Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I hope you are right. But I do know that as far as my investments and speculations are concerned, my hopes are often dashed.
Delays happen because the real world presents continuing difficulties. Anyone who thinks those can all be anticipated has never run a complex project.
There is a big difference between planning and executing a project that is an instance of something that has been done many times before and a project involving major novelty. I think a close examination of NNVC's technology would reveal astonishing novelty, such that each thing done differently leads to unpredictable extra time. It is impossible to accurately predict ahead of time how quickly you can solve a problem you did not know was going to be presented to you.
Have you met every professional goal and timeline?
In my experience, both in my personal life and in the world of business and investing, unforeseeable negative events outnumber unforeseeable positive events by about 10 to 1.
They have the money if everything goes almost perfectly with no significant delays or setbacks.
But unfortunately the past history of NNVC is one long woeful tale of significant delays and setbacks.
I fully agree that a great deal of the delay can be explained by events that were outside of management's control and that were largely unforeseeable. But what's special about now? Surely in the next few years there will be unpredictable negative events that are outside of management's control.
Nanoviricides through the Wayback Machine.
Bunny's recent post inspired me to do a little web crawling.
From DaBoze:
I cannot recall a single instance where the NNVC management has not produced the results they predicted. In the long run that's all that will matter.
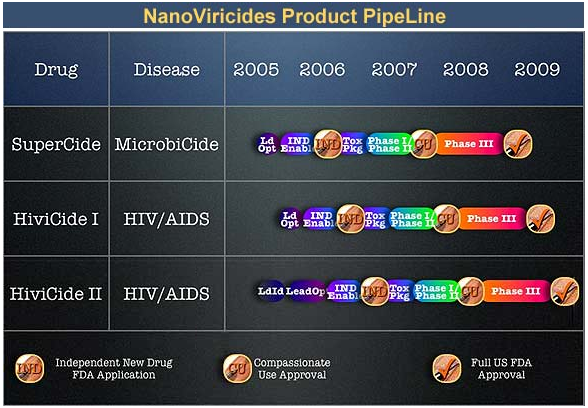
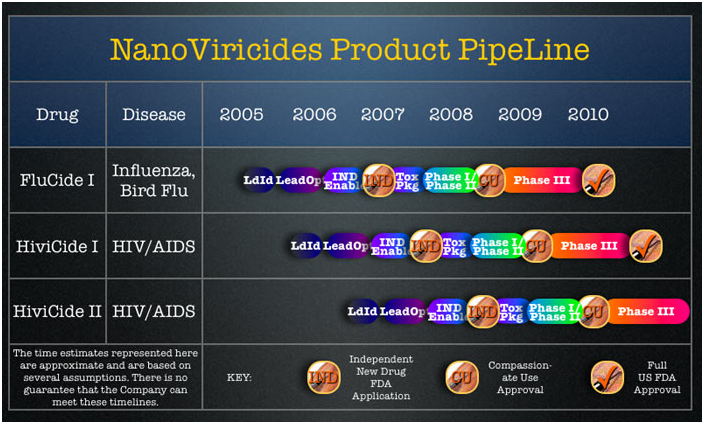


If I were married and bought NNVC stock I think I'd try to hide that fact from my wife -- at least until after phase I/IIa trials are completed.
You are right. I didn't notice that brief plunge down to 0.41. I missed my chance to sell at the bottom, although I'm sure I'll have another opportunity.
Nope, he's still 3 cents away from vindication.
Nope, I always sell at bottoms. I haven't sold, so the bottom isn't in yet.
Because the numbers are for oncology, along with a lot of other numbers much higher.
In fact the number you got from the misquote was actually $23K as an average cost, not $26. The Tufts study said $26. Go look that one up.
Me, I'm just delighted to find that I've invested in the only drug on earth where the most expensive part of the path to approval, phase 3 trials, is irrelevant.
Lucky me.
By the way, I have a copy of the 2008 Cutting Edge Information study and I know exactly what it says. Amazing what a collage professor can access.
And, now you know that even $13,000 is too much.
So your estimate is already more than twice that of Puffer's. You two had better fight this one out.
