Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
the amount of shares churned today was "OBESE"
3.5-Mil. share "low floater" running in the beginning stages of an epic year my friend...
MOO
yo Redspudda
hope you and your alter-ego Moshe were buying at the bottom sporto
lolzzz
MOO ding a ling
iBio and AstralBio Announce Transformative AI drug discovery Collaboration to Rapidly Develop Novel Antibodies for Obesity and Cardiometabolic Diseases
March 27 2024 - 07:00AM
https://ih.advfn.com/stock-market/AMEX/ibio-IBIO/stock-news/93558575/ibio-and-astralbio-announce-transformative-ai-drug
iBio, Inc. (NYSEA:IBIO) (“iBio” or the “Company”) announces today that it has entered into a collaboration agreement with AstralBio, Inc. (“AstralBio”) to discover, engineer and develop novel antibodies to treat obesity and other cardiometabolic conditions.
Yesterday, iBio announced the closing of a $15 million private placement financing with ADAR1 Capital Management, Lynx1 Capital Management, Ikarian Capital and other institutional investors. The company will use a portion of the net proceeds to support new partnerships, such as this collaboration with AstralBio.
The goal of this transformative collaboration is to rapidly build a novel, best-in-class cardiometabolic disease portfolio by combining iBio’s ability to create antibodies against hard-to-drug targets with AstralBio’s significant biologics experience and drug development expertise. As part of the collaboration, iBio has granted an exclusive license to its AI-powered technology to identify and engineer four (4) targets for the treatment of cardiometabolic disease, of which AstralBio may continue the pre-clinical development and deploy its proven drug development expertise to advance candidates to an Investigational New Drug (IND) application. iBio has the exclusive option to license three (3) cardiometabolic targets from AstralBio and will receive the rights to develop, manufacture and commercialize those targets upon exercise. As a result of this collaboration, iBio and AstralBio have agreed to initiate the development of a novel lead program focused on targeting the transforming growth factor beta (TGFb) superfamily for the treatment of muscle wasting and obesity. Upon mutual consent, the parties may also expand the collaboration to include additional targets in other fields.
“We are confident our AI-enabled technology is exceptionally well positioned to develop antibodies against challenging targets in the cardiometabolic space,” said iBio’s Chief Executive Officer and Chief Scientific Officer, Martin Brenner, DVM, Ph.D. “The future of treating millions of patients affected by obesity and cardiometabolic disease globally will rely on next generation therapies aimed at enhancing both the efficacy and the tolerability profiles of current treatments. Notably, the collaboration’s lead program targeting the TGFb superfamily is designed to achieve a best-in-class profile. Our approach, which combines AI-driven epitope steering with the StableHu antibody optimizer and mammalian display, aims to validate proof of concept studies that underscore the immense potential of anti-myostatin strategies. Over the coming months, we expect to announce additional novel targets, each promising a similarly transformative profile.”
Dr. Brenner added, “Leveraging my experience in obesity and cardiometabolic disease drug development from my time at Eli Lilly and Pfizer, I am eager to advance potentially groundbreaking compounds through our partnership with AstralBio, a team renowned for its entrepreneurship and track record to develop best-in-class and first-in-class therapies.”
“We are excited to collaborate with the talented iBio team to rapidly advance exciting targets in cardiometabolic disease and beyond. Using machine learning tools in the discovery of biological drugs has the potential to shorten development timeframes and lower expenses while enhancing the likelihood of successful development. We look forward to working with iBio to mutually advance our goals to deliver meaningful precision therapies for patients efficiently,” said Patrick Crutcher, AstralBio’s Co-founder and Chief Executive Officer.
About iBio
iBio is an AI-driven innovator that develops next-generation biopharmaceuticals using computational biology and 3D-modeling of subdominant and conformational epitopes, prospectively enabling the discovery of new antibody treatments for obesity, cardiometabolic, hard-to-target cancers, and other diseases. iBio’s mission is to decrease drug failures, shorten drug development timelines, and open up new frontiers against the most promising targets. For more information, visit www.ibioinc.com.
About AstralBio
AstralBio is a privately held biotechnology company founded by Patrick Crutcher and several former team members of ValenzaBio and AlmataBio. AstralBio's mission is to develop first-in-class or best-in-class medicines to treat cardiometabolic and immune-mediated diseases. The company plans to leverage iBio’s proven machine learning-enabled platform to accelerate the progression of novel therapeutic targets to transform patient care.
Webcasted Conference Call Details
iBio will host a webcasted conference call with an associated slide presentation today, March 27, at 8:30 a.m. EDT to discuss its collaboration with AstralBio.
The live and archived webcast may be accessed on the Company’s website at www.ibioinc.com under “News and Events” in the Investors section. To participate in the conference call by telephone, please pre-register via this link to receive the dial-in number and your unique PIN.
Forward-Looking Statements
Any statements contained in this press release about future expectations, plans, and prospects, as well as any other statements regarding matters that are not historical facts, may constitute “forward-looking statements.” These statements include statements regarding the intended use of proceeds, the expected gross proceeds from the offering and the expected extension of the Company’s cash runway to fund its operating plan into 2025, the ability to build a diverse set of novel best-in-class obesity treatments, the Company’s AI-enabled technology being exceptionally well positioned to develop antibodies against challenging targets in the cardiometabolic disease space, the collaboration’s lead program targeting the TGFb superfamily achieving a best-in-class profile, the size of the obesity market, aiming to pioneer antibody treatments for obesity and cardiometabolic diseases and advancing potentially groundbreaking compounds through the Company’s partnership with AstralBio. The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including the uncertainties related to market conditions and the completion of the offering on the anticipated terms or at all, the ability to develop antibodies against challenging targets in the cardiometabolic disease space, the ability to finance when needed and the risk factors described in the Company’s Annual Report on Form 10-K for the year ended June 30, 2023, and the Company’s subsequent filings with the SEC, including subsequent periodic reports on Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. Any forward-looking statements contained in this press release speak only as of the date hereof and, except as required by federal securities laws, iBio, Inc. specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events, or otherwise.
Contacts:
Investor Relations
Stephen Kilmer
iBio, Inc.
(646) 274-3580
skilmer@ibioinc.com
Media Relations
Susan Thomas
iBio, Inc.
(619) 540-9195
susan.thomas@ibioinc.com
Moshe isn’t a MONI shareholder…
MONI has a pristine shell and is worth far more than the current pps
awaiting next filing as indicated
BAF MONI…!
MOO
130-Mil shares traded w/3-Mil in the float
$365k cash passing through hands and 2-1/4 hours left till closing bell
dumptins up Buchwheat ….!
MOO
and BOOM that’s how a 3–Mil OS share floater runs…
nice PP financing package - cant wait to see what’s under IiBio’s AI engine
$50 pps was the drop zone - with the right developments
this could easily get bought right back up again
MOO
here you go Joey-the-Forks-Bolo
served up to you ...
just like room service Sporto
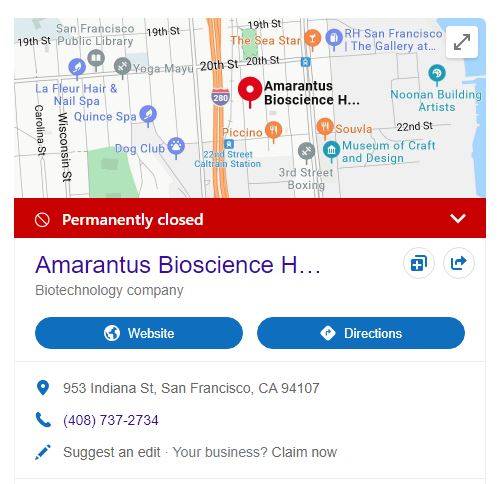
uh oh, Gerald's "WeWorks" cubicle ...
must have gotten emptied out in a hurry - eh JOEY...?

