15 10Q iss. 12-10-15)<=Incl. 18.5mm sh. Dart/EastCAP @1.08
12-9-15: 229,701,808 +3,877,257 (10-31-15 10Q iss. 12-10-15)
1-31-16: 232,231,242 +2,529,434 (1-31-16 10Q iss. 3-9-16)
3-8-16: 233,738,426 +1,507,184 (“ “ “)
4-30-16: 236,930,485 +3,192,059 (4-30-16 10K iss. 7-14-16)
7-11-16: 241,456,721 +4,526,236 (“ “ “)
7-31-16: 241,456,721 -0- (7-31-16 10Q iss. 9-8-16)
9-2-16: 242,381,850 +925,129 (“ “ “)
10-31-16: 251,765,279 +9,383,429 (10-31-16 10Q iss. 12-12-16)
12-8-16: 257,141,534 +5,376,255 (“ “ “)
= = = = = = = = = = = = = = = = = =
ATM Sales Summary (3/2009–12/12/2016). Also, PPHM O/S Shares History Table (’06–curr.) at the bottom of this post. At 12-8-16, common shares O/S = 257,141,534sh.
ATM = “At-The-Market Sales Issuance”
I. WM-SMITH 3-2009:
• $7.5mm ATM/Wm.SMITH 3-26-09: $7,500,000gr. / 2,150,759sh. = $3.49/sh. (commiss: 3%)
• $25mm ATM/Wm.SMITH 7-14-09: $25,000,000gr. / 7,569,314sh. = $3.30/sh. (commiss: 3%/1st$15mm, then 2%)
*Total Raised via WmSmith ATM Sales thru 7-31-10:
. . . . $32,500,000gr. / 9,720,073sh. = $3.34/sh.
II. MLV 6-2010: http://www.mlvco.com
$15mm ATM/MLV 6-22-10 (commiss: 2%) Form424: http://tinyurl.com/24txkxb
• Sold 6/22/10–10/31/10: $6,840,000gr. / 4,031,018sh. = $1.70/sh.
• Sold 11/1/10–11/30/10: $7,407,000gr. / 4,711,611sh. = $1.57/sh.
• Sold 12/1/10–1/31/11: $753,000gr. / 471,744sh. = $1.60/sh.
*Total Raised via MLV June’10 ATM Sales thru 1-31-11:
. . . . $15,000,000gr. / 9,214,373 = $1.63/sh.
III. MLV 12-2010: “Dec’10 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-10 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 12-17-10: http://tinyurl.com/2469b2d )
• Sold 12/29/10-1/31/11: $6,460,000gr. / 2,385,862sh. = $2.71/sh.
• Sold 2/1/11-2/28/11: $2,358,000gr. / 998,142sh. = $2.36/sh.
• Sold 3/1/11-4/30/11: $4,470,000gr. / 1,840,487sh. = $2.43/sh.
• Sold 5/1/11-7/31/11: $3,713,000gr. / 1,912,576sh. = $1.94/sh.
• Sold 8/1/11-10/31/11: $5,582,000gr. / 4,727,840sh. = $1.18/sh.
• Sold 9-2-12 Roth Direct: $6,940,000gr./ 6,252,252sh. = $1.11/sh.
• Sold 11/1/11-1/31/12: $10,961,000gr. / 10,308,025sh. = $1.06/sh.
• Sold 2/1/12-2/29/12: $5,871,000gr. / 5,726,946sh. = $1.03/sh.
• Sold 3/1/12-4/30/12: $1,263,000gr. / 2,198,543sh. = $.57/sh.
• Sold 5/1/12-6/30/12: $1,496,000gr. / 2,752,691sh. = $.54/sh.
• Sold 7/1/12-9/26/12: none**
• Sold 9/27/12-10/31/12: $16,719,000gr./ 18,557,928 = $.90/sh.
• Sold 11/1/12-11/30/12: $7,296,000gr./ 9,220,313 = $.79
• Sold 12/1/12-1/31/13: $1,540,000gr./ 1,131,282 = $1.36
• Sold 2/1/13-3/12/13: $330,000gr./ 201,154 = $1.64
*Total Raised via MLV Dec’10 ATM Sales thru 3-12-2013:
. . . . $75,000,000gr. / 68,214,041 = $1.10sh.
IV. MLV 12-2012: “Dec’12 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-12 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 3-9-12: http://tinyurl.com/7dl7pjm )
• Sold 2/1/13-3/12/13: $4,475,000gr. / 3,132,402sh. = $1.43/sh.
• Sold 3/13/13-4/30/13: $8,897,000gr. / 6,188,273sh. = $1.44/sh.
• Sold 5/1/13-7/11/13: $12,729,000gr. / 7,927,016sh. = $1.61/sh.
• Sold 7/12/13-7/31/13: $2,468,000gr. / 1,690,864sh. = $1.46/sh.
• Sold 8/1/13-9/9/13: $4,372,000gr. / 3,057,431sh. = $1.43/sh.
• Sold 9/10/13-10/31/13: $4,708,000gr. / 3,262,958sh. = $1.44/sh.
• Sold 11/1/13-12/6/13: NONE – see 10Q note below.
• Sold 12/7/13-1/31/14: $28,130,000gr. / 16,045,717sh. = $1.75/sh.
• Sold 2/1/14-4/30/14: $3,017,000gr. / 1,543,383sh. = $1.95/sh.
• Sold 5/1/14-7/31/14: $425,000gr. / 226,700sh. = $1.92/sh.
• Sold 8/1/14-10/31/14: $3,891,000gr. / 2,494,835sh. = $1.56/sh.
• Sold 11/1/14-1/31/15: $1,878,000gr. / 1,261,825sh. = $1.49/sh.
*Total Raised via MLV Dec’12 ATM Sales thru 10-31-2014:
. . . . $75,000,000gr. / 46,831,404 = $1.60sh.
V. MLV 6-2014: “Jun’14 AMI Agreement – up to $25mm” http://www.mlvco.com
• Sold 11/1/14-1/31/15: $1,193,000gr. / 869,504sh. = $1.43/sh.
