| Followers | 1323 |
| Posts | 247,905 |
| Boards Moderated | 57 |
| Alias Born | 12/07/2009 |
| Twitter Profile: | Temporarily Unavailable |
| Follow on Twitter: | Follow @ Temporarily Unavailable |
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$CCTG For more information, please visit the Company’s website: http://ir.ccsc-interconnect.com.
CCTG - The OEM (“original equipment manufacturer”) and ODM (“original design manufacture”) interconnect products for manufacturing companies that produce end products, as well as electronic manufacturing services (“EMS”) companies that procure and assemble products on behalf of such manufacturing companies. The Company has a diversified global customer base located in more than 25 countries throughout Asia, Europe and the Americas.
About CCSC Technology International Holdings Limited
CCSC Technology International Holdings Limited, is a Hong Kong-based company that engages in the sale, design and manufacturing of interconnect products. The Company specializes in customized interconnect products, including connectors, cables and wire harnesses that are used for a range of applications in a diversified set of industries, including industrial, automotive, robotics, medical equipment, computer, network and telecommunication, and consumer products. $CCTG
$CCTG Nasdaq Profile: https://www.nasdaq.com/market-activity/stocks/cctg
$BLIS .041 +20.59% Company Name Change to NAPC Defense, Inc. (OTCPK: BLIS) and Website to NAPCDefense.com among Business Updates https://finance.yahoo.com/news/company-name-change-napc-defense-120500381.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$BLIS .041 +20.59% NAPC Defense, Inc. (OTCPK: BLIS) Website is NAPCDefense.com https://finance.yahoo.com/news/company-name-change-napc-defense-120500381.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$BLIS .041 +20.59% Company Name Change to NAPC Defense, Inc. (OTCPK: BLIS) and Website to NAPCDefense.com among Business Updates https://finance.yahoo.com/news/company-name-change-napc-defense-120500381.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
OTC: $TREIF - Treatment.com AI and Novus Health Partnership Expands Access to its Global Library of Medicine (GLM) @novushealthSTL @TreatmentAI https://seekingalpha.com/pr/19726214-treatment-com-ai-and-novus-health-partnership-expands-access-to-global-library-of-medicine?source=tweet
#BreakingNews? #healthcare #AI #GobalLibrary CSE: $TRUE
OTC: $TREIF - Treatment.com AI and Novus Health Partnership Expands Access to its Global Library of Medicine (GLM) @novushealthSTL @TreatmentAI https://seekingalpha.com/pr/19726214-treatment-com-ai-and-novus-health-partnership-expands-access-to-global-library-of-medicine?source=tweet
#BreakingNews? #healthcare #AI #GobalLibrary CSE: $TRUE
$NCL Northann Corp. (NYSE: NCL) #HotStock #3DPrinting ECO Friendly 3D-Printed Flooring & Wall Panel Products Made Using Recycled Ocean Plastics For more information on NCL, visit: https://icont.ac/4Yp5Z
$NCL Northann Corp. (NYSE: NCL) #HotStock #3DPrinting ECO Friendly 3D-Printed Flooring & Wall Panel Products Made Using Recycled Ocean Plastics For more information on NCL, visit: https://icont.ac/4Yp5Z
$EPAZ Zenadrone -Here are some of the promising advantages of #HomeSecurityDrones for house owners! Say goodbye to hiring guards and installing CCTV cameras! #drones #dronetechnology #homesecurity #security
Here are some of the promising advantages of #HomeSecurityDrones for house owners! Say goodbye to hiring guards and installing CCTV cameras! 🛡️🚁 #drones #dronetechnology #homesecurity #security pic.twitter.com/g5INkT9VaT
— ZenaDrone_Inc (@ZenadroneInc) May 14, 2024
$ACGX NEWS! Q1 2024 report is out with an 88% increase in Net Income from Q1 2023 to Q1 2024 PR at: https://otcmarkets.com/stock/ACGX/news #OTCmarkets #RealEstate #SecuredInvestments #CorporateAcquisitions #Investments #Consulting #PeopleVine #ACGX #MembershipClubs #MemberPayments #CRM $ACGX
