Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
ShipB, just so you know, UTSW's Alan Schroit is THE MAN re: Exosomes, not the good Dr. Brekken.
Alan J Schroit (PPHM SAB, UTSW Profile: http://tinyurl.com/yaqnvcvq )
Dr. Schroit’s Articles: http://tinyurl.com/lqbd8kt
Exosomes: http://tinyurl.com/yacaw8xn
RM, you left out that 7-21-17 is the date by which we need 10 trading days above $1. You almost made it seem that we need the 10days by 7-7-17. I’m sure you didn’t mean to phrase it that way.
RM: “11 trading days until 7/7 RS date and to avoid RS we need 10 days with $1 min. ain't gonna happen.”
THE CORRECT NASDAQ REQUIREMENT:
PPHM’s pps must-reach $1 by 7-7-17 and stay above $1 for 10 days in order to evidence a closing bid price of $1.00 or more for a min. of 10 prior consecutive trading days on or before July 21, 2017.
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131992491
6-20-17/13D: Group Ronin & SW-Partners add 2,647,193 COMMON/Mar10-June16 (since last COMMON 13D filed 3-10-17)
6-20-17/13D: Ronin Cap.(John Stafford III) & SWPartners(Stephen White), a 13D “Group”, adds 2,647,193 COMMON during Mar10-June16.
That brings total ownership of the Group to:
26,313,734 COMMON, 8.8% of 297,709,478 common O/S at 3-10-17.
123,499 PREFERRED, 7.5% of 1,647,760 preferred O/S at 1-31-17 (convertible to 1,029,158 Common @8.333).
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +20,631,977 +51,364 20,631,977 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +3,034,564 +25,661 23,666,541 77,025 http://tinyurl.com/ydxra96u
4-17-17 3/28/17-4/10/17 0 +23,334 23,666,541 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 23,666,541 123,499 http://tinyurl.com/mgnn92x
6-20-17 3/10/17-6/16/17 +2,647,193 0 26,313,734 123,499 http://tinyurl.com/y76q5rqu
SPLITOUT 13D GROUP into Ronin(John Stafford III) and SW-Partners(Stephen White):
(since last COMMON 13D, filed 3-10-17)
Ronin Capital (Stafford) +1,997,194 +39,974 22,213,734 111,699
SW-Partners (White) +650,000 +6,500 4,100,000 11,800
Jim, pps must-reach $1 by 7-7-17 and stay above $1 for 10 days in order to evidence a closing bid price of $1.00 or more for a min. of 10 prior consecutive trading days on or before July 21, 2017.
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131992491
= = = = = = = = = = =
6-7-17: Hearing panel grants PPHM until 7-21-17 to regain compliance (3.3mos extension, from 4-10-17 to 7-21-17)...
Form 8-K: http://tinyurl.com/yauwzhum
Peregrine Pharmaceuticals, Inc. (the “Company”) recently attended a hearing before the NASDAQ Hearings Panel (the “Panel”) pursuant to its previously disclosed intent to request a hearing before the Panel in order to (i) present its plan to regain compliance with the $1.00 minimum closing bid price requirement set forth in the NASDAQ Listing Rule 5550(a)(2) (the “Minimum Bid Price Requirement”) and (ii) request that the Panel allow the Company additional time to implement the plan. On June 1, 2017, the Company received a decision letter from the NASDAQ Office of General Counsel stating that the Panel has granted the Company until no later than July 21, 2017 to regain compliance with the Minimum Bid Price Requirement, subject to certain conditions, including but not limited to, having evidenced a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days.
As previously disclosed, the Company’s stockholders approved a reverse stock split of the Company’s common stock at a ratio of up to 1-for-7, to be determined by the Company’s Board of Directors in its sole discretion. However, if the Company chooses to effect a reverse stock split in order to regain compliance with the Minimum Bid Price Requirement, it must do so no later than July 7, 2017, in order to evidence a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days on or before July 21, 2017.
Hutschi, that PGN635 Imaging article is from 12-2013. Another one of those shady bloggers that copy/pastes in something without stating the real PUB. DATE and making it look like it’s brand new. I hate that sort of stuff. It is a nice article, however, with Dr. Thorpe as Senior author, so thanks for posting.
12-19-2013/PLOS: “Highly Specific PET Imaging of Prostate Tumors in Mice with an Iodine-124-Labeled Antibody Fragment That Targets Phosphatidylserine”
Jason H. Stafford, Guiyang Hao, Anne M. Best, Xiankai Sun, Philip E. Thorpe
“Phosphatidylserine (PS) is an attractive target for imaging agents that identify tumors and assess their response to therapy. PS is absent from the surface of most cell types, but becomes exposed on tumor cells and tumor vasculature in response to oxidative stresses in the tumor microenvironment and increases in response to therapy. To image exposed PS, we used a fully human PS-targeting antibody fragment, PGN635...”
https://doi.org/10.1371/journal.pone.0084864
SHADY COPY/PASTE Blogger:
Posted on June 17, 2017 by Jose Wallace...
(no indication as to Actual Pub. Date or link to the article itself)
http://stopvivisection.info/phosphatidylserine-ps-is-an-attractive-focus-on-for-imaging-agencies-that/
Avid Revs FY17(fye 4-30-17) Guidance $60-65mm. I went back 4 prior years – they’re not in the habit of not beating the upper-ends of March Guidances. With Revs for 1st 3qtrs = (5.6 + 23.4 + 10.7) = $39.7mm, if they follow suit, Q4 Revs will be at least (65mm – 39.7) = $25mm.
...How close would that get us to Cash-Flow positive for Q4, should it happen? OP. CASH BURN for Q3(1-31-17) was: $6,274,000, and that's with Revs=$10.7mm at GM=26%.
3-13-17/PR: “Avid Revenue Guidance Increased to $60-65mm for FY’17; Contracted B/L=$70mm” http://tinyurl.com/grhwjvy
ACTUAL FY17= $??.?mm (reported ~7-13-17)
- - - - - - - - - - - - - - -
PS: ALL of the above BEFORE this:
5-2017: Avid II (Myford) adds 2 MilliporeSigma Mobius 2,000L single-use bioreactors; total mfg. capacity now ">11,000L".
...S.King: “We have already secured customer commitments for the 2,000L capacity as soon as it is installed in our Myford facility [Avid II]… Once they're operating in campaign mode, that brings us to well over $100mm in total potential revenue between our 2 mfg. Facilities.”
http://tinyurl.com/ky7bmu4
5-10-17: Halozyme comments on Avid II(Myford) expansion in their 3-31-17/10Q pub. 5-9-17: http://tinyurl.com/mrl34uk
..."validation of the new facility is scheduled to end in Q2/2017… Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20.”
PPHM REVS-BY-QTR TABLE, now thru FY17'Q3(qe 1-31-17), per the 10-Q ( http://tinyurl.com/zlozrv4 ) issued 3-13-17.
• 3-13-17: FY'17 (May'16-Apr'17) Avid revs guidance $60-65mm (Committed B/L=$70mm).
• Deferred-Revs at 1-31-17 total $26.4mm, UP from $18.0mm at 10-31-16.
• Cust.Deposits at 1-31-17 total $26.2mm, DOWN from $26.9mm at 10-31-16.
• Inventories at 1-31-17 total $33.8mm, UP from $25.9mm at 10-31-16.
• Avid’s Gross-Profit over last 4 qtrs: $22.3mm on revs of $58.5mm (GP%=38%)
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 23,370,000 15,441,000 7,929,000 34%
FY17Q3 1-31-17 10,747,000 7,974,000 2,773,000 26%
PPHM’s Nasdaq $1/min. Non-compliance progression
4-12-16 8-K: “1st Notice of Non-Compliance” (auto 180 day extension): PPS must be $1+ for 10 consecutive days prior to 10-10-16 to become compliant.
https://www.sec.gov/Archives/edgar/data/704562/000101968716005830/peregrine_8k.htm
10-11-16 8-K: “2nd Notice of Non-Compliance” (2nd auto 180 day extension): PPS must be $1+ for 10 consecutive days prior to 4-10-17 to regain compliance.
https://www.sec.gov/Archives/edgar/data/704562/000168316816000298/peregrine_8k.htm
6-7-17 8-K: “Hearing Panel grants PPHM Extension Appeal”: PPS must be $1+ for 10 consecutive days prior 7-21-17 to regain compliance.
...“If the Company chooses to effect a RS in order to regain compliance with the $1 rule, it must do so no later than July 7, 2017, in order to evidence a closing bid price of $1.00 or more for a min. of 10 prior consecutive trading days on or before July 21, 2017.”
https://www.sec.gov/Archives/edgar/data/704562/000168316817001520/peregrine_8k.htm