• Sold 2/1/15-3/12/15: $6,204,000gr. / 4,354,954sh. = $1.44/sh.
• Sold 3/13/15-4/30/15: $6,147,000gr. / 4,457,299sh. = $1.38/sh.
• Sold 5/1/15-7/14/15: $8,896,000gr. / 6,534,400sh. = $1.36/sh.
• Sold 7/15/15-7/31/15: $1,270,000gr. / 1,003,830sh. = $1.27/sh.
• Sold 8/1/15-9/9/15: $1,290,000gr. / 1,091,508sh. = $1.18/sh.
*Total Raised via MLV Jun’14 ATM Sales thru 9-9-2015:
. . . . $25,000,000gr. / 18,311,495 = $1.37/sh.
VI. MLV 8-2015: “Aug’15 AMI Agreement – up to $30mm” http://www.mlvco.com
• Sold 8/7/15-9/9/15: $892,000gr. / 1,091,000sh. = $1.18/sh.
• Sold 9/10/15-10/31/15: $4,294,000gr. / 4,059,478sh. = $1.06/sh.
• Sold 11/1/15-12/10/15: $2,261,000gr. / 1,939,413sh. = $1.17/sh.
• Sold 12/11/15-1/31/16: *NO ATM SALES*, per 1-31-16 10Q iss. 3-9-16
• Sold 2/2/16-4/30/16: *NO ATM SALES*, per 4-30-16 10Q iss. 7-14-16
• Sold 5/1/16-7/14/16: $937,000gr. / 1,876,918sh. = $.50/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $2,513,000gr. / 6,432,439sh. = $.39/sh.
• Sold 11/1/16-12/12/16: $1,174,000gr. / 3,788,346sh. = $.31/sh.
. . . . $12,071,000gr. / 18,849,354 = $.64/sh.
VII. NOBLE 8-2015: “Aug’15 Eq.Dist. Agreement – up to $20mm” http://www.noblelsp.com
• Sold 11/1/15-12/10/16: $2,371,000gr. / 1,925,844sh. = $1.23/sh.
• Sold 12/11/15-1/31/16: $2,857,000gr. / 2,529,434sh. = $1.13/sh.
• Sold 2/1/16-4/30/16: $1,741,000gr. / 4,017,010sh. = $.43/sh.
• Sold 5/1/16-7/14/16: $1,233,000gr. / 2,649,318sh. = $.47/sh.
• Sold 7/15/16-9/8/16: *NO ATM SALES*, per 7-31-16 10Q iss. 9-8-16
• Sold 9/9/16-10/31/16: $1,283,000gr. / 2,988,086sh. = $.43/sh.
• Sold 11/1/16-12/12/16: $496,000gr. / 1,587,909sh. = $.31/sh.
. . . . $9,981,000gr. / 15,697,601 = $.64/sh. '
TOTAL ALL A-T-M SALES – INCEPTION (3-2009) THRU 12-12-2016:
==> $244,552,000gr. / 186,838,341sh. = $1.31/sh.
= = = = = = = = = = = = = = = = = =
O/S WARRANTS & STOCK-OPTIONS A/O 10-31-2016 (10Q pg.14-15):
• WARRANTS: As of 10-31-2016, warrants to purchase 273,280 shares at an exercise price of $2.47 were outstanding and are exercisable through Aug30 2018. These warrants were issued in FY’13 in connection with the Aug.2012 [Oxford] loan agreement, which was paid in full Sept.2012.
• STOCK OPTIONS OUTSTANDING A/O 10-31-2016: 29,674,529 shares at a wgt.avg. exercise price of $1.26.
- - - - - - - - - -
10-31-11 10Q: “During the 6mos. 10-31-11, we sold 6,440,416 shares of our common stock at mkt-prices for gross proceeds of $9,295,000 under the Dec’10 AMI Agreement before deducting commissions and other issuance costs of $260,000”
1-31-12 10Q: “During the 9mos. ended 1-31-12, we sold 16,948,441 shares of our common stock at mkt-prices for gross proceeds of $20,256,000 under the Dec’10 AMI Agreement before deducting commissions and other issuance costs of $482,000. …During Feb.2012, we sold an addl. 5,726,946 shares of common stock at market prices under the Dec’10 AMI Agreement in exchange for aggregate gross proceeds of $5,871,000. As of 2-29-12, gross proceeds of $38,644,000 remained available under our 2 effective shelf registration statements.”
4-30-12 10K: “Under the Dec. 2010 AMI Agreement with MLV …for aggregate gross proceeds of up to $75,000,000… During FY’s 2011 (5’10-4’11) and 2012 (5’11-4’12), we sold 30,098,421 shares of common stock at market prices under the Dec.2010 AMI for aggregate gross proceeds of $40,678,000 before deducting commissions & other issuance costs of $917,000. As of April 30, 2012, aggregate gross proceeds of up to $27,382,000 remained available under the Dec.2010 AMI… Subsequent to April 30, 2012 and through June 30, 2012, we sold 2,752,691 shares of common stock at mkt prices under the Dec.2010 AMI for aggregate gross proceeds of $1,496,000… Under the registered direct public offering dated Sept. 2, 2011, we entered into separate subscription agreements with 3 institutional investors, pursuant to which we sold an aggregate of 6,252,252 shares of our common stock at a purchase price of $1.11/sh. for gross proceeds of $6,940,000 before deducting placement agent fees and other offering expenses of $525,000.”
10-31-12 10Q: “During the 6mos. 10-31-12, we sold 21,310,619 shares… at varying mkt-prices under the Dec’10 AMI Agreement for gross proceeds of $18,215,000 before deducting commissions and other issuance costs of $620,000. From 11-1-12 thru 11-30-12, we sold 9,220,313 shares gross of $7,296,000. As of 11-30-12, aggregate gross proceeds of up to $1,871,000 remained available under the Dec’10 AMI Agreement. As of 11-30-12, gross proceeds of $151,871,000 remained available under 2 effective shelf registration statements.”