Ha ha ha! It is YOU, not me, who keeps dragging in the costs of oncology trials. You said the cost of oncology trials is irrelevant. I agree. So stop bringing them up.
The original study you cited was for oncology costs
You are the one who first said I was greatly overstating the costs of doing phase 3 trials. If the cost of phase 3 trials is irrelevant, why did you make such a big deal about it?
It was you who pounced upon an off hand remark and made a mountainous issue out of it, accusing me of being a liar. The original quote:
I did assume that the costs will only be half the average. (Plenty of phase III trials cost $45,000 per patient or more.)
You keep dragging up oncology, when even for the expensive trials in that Manhattan Institute paper none of the drugs were for oncology. And I never claimed that influenza studies would cost $47,000 per patient. I just said that there were types of phase 3 studies for other conditions that did cost that much. Who's doing the fabricating here?
Good grief. Table 3, for obesity drugs. The specific drugs: Lorqess, Qnexa, Contrave. Costs are broken down by phase.
Also shown are the number of subjects per trial. If you can divide you can get the costs per patient for phase 3. Hint: It's $47,000.
Table 4, for GLP analogs in diabetics. The specific drugs: Byetta, Bydureon, Victoza, Taspo. Costs are broken down by phase.
Also shown are number of subjects per trial.
Jeesh, do you have to be spoon fed?
First, I have a correction to make. I got my $26,000 number from a tertiary source that misquoted the original paper. Here is the original paper:
"Clinical Operations: Accelerating Trials, Allocating Resources &
Measuring Performance"
Cutting Edge Information, September 2006, 182 pages
Unfortunately that paper does not seem to be available on the internet. This is not surprising, given that it is copyrighted material that apparently costs $6995. Here is a review/summary of that paper:
http://firstclinical.com/journal/2006/0612_Clinical_Operations.pdf
According to that review, it is *Phase II* trials that have an average per subject cost of $26,050. The average per subject cost of Phase III trials is a mere $19,320. From the review:
"Per-subject costs vary substantially by therapeutic area, in Phase III ranging from $15,000 for analgesia and rheumatology studies to $42,000 for CNS and psychiatry studies."
So you and Puffer are assuming that the per subject cost of Phase III trials for Flucide will be a mere 40% of the average of the cheapest type of study covered in that paper. *Could* they be done that cheaply? Maybe. Do I see this as a high probability outcome? No.
Your accusations of "complete fabrication" are premature, although eagerly seized by Puffer. I got that from
http://www.manhattan-institute.org/html/fda_05.htm
If you divide the costs by the people you can see that they are using an average phase 3 per person cost of $47,000.
It may be that the Manhattan Institute is fabricating the data, but I didn't.
That's a mighty small number of patients for a phase III trial. Except for exotic diseases where the total number of patients available for trials is tiny, typically phase III trials have a minimum of 1000 patients, with 2000 or more (sometimes a lot more) being common.
If there are only 500 patients in the phase III trial we'll just have to hope that the results are so spectacular that even the FDA agrees they are statistically significant. And the FDA loves to find reasons not to approve drugs.
I did assume that the costs will only be half the average. (Plenty of phase III trials cost $45,000 per patient or more.) A budget of "a few million" assumes you're only going to spend $3000 per patient. That sounds an awful lot like wishful thinking.
So pointing out obvious risks in a highly speculative stock is a "new line of attack". My, my, how defensive you are.
Refrigerators and stoves are built off site, that still doesn't stop contractors from having inordinate delays in putting a kitchen together. And who says that building some new piece of technology off site means there won't be any screw ups and delays in its construction? Now maybe NNVC will surprise us and get this big project done on schedule. I hope so. But delays in construction are commonplace, even when the project doesn't involve any advanced technology; it's not like I'm inventing some exotic and contrived risk.
It must be utterly baffling to you how NNVC can be at 0.44 a share. After all, it's nearly a sure thing! With better than even odds of hitting $100 in two years! Strangely Warren Buffet isn't buying it. George Soros isn't buying it. David Einhorn isn't buying it. There are 6.5 billion people out there too stupid to buy Nanoviricides stock and increase their bank account by a factor of 200! How can this be?
Or maybe, just maybe, the risks are greater than you think.