MORE: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131992491
Stoney, pps must-reach $1 by 7-7-17 (not 6/23) and stay above $1 for 10 days in order to evidence a closing bid price of $1.00 or more for a min. of 10 prior consecutive trading days on or before July 21, 2017.
See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131992491
Got 3.3mos extension, from 4-10-17 to 7-21-17.
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
PL: “Switching gears now, I’d like to address our plans to preserve our NASDAQ listing. At a backdrop, last April, we were notified by the NASDAQ that our share price did not meet the $1 min. bid price for 30 consecutive days, which is a requirement for continued listing on the NASDAQ market. At that time, we were automatically afforded an initial compliance period of 180 days or until Oct. 10, 2016 to regain compliance. At the end of the initial compliance period, we requested and were granted a 180 day extension to regain compliance. This 2nd extension period will end on April 10, 2017. Let me discuss our strategy and future steps we plan to take over the next few months..."
See: http://tinyurl.com/yagoqth5
= = = = =6-7-17 8-K:
"The Panel has granted the Company until no later than July 21, 2017 to regain compliance with the Minimum Bid Price Requirement, subject to certain conditions, including but not limited to, having evidenced a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days.
...
if the Company chooses to effect a RS in order to regain compliance, it must do so no later than July 7, 2017, in order to evidence a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days on or before July 21, 2017.
https://www.sec.gov/Archives/edgar/data/704562/000168316817001520/peregrine_8k.htm
Panel grants PPHM until 7-21-17 to regain compliance...
6-7-17 Form 8-K:
Peregrine Pharmaceuticals, Inc. (the “Company”) recently attended a hearing before the NASDAQ Hearings Panel (the “Panel”) pursuant to its previously disclosed intent to request a hearing before the Panel in order to (i) present its plan to regain compliance with the $1.00 minimum closing bid price requirement set forth in the NASDAQ Listing Rule 5550(a)(2) (the “Minimum Bid Price Requirement”) and (ii) request that the Panel allow the Company additional time to implement the plan. On June 1, 2017, the Company received a decision letter from the NASDAQ Office of General Counsel stating that the Panel has granted the Company until no later than July 21, 2017 to regain compliance with the Minimum Bid Price Requirement, subject to certain conditions, including but not limited to, having evidenced a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days.
As previously disclosed, the Company’s stockholders approved a reverse stock split of the Company’s common stock at a ratio of up to 1-for-7, to be determined by the Company’s Board of Directors in its sole discretion. However, if the Company chooses to effect a reverse stock split in order to regain compliance with the Minimum Bid Price Requirement, it must do so no later than July 7, 2017, in order to evidence a closing bid price of $1.00 or more for a minimum of 10 prior consecutive trading days on or before July 21, 2017.
6-7-17 8K: https://www.sec.gov/Archives/edgar/data/704562/000168316817001520/peregrine_8k.htm
= = = = =
4-12-17/8K: Nasdaq Delisting notice per $1 min. rule; Peregrine to Appeal for Extension See: http://tinyurl.com/yagoqth5
The Hearing-Panel typically issues-a-decision within 30 days of-the-hearing. I do realize the word used is “typically”...
D. DECISION. The Hearing Panel typically issues a decision within 30 days of the hearing.
* For Peregrine, decision could be as late as (6-2-17 + 30days) = 7-2-17 (Sunday).
More: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131733774
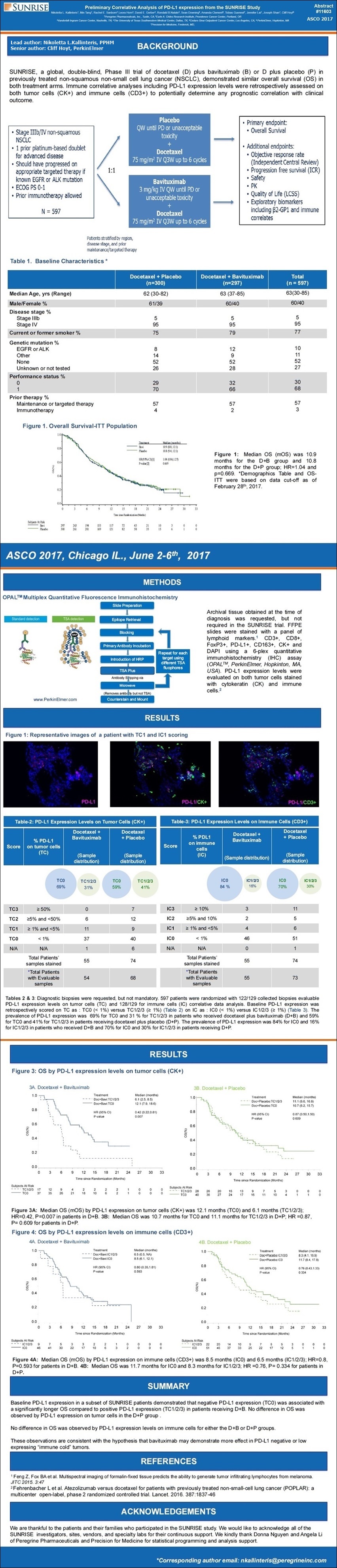
ASCO'17: “Prelim-Correlative-Analysis of PD-L1 Expr. from SUNRISE (Biomarker#4)” - press release, poster image, abstract detail...
6-5-17/PR: Peregrine Pharmaceuticals Presents Preliminary Correlative Analysis of PD-L1 Expression from SUNRISE Trial at ASCO 2017
-- Negative PD-L1 Expression was Associated with a Significantly Longer Median Overall Survival Compared to Positive PD-L1 Expression in Patients Receiving Docetaxel Plus Bavituximab
-- Presented Results Support Hypothesis that Bavituximab May Demonstrate Greater Effect in "Cold" Tumors Expressing Low to No PD-L1
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1028997
TUSTIN, June 05, 2017: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by advancing its proprietary R&D pipeline and manufacturing high quality products for biotechnology and pharmaceutical companies, today announced the presentation of promising new data from its Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC). Presented results demonstrated that patients in the study's bavituximab treatment arm who had low baseline PD-L1 expression levels had a statistically significant improvement in median overall survival (mOS) as compared to patients in the same treatment arm who had higher baseline levels of PD-L1. Data were presented by Peregrine scientists at the Annual Meeting of the American Society of Clinical Oncology (ASCO), being held June 2-6, 2017 in Chicago.
Data presented demonstrated that patients in the study's docetaxel plus bavituximab (D+B) treatment arm with a pre-treatment PD-L1 expression level on tumor cells of < 1% (TC0) had a mOS of 12.1 months compared to a mOS of 6.1 months for patients with PD-L1 expression >=1% (TC1/2/3) (HR = 0.42 p=0.007). There was no difference in mOS based on PD-L1 expression levels observed in the study's docetaxel plus placebo (D+P) control arm (10.7 months for TC0 vs. 11.1 months for TC1/2/3; HR = 0.87; p=0.609).
"We believe that these latest observations from the SUNRISE trial further support the hypothesis that bavituximab, through its immune modulating mechanism, may have more effect on tumors without pre-existent immunity. These ‘cold' tumors suppress normal anti-tumor immune response and are categorized by very low to no PD-L1 expression on tumor cells," said Joseph Shan, VP of Clinical & Regulatory Affairs at Peregrine. "These latest findings, along with other recently announced clinical and preclinical data from our PS-targeting program, inform our clinical development strategy going forward and provide additional rationale for combining bavituximab with checkpoint inhibitors."
As part of the SUNRISE clinical study protocol, researchers requested but did not require that patients provide a tumor tissue sample at the time of diagnosis. In total, tissue samples were collected from 129 of the trial's 597 patients and were assessed retrospectively for baseline PD-L1 expression levels on tumor cells. Of the 129 tissue samples collected, 122 were evaluable for PD-L1 expression on tumor cells (54 in D+B arm and 68 in D+P control arm). Of the evaluable samples in the D+B arm, 69% demonstrated PD-L1 expression levels < 1%, as compared to 59% in the D+P arm.
Bavituximab is an investigational immune-modulatory monoclonal antibody that targets phosphatidylserine (PS). PS inhibits the ability of immune cells to recognize and fight tumors. Bavituximab is believed to reverse PS-mediated immunosuppression by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS-targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes grants awarded by the National Comprehensive Cancer Network (NCCN) to support 3 different clinical trials of bavituximab treatment combinations. These trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center (MSK) with the goal of evaluating combinations of PS targeting antibodies with checkpoint inhibitors and other immune stimulatory agents. Peregrine's intent behind this strategy is to focus its research and development spending to further validate bavituximab's combination potential as the company seeks to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com