1-31-13 10Q/pg.11: “DEC’10-AMI(max=$75mm): During the 9 mos. ended 1-31-13, we sold 31,662,214 shares at varying mkt-prices for gross proceeds of $27,051,000 before deducting commissions/other-costs of $885,000. As of 1-31-13, gross proceeds of up to $330,000 remained available. From 2-1-13 – 3-12-13, we sold 201,154 shares at mkt prices for gross $330,000. As of 3-12-13, we had raised the full amt of gross proceeds available… DEC’12-AMI(max=$75mm): As of 1-31-13, we had not sold any shares. From 2-1-13 - 3-12-13, we sold 3,132,402 shares at mkt prices for gross proceeds of $4,475,000. As of 3-12-13, gross proceeds of up to $70,525,000 remained available.”
4-30-13 10K/pg.F26: Dec’12-AMI(max=$75mm)Agreement – During FY’13, we sold 9,320,675 shares gross proceeds of $13,372,000 before deducting commissions and other issuance costs of $337,000. As of April 30, 2013, gross proceeds of up to $61,628,000 remained available under the Dec’12-AMI. From 5/1/13 – 7/11/13, we sold 7,927,016 shares for gross proceeds of $12,729,000. As of 7-11-13, gross proceeds of $48,899,000 remained available under the Dec’12-AMI. http://tinyurl.com/p58jcbw
7-31-13 10Q/pg.10: During the 3mos ended 7-31-13, we sold 9,617,880 shares at mkt prices under the Dec’12 AMI Agreement for gross proceeds of $15,197,000 before deducting commissions and other issuance costs of $491,000. As of July 31, 2013, gross proceeds of up to $46,431,000 remained available under the Dec’12-AMI. From 8-1-13 – 9-9-13, we sold 3,057,431 shares for gross proceeds of $4,372,000. As of 9-9-13, gross proceeds of $42,059,000 remained available under the Dec’12-AMI.
10-31-13 10Q/pg.10: During the 6mos ended 10-31-13, we sold 15,938,269 at mkt prices under the Dec’12 AMI for gross proceeds of $24,277,000 before deducting costs of $722,000. As of 10-31-13, aggregate gross proceeds of up to $37,351,000 remained available under the Dec’12 AMI.
***NOTE: There is no stmt in the 10-Q regarding AMI Sales subsequent to 10-31-13 (thru 12-6-13) as has been the case in the 10Q’s for years – the assumption being: NO AMI Sales made 11/1/13-12/6/13, a period where O/S shares only went up by 77,149.
1-31-14 10Q/pg.11: During the 9 mos. ended 1-31-14, we sold 31,983,986 shares under the Dec’12/AMI for gross $52,407,000 before deducting commissions & other iss. costs of $1,427,000. As of 1-31-14, gross proceeds of up to $9,221,000 remained available under the Dec’12/AMI. http://tinyurl.com/pxcjocw
4-30-14 10K pg.F25: “During FY14, we sold 33,527,369 shares under the Dec’12/AMI for gross proceeds of $55,424,000 before deducting commissions & other iss. costs of $1,504,000. As of April 30, 2014, aggregate gross proceeds of up to $6,204,000 remained available under the December 2012 AMI Agreement.” http://tinyurl.com/mhva3k3
7-31-14 10Q/Pg5: During the 3mos. ended 7-31-14, we raised $10,000,000 in aggregate gross proceeds from the sale of our 10.50% SeriesE Convertible Preferred Stock under an At Market Issuance Sales Agreement. Pg.11: During the 3mos. ended 7-31-14, we sold 226,700sh. under the Dec’12-2012 AMI Agreement for gross of $435,000 before deducting commissions & other costs of $14,000. As of 7-31-14, gross of up to $5,769,000 remained available under the Dec’12-2012/AMI. On 6-13-14, we entered into an At Market Issuance Sales Agreement (“June2014/AMI”), with MLV, pursuant to which we may sell shares of our common stock through MLV, for aggregate gross proceeds of up to $25,000,000, in registered transactions from our shelf registration statement on Form S-3. As of 7-31-14, we had not sold any shares of common stock under the June 2014 AMI Agreement. http://tinyurl.com/phw6dkp
10-31-14 10Q/Pg12: COMMON: During 6mos. ended 10-31-14, we sold 2,721,535 sh. at mkt prices under the Dec’12-AMI for gross proceeds of $4,326,000 before deducting commissions and other issuance costs of $113,000. As of 12-31-14, gross proceeds of up to $1,878,000 remained available under the Dec’12-AMI. PREFERRED: During 6mos. ended 10-31-14, we sold 402,858 sh. at mkt prices gross proceeds of $10,070,000 before deducting commission and other issuance costs of $552,000. As of 10-31-14, gross proceeds of up to $19,930,000 remained available under the Series E AMI Agreement. http://tinyurl.com/m6ldhg7
1-31-15 10Q/Pg12: COMMON: During the 9mos. ended 1-31-15, we sold 3,983,360 sh. at mkt prices under the Dec’12-AMI for gross proceeds of $6,204,000 before deducting commissions and other issuance costs of $162,000. As of 1-31-15, we had raised the full-amt of gross proceeds available to us under the Dec’12-AMI. PREFERRED:
Jun’14-AMI: On 6-13-14, we entered into an AMI with MLV…up to $25mm… ,000,000, in registered transactions from our shelf registration statement on Form S-3 (File No. 333-180028). During the 9mos. ended 1-31-15, we sold 869,504 sh. at mkt prices under the Jun’14-AMI for gross of $1,193,000 before deducting commissions and other issuance costs of $31,000. As of 1-31-15, gross proceeds of up to $23,807,000 remained available. Subsequent to 1-15-15 and thru 3-12-15, we sold 4,354,954 sh. of common at mkt prices under the Jun’14-AMI for gross of $6,204,000. As of 3-12-15, 2015, gross proceeds of $17,603,000 remained available under the Jun’14-AMI.