but hey, at least you got some toilet paper left
to clean up the mess he left you...!
SMH - can't make this stuff up folks
MOO bad brains
care to provide written proof...?
"Already said in the deal structure, MONI shareholders will get nothing."
who says it was pretend - you - really...?
"Anyone that calls into a investor call like the ones the CEO of this company held and pretends to be a stock analyst pretends to be a stock analyst should pay of heavy penalty and/or go to jail for fraud."


sorry to break it to you Sunny
Grace Carlton is a warm loving caring and kind human being
which is why she prolly ditched NH as soon as she could
BAF MONI...!
AJMHO
its simple - whatever I say or try to explain ...
you will obfuscate and create false narratives around it
that's just how you, Mista Redspudda and Joey-the-Forkit-Bolo operate in the Back Office Boiler Room
and the fact that you were all warned - by me a number of others ...
who knew that Gerald Commissiong is a sham CEO and TOMDF was going to go belly-up
just like he did with AMBS - broke - insolvent - OTC delinquent, dark & non-reporting
in essence, here's all that really matters to YOU sport or anybody else in Tolloup Buttercup Todosville
Todos Medical - permanently dissolved & out-of-business:
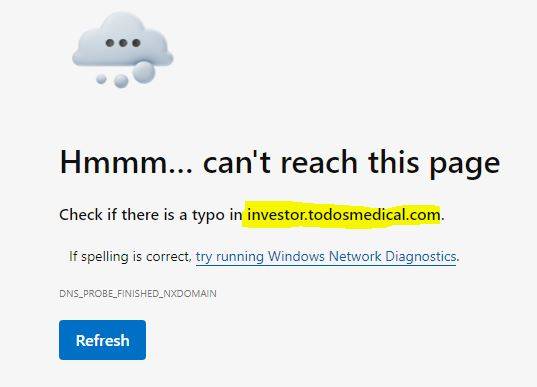
TOMDF Printing $0.000001 and zero volume:

but all is not lost the S wordman
because here is one last opportunity for you to "sell me this pen"

SMH - sad but true
MOO slow brains
yo Swordie it’s been answered
more than a dozen times
but no matter how many times it’s been answered is meaningless…
cause you are in denial - or refuse to listen
but hey, good luck with your TOMDF holdings sporto
you, Redspudda and Joey-the-Forkit-Bolo are going to need it…
printing $0.000001 - Tollo-up ButterCup pulled from all store shelves…!
SMH - sad but true
MOO slow brains
JOEY JOEY JOEY - what am I going to do with you JOEY....?
"despite the failure of ALL previously announced mergers and acquisitions"
yip Velcro precisely right
however the Boiler Room Pumper Boys Redspudda, JoeyForkitBolo and Swordie ...
would rather have everyone focus on everything other than the AMBS ot TOMDF CEO failure
who they were pumping like a tire pressure filling station all along
and thats been the moral of the story here forever
SMH sad but true...!
Tollovid Snake Oil - now outta business folks...

MOO shills touts & dolts
yo Joey - I know understanding this mater is like
maybe like understanding Differential Equations, Determinate & Indeterminate Structures or Calculus 3
but here's the bottom line Sporto
"Your list of analysts covering Amarantus Biosciences is incomplete."





in at $0.72 & $0.76
saw projections over $8 pps for 2024
best wishes to you ...!
waiting on 8K - staying pat - adding more - here's why...
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174056246
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174056783
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174056783
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174056030
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174055948
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=174015309
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173740227
somewhere on some SM site - it was mentioned FO bypassed prior acquisitions ...
because it was thought they would start to dilute
FO has been 'steadfast' in keeping MONI as dilution free as possible over the past several years
IMHO, if that was in fact his criteria in rejecting the former - and accepting the present acquisition target
then safe to say- I'm confident we have a solid acquisition on our hands
also I like the stats on Wikilo, its members and the revenue that SuperApps make
and the future projections that SuperApps will continue to make

BAF MONI...!
MOO
JoeForkitBolos gets it wrong again folks
not sure where these false narratives & fake posts originated from
but it sure seems apparent who they are being disseminated by...!
FYI JOEY - here's the certified "AMBS Stock Analyst List"
3 analysts who are all FINRA licensed & approved to represent AMBS
just lookie here folks - no JWP, Inc firm on it whatsoever:

and do you know why JWP, Inc is not on the approved FINRA stock analyst list for AMBS...?
because JWP, Inc is not a stock analyst firm - never was - never will be - eh JOEY...!
yeah, and those dissolved insolvent liquidated TOMDF shares are'nt looking too good these daze either
not while printing $0.000001 and being permanently "out-of-business"

so lets find out - who do TOMDF shareholders love - maybe this guy - eh JOEY:

