Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Tumors have too many body guards.
https://www.nature.com/articles/s41467-022-33117-y
allo carts need to make a breakthrough to treat solid tumors. Most allo bios targeting BCMA/CD19 won't survive the bear market. They are still trying to figure out the effective dose and target pts. AUTL and AFMD offer the most promising data IMO
With NVS 2 day AUTO cell therapy, ALLO will have to target the AUTO relapsed pts. The ones that are old, frail with damages from prior therapies. A fragile population to say the least. That is why DTIL mgt not to urgent to recruit pts for sLD/19A.
Are you expecting any NK or T cell therapies, ICE .... breakthrough in 2022?
Same as DTIL 19A.
There were 2 deaths reported as related to ADP-A2M4CD8 by investigators. A 60-year-old woman with ovarian cancer with a large tumor burden in her lungs died due to pneumonia and CRS. As reported at ESMO 2021, a 71-year-old man with adenocarcinoma of the esophagus and a history of chronic anemia died due to bone marrow failure.
NKTX seems to repeat what DTIL tried with 19a/eLD. Why they don't increase the NK cells dose to 4 billions same as AUTO NK?
Will the ISPC NK cells work if the mAb is the weakest link? .AFM24 v. cetuximab hold the key to success?
AZ just stopped the trial of monalizumab in combination with cetuximab vs. cetuximab in patients with recurrent or metastatic squamous cell carcinoma of the head and neck (R/M SCCHN).
More data will lead to more questions from FDA? Off target effects are not predictable.
AFMD seemed to have solved the cleaved CD16 issue by pretreating the NK with their AFM13 ex-vivo.
Looking at P1 data from Fate and NKTX, NK cells seem to be effective on indolent lymphoma and nodal DLBCL. For pts with high tumor burden or aggressive disease, CART will still be needed. I listened to NKTX presentation, the number of cycles given to a patient will be up to the oncologist.
Has anyone compared ISPC NK to CBNK?
DTIL no longer mentioned foralumab. I think maintaining a high CD4/CD8 ratio with a high # of CD8 is the key to any Allo success.
Thanks for the list. It is not long compared to the one for CD19. I am trying to figure out how innate engagers affect TAMs thru CD16.
cbNK cells pre-complexed with engager is safe and CAR like.
As of October 31, 2021, a total of 18 patients with CD30-positive relapsed or refractory Hodgkin and non-Hodgkin lymphomas (16 and 2 patients, respectively) were treated with the novel combination of cbNK cells pre-complexed with AFM13.
As of the cutoff date, 16 of 18 patients had achieved an objective response to the treatment according to investigator assessment, with seven complete responses (CR) and nine partial responses.
100% objective response rate (ORR) was observed with a 42% CR rate in 12 patients with Hodgkin Lymphoma, after the 1st of 2 planned cycles at the recommended phase 2 dose of 108 cbNK cells/kg pre-complexed with AFM13.
No cases of serious adverse events such as cytokine release syndrome, neurotoxicity syndrome or graft-versus-host disease were observed.
Treatment was well tolerated with five reported cases of transient infusion related reactions after the monotherapy infusions of AFM13.
More allogenic = less cytotoxic. It is a trade off for elderlies with tamed T cells. No free lunch in real world.
Diorio et al demonstrate that base editing enabled higher cell viability, better proliferation, and optimized CAR T-cell yields when compared with the conventional CRISPR-Cas9 nuclease system. This may be explained by reduced DNA damage response observed after quadruple editing with CBEs and better cellular fitness in the absence of gross genetic rearrangements. With overall yields between 16 × 109and 17.2 × 109 CAR+ T cells, a single manufacturing run may provide up to 200 administrations in adult patients (assuming a treatment dose of 1 × 106 CAR+ T cells per kilogram of body
https://ashpublications.org/blood/article/140/6/526/486105/The-uncut-version-base-edited-allo-CAR-T-cells
recent technical advances using culture conditions with IL-7/IL-15 and
the addition of IL-21 may enhance the enrichment for TSCM cells
in the final CART product (74–76), which can be further increased
with the addition of drugs blocking T-cell differentiation, such as
glycogen synthase-3 inhibitors (77). Nevertheless, robust clinical grade protocols for generating TSCM-enriched CART products
have not been developed so far. Recently, a few CART19 clinical
trials for DLBCL have been conducted in which CART products
were manufactured from CD62L+ isolated T cells to generate
cellular products enriched for TCM cells (21, 78); however, due to
prolonged culture conditions, enrichment for TSCM and TCM
subsets in the infused product could not be demonstrated
https://www.frontiersin.org/articles/10.3389/fimmu.2022.904497/full
Thanks for the link.
Most commonly reported grade 3/4 AEs were neutrophil count decrease (6/9, 67%), anemia (5/9, 56%), thrombocytopenia (2/9,22%), platelet count decrease (2/9, 22%) and no infection was reported. Complete response rate(CRR) was 78%
Compared to T-Charge
http://www.koreabiomed.com/news/articleView.html?idxno=12767
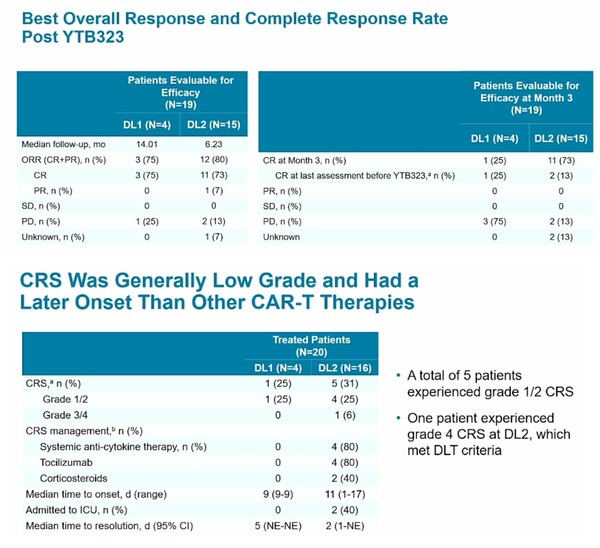
Which will perform better ? BE with 7,8 edits on many genes, Epi secret sauce
or T-Charge which seems to add a CAR only to starting material in order to retain CD4/CD8 ratio.