PREFERRED: During the 9mos. ended 1-31-15, we sold 405,004 sh. of our Series E Preferred Stock at mkt prices under the Series E AMI Agreement for gross proceeds of $10,121,000 before deducting commission and other issuance costs of $553,000. As of 1-31-15, 2015, gross proceeds of up to $19,879,000 remained available under the Series E AMI. http://tinyurl.com/mwedt8w
4-30-15 10K pg.F25: During FY’15 we sold 9,681,757sh. under the June’14/AMI for $13,544,000, before deducting costs of $352,000. As of 4-30-15, $11,456,000 remained available. Pg.F35: Subsequent to 4-30-15 & thru 7-14-15, we sold 6,534,400sh. for $8,896,000. A/o 7-14-15, $2,560,000 remain. http://tinyurl.com/ocrtkuj
7-31-15 10Q/Pg12+18: During the 3mos ended 7-31-15, we sold 7,538,230sh. Under the JUNE2014 AMI for gross of $10,166,000 before deducting commiss/iss-costs of $275,000. As of 7-31-15, gross of $1,290,000 remained available under the JUNE2014 AMI, as amended. JUNE2014 AMI: subsequent to 7-31-15 and thru 9-9-15, we sold 1,091,508sh. gross of $1,290,000. As of 9-9-15, 2015, we had raised the full amount available under the June2014 AMI. AUG2015 AMI: on 8-7-15, we entered into an At Market Issuance Sales Agreement with MLV, for gross proceeds of up to $30,000,000, 2.5% commission; as of 9-9-15, we sold 752,760sh. under the AUG2015 AMI for gross of $892,000. http://tinyurl.com/pemub47
10-31-15 10Q/Pg13+19: During 6mos ended 10-31-15, we sold 4,812,238sh at mkt prices under MLV AUG2015 AMI for gross=$5,186,000 before deducting commiss/costs of $132,000. As of 10-31-15, gross proceeds of up to $24,814,000 remained available. Subsequent to 10-31-15, thru 12-10-15, we sold 1,939,413sh for gross=$2,261,000. As of 12-10-15, $22,553,000 remained.
On 8-7-15, we entered into an “Equity Dist. Agreement” with Noble Life Science Partners, pursuant to which we may sell shares for gross proceeds of up to $20,000,000 (commission=2.5% of gross). As of 10-31-15, we had not sold any shares… Subsequent to 10-31-15 & thru 12-10-15, we sold 1,925,844 shs. at mkt prices for gross=$2,371,000. As of 12-10-15, gross of up to $17,629,000 remained available.
On 10-30-15, we entered into a Common Stock Pur. Agreement with EASTERN CAPITAL Ltd: sold 18,518,518shs @1.08/sh for gross=$20,000,000 before deducting issuance costs of $1,000.
10Q: http://tinyurl.com/zdbo9rv [10-30-15: Kenneth Dart (Eastern Capital) acquires 13.1% stake (30,106,945sh.) in PPHM http://tinyurl.com/24qctos ]
1-31-16 10Q/Pg13: During 9mos ended 1-31-16, we sold 6,751,651sh at mkt prices under MLV AUG2015 AMI for gross=$7,447,000 before commiss/costs =$190,000. As of 1-31-16, gross of $22,553,000 remained available. Subsequent to 10-31-15 thru 12-10-15, we sold 1,939,413sh for gross=$2,261,000. As of 12-10-15, $22,553,000 remained.
NOBLE Eq-Dist-AMI: during 9mos. ended 1-31-16, we sold 4,455,278shs. for gross of $5,228,000, commissions=$131,000. As of 1-31-16, $14,772,000 remained… http://tinyurl.com/hdgto9y
4-30-16 10K pg.F24+25: NOBLE Eq-Dist-AMI - FY’16 sales: 8,472,288sh. For gross=$6,969,000. As of 4-30-16, gross remaining=$13,031,000. Pg.F35/SUBSEQUENT EVENTS: NOBLE/EQ 5/1/16-7/14/16, sold 2,649,318sh. for gross=$1,232,000, a/o 7-14-16, $11,799,000 remain available. MLV AUG2015 AMI: 5/1/16-7/14/16, sold 1,876,918sh. for gross=$937,000, a/o 7-14-16, $21,616,000 remain available. 10K: http://tinyurl.com/zgognwz
7-31-16 10Q p.12: NOBLE Eq-Dist-AMI: 5/1/16-7/31/16, sold 2,649,318sh. for gross=$1,233,000, a/o 7-31-16, $ 11,798,000 remain available. MLV AUG2015 AMI: 5/1/16-7/31/16, sold 1,876,918sh. for gross=$937,000, a/o 7-31-16, $21,616,000 remain available. Pg16/SUBSEQUENT: PREFERRED/SeriesE: 8/1/16-9/8/16, we sold 68,910sh. for gross=$1,601,000; a/o 9-8-16, $9,134,000 remained available. 10Q: http://tinyurl.com/jmy77g3
10-31-16 10Q p.13: MLV AUG2015 AMI $30mm max, 2.5% commiss): 6mos ended 10-31-16, sold 8,309,357sh. for gross=$3,450,000. Pg17/SUBSEQUENT: 11/1/16 - 12-12-16, sold 3,788,346sh. for gross=$1,174,000. A/o 12-12-16, $17,929,000 remain available. NOBLE AUG2015 Eq-Dist-AMI($20mm max, 2.5% commiss): 6mos ended 10-31-16, sold 5,637,404sh. for gross=$2,516,000. Pg17/SUBSEQUENT: 11/1/16 - 12-12-16, sold 1,587,909sh. for gross=$496,000. A/o 12-12-16, $10,019,000 remain available.