SMH folks - can't make this shite up
sell me this pen - lolzzz

MOO shills & touts
would not at all be surprised that "if" Wikilo ...
is MONI's target acquisition candidate - that Wikilo...
a.) already has substantive non-dilutive VC, Angel, or PP funding in-place
b.) already has significant clients lined-up and in Wikilo's basket for launch
c.) already has Wikilo Super App thru beta testing approvals & close to readied for launch
d.) already has an Advertising, Marketing & Public Relationships firm in place
e.) already has collaborated with some existing SuperApp's, e.g.: Alipay, WeChat, Tata Neu, Grab, etc.
after doing some drill down, Wikilo feels like its been in development for the past +5 years - minimum
"SuperApps can consolidate and replace multiple apps for customer or employee use and
support a composable business ecosystem"
"The global Super Apps market size was valued at $59-Bil. in 2022, and is projected to reach $722-Bil. by 2032, growing at a CAGR of 29% from 2023 to 2032. Super App is a mobile application that offers a wide range of services within a single platform."
what shocking is the fact that you have an inexperienced manipulative twice failed hack CEO
broadcasting intellectual prowess stating he's a Stanford "Financial Engineer" who understands finance
yet ran TOMDF to 3.3-BIL. OS Shares, and used toxic death spiral convertibles
employing the Astoria Queens financial mafia to fund themselves
and put out PR after PR about just how great TOMDF and Tollovid Chinese gromwell root extract was
and how Todos Medical was saving the world from COVID and was developing an ANTI-COVID pill

then switched to saving LH-COVID sufferers when the pandemic was declared over by the US-NIH
TOMDF also spent (+/-) $138-Mil. in aprox 5 yr's and would up dissolved
with unaudited books and an OTC Dark or Defunct Non-reporting status
just lookie here 3.3-Bil. OS shares - yes that's BILLION SHARES folks for starters:

now peruse the timing and dates of TOMDF's PR releases
and hold that up against TOMDF's 5 Year TA Chart
then tell me what you see...?