Poster #11603 Image:
http://www.peregrineinc.com/images/stories/pdfs/asco2017kallinteris.pdf
= = = = = = = = = = = = = = = = = = = = = = = = = = = = =
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
Note: this is known Sunrise Biomarker #4 (PD-L1 Expr.)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ORIG. ABSTRACT (superceded by 6-5-17 PPHM PR)...
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab (Opdivo) on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela Dir.Res. (LEAD), Melba Marie Page, Jenny Kreahling ”Nilogen: Bringing the complex tumor microenvironment to life with 3D ex vivo assays”
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org - NOTE: Moffitt’s S.Antonia & S.Altiok are CMO & CSO of Nilogen http://www.nilogen.com/our-team
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. [NOTE: a/o 5-2017, Dr. Antonio is also CMO of Nilogen Oncosystems http://www.nilogen.com ]. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD (also CSO of Nilogen Oncosystems http://www.nilogen.com ) - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic & prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab [BMS’ Opdivo] may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.
Also See: http://tinyurl.com/ydhf6tfx

Sunrise Biomarkers (#1/B2GPI #2/COMPL,IL10 #3/IFN-y #4/PD-L1) – updated per ASCO’17 (PD-L1 added). Also added BioBS2012’s 6-6-17 summary...

CHRONOLOGICAL ORDER:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.”
David R. Spigel: LEAD AUTHOR: CSO/Dir. Lung Cancer Pgm/Sarah Cannon Res.
David E. Gerber: SENIOR AUTHOR: UTSW/Dallas (Sunrise PI)
CO-AUTHORS: R.Natale/CEDARS-SINAI, R.Sanborn/PROVCC, PPHM’s N.Kallinteris, J.Lai, M.Tang, J.Shan, and 7 Intl. Sunrise PI’s: Ger/2, Ukraine, Greece, Spain, Korea, Hungry.
RESULTS:
~200 of 600 pts (~30%) tested positive for B2GPI(200-240). Those 200 received Stat.Sig. Improvement in O/S 7.7=>13.2mos; ie, lived +70% longer.
MORE DETAILS: http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce”
PRESENTATION CANCELLED – IR said,”Data analysis not completed in time.”
David E. Gerber: LEAD AUTHOR: UTSW/Dallas (Sunrise PI)
Rachael Sanborn: SENIOR AUTHOR: Dir./Thoracic-Oncology, Providence CC/Portland
CO-AUTHORS: L.Horn/VANDY, G.Losonczy/BUDAPEST, R.Natale/CEDARS-SINAI, H.Roder, J.Roder/BIODESIX, PPHM’s N.Kallinteris, M.Tang, J.Shan.
RESULTS:
104 of 193 pts tested high for complement activation, and from that, a 2nd subgrp isolated that tested low IL-10: O/S 5.9=>12.5mos. Remaining 397pts will be presented at WCLC’16.
* ”The Complement System is an enzyme cascade that is a collection of blood & cell surface proteins to help the abilities of antibodies to clear pathogens from an organism.”
* ”IL-10 is an anti-inflammatory TH2 cytokine that has a critical role in limiting the immune response to pathogens to prevent host damage.”
MORE DETAILS: http://tinyurl.com/z8cq8vx
. . . .
12-12-16 CEO/S.King: “We are actively evaluating addl. potential biomarkers and we hope to identify a profile for patients who will receive therapeutic benefit from treatment with bavituximab.” VP/JOE SHAN: ”Numerous addl. biomarkers are currently being evaluated.” http://tinyurl.com/hhn4gga
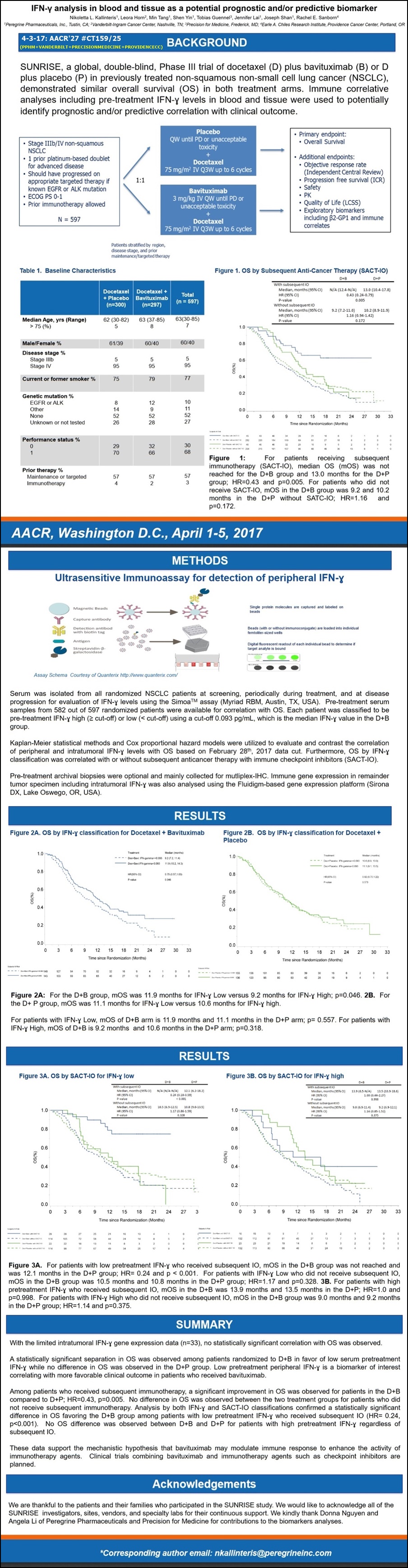
#3 4/3/17 AACR’17 CT159/25 (Session: Phase II/III Clinical Trials in Progress)
“IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
Lead Author: Nikoletta Kallinteris (PPHM); Senior Author: Rachael Sanborn (Dir./Thoracic-Oncology, Providence CC); CO-AUTHORS: L.Horn/VANDY, T.Guennel/PRECISION-FOR-MEDICINE, PPHM’s N.Kallinteris, M.Tang, S.Yin, J.Lai, J.Shan.
NOTE: “IFNy, or type II interferon gamma, is a cytokine that is critical for innate & adaptive immunity against viral, some bacterial & protozoal infections.”
4-4-17/PR: ( http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046 )
“SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy…”
Pts in the study's BAVI+DOCE arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs (4-4-17/PR):
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab & pembrolizumab in Head & Neck cancer through our ongoing collaboration with the NCCN.”
---------
**NOTE: Per JDM find, we know that 96 Sunrise Pts received “Subsequent Immunotherapy” - see 10-13-16 ASM CEO S.King Slide#14: http://tinyurl.com/n2bajew
-----------
Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn [Providence CC, Dir./Thoracic-Oncology http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN]
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
ORIG. AACR’17 ABSTRACT:
BACKGROUND: SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer (NSCLC), demonstrated similar overall survival (OS) in both treatment arms. Immune correlate analyses including pre-treatment IFN-y levels in blood and tumor tissue were used to potentially identify prognostic and/or predictive correlation with clinical outcome.
METHODS: Serum was isolated from all randomized NSCLC patients at screening, periodically during treatment and at disease progression for evaluation of IFN-y levels using the Simoa TM assay (Myriad RBM, Austin, TX). Available archival tissue was also tested for 91- immune gene activation markers, including IFN-y by the Fluidigm-based gene-expression platform (Sirona Dx, Lake Oswego, OR). Kaplan-Meier statistical methods and Cox proportion hazards models were utilized to evaluate and contrast the correlation of peripheral and intratumoral IFN-y levels with OS. Patients were classified paradoxically as IFN-y "low" with a favorable disease prognosis vs. "high" associated with more aggressive disease based on the median.
RESULTS: Pretreatment serum results were available for 582 out of the 597 randomized patients. Each patient was classified to be pre-treatment IFN-y high or low (< cut-off) using cut-off 0.093 pg/ml, which is the median IFN-y value in the D+B group. Median overall survival (mOS) in all patients with IFN-y low is 11.3mos. (95% CI, 10.1-13.5) vs. 10.4mos. (95% CI, 8.4-11.3) in all IFN-y high; p=0.047. mOS of D+B arm is 11.6mos. (95% CI, 10.2-13.9) and 11.1mos. (95% CI, 9.1-14.7) in the D group; p=0.982 for IFN-y low. mOS of D+B arm is 9.0mos. (95% CI, 6.7-11.2) and 10.6mos. (95% CI, 8.9-13.0) in the D group; p=0.252 for IFN-y high. With the limited intratumoral IFN-y gene expression data (n=33), no statistically significant correlation with OS was observed.
CONCLUSIONS: Correlative approaches identified low peripheral low IFN-y at pretreatment as a biomarker of interest correlating with more favorable clinical outcomes and is consistent with the hypothesis that bavituximab may demonstrate more immunomodulatory effects in patients with “immune cold” tumors.
POSTER #159/25 IMAGE:
http://www.peregrineinc.com/images/stories/pdfs/aacr2017kallinteris.pdf
#4 6/3/17 ASCO’17 #11603 (Session: Tumor Biology)
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study”
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
- - - - - - - - - - - - - -
6-5-17/PR Excerpts – see http://tinyurl.com/yd2c9tty
“Peregrine Presents Prelim. Correlative Analysis of PD-L1 Expression from SUNRISE Trial at ASCO 2017”
**Negative PD-L1 Expression was Associated with a Significantly Longer mOS Compared to Positive PD-L1 Expression in Patients Receiving Docetaxel+Bavituximab
**Presented Results Support Hypothesis that Bavituximab May Demonstrate Greater Effect in "Cold" Tumors Expressing Low to No PD-L1
“...today announced the presentation of promising new data from its Phase III SUNRISE trial of bavituximab in patients with previously treated locally adv./metastatic NSCLC. ...patients in the bavituximab arm who had low baseline PD-L1 expression levels had a statistically significant improvement in mOS as compared to patients who had higher baseline levels of PD-L1… patients in the D+B arm with a pre-treatment PD-L1 expr. level of < 1% (TC0) had a mOS of 12.1 mos. compared 6.1 mos. for patients with PD-L1 expr. >=1% (TC1/2/3) (HR=.42 p=0.007). There was no difference in mOS based on PD-L1 expr. levels observed in the D+P ctl. arm (10.7 mos. for TC0 vs. 11.1 mos. for TC1/2/3; HR=.87; p=0.609).
"We believe that these latest observations from the SUNRISE trial further support the hypothesis that bavituximab, through its immune modulating mechanism, may have more effect on tumors without pre-existent immunity. These ‘cold' tumors suppress normal anti-tumor immune response and are categorized by very low to no PD-L1 expression on tumor cells," said Joseph Shan, VP/Clin+Reg Affairs. "These latest findings, along with other recently announced clinical & preclin. data from our PS-targeting program, inform our clinical development strategy going forward and provide additional rationale for combining bavituximab with checkpoint inhibitors."
As part of the SUNRISE clinical study protocol, researchers requested but did not require that patients provide a tumor tissue sample at the time of diagnosis. In total, tissue samples were collected from 129 of the trial's 597 patients and were assessed retrospectively for baseline PD-L1 expression levels on tumor cells. Of the 129 tissue samples collected, 122 were evaluable for PD-L1 expression on tumor cells (54 in D+B arm and 68 in D+P ctl. arm). Of the evaluable samples in the D+B arm, 69% demonstrated PD-L1 expression levels < 1%, as compared to 59% in the D+P arm.