= = = = = = = = = = =PREFERRED STOCK:
...2-5-14: PPHM Announces Public Offering of 10.5% Series E Convertible Preferred Stock http://tinyurl.com/lkxsna6
...2-11-14: PPHM Raises net $16.2mm selling 700k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/jwmsnsk (8-K)
...6/14/14-7/14/13: PPHM Raises net $9.5mm selling 400k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/mhva3k3 (4-30-14 10-K pg. F-34; $20mm gross remaining)
PREFERRED SHARES:
4-30-15 10K/p.F26: On 6-13-14, we entered into an At Market Issuance Sales Agreement with MLV, pursuant to which we may issue & sell shares of our Series E Preferred Stock through MLV, as agent, for gross proceeds of up to $30,000,000, in registered transactions from our Jan2014 Shelf. During FY’2015, we sold 799,764 shares of our Series E Preferred Stock at mkt prices for gross $19,205,000 before deducting commissions/costs of $1,002,000. As of 4-30-15, gross proceeds of up to $10,795,000 remained available under the Series E AMI Agreement.
NOTE: One of the holders of Preferred is KENNETH DART (EASTERN CAPITAL), who in a 2-13-15 SG14G ( http://tinyurl.com/k4nsfuu ) reported their 240,000sh. Preferred holdings as 2,000,000 common shares (SHARED VOTING POWER: 9,921,760,% OF CLASS REPRESENTED: 5.4%).
“which has a liquidation preference of $25.00/sh. and a conversion price of $3.00/sh.” 2,000,000 / 240,000 = 8.333
10Q: http://tinyurl.com/j2u2bjk
**7-31-16 10Q(iss. 9-8-16): 8/1/16-9/8/16, we sold 68,910sh. for gross=$1,601,000; a/o 9-8-16, $9,134,000 remained available.
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-16 iss. 7-14-16 http://tinyurl.com/zgognwz PR: http://tinyurl.com/h8eqtg5 (Cash 4-30-16=$61.4mm)
Latest 10Q 10-31-16 iss. 12-12-16 http://tinyurl.com/j2u2bjk PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1003834 (Cash 10-31-16=$49.1mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
Cash Burn for Q2(qe/10-31-16): $2.6mm (down from $9.6mm for Q1/7-31-16).
Q2 Avid Revs (q/e 10-31-16): $23.4mm (curr. B/L = $73mm, guidance FY17(fye 4-30-16: $50-55mm).
Cash at 10-13-16: $49.1mm (up from Q1/7-31-16 $44.2mm).
O/S Shares at 10-31-16: 251,765,279 (up from Q1/7-31-16 241,456,721).
O/S Shares at 12-8-16: 257,141,534 (from 10Q cover pg. - http://tinyurl.com/j2u2bjk )
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q2(qe 10-7-31-16), per the 10-31-16 10-Q ( http://tinyurl.com/j2u2bjk ) issued 12-12-16.
• Total Revs since May’06: ($202.5mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $229.2mm
• 12-12-16: FY'17 (May'16-Apr'17) Avid revs guidance $50-55mm (Committed B/L=$73mm).
• Deferred-Revs at 10-31-16 total $18.0mm, DOWN from $21.5mm at 7-31-16.
• Cust.Deposits at 10-31-16 total $26.9mm, UP from $21.7mm at 7-31-16.
• Inventories at 10-31-16 total $25.9mm, UP from $25.3mm at 7-31-16.
• Avid’s Gross-Profit over last 4 qtrs: $22.3mm on revs of $54.4mm (GP%=41%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.25)
FY16Q2 10-31-15 11,701,000 (Q1+Q2: 24,007,000 10Q pg.26)
FY16Q3 1-31-16 15,086,000 (Q1+Q2+Q3: 39,093,000 10Q pg.27)
FY16Q4 4-30-16 10,112,000 (FY'16: 49,205,000 10K pg.39)
FY17Q1 7-31-16 9,607,000 (from 10Q pg.22)
FY17Q2 10-31-16 2,565,000 (Q1+Q2: 12,172,000 10Q pg.24)
Thx/CP: 1st (of 3) NCCN Trial hits ClincalTrials.gov.
That’s this one:
#1: Ph1/HepC-Related Hepatocellular (Bavi+RAD+Sorafenib), MOFFITT CANCER CENTER - PI: Jessica Frakes, MD
"A Phase I Trial of Sorafenib & Bavituximab + Stereotactic Body Radiation Therapy (SBRT) for Unresectable Hepatitis C Associated Hepatocellular Carcinoma"
=> 9-8-16/CC/JoeShan: “The 1st award is for a Phase I trial of Sorafenib [Bayer’s Nexavar] and bavituximab + radiation in Hepatocellular Carcinoma. This will build on a previously reported investigator sponsored Phase II trial of bavituximab & sorafenib in Liver Cancer. [3-25-15/Dr.Adam.Yopp(UTSW)/Ph2data, N=38: http://tinyurl.com/opkh5qy ]”
- - - - - - -
9-6-16: NCCN to Initiate 3 Bavi-Trials Early’17 (Moffitt,MassGEN,JohnHopkins) http://tinyurl.com/gutgwb5
1. Ph1/HepC-Related-Hepatocellular/MOFFITT (Bavi+RAD+Sorafenib)
2. Ph1-2/Glioblastoma/MASS-GEN (Bavi+RAD+Merck’s Temodar)
3. Ph2/Head+Neck/JOHN-HOPKINS (Bavi+Merck’s Keytruda)
NCCN PR:
https://www.nccn.org/about/news/newsinfo.aspx?NewsID=785
”NCCN is excited to initiate 3 studies by accomplished investigators at Member Institutions that will explore the effect of this novel immunotherapy in 3 different cancers with significant unmet need,” said Robert C. Young, MD, Interim VP, NCCN ORP.
= = = = = = = = = = = = =
12-12-16: First Bavituximab clinical trial with NCCN on ClinicalTrials.gov:
Sorafenib & Bavituximab Plus SBRT in Unresectable Hepatocellular Carcinoma”
Entered 12-8-16: NCT02989870 https://clinicaltrials.gov/ct2/show/NCT02989870
This study involves a course of radiation to up to 5 tumors in the participant's liver followed by systemic therapy. (Treatment using substances that travel through the bloodstream, reaching and affecting cells all over the body.) The type of radiation is called stereotactic body radiation therapy (SBRT). The purpose of this study is to compare the effects, good and/or bad, of different doses of SBRT given along with the systemic therapies, sorafenib and bavituximab. The researchers want to see which dose of radiation will work best in stimulating the immune response and provide local control to the participant's liver. The usual treatment for hepatocellular carcinoma that is unresectable can be transarterial therapy, sorafenib alone and/or clinical trial.