TOMDF Sham Exposed in PR Chronological Order
Todos Medical Announces Insolvency Proceedings in Israel
Dec 22, 2023 5:28pm EST
Todos Medical Provides Corporate Update: Retooling to Focus on Long COVID Epidemic
Sep 26, 2023 11:01am EDT
Todos Announces Notice of Allowance From USPTO for Patent Application Covering Use of Compositions In Tollovid™ and Tollovir™ Ingredients
Aug 30, 2023 8:19am EDT
Todos Medical Enters Into Letter of Intent for Provista Diagnostics to License PCR-based Sepsis Diagnostic Test AcuSept LDT Rights From Acumen Diagnostics
Mar. 28, 2023
Todos Medical Begins Trading on the OTC Pink Marketplace
Mar. 27, 2023
Todos Medical Supports Long COVID Awareness Day
Mar. 15, 2023
Todos Medical Enters Into Moratorium on Conversions Agreement With Convertible Note Holders
Mar. 13, 2023
Todos Medical Initiates Validation Plan for PCR-based Avian Influenza A (H5N1, Bird Flu) Test at CLIA/CAP Testing Lab Provista Diagnostics
Feb. 14, 2023
Todos Medical to Present at the RHK 2022 Disruptive Growth Conference
Dec. 06, 2022
Todos Medical’s 3CL Pharma Awarded Oral and Poster Presentations at World Antiviral Congress 2022
Nov. 29, 2022
Todos Medical to Attend World Antiviral Congress
Nov. 22, 2022
Todos Medical Enters Into Exclusive Worldwide Automated Retail Supply Agreement With Smart Automated Retail Network
Nov. 16, 2022
Todos Medical Terminates Tollovid™ Products License & Distribution Agreement with T-Cell Protect Hellas S.A.
Nov. 10, 2022
Todos Medical CLIA/CAP Lab Provista Submits Emergency Use Authorization Application for MonkeyPox PCR Test to US FDA
Oct. 14, 2022
Todos Medical Announces Successful Pilot Launch of Suite of PCR Testing Services by CLIA/CAP Lab Provista Diagnostics
Oct. 06, 2022
Todos Medical Receives Two Notices of Allowance from USPTO for AI-Based TBIA Pan-Cancer Blood Diagnostics Platform
Sep. 30, 2022
Todos Medical Announces USPTO Trademark Notice of Allowance for 3CL Protease Biomarker Diagnostics TolloTest™
Sep. 27, 2022
Todos Medical Enters Into $50M Contract to Supply Tollovid™ and CBD Products to Retail Distributor Company Nerd Hemp
Sep. 22, 2022
Todos Medical Announces NFL Hall of Famer Michael Irvin as Tollovid™ Sports Ambassador for #TolloUp Campaign
Sep. 16, 2022
Todos Medical Announces Preprint of Long COVID Case Study Participant with Confirmed Microclot and Hyperactivated Platelets who Benefitted from Tollovid
Sep. 15, 2022
Todos Medical & The Alchemist’s Kitchen to Host “Long COVID: An Integrated Approach” Event Benefitting Survivor Corps
Sep. 13, 2022
Todos Medical’s 3CL Protease Inhibitor Supplement Tollovid™ Featured in Wellness Magazine
Sep. 13, 2022
Todos Medical Partner NLC Pharma Assigns IP for Tollovir™, Tollovid™, and TolloTest™ into JV 3CL Pharma
Sep. 12, 2022
Todos Medical CEO Discusses Long COVID Economic Impact with Moneta Advisory Managing Partner Marc LoPresti
Sep. 09, 2022
Todos Medical Announce Kingcarlx as Brand Ambassador for the Tollovid #TolloUp Lifestyle Campaign
Sep. 02, 2022
Todos Medical Establishes Botanical Supplement Manufacturing Capabilities to Support Tollovid Global Expansion and Initiates CBD-A Production
Aug. 31, 2022
Todos Medical Expands COVID Onsite School Sample Collection to Include MonkeyPox Testing
Aug. 29, 2022
Todos Medical Announces Saliva-Only MonkeyPox Testing at CLIA/CAP Clinical Lab Provista Diagnostics
Aug. 26, 2022
Todos Medical Enrolls Female Long COVID Patient Unable to Secure Testing While Symptomatic for MonkeyPox into Provista Diagnostics Case Study Series
Aug. 25, 2022
Todos Medical Initiates MonkeyPox Diagnostic Case Studies After Being Approached by 4 Patients, Including 3 Women
Aug. 23, 2022
Todos Medical Announces Commercialization of Lesion and Saliva-Based MonkeyPox PCR Testing
Aug. 22, 2022
Todos Medical Announces Preprint of Data from First 100 Participants in IRB-waived Market Research Study of Supplementation with Tollovid in Long COVID
Aug. 19, 2022
Todos Medical Reports Pre-Print of Publication Detailing Tollovid Supplementation Following COVID-19 mRNA Vaccination in Long COVID
Aug. 15, 2022
Todos Medical Completes Trial Design for Tollovid™ Long COVID Clinical Study in Adults
Aug. 11, 2022
Todos Medical Announces PCR Validation of Wound and Respiratory Pathogen Panels at CLIA/CAP Lab Provista Diagnostics
Aug. 10, 2022
Todos Medical Announces First Two Contracts for PCR-based MonkeyPox Testing at CLIA/CAP Clinical Testing Laboratory Provista Diagnostics
Aug. 08, 2022
Todos Medical Announces First Long COVID Clinic Laboratory Services Agreement for Long COVID Panel Biomarker Partnership with Amerimmune Diagnostics
Aug. 05, 2022
Todos Medical Initiates Validation Plan for PCR-based MonkeyPox Test at CLIA/CAP Clinical Testing Laboratory Provista Diagnostics
Aug. 03, 2022
Todos Medical Announces Preprint of Acute COVID-19 Paxlovid Rebound Rescued by Tollovid
Aug. 01, 2022
Todos Medical Partners with Amerimmune Diagnostics on Long COVID Biomarker Panel
Jul. 28, 2022
Todos Medical Announces Preprint of Chronic Long COVID Case Study #8 of Paxlovid® Treatment Followed by Tollovid™ Dietary Supplementation
Jul. 22, 2022
Todos Medical Reports Case Study #7 in Acute and Long COVID
Jul. 12, 2022
Todos Medical Announces Positive Biomarker Data from Phase 2 Trial of Tollovir in Hospitalized COVID-19 Patients
Jun. 30, 2022
Todos Medical Releases Preliminary Data From IRB-Waived Tollovid® Market Research Study in Acute and Long COVID
Jun. 23, 2022
Todos Medical Retains Moneta Advisory Partners as Strategic Advisors for National Exchange Listing and 3CL Pharma Spinoff
Jun. 21, 2022
Todos Medical Completes Validation of 27 Pathogen UTI PCR Panel at Its CLIA/CAP Laboratory Provista Diagnostics
Jun. 13, 2022
Todos Medical Reports Day 45 Update for Case Study #6
Jun. 10, 2022
Todos Medical Reports Day 28 Update for Case Study #5 with Erectile Dysfunction
Jun. 07, 2022
Todos Medical Announces Enrollment Complete for Confirmatory Cohort of LymPro Alzheimer’s Blood Test Clinical Validation Trial vs. Amyloid PET
Jun. 03, 2022
Todos Medical Reports Day 26 Update For Case Study #4
Jun. 01, 2022
Todos Medical to Present Final Data from the Tollovir Phase 2 Clinical Trial in Hospitalized COVID-19 Patients at the Personalized Medicine World Conference
May 27, 2022
Todos Medical Reports Day 14 Update from Ongoing 30-Day Case Study of Paxlovid Rebound Patient
May 06, 2022
Todos Medical Receives New FDA Certificate of Free Sale for Tollovid® Max Strength Including 5 Day Dosing, 30 Day Dosing and 3CL Protease (3CLpro, Mpro, Nsp5) Inhibitor Claim
May 02, 2022
Todos Medical Initiates IRB-Waived Tollovid Market Research Study and Announces 50% Price Reduction Until May 30th, 2022