Poster Image:
http://www.peregrineinc.com/images/stories/pdfs/asco2017kallinteris.pdf

- - - - - - - - - - - - - -
ORIG. ABSTRACT (superceded by 6-5-17 PPHM PR)...
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

.
.
BIOBS2012’S 6-6-17 SUMMARY OF THE 4 KNOWN SUNRISE BIOMARKERS:
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131942267
SUNRISE Topline Results (at 70% cut-off ; presented at ESMO October, 2016)
BAVI Arm = 10.7
Placebo Arm = 10.8
Biomarker #1 (B2GP1)
B2GP1 Levels between 200 and 240 ug/ml (~30% of randomized patients)
BAVI Arm = 13.2
Placebo Arm = 7.7
---
B2GP1 Levels > or = 240 ug/ml (~50% of randomized patients)
BAVI Arm = 11.9
Placebo Arm = 10.1
---
Biomarker #2 (197 patients)
Subgroup with high Complement Activation (subgroup IL-10 activation)
BAVI Arm = 12.5 (n=50)
Placebo Arm = 5.9 (n=54)
---
Remaining subgroup
BAVI Arm = 5.6
Placebo Arm = 10.4
Data from remaining 397 patients remains unknown at this time.
Biomarker #3 (IFN-y)
Low pre-treatment IFN-gamma levels
BAVI Arm = 11.9
Placebo Arm = 11.1
---
High pre-treatment IFN-gamma levels
BAVI Arm = 9.2
Placebo Arm = 10.6
---
Subsequent I/O therapy (low pre-treatment IFN-gamma levels)
BAVI Arm = NOT REACHED
Placebo Arm = 12.1
---
Subsequent I/O therapy (high pre-treatment IFN-gamma levels)
BAVI Arm = 13.9
Placebo Arm = 13.5
---
NO Subsequent I/O therapy (low pre-treatment IFN-gamma levels)
BAVI Arm = 10.5
Placebo Arm = 10.8
---
NO Subsequent I/O therapy (high pre-treatment IFN-gamma levels)
BAVI Arm = 9.0
Placebo Arm = 9.2
Biomarker #4 (PD-L1 Expr.)
PD-L1 <1%
BAVI Arm = 12.1
Placebo Arm = 10.7
---
PD-L1 >1%
BAVI Arm = 6.1
Placebo Arm = 11.1
NOTES:
= = = = = = = =BIOMARKER #3 (IFN-y) AND PPHM/NCCN JOHNS-HOPKINS TRIAL TIE-IN (???):
Dr. Ranee Mehra’s (Johns-Hopkins/SidneyKimmelCC) work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 that was presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
-------
1-6-17 Dr. Ranee Mehra (Johns-Hopkins/SidneyKimmelCC), P.I. for the upcoming NCCN Ph2 Bavi+Keytruda Head&Neck trial. Excerpts from her 1-6-17 talk at GBMC/Greater Balt. MC “H+N Grand Rounds”… I do believe she views this trial is an important part of Johns-Hopkins future anti-cancer direction. Also, look at her Slide #36: “Interferon-y Signature”. Is that an exact tie-in to the newly-revealed AACR’17(4-3-17) Sunrise Biomarker #3 Abstract, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (embargoed)? I still totally believe that PPHM has been working with the 3 NCCN Bavi Awardees (Moffitt, MassGEN, JohnsHopkins) to weave in the “chosen” Sunrise Biomarker that is associated with generating “improved outcomes for bavituximab-containing treatments” in future trials.
PDF Link(1-6-17): http://www.gbmc.org/workfiles/HeadNeck/Grand%20Rounds/IO_Therapy_SCCHN02017.pdf
-------
Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176


PPHM’s NCCN#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD
”Phase II Study of Pembrolizumab[Keytruda] & Bavituximab for Progressive Recurrent/Metastatic Squamous Cell Carcinoma of the Head & Neck" http://tinyurl.com/gutgwb5
Ranee Mehra, MD: Dir., Head & Neck Oncology Therapeutics, Johns Hopkins Medicine https://www.linkedin.com/in/ranee-mehra-34a0467
RANEE MEHRA Disclosures(ASCO’16): GSK, Bayer, BMS, Genentech, Novartis, Mirati Ther.