Sponsor: H. Lee Moffitt Cancer Center & Res. Institute
Collaborator: National Comprehensive Cancer Network
Info provided by (Responsible Party): H. Lee Moffitt Cancer Center & Res. Institute
If I make an error in a post (whether I find it or somebody else points it out) and the 15min Edit time hasn't passed, I GO FIX IT DIRECTLY and make it right. No need to post, "I fixed my error" at the bottom.
I’ve been thinking about Avid Q2/Revs coming Monday… Per the 9-8-16/CC, they will "be in excess of $20mm". The question is, How Much In Excess?
Ponder the last 6 months revs & gross-profit history...
AVID PROFITABILITY (GROSS) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 “we remain on track to gen. revs in excess of $20mm in Q2.”

From the 9-8-16/PR-CC, Q2(10-31-16) Revs coming Monday will "be in excess of $20mm". Peregrine's Op.Cash.Burn for Q1(7-31-16) was $9.6mm (vs. Net Loss of $12.4mm) - see http://tinyurl.com/jydtkoy . With Avid's GP% running around 47% and depending on how much "in excess" of $20mm revs are, it'll be interesting how close we come to Positive Cash Burn for Q2. We'll find out Monday around 4:30pm!
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q1(qe 7-31-16), per the 7-31-16 10-Q ( http://tinyurl.com/jmy77g3 ) issued 9-8-16.
• Total Revs since May’06: ($179.2mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $205.8mm
• 9-8-16: FY'17 (May'16-Apr'17) Avid revs guidance $50-55mm (Committed B/L=$71mm).
• Deferred-Revs at 7-31-16 total $21.5mm, UP from $10.0mm at 4-30-16.
• Cust.Deposits at 7-31-16 total $21.7mm, DOWN from $24.2mm at 4-30-16.
• Inventories at 7-31-16 total $25.3mm, UP from $16.2mm at 4-30-16.
• Avid’s Gross-Profit over last 4 qtrs: $19.2mm on revs of $40.6mm (GP%=47%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45% <=low due to 3rd-party testing delays.
FY17Q2 10-31-16 “we remain on track to gen. revs in excess of $20mm in Q2.” <=to report 12-12-16.

Remember, Dr.Wolchok spoke about Bavi in the 11-14-16/PR (SITC’16, Joint MSKCC & PPHM Poster, “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”)…
11-14-16: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model," stated Dr. Jedd Wolchok. "It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact." See: http://tinyurl.com/js3fca4
As well as Dr. Wolchok's Sloan Kettering associate, Dr. Merghoub:
11-14-16: "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model," said Dr. Taha Merghoub, PhD, co-director of the Ludwig Collaborative Laboratory at MSK. "We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers." See: http://tinyurl.com/js3fca4
Note: FULL SITC’16 ABSTRACT #199: http://bit.ly/2dHTEVn
Only via my Googling capabilities. I do think we'll get a Gerber/IASLC'16 New-Biomarker PR, and hopefully they'll put his Poster image on the website.
Dec7/WED: WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer, Vienna, Austria
…2:30-3:45pm (8:30-9:45am USA/ET) UTSW’s Dr. David Gerber to present addl. Biomarker data from Ph3.SUNRISE Trial: “A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel” See: http://tinyurl.com/z8cq8vx
= = = = = = = = = = = =
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
http://wclc2016.iaslc.org
IASLC = Intl. Association for the Study of Lung Cancer - http://iaslc.org
WCLC = World Conference on Lung Cancer (IASLC)
Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with high complement activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [median OS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded independent validation.
TODAY/12-7-16 8:30-9:45amET IASLC/WCLC'16: UTSW’s David.Gerber, ADDL. Biomarker Data from Ph3.SUNRISE Trial in Vienna. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
FROM DR. GERBER's ABSTRACT ( http://tinyurl.com/z8cq8vx ):
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...”
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
DR. GERBER’s ABSTRACT CONCLUSION:
Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded independent validation.
MORE See: http://tinyurl.com/z8cq8vx
10-10-16: ESMO’16 Topline/Ph3.SUNRISE Data: INITIAL Biomarker data, B2GPI/200-240(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
= = = = = = = = = = = =KNOWN DECEMBER:
Dec7/WED: WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer, Vienna, Austria
…2:30-3:45pm (8:30-9:45am USA/ET) UTSW’s Dr. David Gerber to present addl. Biomarker data from Ph3.SUNRISE Trial: “A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel” See: http://tinyurl.com/z8cq8vx
Dec7/WED 10am: MSKCC’s Dr. Jedd Wolchok lecture at Wistar/Philadelphia on “Combination Checkpoint Blockade" (North40000/Eb0783/Wook attending!)
Dec8/THU: 39th San Antonio Breast Cancer Symposium”
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology) poster: “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors” See: http://tinyurl.com/zb5sopc
Dec12/MON/AfterMkt: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Mem.Sloan’s Jedd.Wolchok Video/July2014: “Immunotherapy for Metastatic Melanoma, PD-1 Clinical Trials”… Dr. Wolchok (1:42), “We will not rest until every patient can have a durable response.” http://cyberspaceandtime.com/kvWeW8Zt0sU.video (1:49)
TOMORROW:
12-7-16/WED 10am: JEDD D. WOLCHOK, MD/PhD(Mem. Sloan Kettering)
Lecture: ”Combination Checkpoint Blockade" (free to public)
Location: The Wistar Institute, Philadelphia (Jedd D. Wolchock: Chief of Melanoma & Immunotherapeutics Service, MSKCC)
https://www.wistar.org/events/2016/12/combination-checkpoint-blockade
...We don’t know if Dr. Wolchok will mention Peregrine/Bavi in his talk (North40000/Eb0783/Wook will let us know!), but RECALL that on 11-14-16, Dr. Wolchok’s MSKCC Lab Team, jointly with PPHM Scientists, presented a poster at SITC’16 on Triple Combo Rad+Bavi+aPD1 vs. Melanoma:
...11-14-16/SITC’16: Dr. Jedd Wolchok states, “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact.”