Apr. 27, 2022
Todos Medical Appoints Philippe Goix as Chief Commercial Officer for Provista Diagnostics
Apr. 21, 2022
Todos Medical Reports 2nd Long COVID Case Study and Launches Website for Physicians and Pediatricians to Indicate Interest in Participating in Tollovid Long COVID Clinical Study
Apr. 20, 2022
Todos Medical Targets Athlete Support with Informed Sport Certification for Tollovid by LGC Group
Apr. 19, 2022
Todos Medical Applauds White House Initiative on Long COVID and Announces New Tollovid® US FDA Certificate of Free Sale with Daily and Acute Dosing on the Label
Apr. 06, 2022
Todos Medical Announces Positive 3CL Protease Inhibition In Vitro Data Against BA.1 and BA.2 Omicron SARS-CoV-2 Variants
Apr. 04, 2022
Todos Medical Reports Case Study of Patient with Long COVID
Mar. 31, 2022
Billy Blanks® Endorses Todos Medical’s Line of Tollovid® Products
Mar. 28, 2022
Todos Medical Completes Acquisition of Key Assets and Intellectual Property from NLC Pharma
Mar. 16, 2022
Todos Medical Appoints Greg Meiselbach as Vice President of Government Affairs
Mar. 08, 2022
Todos Medical and NLC Pharma Announce Primary and Secondary Endpoints Met in NLC-V-01 Phase 2 Clinical Trial of Oral Antiviral 3CL Protease Inhibitor Tollovir™ in the Treatment of Hospitalized COVID-19 Patients
Jan. 27, 2022
Todos Medical Announces Data Lock in Tollovir Phase 2 Clinical Trial for the Treatment of Hospitalized COVID-19 Patients
Jan. 24, 2022
Pepperdine University Hosts the 22nd Annual Celebrity Flag Football Challenge®, Presented by Todos Medical’s Tollovid®
Jan. 19, 2022
Todos Medical Releases Videos of Recent CEO Interviews with Fox Business, NewsMax and Black News Channel on COVID Testing & Treatment Strategies
Jan. 18, 2022
Todos Medical to Announce Topline Results from Tollovir™ Phase 2 Clinical Trial from the Treatment of Hospitalized COVID-19 Patients on January 27th, 2022
Jan. 12, 2022
Todos Medical CEO to Appear on Fox Business to Talk COVID Testing & Tollovir on Tuesday, January 11, 2022 and is Presenting at Biotech Showcase 2022
Jan. 10, 2022
Todos Medical Records Record Weekly COVID PCR Testing Volumes at Provista
Dec. 27, 2021
Todos Medical CEO to Appear on Yahoo Finance Live! at 12:10PM EST with Akiko Fujita to Talk Pfizer’s Paxlovid Approval, Tollovir™ Phase 2 & COVID Testing
Dec. 23, 2021
Todos Medical Announces New cPass Neutralizing Antibody Testing Reference Lab Agreement for CLIA/CAP Lab Provista with Physician Group in 40 States
Dec. 22, 2021
Todos Medical Appoints Valentino Smith, MBA as Vice President of Marketing
Dec. 20, 2021
Todos Medical Announces “Test & Tollovid®” Holiday Package for Atlanta Area at Provista Diagnostics
Dec. 17, 2021
Todos Medical to Host Key Opinion Leader Webinar Entitled Tollovir™: a Potential Treatment for Covid-19
Dec. 16, 2021
Todos Medical Enters 90-Day Extension of Agreement with Convertible Note Holders on Moratorium on Conversions of Convertible Notes
Dec. 13, 2021
Todos Medical Interview and Presentation on Benzinga's All Access Conference Live December 14th at 10:20 AM EST
Dec. 10, 2021
Todos Medical Announces 2 New COVID-19 PCR Testing Reference Lab Agreements for CLIA/CAP Lab Provista Diagnostics
Dec. 07, 2021
CEOs Presenting at the Emerging Growth Conference on December 8; Register Now
Dec. 07, 2021
Todos Medical to Present at Upcoming Emerging Growth Conference
Dec. 06, 2021
Todos Medical Announces All 31 Patients Enrolled to Date in Tollovir® Phase 2 Clinical Trial in Hospitalized COVID-19 Patients Have Completed Study Participation
Dec. 02, 2021
Todos Medical Confirms SARS-CoV-2 PCR Test Kits Used at CLIA/CAP Lab Provista Diagnostics Detect Omicron Variant
Nov. 30, 2021
Todos Medical Enters into Binding Agreement to Acquire All 3CL Protease Biology-Related Assets and Intellectual Property from NLC Pharma
Nov. 29, 2021
Todos Medical Receives Purchase Order for 50,000 Bottles of Tollovid Daily from Its European Distribution Partner T-Cell Protect Hellas S.A for Initial Market Launch in Greece
Nov. 22, 2021
Todos Medical Announces Completion of Enrollment for COVID-19 Oral Antiviral 3CLPro (Mpro) Inhibitor Tollovir Phase 2 Clinical Trial Interim Analysis in Severe and Critical Hospitalized Patients
Nov. 19, 2021
Todos Medical Announces Tollovid® Products Exclusive License & Distribution Agreement with T-Cell Protect Hellas S.A. Covering 30 Countries in Europe
Nov. 18, 2021
Todos Medical Announces Addition of Semi-Quantitative Titer Claims to the Emergency Use Authorization for the cPass Neutralizing Antibody Test
Nov. 17, 2021
Todos Medical’s Provista Laboratory to Boost Top Line Through New Atlanta-based Reference Lab Agreement for COVID PCR, cPass Neutralizing Antibody & Respiratory Pathogen Panel Tests
Nov. 16, 2021
Todos Medical Reports Third Quarter 2021 Business and Financial Results
Nov. 15, 2021
Todos Medical Sees Significant Ramp Up in COVID PCR Testing Volume at Its Provista CLIA/CAP Lab with New Automation in Place
Nov. 02, 2021
Todos Medical Announces 2nd Clinical Trial Site for Tollovir Phase 2 Clinical Trial for the Treatment of Hospitalized COVID-19 Patients
Nov. 01, 2021
Todos Medical Announces Publication in Nature Neuroscience That Describes the Main Protease (3CL Protease) Causes Microvascular Brain Pathology
Oct. 25, 2021
Todos Medical Announces Publication in Journal of Clinical Virology Highlighting Clinical Utility of cPass Semi-Quantitative Neutralizing Antibody Test in SARS-CoV-2 Risk Assessment Post-Vaccination or Recovery from Infection
Oct. 21, 2021
Todos Medical to Conduct Webinar Highlighting the cPass SARS-COV-2 Neutralizing Antibody Test for AMDA, The Society for Post-Acute and Long-Term Care Medicine, on October 22, 2021 at 12:00 PM EDT
Oct. 14, 2021
Todos Medical Announces Positive Data in Hospitalized and Outpatient Setting for TolloTest™, a Novel SARS-CoV-2 3CL Protease Biomarker Assay
Oct. 06, 2021
Todos Medical Provides Update on COVID-19 Oral Antiviral 3CL Protease (Main Protease) Inhibitor Tollovir Clinical Development Program in Light of Molnupiravir Clinical Trial Data from Merck
Oct. 04, 2021
Todos Medical Announces Positive Observational Trial Results for Oral Antiviral 3CL Protease (MPro) Inhibitor Tollovir®
Sep. 30, 2021
Todos Medical Receives Approval by Amazon to Sell Immune Supplements Tollovid® & Tollovid Daily™
Sep. 27, 2021
Todos Medical Reports Second Quarter 2021 Financial Results
Fri, Sep. 24, 2021
UPDATE: Todos Medical Announces 90-Day Moratorium on Conversions of Convertible Notes and Lockup of Common Stock Sales with Convertible Note Holders
Sep. 24, 2021
Todos Medical Launches Tollovid Daily™ via Subscription at MyTollovid.com