NCCN Bavituximab Trials Announced 9-6-16 - To Begin "Early 2017" http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Sorafenib), MOFFITT CANCER CENTER - PI: Jessica Frakes, MD - https://clinicaltrials.gov/ct2/show/NCT02989870
...#2: P1-2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. CANCER CENTER - PI: Elizabeth Gerstner, MD
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD

9-9-16/CC/JoeShan: “The 3rd award is for a Phase II study of pembrolizumab [Merck’s Keytruda, anti-PD-1] & bavituximab in Head & Neck Cancer. We are particularly excited about this project, as it will be the 1st clinical trial of bavituximab with a checkpoint inhibitor. In multiple previous preclin. studies, we have observed bavituximab's potential to work synergistically with PD1 inhibitors such as pembrolizumab (Merck’s Keytruda).” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC: “Our collaboration with the NCCN has been an important part of our strategy for advancing the bavituximab clinical program in a cost effective way. We earlier provided NCCN with a $2mm grant to support bavituximab related clinical research with no further financial obligations, and these grant awards represent the outcome of a competitive selection process for the best proposals. These studies will evaluate novel bavituximab combinations in Glioblastoma, Head & Neck Cancer, and Hepatocellular Carcinoma, including an immunotherapy combination [Bavi + Merck’s Keytruda], which is a major focus for advancing the program.” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC/Q&A: “I’m very excited about the combinations that were chosen because the Radiation combination is one that in preclinical studies, as was mentioned during the prepared remarks, has always shown a lot of promise. It’s great to be able to now see that put into a clinical setting in a couple of different clinical trials. And the I-O combinations, as Jeff mentioned during his prepared remarks, is a major focus of ours. So to see a Pembro [Merck’s Keytruda] combination picked as well, we are just really excited that these were the 3 winners out of the NCCN selection process.” http://tinyurl.com/ktrfswj
= = = = = = = = = =DETAILS ON SUNRISE BIOMARKERS #1(B2GPI) & #2(COMPLEMENT/IL-10):
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
10-10-16/PR:
“Peregrine Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at Eur. Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Addl. Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Addl. patient sample testing & analysis is ongoing and may result in other biomarkers of importance.
--------------
** Data presented at ESMO’16 demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approx. 30% of randomized patients) achieved a statistically significant, 5.5-mo. improvement (13.2 mos. vs. 7.7 mos.) in MOS as compared to patients in the ctl. group with the same range of B2GP1 levels [p = 0.049; HR=.67].
-------------
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP/Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific & medical conferences as the more results become available.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
= = = = = = = = = = = = = = = =
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefitting from Bavi+Doce” <=CANCELLED/”Anal.Not.Done(IR)”
...Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
ABSTRACT CONCLUSION:
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...” [Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9mos => 12.5mos.]
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
http://wclc2016.iaslc.org Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
”A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
ABSTRACT Book PDF: http://wclc2016.iaslc.org/wp-content/uploads/2016/12/WCLC2016-Abstract-Book_vF-WEB_revDec12.pdf
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with High Complement Activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [MOS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded indep. validation.
- - - - - - - - -
12-20-16/S.Diaz(per Cheynew post #282068): “There was no poster in Vienna. The team was working on a very tight timeframe and, despite their best efforts, couldn’t complete the data analysis in time. Given the crunch, we knew there was a possibility that we wouldn’t meet the timing, so we never issued a PR announcing that we would present. Hence no PR announcing that we did not present.” S.DIAZ/FOLLOWUP: ”Unfortunately, I don’t have a timeline for completion of the analysis or where/when it might be presented. We’ll certainly announce this as soon as we know for sure. Only Joe Shan attended the conference for Peregrine.”
= = = = = = = = = =NOTE:
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ] “Founded in 2005, Biodesix discovers & commercializes cancer tests (diagnostics) that help patients & their doctors make more informed decisions about treatment based on a patient’s unique molecular profile.”
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
- - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
Reposting Biomarkers Summary w/Poster-Image & BioBS2012's Summary
The ASCO'17 Biomarker#4 PD-L1 Poster now on PPHM.com
http://www.peregrineinc.com/images/stories/pdfs/asco2017kallinteris.pdf
I'm going to put this into my Biomarkers summary post and re-issue later.
JeffH’s 7-14-16, “We-Plan-to-Init Partnering-Discussions in 2017” somehow has gotten morphed on iHub into “Our Deadline to Complete an Exosomes Deal is 6-30-17”.
7/14/16 CC, Jeff Hutchins:
“Once we have successfully validated this assay, we plan to establish proof-of-concept through an efficient preclinical and clinical testing program. We have no intention of conducting further development work beyond the proof-of-concept stage, rather we expect to initiate partnering discussions for commercialization of this program in 2017. We're very excited to begin this work on this new program and we will have more details to offer in the coming months.” http://tinyurl.com/h8eqtg5
CP/iHub 6-3-17:
5) I think it was Dr. Jeff Hutchins (ex PPHM), but it could have been another PPHM person, that said the program would be ready to START partnership talks in begin 2017 (let's make that H1/2017).
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131879091
And so, “Ready to Start Discussions in 2017” has now become “Our Dealine to close a Deal is 6-30-17” here on iHub.
I can’t find any “June30 Exosome-Partner Deadline”, as stated by SK or anybody else at Peregrine. I admit, I may have missed it. I’m not sure who 1st posted it here, it’s been awhile, and then it got repeated a few times, and now seems to have caught on as fact. Appreciate somebody enlightening me, and I’ll update the following post...
2-9-17/PR: PS+ Exosomes Proof-of-Concept Data Published
...See below for known history of PPHM’s Exosome’s pgm – chronological order.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=128560007
PPHM+PerkinElmer ASCO Poster Today: “Prelim-Correlative-Analysis of PD-L1 Expression from the Sunrise Study (Sunrise Biomarker #4)”, 6-3-17 1:15-4:45pm, Chicago.
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
DETAILS – see: http://tinyurl.com/kkpqvra
=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.

