Tuesday, July 13, 2021 5:30:55 PM
Blarcamesine's Overwhelming Phase 2a AD Efficacy: Recap
The following was a project to determine Blarcamesine's most optimal patient size and provide an ADCS-ADL analysis compared to standard of care. I think you will want to read this. All links to sourcing is below.
Metrics:
Number of Alzheimer's patients in the United States (all severity): 6,200,000
Number of Alzheimer's patients in the United States (mild): 3,124,800
Number of Mild Cognitive Impairment due to Alzheimer's patients (MCI-AD) in the United States: 5,000,000
Combined MCI-AD and mild-AD (M-AD) patients in the United States: 8,124,800
Prevalence of S1R WT in population: 84%
Prevalence of APOE3 allele in population: 78%
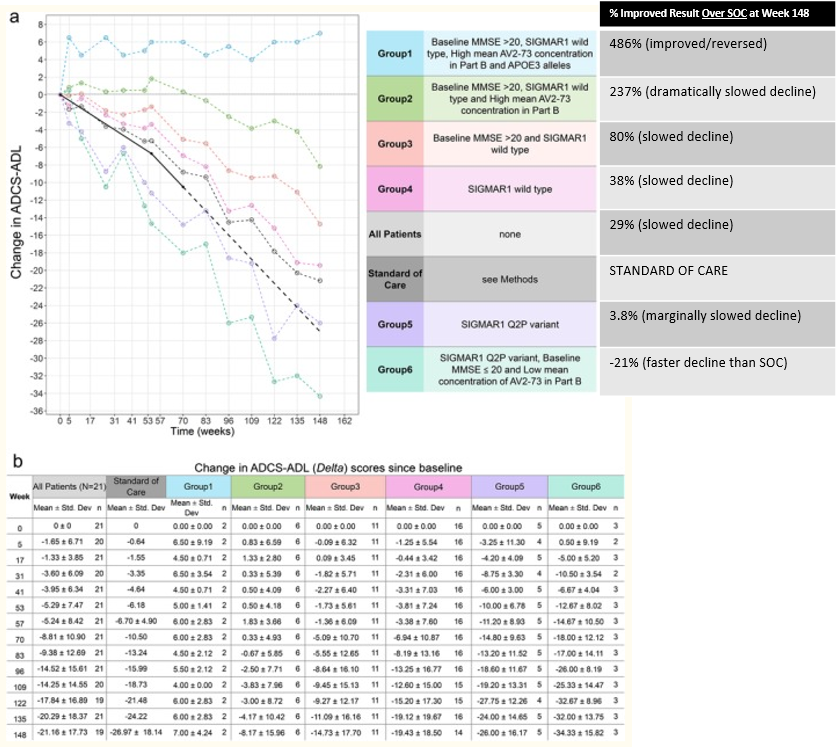
For Absolute Most Optimized Responders (top blue line/group 1)
According to the Alzheimer’s Association, there are 11,200,000 total MCI-AD and all-stage Alzheimer’s patients in the United States. Of the 11,200,000 patients, 8,124,800 are considered MCI-AD or M-AD. For these early patients, Anavex would be able to provide potentially reversible treatment to 5,323,368 (66%). These 5,323,368 patients have the S1R WT variant (84% probability) and APOE3 allele (78% probability). Individuals have approximately 66% chance of having both S1R WT and APOE3 allele.
This analysis indicates that out of ALL U.S. MCI-AD and all-stage Alzheimer’s Dementia patients, Blarcamesine is able to most optimally treat 48%. Most optimal treatment would likely result in reversal of disease pathology. This patient subset performed approximately 486% better than SOC.
For Second-most Optimized Responders (top green line/group 2)
Of 8,124,800 MCI-AD or M-AD patients, Blarcamesine can provide therapeutic effect vastly outpacing placebo and SOC to 1,501,463 patients (18%). The 18% is in addition to the aforementioned 66% (combined 84% for MCI-AD and M-AD patients). These 1,501,463 patients have the S1R WT (84% probability) and non-APOE3 allele (22% probability). Individuals have approximately 18% chance of having both S1R WT and non-APOE3 allele.
This analysis indicates that out of ALL U.S. MCI due to Alzheimer’s and all-stage Alzheimer’s Dementia patients, Anavex is able to provide great therapeutic effect to 13% (61% when combined with most optimized group). Great therapeutic effect would slow decline some 237% more effectively than SOC. Such an effect would allow patients to live relatively normal lives for a dramatically increased time horizon over SOC.