Sep. 23, 2021
Todos Medical Receives Trademark Notice of Allowance from USPTO for Its 3CL Protease Inhibitor Oral Antiviral Drug Candidate Tollovir™
Sep. 20, 2021
Todos Medical Now Accepting Cryptocurrency for the Purchase of Tollovid® and Tollovid Daily™ Through Coinbase Commerce
Sep. 17, 2021
Todos Medical Announces 90-Day Moratorium on Conversions of Convertible Notes and Lockup of Common Stock Sales with Convertible Note Holders
Sep. 15, 2021
Todos Medical’s CLIA/CAP Lab Provista Completes Validation of Combination COVID, Influenza A & B, RSV & hMPV Respiratory Panel Test
Sep. 01, 2021
Todos Medical’s CLIA/CAP Lab Provista Completes Validation of High-Capacity SARS-CoV-2 Variant Testing Service Capable of Identifying Known Variants
Aug. 26, 2021
Todos Medical’s CLIA/CAP Lab Provista Enters Into Reference Lab Agreement with Meadowlands Diagnostics to Provide cPass Neutralizing Antibody Blood Testing for Quantifying and Monitoring Key COVID-19 Immunity Biomarkers
Aug. 24, 2021
Todos Medical Completes Validation of cPass Neutralizing Antibody Blood Test at Provista Diagnostics to Quantify and Monitor Key Biomarkers of COVID-19 Immunity
Aug. 23, 2021
Todos Medical CEO to Appear on Fox Business Network Varney & Company Today at 11:40am to Discuss the Company’s Launch of cPass Neutralizing Antibody Testing for Monitoring COVID-19 Immunity
Aug. 23, 2021
Update: Todos Medical Receives Trademark Notice of Allowance from USPTO for Its 3CL Protease Inhibitor Dietary Supplement Tollovid™
Aug. 05, 2021
Todos Medical Receipt Notice of Allowance from USPTO for 3CL Protease Inhibitor Dietary Supplement Tollovid™
Aug. 05, 2021
Todos Medical Makes Final Payment to Close Provista Diagnostics Acquisition
Aug. 02, 2021
Todos Medical Retains CRO for COVID-19 Outpatient and Inpatient Phase 2/3 Clinical Trials for Oral Antiviral 3CL Protease Inhibitor Tollovir
Jul. 28, 2021
Todos Medical Receives New FDA Certificate of Free Sale for Tollovid Daily™ Including 3CL Protease Inhibitor Claim
Jul. 22, 2021
Todos Medical to Launch EUA Authorized cPass SARS-CoV-2 Neutralizing Antibody Kit Through Agreement with Fosun Pharma
Jul. 19, 2021
Todos Medical Appoints Ilanit Halperin, CPA as Corporate Controller
Jul. 07, 2021
Todos Medical Receives FDA Certificate of Free Sale for New 5-Day Tollovid Dosing Regimen
May 04, 2021
Todos Medical Launches Phase 2 Clinical Trial of Its Antiviral 3CL Protease Inhibitor NLC-V-01 (Tollovir) in Hospitalized COVID-19 Patients
Apr. 19, 2021
Todos Medical Receives Notice of Allowance from European Patent Office for Patent Application Covering Diagnosis of Cancer Using Proprietary Artificial Intelligence TBIA Immune Profiling Platform
Apr. 08, 2021
Todos Medical Completes Automation Equipment Installation and Training for Meadowlands Diagnostics
Apr. 01, 2021
Todos Medical Enters Into Distribution Partnership with Osang Healthcare for the GeneFinder Plus™ COVID-19 Plus RealAMP Kit in the United States
Mar. 30, 2021
Todos Medical Completes Automation Equipment Installation and Training for a Laboratory Client in Brooklyn, NY
Mar. 29, 2021
Todos Medical Enters Into Automation and Reagent Supply Agreement with MAJL Diagnostics
Mar. 23, 2021
Todos Medical Announces $7.2M in Sales for February 2021, a 38% Month Over Month Increase From Sales of $5.2M in January 2021
Mar. 12, 2021
Todos Medical Applauds Senate and House Passage of the American Recovery Act
Mar. 11, 2021
Todos Medical Announces Strategic Investment by Yozma Group Korea
Jan. 25, 2021
Todos Medical Announces $4.8 Million in Sales for December 2020, a 50% Month Over Month Increase in Sales from November 2020
Jan. 11, 2021
Todos Medical Announces Agreement to Supply Natural Wellness Clinics with COVID-19 Testing Products to Support Initiatives in the Commonwealth of Kentucky
Dec. 23, 2020
Aditxt Signs Distribution Agreement with Todos Medical Ltd. For AditxtScore™ for COVID-19 Immune Monitoring Service
Dec. 14, 2020
Todos Medical Announces Commercial Launch of Proprietary 3CL Protease Inhibitor Dietary Supplement Tollovid™ at The Alchemist’s Kitchen
Dec. 07, 2020
Todos Medical Announces $3.2 Million in Sales for November 2020, a 269% Increase Sequential Month over Month Sales Growth from October 2020
Dec. 02, 2020
Todos Medical Provides Strategic Outlook
Dec. 01, 2020
Todos Medical Completes Installation of Lab Automation Equipment at Wisconsin Lab Client to Support COVID-19 PCR Testing
Nov. 24, 2020
Todos Medical Announces Positive In Vitro Data for Tollovid™ Confirming 3CL Protease Inhibition Mechanism of Action
Nov. 20, 2020
Todos Medical Announces Positive Initial Clinical Proof-of-Concept Data for a Rapid SARS-CoV-2 3CL Protease Detection Assay
Nov. 18, 2020
Todos Medical Announces Clinical Validation and National CLIA Certification for MOTO+PARA Mobile High Complexity Labs
Nov. 17, 2020
Todos Medical Announces $867,000 in Sales for October 2020
Nov. 12, 2020
Todos Medical Trademarks Tollovid for Dietary Supplement NLC-001 and Enters Into Private Label & Distribution Agreement With The Alchemists Kitchen
Oct. 29, 2020
Todos Medical Announces Instrument Validation Complete at MOTO+PARA Mobile Lab
Oct. 26, 2020
Todos Medical Announces Distribution Agreement with Adial Pharmaceuticals to Market the FDA, EUA Authorized, Assure/FaStep Point-of-Care Covid-19 Antibody Tests
Oct. 22, 2020
Todos Medical Receives Notices of Allowance From the European Patent Office Covering Use of TBIA Cancer Platform to Detect Benign Colon Cancer
Oct. 20, 2020
Todos Medical Reports $2.0 Million in Revenue for the Third Quarter of 2020
Oct. 13, 2020
Todos Medical Enters into Exclusive COVID-19 Testing Supply Agreement with MOTO+PARA’s National Mobile CLIA Lab Partner Integrated Health
Oct. 07, 2020
Todos Medical Enters into Exclusive Branding and Distribution Agreement with Melbourne Biotech for 96 and 384 Well RT-PCR Machines in the United States
Sep. 30, 2020
Todos Medical Enters Into COVID-19 PCR Testing Implementation and Equipment Financing Partnership with AID Genomics
Sep. 25, 2020
Todos Medical Expands Existing Contract for COVID-19 PCR Testing Equipment & Supplies with Wisconsin-based Laboratory to $47.5 Million
Sep. 22, 2020
Todos Medical CEO Releases Letter to Shareholders
Sep. 18, 2020
3CL Protease Inhibitor NLC-001 Added to COVID-19-focused Joint Venture Between Todos Medical and NLC Pharma
Sep. 17, 2020
Todos Medical Expands Partnership with Care G.B. Plus for Proprietary TBIA Cancer Tests to Include Europe, Israel and Africa
Sep. 16, 2020
Todos Medical Announces Commercial Launch of Todos Branded COVID-19 qPCR Test Kits in the United States
Sep. 11, 2020