Nasdaq appeal decision could come as-late-as 7-2-17
I did not know this: “The Hearing Panel typically issues a decision within 30 days of the hearing.” If anybody sees any errors I’ve made, let me know and I’ll fix & re-post.
A. DETERMINATION LETTER (company does not meet the stds. for continued listing).
...Peregrine recv’d this 4-11-17, per subsequent Form 8-K: https://www.sec.gov/Archives/edgar/data/704562/000168316817000902/peregrine_8k.htm
B. NASDAQ APPEAL REQUEST: The company then has 7 days to appeal the determination. The company is typically scheduled for a hearing 30-45 days from the date of its request for a hearing, and informed of this date & time within a week of its request.
* The Latest date possible for the Hearing would be 4-18-17 + 45days = 6-2-17 (Friday).
C. HEARING: A company is allotted approx. 50 mins. to make its presentation. The hearing takes place in a conference room before 2 panelists (2 professionals from the business community, who are indep. of Nasdaq – the pool of panelists includes academics, accountants, auditors, corporate officers, investor advocacy representatives, investment bankers, securities lawyers, and venture capitalists), a Hearings Advisor, and a court reporter. Nasdaq does not typically attend, but may; if Nasdaq decides to attend, the company will be informed. A company may make its presentation in any form it wishes and should expect questions from the Panel. The Panel will want to understand the reasons for the company's deficiency and the specific details of its plan of compliance, including a timeline for regaining compliance... Nasdaq does not make a Panel's decision letter publicly available. However, Nasdaq cannot guarantee confidentiality since documents & information provided to Nasdaq may be subject to subpoenas from private parties or requests for access from federal or state gov’t agencies & self-regulatory bodies.
D. DECISION. The Hearing Panel typically issues a decision within 30 days of the hearing.
* For Peregrine, decision could be as late as (6-2-17 + 30days) = 7-2-17 (Sunday).
E. FURTHER APPEALS. May a company appeal a Hearing Panel's decision? Yes. A company may appeal a Panel's decision to the Listing Council. The Listing Council may also decide to call the decision for review. The appeal to the Listing Council does not stay a Panel's decision or the suspension of the trading of a company's shares pending a final determination by the Listing Council. If the Listing Council overturns a Panel decision, then the company's securities may be reinstated on Nasdaq. After a determination by the Listing Council, a company may appeal to the SEC and, from there, it may proceed to the federal court system.
SOURCE: https://listingcenter.nasdaq.com/Material_Search.aspx?cid=14&mcd=LQ
= = = = = = = = = = = = = = = = = = = = = = = = =
Peregrine’s 4-11-17 Form 8-K, “Notice of Delisting: Received:
“On April 11, 2017, Peregrine received a letter from NASDAQ stating that the Company has not regained compliance with the $1.00 min. closing bid price requirement… The letter further states that the Nasdaq has determined to delist the Company’s securities on April 20, 2017, unless the Company requests a hearing before the NASDAQ Hearings Panel by April 18, 2017 to appeal the Staff’s determination to delist its securities. The Company intends to timely request such a hearing before the Panel, and such request will stay any delisting action by the Staff pending the issuance of a written Panel decision. The Panel will typically hold a hearing to consider an appeal within 45 days after the request for a hearing is made. At the hearing, the Company will present its plan to regain compliance with the requirements for continued listing on The NASDAQ Capital Market. The Company is considering several paths to regain compliance with the Minimum Bid Price Requirement, including, among other things, a reverse stock split. As previously disclosed, the Company’s stockholders approved a reverse stock split of the Company’s common stock at a ratio of up to 1-for-7, to be determined by the Company’s Board of Directors in its sole discretion and effected, if at all, at any time until the Company’s 2017 annual meeting of stockholders [~10-12-18].”
https://www.sec.gov/Archives/edgar/data/704562/000168316817000902/peregrine_8k.htm
= = = = = = = = = =
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
PL: “Switching gears now, I’d like to address our plans to preserve our NASDAQ listing. At a backdrop, last April, we were notified by the NASDAQ that our share price did not meet the $1 min. bid price for 30 consecutive days, which is a requirement for continued listing on the NASDAQ market. At that time, we were automatically afforded an initial compliance period of 180 days or until Oct. 10, 2016 to regain compliance. At the end of the initial compliance period, we requested and were granted a 180 day extension to regain compliance. This 2nd extension period will end on April 10, 2017. Let me discuss our strategy and future steps we plan to take over the next few months. First & foremost, we have determined that it is not in the best interest of our stockholders to affect a reverse stock split prior to April 10, 2017. If our share price does not trade above $1 for 10 consecutive trading days by April 10th, we expect to receive a notice of delisting. If we received this notice, we will appeal this decision within the required 7 days and this appeal will stay any delisting actions by NASDAQ. At that point, NASDAQ will schedule a hearing, which is typically held within 45 days, and we will present our plan to regain compliance. We are considering several paths to regain compliance, including having addl. time to continue to allow the market to adjust, to reflect the current value of Avid as evidence with the recent movement in our share price. The hearing panel could afford us up to 180 addl. days to regain compliance, although this addl. time is not guaranteed and this decision is at the discretion of the NASDAQ’s hearing panel. If we are not afforded addl. time from this panel, it is important to note that our stockholders approved the Board’s ability to implement a reverse stock split at a ratio of up to 1:7 as a means to regain compliance and to preserve our listing on NASDAQ. During this entire process, Peregrine will continue to be listed on that NASDAQ capital market as it does today. We will keep you informed on any material developments through our SEC filings on Form 8-K as we execute on this plan.”
*ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
= = = = = = = = = = = = = = = = = = =
Known Upcoming Events (incl. ASCO’17: Analysis/Sunrise PD-L1 Expression)
Jun2-6: ASCO 2017, Chicago https://am.asco.org See: http://tinyurl.com/kkpqvra
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
#1: 6-3-17 1:15-4:45pm, #11603, “Prelim. Correlative Analysis of PD-L1 Expression from the Sunrise Study” (Sunrise Biomarker #4)
...11 Authors: LEAD: Nikoletta Lea Kallinteris (PPHM); SENIOR: Cliff Hoyt (PerkinElmer, "Technology Strategist", see http://tinyurl.com/kk5ald6 )
#2: #e23091 (pub.only): “Effect Of Bavituximab In Combination With Nivolumab (Opdivo) on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
...Authors: LEAD is from Nilogen Oncosystems, SENIOR from Moffitt w/Scott Antonio and PPHM co-authors.
**See ASCO 2017 DETAILS BELOW**
Jun18-20: ICDS’17, Annual ICDS Conf. on Cell Death in Development & Disease, Israel http://www.weizmann.ac.il/conferences/CDDD2017
...Jun20 11:30-12:00pm: Raymond Birge (Rutgers), “Regulation of Efforocytosis by Phosphatidyserine Receptor”
Jul2: Latest date Nasdaq gives decision on PPHM’s Delisting Appeal http://tinyurl.com/yagoqth5
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Aug21-25/Avid Booth #403: CHI's 9th Annual BioProcessing Summit, Boston http://www.bioprocessingsummit.com
~Sep7: FY'18Q1 (qe 7-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Sep25-28/Avid Exhibiting: Informa’s BPI’17: BioProcess Intl. Conf. & Exhibition, Boston https://lifesciences.knect365.com/bioprocessinternational
~Oct12: Peregrine's Annual Shareholder’s Meeting (2016 attendee reports: http://tinyurl.com/jx7ouay )
Peregrine's Anti-PS Mabs (from iBox)...
• PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”
• PGN632 (B2GPI-indep.) is the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p
• PGN650 is a human F(ab’)2 fragment that targets PS expression (1st ref’d in AACR’12 #2452) – see http://tinyurl.com/76nqqkm . 124I-PGN650 is Peregrine’s PS-Imaging candidate (see http://tinyurl.com/7p7jovt )
• BETABODIES (Clipped/Nicked B2GPI - ex: KL15, “2nd-gen. PS-Targeting”) - bind to PS directly, are smaller in size (100 vs. 250KDa) and have a longer serum half-life (~5days) than natural antibodies (Bavi=~1day) – see http://tinyurl.com/khopa3d
…2/17/15: UTSW/PPHM’s BetaBodies patent#8,956,616 Awarded(Granted) http://tinyurl.com/p75uyfu
With PerkinElmer emerging in ASCO’2017 as a PPHM-Collaborator for The Biomarkers Project, a smart friend jiggled my mind with this possibility: PerkinElmer, Our Exosomes Partner?? I think it makes perfect sense. PKI's ($6.8B MktCap) big area in Health is Diagnostics...
PerkinElmer Introduction… "Diagnose & Treat Disease Earlier"
2013 Vid(1:28):


5-19-17/13D: Group Ronin & SW-Partners add 23,140 PREFERRED/May1-17
5-19-17/13D: Ronin(John Stafford III) & SWPartners(Stephen White), a 13D “Group”, adds 23,140 PREFERRED during May1-May17.
That brings total ownership of the Group to:
23,666,541 COMMON, 7.9% of 297,709,478 common O/S at 3-10-17.
123,499 PREFERRED, 7.5% of 1,647,760 preferred O/S at 1-31-17.
13D TransDates COMMON-CHG PREF-CHG ENDING-COMMON ENDING-PREF.
3-2-17 1/20/17-3/1/17 +20,631,977 +51,364 20,631,977 51,364 http://tinyurl.com/jr42u23
3-10-17 3/2/17-3/9/17 +3,034,564 +25,661 23,666,541 77,025 http://tinyurl.com/huundwu
4-17-17 3/28/17-4/10/17 0 +23,334 23,666,541 100,359 http://tinyurl.com/lanjddc
5-19-17 5/1/17-5/17/17 0 +23,140 23,666,541 123,499 http://tinyurl.com/mgnn92x
Sunrise Biomarkers (#1/B2GPI #2/COMPL,IL10 #3/IFN-y #4/PD-L1) – updated per ASCO’17 Abstracts (PD-L1 added)...