Final Summation & Thoughts
You may be asking yourself why I didn’t include groups 3 (peach color) through group 6 (sea foam green color) in this analysis. Simply, it is difficult to make a meaningful assessment because it is unclear if these patients were on high or lose dose. What is clear, is Blarcamesine’s overwhelming effect on early pathology patients when used in high doses and when those patients have either S1R WT, APOE3 alleles, or both. Conveniently, approximately 84% of the population has S1R WT, 78% have APOE3 alleles, and statistically 66% of the population have both. In case there is any remaining doubt as to Blarcamesine’s efficacy, remember that ALL high dose patients from the phase 2a Alzheimer’s trial improved over SOC regardless of genomic makeup (29% better when combined). This result has been proven in the relatively large PDD trial. If Anavex can closely duplicate these results in their large 2b/3 Alzheimer’s trial, Anavex will nearly certainly become approved by all relevant regulatory agencies (when applied) and reap significant gains for its shareholders.
Sources:
https://www.nia.nih.gov/news/half-alzheimers-disease-cases-may-be-mild
https://www.ncbi.nlm.nih.gov/snp/rs1800866?fbclid=IwAR0RgXA_0yBUuw6RUDHi4D9bbprCqwPa6cGmAUrGS8vqLm0VykOpzlAFxf8#frequency_tab
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3726719/
https://alz-journals.onlinelibrary.wiley.com/doi/10.1002/alz.12328
https://www.alz.org/media/documents/alzheimers-facts-and-figures.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7167374/
The following was a project to determine Blarcamesine's most optimal patient size and provide an ADCS-ADL analysis compared to standard of care. I think you will want to read this. All links to sourcing is below.
Metrics:
Number of Alzheimer's patients in the United States (all severity): 6,200,000
Number of Alzheimer's patients in the United States (mild): 3,124,800
Number of Mild Cognitive Impairment due to Alzheimer's patients (MCI-AD) in the United States: 5,000,000
Combined MCI-AD and mild-AD (M-AD) patients in the United States: 8,124,800
Prevalence of S1R WT in population: 84%
Prevalence of APOE3 allele in population: 78%
For Absolute Most Optimized Responders (top blue line/group 1)
According to the Alzheimer’s Association, there are 11,200,000 total MCI-AD and all-stage Alzheimer’s patients in the United States. Of the 11,200,000 patients, 8,124,800 are considered MCI-AD or M-AD. For these early patients, Anavex would be able to provide potentially reversible treatment to 5,323,368 (66%). These 5,323,368 patients have the S1R WT variant (84% probability) and APOE3 allele (78% probability). Individuals have approximately 66% chance of having both S1R WT and APOE3 allele.
This analysis indicates that out of ALL U.S. MCI-AD and all-stage Alzheimer’s Dementia patients, Blarcamesine is able to most optimally treat 48%. Most optimal treatment would likely result in reversal of disease pathology. This patient subset performed approximately 486% better than SOC.
For Second-most Optimized Responders (top green line/group 2)
Of 8,124,800 MCI-AD or M-AD patients, Blarcamesine can provide therapeutic effect vastly outpacing placebo and SOC to 1,501,463 patients (18%). The 18% is in addition to the aforementioned 66% (combined 84% for MCI-AD and M-AD patients). These 1,501,463 patients have the S1R WT (84% probability) and non-APOE3 allele (22% probability). Individuals have approximately 18% chance of having both S1R WT and non-APOE3 allele.
This analysis indicates that out of ALL U.S. MCI due to Alzheimer’s and all-stage Alzheimer’s Dementia patients, Anavex is able to provide great therapeutic effect to 13% (61% when combined with most optimized group). Great therapeutic effect would slow decline some 237% more effectively than SOC. Such an effect would allow patients to live relatively normal lives for a dramatically increased time horizon over SOC.
Final Summation & Thoughts
You may be asking yourself why I didn’t include groups 3 (peach color) through group 6 (sea foam green color) in this analysis. Simply, it is difficult to make a meaningful assessment because it is unclear if these patients were on high or lose dose. What is clear, is Blarcamesine’s overwhelming effect on early pathology patients when used in high doses and when those patients have either S1R WT, APOE3 alleles, or both. Conveniently, approximately 84% of the population has S1R WT, 78% have APOE3 alleles, and statistically 66% of the population have both. In case there is any remaining doubt as to Blarcamesine’s efficacy, remember that ALL high dose patients from the phase 2a Alzheimer’s trial improved over SOC regardless of genomic makeup (29% better when combined). This result has been proven in the relatively large PDD trial. If Anavex can closely duplicate these results in their large 2b/3 Alzheimer’s trial, Anavex will nearly certainly become approved by all relevant regulatory agencies (when applied) and reap significant gains for its shareholders.