$ACGX Q1 2024 report is out with an 88% increase in Net Income from Q1 2023 to Q1 2024 PR at: https://t.co/n85aS5OcyN #OTCmarkets #RealEstate #SecuredInvestments #CorporateAcquisitions #Investments #Consulting #PeopleVine #ACGX #MembershipClubs #MemberPayments #CRM $ACGX
— $ACGX (@ACGpaul) May 15, 2024
$ACGX NEWS! Q1 2024 report is out with an 88% increase in Net Income from Q1 2023 to Q1 2024 PR at: https://otcmarkets.com/stock/ACGX/news #OTCmarkets #RealEstate #SecuredInvestments #CorporateAcquisitions #Investments #Consulting #PeopleVine #ACGX #MembershipClubs #MemberPayments #CRM $ACGX
$ACGX Q1 2024 report is out with an 88% increase in Net Income from Q1 2023 to Q1 2024 PR at: https://t.co/n85aS5OcyN #OTCmarkets #RealEstate #SecuredInvestments #CorporateAcquisitions #Investments #Consulting #PeopleVine #ACGX #MembershipClubs #MemberPayments #CRM $ACGX
— $ACGX (@ACGpaul) May 15, 2024
$ACGX NEWS! Alliance Creative Group (ACGX) Releases Q1 2024 Financial and Disclosure Report with an 88% increase in Net Income from Q1 2023 to Q1 2024 https://finance.yahoo.com/news/alliance-creative-group-acgx-releases-123000832.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance @ACGpaul
$ACGX NEWS! Alliance Creative Group (ACGX) Releases Q1 2024 Financial and Disclosure Report with an 88% increase in Net Income from Q1 2023 to Q1 2024 https://finance.yahoo.com/news/alliance-creative-group-acgx-releases-123000832.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance @ACGpaul
News: Alliance Creative Group (ACGX) Releases Q1 2024 Financial and Disclosure Report with an 88% increase in Net Income from Q1 2023 to Q1 2024
PR Newswire
Wed, May 15, 2024, 8:30 AM EDT
In this article:
ACGX
West Devon Property listed for sale at $3.950,000 with a 7.97% Cap Rate
CHICAGO, May 15, 2024 /PRNewswire/ -- Alliance Creative Group, Inc., (http://www.ACGX.us) (Stock Symbol OTC: ACGX) is pleased to announce the results of Operations for the Quarter Ending March 31, 2024.
Revenues for the Quarter ending March 31, 2024 ("Q1 2024") were $217,916 Gross Profits for the Quarter ending March 31, 2024 ("Q1 2024") were $217,916 Net Income for the Quarter ending March 31, 2024 ("Q1 2024") were $142,330
The total assets on the Balance Sheet for the Alliance Creative Group as of 3/31/24 were $4,531,689
The total outstanding common shares as of March 31, 2024 were 4,454,211 with 2,799,023 of those shares in the float - Same as the end of the third quarter.
The Company ended the quarter with $156,554 Cash on hand.
The property located at 1324 W. Devon Ave, Chicago, IL. 60660 has been listed for sale at $3,950,000 ($246,975 per Unit) with a 7.97% Cap Rate - Link below:
https://www.loopnet.com/Listing/1324-W-Devon-Ave-Chicago-IL/31610262/
The full financial statement, balance sheet, statement of operations, cash flow statement, and disclosure statements are posted on the OTC Market Company website at www.OTCmarkets.com under the stock symbol ACGX in the section for filings and disclosure and on www.ACGX,us in the investor relations section.
Paul Sorkin, CEO of the Alliance Creative Group, Inc. said, "This quarter was a continuation of keeping our expenses as low as possible while we attempt to sell the 1324 W. Devon property and look for the next best strategic investment, acquisition, or partnership to continue to grow our positive cashflow and position ourselves for potentially larger opportunities.
$CBDW CEO Greg Lambrecht and crew presenting at the @PlanetMicroCap Showcase in Las Vegas in April 2024 was a success for their new #IR #chatbot.
A message from CEO Greg Lambrecht recapping the Planet Microcap show in Las Vegas. #ai #chatbots #investing pic.twitter.com/YiRKF4mTJB
— CBDW.ai | A Brand by 1606 Corp (@CBDWInc) May 2, 2024
$CBDW CEO Greg Lambrecht and crew presenting at the @PlanetMicroCap Showcase in Las Vegas in April 2024 was a success for their new #IR #chatbot.