CHRONOLOGICAL ORDER:
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.”
David R. Spigel: LEAD AUTHOR: CSO/Dir. Lung Cancer Pgm/Sarah Cannon Res.
David E. Gerber: SENIOR AUTHOR: UTSW/Dallas (Sunrise PI)
CO-AUTHORS: R.Natale/CEDARS-SINAI, R.Sanborn/PROVCC, PPHM’s N.Kallinteris, J.Lai, M.Tang, J.Shan, and 7 Intl. Sunrise PI’s: Ger/2, Ukraine, Greece, Spain, Korea, Hungry.
RESULTS:
~200 of 600 pts (~30%) tested positive for B2GPI(200-240). Those 200 received Stat.Sig. Improvement in O/S 7.7=>13.2mos; ie, lived +70% longer.
MORE DETAILS: http://tinyurl.com/hp73njt
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefiting from Bavi+Doce”
PRESENTATION CANCELLED – IR said,”Data analysis not completed in time.”
David E. Gerber: LEAD AUTHOR: UTSW/Dallas (Sunrise PI)
Rachael Sanborn: SENIOR AUTHOR: Dir./Thoracic-Oncology, Providence CC/Portland
CO-AUTHORS: L.Horn/VANDY, G.Losonczy/BUDAPEST, R.Natale/CEDARS-SINAI, H.Roder, J.Roder/BIODESIX, PPHM’s N.Kallinteris, M.Tang, J.Shan.
RESULTS:
104 of 193 pts tested high for complement activation, and from that, a 2nd subgrp isolated that tested low IL-10: O/S 5.9=>12.5mos. Remaining 397pts will be presented at WCLC’16.
* ”The Complement System is an enzyme cascade that is a collection of blood & cell surface proteins to help the abilities of antibodies to clear pathogens from an organism.”
* ”IL-10 is an anti-inflammatory TH2 cytokine that has a critical role in limiting the immune response to pathogens to prevent host damage.”
MORE DETAILS: http://tinyurl.com/z8cq8vx
. . . .
12-12-16 CEO/S.King: “We are actively evaluating addl. potential biomarkers and we hope to identify a profile for patients who will receive therapeutic benefit from treatment with bavituximab.” VP/JOE SHAN: ”Numerous addl. biomarkers are currently being evaluated.” http://tinyurl.com/hhn4gga
#3 4/3/17 AACR’17 CT159/25 (Session: Phase II/III Clinical Trials in Progress)
“IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
Lead Author: Nikoletta Kallinteris (PPHM); Senior Author: Rachael Sanborn (Dir./Thoracic-Oncology, Providence CC); CO-AUTHORS: L.Horn/VANDY, T.Guennel/PRECISION-FOR-MEDICINE, PPHM’s N.Kallinteris, M.Tang, S.Yin, J.Lai, J.Shan.
NOTE: “IFNy, or type II interferon gamma, is a cytokine that is critical for innate & adaptive immunity against viral, some bacterial & protozoal infections.”
4-4-17/PR: ( http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=1020046 )
“SUNRISE Data Analysis Demonstrates Stat. Significant Overall Survival (OS) Improvement in Patients Receiving Bavituximab+Docetaxel and Subsequent Immunotherapy Compared to Placebo+Docetaxel and Subsequent Immunotherapy…”
Pts in the study's BAVI+DOCE arm who received subsequent immunotherapy, the mOS was not reached, while mOS was 13.0mos. for patients in the study's DOCE+PLACEBO arm who received subsequent immunotherapy [HR=0.43; p=.005]. These are the first clinical results reported supporting the hypothesis that bavituximab may modulate the tumor microenvironment to enhance the anti-tumor activity of immunotherapy agents... The presentation highlighted an analysis in which the company evaluated the impact of subsequent immunotherapy treatment, as well as patients' pre-treatment interferon gamma (IFN-y) levels on overall survival. Overall, low peripheral IFN-y correlated with more favorable OS in the patients receiving BAVI+DOCE and is a biomarker of interest. Data were also analyzed by low vs. high IFN-y levels. For patients with low pre-treatment IFN-y levels who received subsequent immunotherapy, those in the BAVI+DOCE arm did not reach mOS compared to mOS of 12.1mos. for the DOCE+PLACEBO arm [HR=0.24; p<.001].
Joseph Shan, Peregrine’s VP of Clinical & Regulatory Affairs (4-4-17/PR):
"We are extremely encouraged by the results of these exploratory analyses which provide further clinical rationale for combining bavituximab and checkpoint inhibitors. This will be the key focus for upcoming early phase clinical trials, which includes a study of bavituximab & pembrolizumab in Head & Neck cancer through our ongoing collaboration with the NCCN.”
---------
**NOTE: Per JDM find, we know that 96 Sunrise Pts received “Subsequent Immunotherapy” - see 10-13-16 ASM CEO S.King Slide#14: http://tinyurl.com/n2bajew
-----------
Nikoletta Kallinteris 1, Leora Horn 2, Min Tang 1, Tobias Guennel 3, Shen Yin 1, Jennifer Lai 1, Joseph Shan 1, Rachel E. Sanborn [Providence CC, Dir./Thoracic-Oncology http://cancergrace.org/faculty/rachel-sanborn-md - Dr. Sanborn's Conflicts of interest: DNA, AZN]
1=Peregrine Pharmaceuticals
2=Vanderbilt-Ingram Cancer Center, Nashville, TN
3=Precision for Medicine, Frederick, MD
4=Providence Cancer Center, Portland, OR
ORIG. AACR’17 ABSTRACT:
BACKGROUND: SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer (NSCLC), demonstrated similar overall survival (OS) in both treatment arms. Immune correlate analyses including pre-treatment IFN-y levels in blood and tumor tissue were used to potentially identify prognostic and/or predictive correlation with clinical outcome.
METHODS: Serum was isolated from all randomized NSCLC patients at screening, periodically during treatment and at disease progression for evaluation of IFN-y levels using the Simoa TM assay (Myriad RBM, Austin, TX). Available archival tissue was also tested for 91- immune gene activation markers, including IFN-y by the Fluidigm-based gene-expression platform (Sirona Dx, Lake Oswego, OR). Kaplan-Meier statistical methods and Cox proportion hazards models were utilized to evaluate and contrast the correlation of peripheral and intratumoral IFN-y levels with OS. Patients were classified paradoxically as IFN-y "low" with a favorable disease prognosis vs. "high" associated with more aggressive disease based on the median.
RESULTS: Pretreatment serum results were available for 582 out of the 597 randomized patients. Each patient was classified to be pre-treatment IFN-y high or low (< cut-off) using cut-off 0.093 pg/ml, which is the median IFN-y value in the D+B group. Median overall survival (mOS) in all patients with IFN-y low is 11.3mos. (95% CI, 10.1-13.5) vs. 10.4mos. (95% CI, 8.4-11.3) in all IFN-y high; p=0.047. mOS of D+B arm is 11.6mos. (95% CI, 10.2-13.9) and 11.1mos. (95% CI, 9.1-14.7) in the D group; p=0.982 for IFN-y low. mOS of D+B arm is 9.0mos. (95% CI, 6.7-11.2) and 10.6mos. (95% CI, 8.9-13.0) in the D group; p=0.252 for IFN-y high. With the limited intratumoral IFN-y gene expression data (n=33), no statistically significant correlation with OS was observed.
CONCLUSIONS: Correlative approaches identified low peripheral low IFN-y at pretreatment as a biomarker of interest correlating with more favorable clinical outcomes and is consistent with the hypothesis that bavituximab may demonstrate more immunomodulatory effects in patients with “immune cold” tumors.
POSTER #159/25 IMAGE:
http://www.peregrineinc.com/images/stories/pdfs/aacr2017kallinteris.pdf

#4 6/3/17 ASCO’17 #11603 (Session: Tumor Biology)
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study”
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
…Note (Nov2016):
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
Interesting: 5-1-15/1hr. Webinar Video: Clifford Hoyt (PerkinElmer) & Bernard Fox (Providence CC, Portland) - “Cancer immunology: Charting The Course Forward For Immunoprofiling” (Dr. Hoyt speaks at 31:30)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ABSTRACT - BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

.
.
NOTES:
= = = = = = = =BIOMARKER #3 (IFN-y) AND PPHM/NCCN JOHNS-HOPKINS TRIAL TIE-IN (???):
Dr. Ranee Mehra’s (Johns-Hopkins/SidneyKimmelCC) work with Biomarker IFN-y seems to dovetail into PPHM’s newly revealed Sunrise Biomarker #3 that was presented 4-3-17 at AACR’17: #CT159/25, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker”
-------
1-6-17 Dr. Ranee Mehra (Johns-Hopkins/SidneyKimmelCC), P.I. for the upcoming NCCN Ph2 Bavi+Keytruda Head&Neck trial. Excerpts from her 1-6-17 talk at GBMC/Greater Balt. MC “H+N Grand Rounds”… I do believe she views this trial is an important part of Johns-Hopkins future anti-cancer direction. Also, look at her Slide #36: “Interferon-y Signature”. Is that an exact tie-in to the newly-revealed AACR’17(4-3-17) Sunrise Biomarker #3 Abstract, “IFN-y Analysis In Blood & Tissue as a Potential Prognostic and/or Predictive Biomarker” (embargoed)? I still totally believe that PPHM has been working with the 3 NCCN Bavi Awardees (Moffitt, MassGEN, JohnsHopkins) to weave in the “chosen” Sunrise Biomarker that is associated with generating “improved outcomes for bavituximab-containing treatments” in future trials.
PDF Link(1-6-17): http://www.gbmc.org/workfiles/HeadNeck/Grand%20Rounds/IO_Therapy_SCCHN02017.pdf
-------
Note: DR. RANEE MEHRA was co-author of ASCO’16, “Biomarkers & Response to Pembro(Keytruda) in Recurrent/Metastatic Head & Neck Cancer” - Conclusion: “The IFN-y signature score was significantly associated with ORR, PFS, and OS (all, P< .001)… PD-L2 & IFN-y signature may be associated with clinical response to Pembro[Keytruda] and may offer addl. strategies to improve prediction of response.” http://meetinglibrary.asco.org/content/165708-176


PPHM’s NCCN#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD
”Phase II Study of Pembrolizumab[Keytruda] & Bavituximab for Progressive Recurrent/Metastatic Squamous Cell Carcinoma of the Head & Neck" http://tinyurl.com/gutgwb5
Ranee Mehra, MD: Dir., Head & Neck Oncology Therapeutics, Johns Hopkins Medicine https://www.linkedin.com/in/ranee-mehra-34a0467
RANEE MEHRA Disclosures(ASCO’16): GSK, Bayer, BMS, Genentech, Novartis, Mirati Ther.