YTB323 CAR-T cell products generated via this novel expansionless manufacturing process retained the immunophenotype of the input leukapheresis materia
Do you need Epigenetic Reprogramming if pts can be treated with healthy, young donor T cells?
I am glad you found something scary. 0.49/100K was the latest data point. It wasn't non-sense as you wished.
You are calling all the PhDs at NYC Health fake news?
https://www1.nyc.gov/site/doh/covid/covid-19-data.page#daily
0.5/100K unvaccinated died of covid in NYC. What is the point?
Every American should celebrate on these news. Mother nature is saving the human race by taming the virus. Avg. Covid deaths in NYC is 9 yet the infection rate has reached >20% in Queens.
Did you see see any study that looked at the ratio of exhausted T cells and naive T cells in people who have been boosted 3, 4 times?
Base editing reminds me of vaccine boosters. It can be done safely every few months with no side effects according to the science gods at NIH. Really?
120 mil cash + 67 mil deferred revenue
From 10K:
Importantly, our one-step genome editing
approach avoids making multiple breaks to the T cell’s DNA and also contributes to minimizing cell processing time, which helps prevent the
CAR T cells from differentiating during the process.
• Novel co-stimulatory domain. Our genetically engineered CAR T cells incorporate a novel, proprietary, costimulatory domain called N6, which
may enable us to enhance cell proliferation and effector function while preserving cell phenotype. We engineered N6 to improve on the function
of the 4-1bb costimulatory domain commonly used in autologous CAR T products. Our preclinical data suggests that, compared to 4-1bb, N6
provides an activation signal to the CAR T cells that better preserves cell expansion potential while maintaining naïve cell phenotype following
exposure to cancer cells. We also believe N6 can help avoid CAR T cell hyperstimulation, which can contribute to adverse events seen with
autologous products.
This allows us to “fine-tune” the CAR T cells to ensure that they respond appropriately to the cancer but do not become
hyper-activated or exhausted. The below comparison demonstrates the difference in consistency achieved by using lentivirus delivery compared
with targeted delivery through an ARCUS nuclease. CAR T cells produced using ARCUS exhibit reduced cell-to-cell variability as well as more
controlled levels of CAR gene expression depending on whether the cells are tuned for high expression or low expression.
The most popular first salvage regimen for relapse or progression was an alternate CAR-T therapy (dual or alternate target) regardless of CD19 positivity. 27% of responders chose this regimen for CD19 positive relapse, while 31% of responders did so for CD19 negative relapse. 88.5% of responders favored consolidative allogeneic hematopoietic cell transplantation (alloHCT) after response to salvage, whereas 51.2% of physicians would consider autologous hematopoietic cell transplant (AHCT) in transplant naïve patients.
https://www.astctjournal.org/article/S2666-6367(22)01360-4/fulltext#.Yp5U3ghW7h4.twitter
Too bad GNCA NPT with CAR knock in + PD1 will never be tested. ACT with CART targeting multiple neoantigens will be as potent and more durable.
500 clones from different editing sequences? What gets knock in/out first? Are these trade secrets?
Targeting PARP11 to avert immunosuppression and improve CAR T therapy in solid tumors
Response MCL AUTO limited by:
1. T cells fitness and dose of CD8 and CCR71 CD45RA1 T cells too low.
2. High TB
P2 trial of the autologous anti-CD19 chimeric antigen receptor (CAR)
T-cell therapy brexucabtagene autoleucel (KTE-X19) in patients with heavily pretreated MCL. The median DOR was 46.7 months among patients with CR (46/71) and 2.2 months in patients with PR (16/71)
https://ashpublications.org/bloodadvances/article/4/19/4898/464200/Tumor-burden-inflammation-and-product-attributes
phase II trial of the autologous anti-CD19 chimeric antigen receptor (CAR)
T-cell therapy brexucabtagene autoleucel (KTE-X19) in patients with heavily pretreated MCL
The median DOR was 46.7 months among patients with CR (46/71) and 2.2 months in patients with PR (16/71)
https://ascopubs.org/doi/pdf/10.1200/JCO.21.02370#.YptkxkBhQis.twitter
That is a lot of man made mutations. Hard to believe there is no risk or downside when the T cell genome is cut and paste at will. How do they determine the sequence of the edits?
How many gene edit/inserts will be required in that process?
“The CAR T relapse setting is a rapidly growing area with dire unmet medical need that is not adequately addressed by current treatment options.”
https://ascopubs.org/doi/full/10.1200/JCO.21.02370
These pharma execs didn't want to talk about allo. DTIL has shown T cells from a healthy donor beat exhausted T cells from the lymphoma pts. who relapsed on auto. Increasing the dose from 300 to 500 M should result in high CR @ 28 days.
Efficient reprogramming of T cells with a CAR in as little as 24 hours in a more simplified manufacturing process without T cell activation or extensive culture outside the body also offers the possibility of expanding where and when these therapies are produced
https://www.pennmedicine.org/news/news-releases/2022/march/penn-researchers-shorten-manufacturing-time-for-car-t-cell-therapy
NEA just ran GNCA to the ground. Sending ADAP VP to GNCA was a gimmick.