A message from CEO Greg Lambrecht recapping the Planet Microcap show in Las Vegas. #ai #chatbots #investing pic.twitter.com/YiRKF4mTJB
— CBDW.ai | A Brand by 1606 Corp (@CBDWInc) May 2, 2024
$GNPX NEWS! @Genprex dosing subjects in trial of combination therapy for small-cell lung cancer
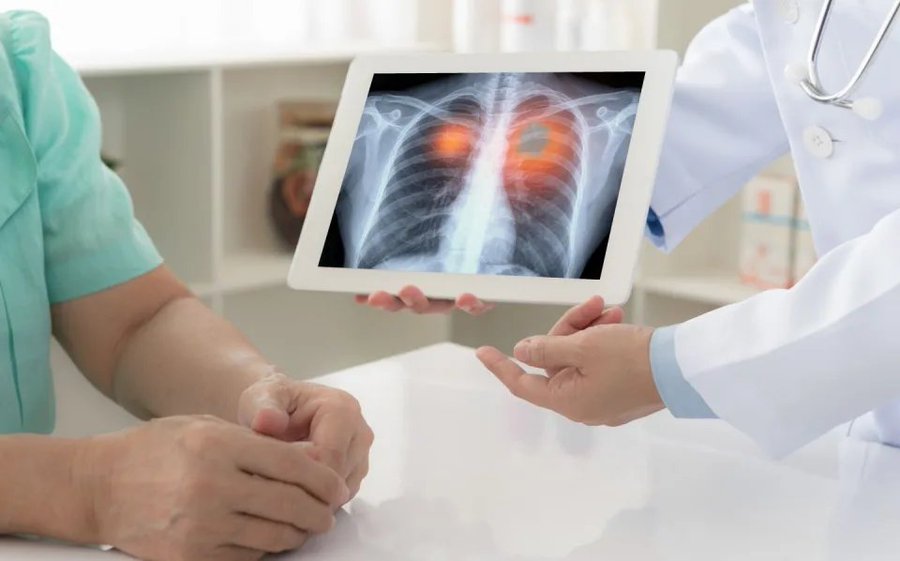
GlobalData
Wed, May 15, 2024, 4:42 AM EDT
In this article:
GNPX
+7.53%
US-based gene therapy company Genprex has enrolled and dosed the first subject in a Phase I/II trial to assess a combination therapy for extensive-stage small cell lung cancer (ES-SCLC).
The Acclaim-3 trial aims to determine the safety and optimal dosage of Reqorsa (quaratusugene ozeplasmid) Immunogene Therapy when given in combination with Genentech’s Tecentriq.
The open-label dose escalation and clinical response study is enrolling patients who have not experienced tumor progression following initial treatment with Tecentriq and chemotherapy.
Its first phase aims to enroll up to 12 subjects across roughly ten clinical sites in the US to identify the maximum tolerated dose.
Should the Phase I study proceed without dose-limiting toxicities, the highest dose tested will be the recommended Phase II dose.
The Phase II portion is anticipated to involve around 50 patients at up to 15 US locations.
Treatment with Reqorsa and Tecentriq will continue until disease progression or unacceptable toxicity occurs.
Genprex plans to begin the Phase II expansion study in the second half of this year.
A futility analysis is scheduled for after the 25th participant reaches an 18-week follow-up period.
Reqorsa has received orphan drug designation and fast track status from the US Food and Drug Administration (FDA) for use in combination with Tecentriq in ES-SCLC patients whose disease has not progressed after initial standard treatment.
Genprex chief medical officer Mark Berger said: “The Phase I dose escalation portion of the Acclaim-3 study is expected to determine the maximum tolerated dose for the Phase II expansion study.
“The favourable results from our Phase I Acclaim-1 study in non-small cell lung cancer enabled us to shorten the Phase I portion of Acclaim-3.
“This should allow us to complete the Phase I portion of the study during the second half of 2024 and to advance more quickly into the Phase II expansion portion of Acclaim-3 in the second half of 2024.”
"Genprex dosing subjects in trial of combination therapy for small-cell lung cancer" was originally created and published by Clinical Trials Arena, a GlobalData owned brand.