#199/SITC’16/11-11-16(MSK+PPHM): Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” See: http://tinyurl.com/js3fca4
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
= = = = = = = = = = = = = = = =KNOWN DECEMBER:
Dec7/WED: WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer, Vienna, Austria http://tinyurl.com/z8cq8vx
…2:30-3:45pm UTSW’s Dr. David Gerber to present addl. Biomarker data from Ph3.SUNRISE Trial: “A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
Dec7/WED 10am: MSKCC’s Dr. Jedd Wolchok lecture at Wistar on “Combination Checkpoint Blockade"
Dec8/THU: 39th San Antonio Breast Cancer Symposium” http://tinyurl.com/zb5sopc
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology) poster: “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors”
Dec12/MON/AfterMkt: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
CONCLUSION SECTION FROM THE JOINT MSKCC/PPHM SITC’16 POSTER:


11-14-16/SITC'16: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
http://tinyurl.com/js3fca4
= = = = = = =
Oct9-11 2017: “EACR Conf. - Defense is the Best Attack: Immuno-Oncology Breakthroughs”, Barcelona
https://www.eacr.org/meeting/defence-is-the-best-attack-immuno-oncology-breakthroughs
EACR = Eur. Association for Cancer Research https://www.eacr.org
Keynote Speaker: Dr. Jedd Wolchok, MSKCC
12-7-16/WED/10am: Jedd Wolchok/MSKCC Lecture: "Combination Checkpoint Blockade"
12-7-16/WED 10am: Jedd D. Wolchok, MD/PhD(Mem. Sloan Kettering)
Lecture: "Combination Checkpoint Blockade" (free to public)
Location: The Wistar Institute, Philadelphia (Jedd D. Wolchock: Chief of Melanoma & Immunotherapeutics Service, MSKCC)
https://www.wistar.org/events/2016/12/combination-checkpoint-blockade
We don’t know if Dr. Wolchok will mention Peregrine/Bavi in his talk (North40000/Eb0783/Wook will let us know!), but RECALL that on 11-14-16, Dr. Wolchok’s MSKCC Lab Team, jointly with PPHM Scientists, presented a poster on Triple Combo Rad+Bavi+aPD1 vs. Melanoma:
...11-14-16: Dr. Jedd Wolchok states, “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact." See: http://tinyurl.com/js3fca4
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
Dec7/WED: WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer, Vienna, Austria http://tinyurl.com/z8cq8vx
…2:30-3:45pm UTSW’s Dr. David Gerber to present addl. Biomarker data from Ph3.SUNRISE Trial: “A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
Dec7/WED 10am: MSKCC’s Dr. Jedd Wolchok lecture at Wistar on “Combination Checkpoint Blockade"
Dec8/THU: 39th San Antonio Breast Cancer Symposium” http://tinyurl.com/zb5sopc
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology) poster: “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors”
Dec12/MON??: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Thx JDM, will add: 4-11-17/J.Hutchins, Immune Profiling World-Congress'17
Apr10-12 2017: “Immune Profiling World Congress 2017”, WashDC
Part of “World Vaccine Congress Washington 2017” (1 of 8 Co-Conf’s)
“The Immune Profiling World Congress USA brings together experts to discuss profiling of the immune system, how using high throughput technologies can aid therapeutic design & treatment strategies in infectious & non-infectious diseases, including cancer.”
http://www.terrapinn.com/conference/world-vaccine-congress-washington/index.stm
4-11-17/Day1 12:10pm: Dr. Jeff Hutchins (VP/PreClinRes., PPHM)
“Increasing the Benefits of Immune Checkpoint Therapies Through Increased Tumor Infiltrating Activated T-Cells”
*Expanding the responding population
*Sharing pre-clinical & translation data
- - - - - -
Chair: Dr Alison Deckhut Augustine, Branch Chief Basic Immunology, NIAID/NIH
DAY1 SPEAKERS 4-11-17(10):
...Bali Pulendran – Emory
...Dr Masahide Yano - FDA
...Dr Jeff Hutchins – Peregrine
...Dr Ali M. Harandi – Univ. of Gothenburg
...Dr Mahesh Yadav - Genentech
...Giuseppe Del Giudice - GSK Vaccines
...Mark M Davis - Stanford Univ.
...Alessandro Sette - La Jolla Infectious Disease Inst.
...Peter W. Che - Repertoire Genesis, Inc
...Dr Wayne C Koff - Human Vaccines Project, Intl. Aids Vaccine Initative
DAY2 SPEAKERS 4-12-17(4):
...Dr Roy Baynes (PD-1 Antibodies) – Merck
...Mr Bruno Gomes - iTeos Therapeutics
...Mr Lei Zheng - Johns Hopkins
...Dr Rakesh Dixit - MedImmune

Known Upcoming Events
Dec7: WCLC’16 - IASLC's 17th World Conf. on Lung Cancer, Vienna, Austria http://tinyurl.com/z8cq8vx
…2:30-3:45pm UTSW’s Dr. David Gerber to present addl. Biomarker data from Ph3.SUNRISE Trial: “A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
Dec8: 39th San Antonio Breast Cancer Symposium” http://tinyurl.com/zb5sopc
...Dr. Bruce Freimark(Dir.Res/Preclin.Oncology) poster: “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors”
Dec12: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Jan24-26/Avid/Table#22: WCBP’17: 21st Symposium on Interface of Reg. & Analytical Sciences, WashDC http://www.casss.org/page/WCBP1700 (Theme: “Convergence & Harmonization”)
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & I-O”, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Bruce Freimark(Dir.Res/Preclin.Oncology), “Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
.
.