Todos Medical Announces August Sales of $1,190,000, Up 100% Compared with July
Sep. 03, 2020
Todos Medical Enters Into Partnership With Pangea for COVID-19 Testing and Contact Tracing
Sep. 02, 2020
Todos Medical Announces $23,760,000 Contract for COVID-19 PCR Testing Equipment & Supplies With Wisconsin-based Laboratory
Aug. 31, 2020
Todos Medical to Present at The LD 500 Virtual Conference
Aug. 31, 2020
Todos Medical Announces $4,500,000 Contract for COVID-19 PCR Testing Equipment & Supplies with New York-based Laboratory
Aug. 27, 2020
Todos Medical Appoints Marsha Fontanive as VP of Sales and Chris Gross as VP of Sales Operations
Aug. 25, 2020
Todos Medical Announces $1.2 Million Contract for COVID-19 PCR Testing Equipment & Supplies with Texas-based Laboratory
Aug. 24, 2020
Todos Medical Announces $6,000,000 Contract for COVID-19 PCR Testing Equipment & Supplies with New York-based Laboratory
Aug. 21, 2020
Todos Medical Announces Positive Proof-of-Concept Data for Novel 10-Minute Point-of-Care Saliva-based Test Detecting Active SARS-CoV-2 Infection
Aug. 17, 2020
Todos Medical Acquires Distribution Rights to SARS-CoV-2 Rapid Point-of-Care Antigen Test and Rapid Point-of-Care PCR Test
Aug. 14, 2020
Todos Medical Announces July Sales of COVID-19 Tests
Aug. 07, 2020
Todos Medical Announces Common Stock Purchase Agreement with Lincoln Park Capital Fund, LLC
Aug. 06, 2020
Todos Medical Appoints Dr. Jorge Leon as Consulting Chief Medical and Scientific Officer of Infectious Disease and Oncology
Aug. 04, 2020
Pathnova Laboratories (PATHNOVA) Enters into Partnership to Commercialize Todos Medicals COVID-19 and Breast Cancer Tests in Singapore
Aug. 03, 2020
Todos Medical Completes Acquisition of Breakthrough Diagnostics, Gains Full Rights to the Alzheimer’s Blood Diagnostic LymPro™ Test from Amarantus
Jul. 28, 2020
Todos Medical Announces SARS-nCoV-19 Testing Kit Contract with NJ-Based Best Supply Clinical Laboratory
Jul. 27, 2020
Todos Medical Expands Management Team and Advisory Board
Jul. 20, 2020
Todos Medical Announces ANVISA Authorization in Brazil for 3D Med qPCR Kits and ANDis Extraction S
Jul. 16, 2020
Todos Medical Releases Letter to Shareholders
Feb 07, 2020 9:15am EST
Todos Medical Enters into Exclusive Option Agreement to Acquire Provista Diagnostics and Announces Management Changes
Jan 09, 2020 8:00am EST
Todos To Present At Upcoming 12th Annual LD Micro Main Event
Dec 06, 2019 7:29am EST
Todos and Amarantus JV Announces Full Enrollment for Clinical Trial of LymPro Alzheimer’s Blood Test Relationship with Amyloid PET
Nov 14, 2019 11:02am EST
Todos Medical to Present at Dawson James Small Cap Growth Conference
Oct 24, 2019 9:25am EDT
Todos Medical and Orot+ Announce First Patient Enrolled in Pre-Commercial Launch Preparations of TM-B1 and TM-B2 Breast Cancer Blood Screening Tests in Europe
Sep 24, 2019 9:14am EDT
Todos Medical Signs Binding Term Sheet with HWH World for Network Marketing of Blood Tests for the Early Detection of Breast Cancer
Sep 13, 2019 7:30am EDT
Todos Medical Enters into MOU to Expand Existing Early Detection Breast Cancer Blood Test Distribution Partnership with Orot+ into Japan
Sep 03, 2019 1:29pm EDT
Todos Medical Appoints Dr. Jorge Leon as Medical Advisor
Aug 06, 2019 7:00am EDT
Todos Medical Announces Positive Alzheimer's Biomarker Clinical Data from LymPro vs. Amyloid PET Interim Analysis
Jul 15, 2019 7:00am EDT
Todos Medical CEO Presented Corporate Update And Roadmap at The LD Micro Conference
Jun 17, 2019 8:30am EDT
Todos Medical Announces Positive Clinical Trial Data for Breast Cancer Blood Test TM-B1 in Dense Breasts
Jun 03, 2019 8:00am EDT
Todos Medical Exercises Option to Acquire Remaining 80.01% of Breakthrough Diagnostics from Amarantus After Review of Alzheimer's Blood Test Data
May 28, 2019 8:57am EDT
Todos Medical Receives Approval from Israeli Ministry of Health (AMAR) to Market and Sell its Breast Cancer Screens in Israel
May 23, 2019 8:00am EDT
Todos Medical Enters Exclusive Distribution Agreement with Care G.B. Plus, Ltd. for its Breast Cancer Screening Tests in Israel
May 21, 2019 8:00am EDT
Todos Medical Announces Voting Results from 2019 Annual Shareholder Meeting
May 03, 2019 8:52am EDT
Todos Medical Receives Notice of Allowance from the USPTO Covering the Early Diagnosis of Lung Cancer Using the Todos Biochemical Infrared Analysis (TBIA)
May 01, 2019 8:00am EDT
Todos Medical Enters Into Definitive Agreement With Orot+ for Exclusive Distribution Rights of its Breast Cancer Screening Tests in 2 European Countries
Apr 15, 2019 8:00am EDT
Todos and Amarantus Announce Completion of LymPro PET 1 Study by Leipzig University for Alzheimer’s Blood Diagnostic LymPro Test 2.0
Apr 08, 2019 7:30am EDT
Todos Medical to Present at Two Upcoming Investor Conferences
Mar 28, 2019 8:00am EDT
Todos Medical Appoints Dr. Colin Bier to Its Board of Directors
Mar 27, 2019 8:00am EDT
Todos Medical Raises $1,350,500 In Funding and Finalizes Joint Venture Agreement with Amarantus Bioscience
Feb 28, 2019 9:30am EST
Todos Medical Presents Encouraging Scientific Abstract at the San Antonio Breast Cancer Symposium (SABCS)
Dec 06, 2018 4:00pm EST
Todos Medical Enters into Joint Venture Agreement with Amarantus to develop Alzheimer’s Blood Diagnostic LymPro Test 2.0
Dec 03, 2018 8:30am EST
Todos Medical to Present Scientific Abstract at the Upcoming San Antonio Breast Cancer Symposium (SABCS)
Nov 29, 2018 8:30am EST
Todos Medical, Ltd. Announces Additional Patent Issuance Covering the Use of IR Spectrum of Blood Plasma For Indicating the Presence of Breast Cancer
Oct 17, 2018 8:30am EDT
Todos Medical, Ltd. Signs Non-Binding Memorandum of Understanding with Orot+ for Distribution Rights of its Breast Cancer Screening Tests in Romania and Austria
Sep 13, 2018 8:30am EDT
Todos Medical, Ltd. Appoints Herman Weiss, MD MBA as New Chief Executive Officer
Jul 31, 2018 4:03pm EDT
Todos Medical reports on results of clinical trials for its blood test for breast cancer screening (TM - B1) conducted at Kaplan Hospital in Rehovot, Israel
Mar 22, 2018 8:00am EDT
Todos Medical initiates clinical trial of its blood test for breast cancer in Kaplan Hospital, Rehovot, Israel for subjects undergoing breast cancer screening
Mar 05, 2018 8:00am EST
U.S. Patent and Trademark Office has granted to Todos Medical two additional patents
Feb 12, 2018 8:00am EST
Mr. David Ben Naim, CPA, MBA Joins Todos Medical as Chief Financial Officer
Feb 06, 2018 8:00am EST
Todos Medical Appoints Dr. Meir Silver as Vice President of Clinical Research and Regulatory Affairs
Aug 29, 2017 8:30am EDT
SMH - no need to look any further than the TOMDF Boiler Room Pump Boys