You must obtain professional or specialist advice before taking, or refraining from, any action on the basis of the content on our site.
https://finance.yahoo.com/news/genprex-dosing-subjects-trial-combination-084204787.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
$GNPX NEWS! @Genprex dosing subjects in trial of combination therapy for small-cell lung cancer

GlobalData
Wed, May 15, 2024, 4:42 AM EDT
In this article:
GNPX
+7.53%
US-based gene therapy company Genprex has enrolled and dosed the first subject in a Phase I/II trial to assess a combination therapy for extensive-stage small cell lung cancer (ES-SCLC).
The Acclaim-3 trial aims to determine the safety and optimal dosage of Reqorsa (quaratusugene ozeplasmid) Immunogene Therapy when given in combination with Genentech’s Tecentriq.
The open-label dose escalation and clinical response study is enrolling patients who have not experienced tumor progression following initial treatment with Tecentriq and chemotherapy.
Its first phase aims to enroll up to 12 subjects across roughly ten clinical sites in the US to identify the maximum tolerated dose.
Should the Phase I study proceed without dose-limiting toxicities, the highest dose tested will be the recommended Phase II dose.
The Phase II portion is anticipated to involve around 50 patients at up to 15 US locations.
Treatment with Reqorsa and Tecentriq will continue until disease progression or unacceptable toxicity occurs.
Genprex plans to begin the Phase II expansion study in the second half of this year.
A futility analysis is scheduled for after the 25th participant reaches an 18-week follow-up period.
Reqorsa has received orphan drug designation and fast track status from the US Food and Drug Administration (FDA) for use in combination with Tecentriq in ES-SCLC patients whose disease has not progressed after initial standard treatment.
Genprex chief medical officer Mark Berger said: “The Phase I dose escalation portion of the Acclaim-3 study is expected to determine the maximum tolerated dose for the Phase II expansion study.
“The favourable results from our Phase I Acclaim-1 study in non-small cell lung cancer enabled us to shorten the Phase I portion of Acclaim-3.
“This should allow us to complete the Phase I portion of the study during the second half of 2024 and to advance more quickly into the Phase II expansion portion of Acclaim-3 in the second half of 2024.”
"Genprex dosing subjects in trial of combination therapy for small-cell lung cancer" was originally created and published by Clinical Trials Arena, a GlobalData owned brand.
You must obtain professional or specialist advice before taking, or refraining from, any action on the basis of the content on our site.
https://finance.yahoo.com/news/genprex-dosing-subjects-trial-combination-084204787.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via @YahooFinance
OTC: $TREIF NEWS OUT! Treatment.com AI CSE: $TRUE Expands Access to GLM
Treatment.com AI and Novus Health Partnership Expands Access to its Global Library of Medicine (GLM) https://finance.yahoo.com/news/treatment-com-ai-novus-health-120000717.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via YahooFinance
OTC: $TREIF NEWS OUT! Treatment.com AI CSE: $TRUE Expands Access to GLM
Treatment.com AI and Novus Health Partnership Expands Access to its Global Library of Medicine (GLM) https://finance.yahoo.com/news/treatment-com-ai-novus-health-120000717.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr via YahooFinance
$NRXP NEWS: NRx Pharmaceuticals (Nasdaq: $NRXP) Reports First Quarter 2024 Financial Results & Provides Business Update
2024 Catalysts: Positive Clinical Data, Two Planned NDAs, Company Launch of Hope Therapeutics; FDA QIDP award in cUTI and New Schizophrenia Opportunity
https://ir.nrxpharma.com/2024-05-14-NRx-Pharmaceuticals-Nasdaq-NRXP-Reports-First-Quarter-2024-Financial-Results-and-Provides-Business-Update
#PharmaNews #mentalillness #mentalhealth #mentalhealthawareness #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101
$NRXP NEWS: NRx Pharmaceuticals (Nasdaq: $NRXP) Reports First Quarter 2024 Financial Results & Provides Business Update
2024 Catalysts: Positive Clinical Data, Two Planned NDAs, Company Launch of Hope Therapeutics; FDA QIDP award in cUTI and New Schizophrenia Opportunity
https://ir.nrxpharma.com/2024-05-14-NRx-Pharmaceuticals-Nasdaq-NRXP-Reports-First-Quarter-2024-Financial-Results-and-Provides-Business-Update
#PharmaNews #mentalillness #mentalhealth #mentalhealthawareness #mentalhealthmatters #depression #ketamine #stress #PTSD #bipolar #NRX100 #NRX101
NEWS: NRx Pharmaceuticals (Nasdaq: $NRXP) Reports First Quarter 2024 Financial Results & Provides Business Update
2024 Catalysts: Positive Clinical Data, Two Planned NDAs, Company Launch of Hope Therapeutics; FDA QIDP award in cUTI and New Schizophrenia Opportunity