= = = = = = = = = = = = = = = = = = = = =PRIOR:
Sep25-28: 2nd AACR-CRI Intl. Cancer Immunotherapy Conf., NYC http://tinyurl.com/hxtbdfm <=9-27-16/PR re: B019*: http://tinyurl.com/zy9yv78 (Bavi+PD1+LAG3 TNBC)
...Sep26 5:15-7:45pm: Michael J. Gray(PPHM Scientist) poster *B019, “LAG3 is an Immunotherapeutic Target in Murine Triple- Breast Cancers, Whose Activity is Significantly Enhanced in Combo with PS-Targeting Antibodies”
...Sep26 5:15-7:45pm: Dr. Raymond Birge(Rutgers) & PPHM poster B119, “Characterization of a PS, TAM Receptor (Tyro3, Axl, Mertk), PDL1 Axis in Breast Cancer”
Oct4-7/Avid/Booth#716: IBC's BioProcess Intl. Conf., Boston http://www.ibclifesciences.com/BPI/overview.xml & http://tinyurl.com/zyblds7
...10-5-16/8-8:30am: Peter Gagnon (Avid's VP/Process-Svcs), “Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”
Oct7-11: 41th ESMO European Cancer Congress, Copenhagen, Denmark http://tinyurl.com/jxdppyo (Oral Pres. of Topline Data from Ph3 SUNRISE trial, incl. initial Biomarker analysis)
...10-10-16 9:15-9:30am: David R. Spigel (CSO/Dir./Sarah Cannon Res. Inst., Nashville) Proffered Oral Presentation, “Top-line Results from Ph3/SUNRISE..." (Senior-Author: David Gerber/UTSW)
......10-10-16 ESMO’16 Topline/Ph3.SUNRISE Data: Biomarker B2GPI/200-240(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
...10-9-16 1:00-2:00pm: Jeff Hutchins(VP/PreClin.Res), “Antibody Mediated Blockade of PS Improves Immune Checkpoint Blockade...”
Oct13/10am: Annual SHM, Avenue of the Arts Hotel, Costa Mesa – Final Proxy: http://tinyurl.com/gsrmgs2 - ATTENDEE Reports: http://tinyurl.com/jx7ouay
Oct22: AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok - Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data: ”Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing PS Expression on the Tumor Surface”
Nov9-13: (SITC) Society for Immunotherapy of Cancer 31st Annual Meeting, Natl-Harbor MD => See posters & Dr. Wolchok's comments: http://tinyurl.com/js3fca4
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (Memorial Sloan Kettering) to be presented” (per 9-8-16/Ccall/Hutchins)
...I. Nov11 12:15-1:30pm: “PS Targeting Antibody in Combo w/Checkpoint Blockade & Tumor Rad. Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC
...II. Nov12 11:45-1pm: “Antibody Targeting of PS Enhances the Anti-Tumor Responses of Ibrutinib & anti-Pd-1 Therapy in a Mouse Triple Neg. Breast Tumor Model” - Jian Gong, PhD, PPHM
...III. Nov11 12:15-1:30pm: "Mabs Targeting PS Enhance Combinational Activity of the the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD, PPHM
Catching up w/you. YES, that is the ABSTRACT for Dec7/IASLC/WCLC'16 Gerber/Sanborn/Biodesix/PPHM "Addl. Sunrise Biomarker Data"...
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
------
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6 [Search/Topic=Bavituximab]
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with high complement activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [median OS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded independent validation.
MORE: http://tinyurl.com/z8cq8vx
JDM, the Dec7 IASLC Gerber/Sanborn/Addl-BioM’s poster includes Biodesix’s CTO, Dr.Heinrich Roder. Plus, look at something we missed: the Nov4-6 2016 “ ESMO Symposium on Immuno-Oncology” presentation by Drs. Gerber, Sanborn, H.Roder, etc – about “Proteomic Signature Analysis” (Biomarker) of Ph.3 SUNRISE data. Basically the same author list as the upcoming Dec7 “New SUNRISE Biomarker” pres. at IASLC/WCLC’16...
= = = = = = = = = =
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ]
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
= = = = = = = = = = = = =
12-7-16/IASLC/WCLC’16: UTSW’s David.Gerber to Present addl. Biomarker Data from Ph3.SUNRISE Trial. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
=>11/30/16 update: ABSTRACT published – see below.
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...”
SEE: http://tinyurl.com/z8cq8vx
12-7-16/IASLC/WCLC: UTSW’s David.Gerber to Present addl. Biomarker Data from Ph3.SUNRISE Trial. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
=>11/30/16 update: ABSTRACT published – see below.
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...”
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
http://wclc2016.iaslc.org
IASLC = Intl. Association for the Study of Lung Cancer - http://iaslc.org
WCLC = World Conference on Lung Cancer (IASLC)
Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with high complement activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [median OS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded independent validation.
= = = = = = = = = =
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ]
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
= = = = = = = = = =
10-10-16/PR: ESMO’16 Topline/SUNRISE; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.
10-10-16: “Peregrine Pharmaceuticals Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at European Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Additional Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Additional patient sample testing and analysis is ongoing and may result in other biomarkers of importance.
** Data presented at ESMO demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approximately 30% of randomized patients) achieved a statistically significant, 5.5-month improvement (13.2 months vs. 7.7 months) in median overall survival (OS) as compared to patients in the control group with the same range of B2GP1 levels [p = 0.049; hazard ratio (HR) = 0.67].
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP of Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific and medical conferences as the more results become available. It is not uncommon in the cancer field for therapeutic candidates to suffer clinical trial setbacks as researchers continue to learn more about the most appropriate patient populations for those drugs. In this landscape, biomarkers play an increasingly important role in helping identify specific patient characteristics that may impact responses to a treatment. This has been seen historically with targeted cancer treatments, as well as more recently with checkpoint inhibitors including PD-1 inhibitors. We look forward to identifying the equivalent markers for bavituximab that will help guide its clinical development.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
- - - - - - - - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
10-10-16 ESMO’16 Topline/Ph3.SUNRISE Data: Biomarker B2GPI/200-240(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") - Excellent Exosome info: http://www.exosome-rna.com
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
...“relates to assays that are able to detect small amts of PS+ Exosomes in a patient's blood sample as a way to detect cancer at a very early stage of development.”