and we all know who those shills touts and dolts are...!
MOO sham
Man Behind the Buzz
Gerald Commissiong - twice failed Stanford University "Financial Engineer" CEO hack
now looking for your money - so he can do it a 3rd time
my-o-my ... how just a few years can change things on a NY dime
especially when things are built on illusions, deceiving SH's and a Make It Up as We Go Along portfolio

SMH - only in America can you rip people off and get away with it
all that's left are broke AMBS and TOMDF SH's, and an indelible 'stain' on Wall St.
MOO sad
nice...!
EOM
strong like bullshite more like it ... down -99% in 1 day
Printing $0.000001
52 Week Range
$0.000001 - $0.004
Holy bat-cack Batman...!

all the shill, tout, and dolt TOMDF Boiler Room Pumper Boy's sure helped bigley this turd - eh...?
of course, along with the $720k/yr. salaried CEO twice failed Stanford University "Financial Engineer" hack
not including CEO travel, auto, speaking engagement's, retirement bene's, options, etc etc etc
but hey, where did (+/-) $138-Mil. go in just 5 years time - do we have audited books to tell the tail...?
SMH - you simply can't make this stuff up folks

oh and they don't like me - ha...!
MOO - TOMDF death knell
God's Devine supernatural abundance & Grace to you & yours this 2024
All the best,
source...?
of course you can't provide one - because you make things up Mista Redspudda
including all the hype you and your ilk made-up about Tolloup Buttercup 9 horsie TOMDF
oh, did you state it was too late to provide your Tolloup testimonial for Todos Medical's CrowdFunding Launch...?

sorry Sporto - you are seriously delusional...!
SMH - if it weren't so pathetic, it would be funnier than hell...
MOO putz
not sure what you expect me to do ...
regarding putting a "good taste" in MONI shareholder's mouths...
I'm just looking to these top 3 basic acquisition milestones:
a.) final achievement on the OTC 8K filing
b.) understand the FMV of the incoming acquisition
3.) get to parse thru what it's Business Plan is
obviously, there are way many other things that could be considered 'positive catalysts'
all of which could easily contribute to a MONI run
but after what appears to be a 3rd and hopefully final acquisition within a lengthy 14-month period
the curent pps pretty much says it all...
hoping for the very best, open to being underwhelmed at the very worst
BAF MONI...!
MOO
no one - and I mean no one - believes a single word you say
except your faithless shoe shine Gerry loving multiple alias burner phone fake IP addy lap dawg JOEY "Lunch Bucket" FORKIT
and especially if its coming from the AMBS TOMDF or VERB threads
shill tout and dolt

you credibility is permanently shot Sporto
SMH - cant make this stuff up folks...!
but hey, here's to you brah:

MOO hooie
complete and unadulterated horse manure Redspudda
but then again - what would anyone expect from the AMBS TOMDF & VERB etc. shill tout & dolt
printing $0.0001 and DISSOLVED - you did real good on this one Sport - lolzzz
Giddyup Tolloup Buttercup is nothing more than a dung ball
MOO shite
and IMHO herein "lies" your problem Sporto...
"problem is, nothing is ever realized ... how many times do they have to deceive investors before someone figures out it's all a scam"
"Crypto is a volatile asset in general, prone to significant price swings. Some crypto crashes are because of systemic issues within crypto, such as the collapse of FTX in 2022. Other times, macroeconomic factors such as interest rates and inflation can push values down. Jan 11, 2024"



Printing $0.0001 and no volume - nice
That’s all that really matters…
Everything else is all TOMDF pumper boy shill tout and dolt hooie…!
TOMDF dissolved
Lolzzz
proof please
"They can't purchase stock according to you.
They can't purchase Tollovid according to you"



here are the embarrassing, sad and pathetic excuses Gerald Commissiong has to offer the Street
as a twice failed AMBS & TOMDF Stanford University "Financial Engineer"
for his ineptness and catastrophic mis-management of TOMDF - now a "DISSOLVED" public security
what an absolute "Shite Show" this CEO created for his loyal AMBS & TOMDF shareholders
this piece reads like a bad Harlequin Romance novel w/ no happy ending for any one
except maybe GC's $720k/yr. salary bank account, + perks, bene's, travel, speaking engagements, etc.

MOO pathetic