-Executed Term Sheet from an institutional investor for an initial $7.5 million note, subject to common closing requirements, primarily to replace current debt, clearing the path to a Hope Therapeutics share distribution, with provision for funding up to $30 million to fund pipeline opportunities
-Positive data from a Phase 2b/3 trial of NRX-101 in Treatment Resistant Bipolar Depression (TRBD); trial demonstrated depression efficacy comparable to standard of care and significant reduction of akathisia (P=0.025). Akathisia is a potentially lethal side effect of all serotonin-targeted antidepressants and is associated with suicide. The study additionally demonstrated a 30% advantage in sustained remission from suicidality that was not statistically-significant at this sample size
-Above findings of reduced suicidality mirror the results of the Company's prior STABIL-B trial in acutely suicidal patients and also mirror the results of an independent published trial
-Company plans to file a New Drug Application (NDA) for Accelerated Approval under Breakthrough Therapy and Priority Review of NRX-101 in treatment of bipolar depression in people at risk of akathisia, based on the Phase 2b/3 and STABIL-B data
Company has developed patentable pH neutral formulation for ketamine that will be suitable for both intravenous and subcutaneous administration. Ketamine efficacy data are in hand from 4 clinical trials. Three manufacturing lots are now initiated (required for NDA) and Company plans to initiate the NDA by July
-HOPE Therapeutics (which focuses on care delivery, not drug development) has partnered with representatives of ketamine clinic providers nationwide to construct a care platform that will include ketamine, operational support, and digital therapeutic extensions. In advance of FDA approval, HOPE is actively in the sales process to supply ketamine under 503b pharmacy licensure to meet the national ketamine shortage declared by FDA. HOPE is planned to be spun out as a separate company to be owned by NRx, current NRx shareholders, and new investors; Term Sheets received from prospective anchor investors for $60 million of new investment, once publicly listed
Data expected shortly in 200-person DOD-funded trial of D-cycloserine (DCS), the key component of NRX-101, to treat chronic pain, conducted by Northwestern University. Statistical analysis plan and data unlock have been approved by Northwestern IRB
NRX-101 in the treatment of Complicated Urinary Tract Infection (cUTI) granted Qualified Infectious Disease Product (QIDP), Fast Track, and Priority Review designations. Company has now demonstrated that NRX-101 does not damage the microbiome of the gut, in contrast to all other advanced antibiotics and is less likely to cause C. Difficile infection (a potentially lethal side effect of antibiotic treatment). NRx is reviewing partnership options
-Executed Memorandum of Understanding with Fondation FundaMental for rights to develop potential disease modifying drug for Schizophrenia. If successful, this would represent the first drug to reverse the underlying disease mechanism of schizophrenia, rather than simply treating symptoms.
-Management to host a conference call, May 14, 2024, at 4:30 PM ET
RADNOR, Pa., May 14, 2024 /PRNewswire/ -- NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) ("NRx Pharmaceuticals", the "Company"), a clinical-stage biopharmaceutical company, today announced its financial results for the quarter ended March 31, 2024, and provided a business update.
"NRx has had an incredibly productive start to 2024, delivering positive data from our phase 2b/3 trial of NRX-101 in suicidal bipolar depression, where we demonstrated what could potentially be a best-in-class medication for people with bipolar disorder. To our knowledge, this is the first successful clinical trial ever conducted with an oral antidepressant in patients with suicidal depression who have been excluded from all antidepressant trials in the past. We plan to file a New Drug Application based on these and previous findings. In addition, we have advanced Hope Therapeutics and IV Ketamine, HTX-100, towards the filing of an NDA this summer, and continue to prepare to capitalize on the opportunity for NRX-101 in chronic pain and complicated UTI," said Stephen Willard, J.D., Chief Executive Officer and Director of NRx Pharmaceuticals. "Together these opportunities constitute treatments that can meet considerable unmet medical need in up to 75,000,000 patients across the country. Obviously, this would build incredible value for our shareholders. We are dedicated to bringing hope to life and I thank our team and shareholders for their ongoing hard work and support. Lastly, we are pleased to have signed a term-sheet to replace the toxic debt on our balance sheet with a new commitment from a solid, institutional investor."