That sounds like a real low ball estimate. The average cost of a phase III trial is $26,000 per patient. Let's assume that because flu trials are relatively fast that the cost per patient is half that -- $13,000 per patient. Remember that there are many fixed costs that are independent of the length of the trial. With a minimal 1000 patients (most phase III trials have more patients than that) the cost is $13,000,000. I would say that's a realistic estimate, and I wouldn't be surprised if the actual cost turned out to be somewhat higher than that.
Those are figures on the IV formulation (for hospitalized patients), not oral. I would expect Flucide oral to sell around $150-$200/dose.
What has big pharma contributed lately?
Well, more precisely, there's enough money to get through phase I and something called phase IIa. The latter is not a full phase II trial, it's a small scale trial intended to give some evidence of efficacy but it won't be as statistically significant as a larger trial would be. Hopefully the results will be good enough to entice some big pharma companies to offer partnerships, although nanopatent doesn't care because he doesn't want to make a deal with the devil anyway.
A big risk factor in all this is the manufacturing facility. If there are significant problems and delays in getting it built and certified that will blow a big hole in the budget. When I think how many delays can happen if you just want a contractor to put in a new kitchen I can only imagine how many "Gotcha's" are waiting for us in this project.
So buy some shares of Pfizer and Merck. Then their ill gotten gains will go to your pockets too.
Your calculation assumes no increase in the number of shares between now and then. I think that's very optimistic. Oh, if only every start up I invested in never had to issue new shares! How rich I'd be!
From Dr Diwan, via Dr. Seymour:
"I estimate that we could charge at least $2200/dose (hopefully only one dose needed) which is what HHS paid Peramivir for its drug that didn't work (under Emergency Use Authorization) during the 2009-10 flu emergency. Because of the DRG payment system, either after US approval or under compassionate use authorization, I think we could charge as much as $5000/dose ..."
So NNVC will charge what the market will bear, just like all those big bad pharmaceutical companies do.
In a recent presentation Dr. Seymour stated that they could complete PhaseI/IIa combined in 4 to 6 weeks depending on the number of rooms they could get. That would probably take about $1M. Assuming success, that is the point that big pharma descends, opening up all kinds of options for additional funding. So you see, for about $1M worth of clinical trials we get to where we need to be.... a whole new ballgame.
Well then dump your shares because if NNVC succeeds and its share price is at $100 (as all you boosters fantasize) it will then be big, bad pharma itself. With projected Flucide prices of $2000 per dose NNVC will be extracting plenty of pounds of flesh for its own. (And I will add that only the ability to get such outrageous prices will support a stock price in the $100 range.)
Now of course the pharmaceutical companies will try to make a deal that's as good for themselves as possible. And if Flucide looks promising NNVC will be able to pick its partner and try to get the best deal for itself. This is called capitalism. If you oppose that, get out of the stock market.
Jeesh, if things were as cheap and easy and you think we'd already have Flucide on the market by now.
The only sources of money that don't involve giving away part of the profits in license agreements require vast amounts of further share dilution. Something you as an existing share holder should not be too keen on. And yes, most likely it will be a licensing agreement with a big pharmaceutical company rather than an outright buyout of NNVC. Me, I'd see such a agreement as a very positive result. It could ensure enough money to make NNVC profitable and also enable the development of other viricides that NNVC could retain full profits on, as it could then afford to do the trials on its own if it wants too.
The only other supplier of cash is stockholders. By tripling its share count a drug developer can raise the cash to do its own phase 3 trials. That makes every share worth 1/3 of what it was worth before. It usually is better for the existing shareholders to make a deal with big pharma. BTW, if NNVC succeeds it will become one of those big pharma companies itself. Are you going to still hate on big pharma then?
You make it sound like "buying innovation" is a bad thing. For many small drug development companies having their innovation bought out is a desirable outcome. They don't have the cash to do phase 3 trials, they don't have a distribution system, they don't want to be in the pill manufacturing business. If there was no possibility of getting funding from big pharma in the latter stages of testing NNVC stock would be even lower right now, because investors would know that another big stock distribution would be required to fund it. Take away the option of selling the results to big pharma and you have taken away one more possible way for this investment to have a positive outcome.
50x from here is about $24 a share, quite a ways from $100.