Sources:
https://www.nia.nih.gov/news/half-alzheimers-disease-cases-may-be-mild
https://www.ncbi.nlm.nih.gov/snp/rs1800866?fbclid=IwAR0RgXA_0yBUuw6RUDHi4D9bbprCqwPa6cGmAUrGS8vqLm0VykOpzlAFxf8#frequency_tab
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3726719/
https://alz-journals.onlinelibrary.wiley.com/doi/10.1002/alz.12328
https://www.alz.org/media/documents/alzheimers-facts-and-figures.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7167374/
Recent AVXL News
- Form 8-K - Current report • Edgar (US Regulatory) • 05/22/2026 12:15:26 PM
- Anavex Life Sciences Receives Expected Nasdaq Delinquency Notification • GlobeNewswire Inc. • 05/22/2026 12:00:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 05/15/2026 08:15:25 PM
- Form 3 - Initial statement of beneficial ownership of securities • Edgar (US Regulatory) • 05/14/2026 08:15:30 PM
- Form NT 10-Q - Notification of inability to timely file Form 10-Q or 10-QSB • Edgar (US Regulatory) • 05/11/2026 08:30:22 PM
- CEO Transition and Delayed SEC Filing Put Anavex (AVXL) Leadership Changes in Focus • IH Market News • 05/06/2026 02:52:36 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 05/06/2026 11:04:59 AM
- Anavex Life Sciences Board of Directors Appoints Former Senior Vice President of Clinical Development Terrie Kellmeyer, PhD, as Interim Chief Executive Officer • GlobeNewswire Inc. • 05/06/2026 11:00:00 AM
- Form 3 - Initial statement of beneficial ownership of securities • Edgar (US Regulatory) • 05/01/2026 11:18:47 PM
- Anavex Life Sciences Highlights New Scientific Findings on Shared Biology Between Autism and Alzheimer’s Disease • GlobeNewswire Inc. • 04/14/2026 11:30:00 AM
- Anavex Life Sciences to Present at the 25th Annual Needham Virtual Healthcare Conference • GlobeNewswire Inc. • 04/07/2026 11:30:00 AM
- Anavex withdraws EU approval filing for Alzheimer’s therapy • IH Market News • 03/30/2026 12:39:26 PM
- Anavex Life Sciences Provides Comprehensive Regulatory Update • GlobeNewswire Inc. • 03/30/2026 11:30:00 AM
- Form 8-K - Current report • Edgar (US Regulatory) • 03/25/2026 08:06:00 PM
- Anavex withdraws EU marketing application for Alzheimer’s therapy blarcamesine • IH Market News • 03/25/2026 02:06:58 PM
- Anavex Life Sciences Provides Update on Regulatory Review in the EU for Blarcamesine to Treat Early Alzheimer’s Disease • GlobeNewswire Inc. • 03/25/2026 11:30:00 AM
- Anavex Life Sciences Presents New Data from its AD-004 Phase IIb/III Trial at AD/PD 2026 Conference Demonstrating Consistent Correlation Between the Treatment Effect of Oral Blarcamesine and Preservation of Brain Volume in Early Alzheimer’s Disease • GlobeNewswire Inc. • 03/23/2026 11:30:00 AM
- New Scientific Findings Highlight Hypothesis of Autophagy Failure as a Precursor of Amyloid Beta and Tau Pathology in Alzheimer’s Disease • GlobeNewswire Inc. • 03/20/2026 11:30:00 AM
- Anavex Life Sciences Presents Significant Treatment Effects of Blarcamesine in New Advanced Alpha-Synuclein Model of Parkinson’s Disease at AD/PD 2026 Conference • GlobeNewswire Inc. • 03/17/2026 11:30:00 AM
- Anavex Life Sciences to Present at the Citizens Life Sciences Conference • GlobeNewswire Inc. • 03/03/2026 12:30:00 PM
- Anavex Life Sciences to Present at the 46th TD Cowen Annual Health Care Conference • GlobeNewswire Inc. • 02/25/2026 12:30:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/25/2026 11:07:01 AM
- Anavex Life Sciences Appoints Seasoned Healthcare Leader to Board of Directors • GlobeNewswire Inc. • 02/23/2026 12:30:00 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 02/09/2026 09:40:27 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 02/09/2026 12:31:17 PM