$BLIS Strong close .034 +5.59% on .5 million volume today.
$NCL .4403 +2.53% #TopStock #3DPrinting
$NCL ECO Friendly 3D-Printed Flooring & Wall Panel Products Made Using Recycled Ocean Plastics: Northann Corp. (NYSE: NCL)For more information on $NCL visit: https://icont.ac/4Yp5Z
$NCL .4403 +2.53% #TopStock #3DPrinting
$NCL ECO Friendly 3D-Printed Flooring & Wall Panel Products Made Using Recycled Ocean Plastics: Northann Corp. (NYSE: NCL)For more information on $NCL visit: https://icont.ac/4Yp5Z
$GNPX $2.55 +6.69% #BULLISH on #NEWS! Genprex Doses First Patient in Acclaim-3 Clinical Study of Reqorsa® Immunogene Therapy in Combination with Tecentriq® to Treat Small Cell Lung Cancer: https://genprex.com/news/genprex-doses-first-patient-in-acclaim-3-clinical-study-of-reqorsa-immunogene-therapy-in-combination-with-tecentriq-to-treat-small-cell-lung-cancer/ $GNPX
Genprex Doses First Patient in Acclaim-3 Clinical Study of Reqorsa® Immunogene Therapy in Combination with Tecentriq® to Treat Small Cell Lung Cancer: https://t.co/tkW6c8sK89 $GNPX
— Genprex, Inc. (@genprex) May 14, 2024
$GNPX $2.55 +6.69% #BULLISH on #NEWS! Genprex Doses First Patient in Acclaim-3 Clinical Study of Reqorsa® Immunogene Therapy in Combination with Tecentriq® to Treat Small Cell Lung Cancer: https://genprex.com/news/genprex-doses-first-patient-in-acclaim-3-clinical-study-of-reqorsa-immunogene-therapy-in-combination-with-tecentriq-to-treat-small-cell-lung-cancer/ $GNPX
Genprex Doses First Patient in Acclaim-3 Clinical Study of Reqorsa® Immunogene Therapy in Combination with Tecentriq® to Treat Small Cell Lung Cancer: https://t.co/tkW6c8sK89 $GNPX
— Genprex, Inc. (@genprex) May 14, 2024
$CBDW #CBDWInc Email capture for ongoing communication. Real-time, personalized interactions to keep you informed and engaged.
New IR Chat •Instant access to crucial documents like 8K filings and quarterly reports. Experience the ease of IR Chat now on http://cbdw.ai
$CBDW #CBDWInc Email capture for ongoing communication. Real-time, personalized interactions to keep you informed and engaged.
New IR Chat •Instant access to crucial documents like 8K filings and quarterly reports. Experience the ease of IR Chat now on http://cbdw.ai
$NRXP $3.24 +8.36% NRxPharma Eyes First Commercial Revenue in 2024, Announces Breakthroughs in Bi-Annual Milestone Update https://finance.yahoo.com/news/nrx-pharmaceuticals-eyes-first-commercial-135500896.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr
@YahooFinance
$NRXP $3.24 +8.36% NRxPharma Eyes First Commercial Revenue in 2024, Announces Breakthroughs in Bi-Annual Milestone Update https://finance.yahoo.com/news/nrx-pharmaceuticals-eyes-first-commercial-135500896.html?soc_src=social-sh&soc_trk=tw&tsrc=twtr
@YahooFinance
$META $PALI $DYNT $GBR $MSFT $NFLX $ROKU $SQ $TSLA $NVDA $AAPL $INTC $AMZN $SOFI $GOOGL $AMST $BOF $CSSE $IMCC $GMM $RENB
OTC: $TREIF CSE: $TRUE Empowering healthcare globally with instantaneous and precise medical insights, through AI
OTC: $TREIF CSE: $TRUE Empowering healthcare globally with instantaneous and precise medical insights, through AI
$BLIS Looking for that buying volume.
$BLIS yeah watching for that. OTC picking up.
$GNPX Genprex, Inc. "We are very proud of what we accomplished in 2023, particularly with the successful completion of the Phase 1 portion of our Acclaim-1 clinical trial in lung cancer, " By Rodney Varner, Chairman, President and Chief Executive Officer at Genprex. https://compasslivemedia.com/gnpx/