NCCN Bavituximab Trials Announced 9-6-16 - To Begin "Early 2017" http://tinyurl.com/gutgwb5
...#1: Ph1/HepC-Related Hepatocellular(Liver) (Bavi+RAD+Sorafenib), MOFFITT CANCER CENTER - PI: Jessica Frakes, MD - https://clinicaltrials.gov/ct2/show/NCT02989870
...#2: P1-2/Newly Diag. Glioblastoma (Bavi+RAD+Merck’s Temodar), MASS-GEN. CANCER CENTER - PI: Elizabeth Gerstner, MD
...#3: Ph2/Progressive Squamous Head+Neck (Bavi+Merck’s Keytruda), JOHNS-HOPKINS(Sidney Kimmel CC) - PI: Ranee Mehra, MD

9-9-16/CC/JoeShan: “The 3rd award is for a Phase II study of pembrolizumab [Merck’s Keytruda, anti-PD-1] & bavituximab in Head & Neck Cancer. We are particularly excited about this project, as it will be the 1st clinical trial of bavituximab with a checkpoint inhibitor. In multiple previous preclin. studies, we have observed bavituximab's potential to work synergistically with PD1 inhibitors such as pembrolizumab (Merck’s Keytruda).” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC: “Our collaboration with the NCCN has been an important part of our strategy for advancing the bavituximab clinical program in a cost effective way. We earlier provided NCCN with a $2mm grant to support bavituximab related clinical research with no further financial obligations, and these grant awards represent the outcome of a competitive selection process for the best proposals. These studies will evaluate novel bavituximab combinations in Glioblastoma, Head & Neck Cancer, and Hepatocellular Carcinoma, including an immunotherapy combination [Bavi + Merck’s Keytruda], which is a major focus for advancing the program.” http://tinyurl.com/ktrfswj
- - - - - - - - - -
Steve King 9-9-16/CC/Q&A: “I’m very excited about the combinations that were chosen because the Radiation combination is one that in preclinical studies, as was mentioned during the prepared remarks, has always shown a lot of promise. It’s great to be able to now see that put into a clinical setting in a couple of different clinical trials. And the I-O combinations, as Jeff mentioned during his prepared remarks, is a major focus of ours. So to see a Pembro [Merck’s Keytruda] combination picked as well, we are just really excited that these were the 3 winners out of the NCCN selection process.” http://tinyurl.com/ktrfswj
= = = = = = = = = =DETAILS ON SUNRISE BIOMARKERS #1(B2GPI) & #2(COMPLEMENT/IL-10):
#1 10-10-16/ESMO’16: “B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.” http://tinyurl.com/hp73njt
10-10-16/PR:
“Peregrine Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at Eur. Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Addl. Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Addl. patient sample testing & analysis is ongoing and may result in other biomarkers of importance.
--------------
** Data presented at ESMO’16 demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approx. 30% of randomized patients) achieved a statistically significant, 5.5-mo. improvement (13.2 mos. vs. 7.7 mos.) in MOS as compared to patients in the ctl. group with the same range of B2GP1 levels [p = 0.049; HR=.67].
-------------
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP/Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific & medical conferences as the more results become available.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
= = = = = = = = = = = = = = = =
#2 12-7-16/WCLC’16(IASLC): “Complement & IL-10 Pathways Id Pts Benefitting from Bavi+Doce” <=CANCELLED/”Anal.Not.Done(IR)”
...Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR. Interestingly, one co-author is Heinrich Roder, CTO of Biodesix, Boulder CO...
ABSTRACT CONCLUSION:
“...Proteomic & correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with Doce+Bavi, in line with preclinical work on Bavi’s MOA...” [Pts=193, within the 1st subgroup of N=104, a 2nd subgroup isolated: MOS 5.9mos => 12.5mos.]
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
http://wclc2016.iaslc.org Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, Heinrich Roder [CTO, Biodesix http://www.biodesix.com/project/heinrichroder ], Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
”A Pre-Treatment Serum Test Based on Complement & IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
ABSTRACT Book PDF: http://wclc2016.iaslc.org/wp-content/uploads/2016/12/WCLC2016-Abstract-Book_vF-WEB_revDec12.pdf
ABSTRACT: http://library.iaslc.org/virtual-library-search?product_id=6
BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) plus bavituximab (B) or D plus placebo (P) in previously treated non-squamous non-small cell lung cancer, demonstrated similar overall survival (OS) in both treatment arms. Mass spectrometry and correlative analysis were used to create a test able to identify a subgroup of patients benefitting from the addition of B to D.
METHODS:
Pre-treatment serum samples were available for 197 of the first 200 subjects enrolled in the trial. Mass spectra could be generated for 193 samples using the Deep MALDI method (Duncan et al, ASMS 2013), processed and features (peaks) identified. Mass spectral (MS) features associated with various biological functions were identified using a gene set enrichment analysis approach. Analysis of scores based on these MS feature, subsets indicated that in patients with High Complement Activation outcome depended on IL-10 activation in D+B but not in D+P. A test using the MS features associated with these functions was created to reliably identify a patient subgroup associated with clinical benefit using modern machine learning methods.
RESULTS:
Complement activation, as assessed by a classifier trained using related MS features, was a prognostic factor in both treatment arms, with high activation associated with poorer clinical outcome (OS HR = 0.54, log-rank p = 0.013 for D+B; OS HR = 0.60, log-rank p = 0.040 for D+P). Within the subgroup with high complement activation [N=50 (D+B); N=54 (D+P)], a second classifier using features related to IL-10 activation was able to isolate a subgroup of patients showing numerical benefit from the addition of B [Bavituximab] [median OS 5.9mos.(D+Placebo), 12.5mos.(D+Bavituximab)]. The remaining subgroup showed no benefit from addition of B [MOS 10.4mos.(D+P), 5.6mos.(D+B)]. Blinded validation of the test in the remainder 397 patients randomized in SUNRISE is will be presented.
CONCLUSION:
Proteomic and correlative approaches identified complement activation and low IL-10 levels as important pathways for predicting improved outcomes of patient treatment with D+B, in line with preclinical work on B’s mechanism of action. The test resulting from this work will undergo blinded indep. validation.
- - - - - - - - -
12-20-16/S.Diaz(per Cheynew post #282068): “There was no poster in Vienna. The team was working on a very tight timeframe and, despite their best efforts, couldn’t complete the data analysis in time. Given the crunch, we knew there was a possibility that we wouldn’t meet the timing, so we never issued a PR announcing that we would present. Hence no PR announcing that we did not present.” S.DIAZ/FOLLOWUP: ”Unfortunately, I don’t have a timeline for completion of the analysis or where/when it might be presented. We’ll certainly announce this as soon as we know for sure. Only Joe Shan attended the conference for Peregrine.”
= = = = = = = = = =NOTE:
Ann Oncol (11-8-16/suppl8): ESMO Symposium on Immuno-Oncology, Nov4-6 2016, Lausanne, Switzerland
#30P: “Proteomic Signature Analysis & Application in Clinical Development of the Novel Phosphatidylserine-Targeting Immunotherapy, Bavituximab”
http://annonc.oxfordjournals.org/content/27/suppl_8/mdw525.30
David E. Gerber [UTSW] 1, N.L. Kallinteris 2, L. Horn 3, G. Losonczy 4, R. Natale 5, Heinrich Roder 6 [CTO, Biodesix], M. Tang 7, J. Lai 2, J. Shan 8, Rachel E. Sanborn [9=Providence Portland Medical Ctr]
1 Oncology, UTSW-MC/Dallas
2 Clinical, Peregrine Pharmaceuticals Inc.
3 Oncology, Vanderbilt Ingram CC, Nashville, TN
4 Oncology, Semmelweis Univ., Budapest, Hungary
5 Oncology, Cedars-Sinai M/C, Los Angeles
6 Biodesix, Boulder, CO [CTO, Dr. Heinrich Roder: http://www.biodesix.com/project/heinrichroder ] “Founded in 2005, Biodesix discovers & commercializes cancer tests (diagnostics) that help patients & their doctors make more informed decisions about treatment based on a patient’s unique molecular profile.”
7 Biostatistics, Peregrine Pharmaceuticals Inc.
8 Clinical & Regulatory Affairs, Peregrine Pharmaceuticals, Inc.
9 Thoracic Oncology, Providence Cancer Care, Providence, OR
Aim/Background:
Understanding the multi-dimensional characteristics of cancer is essential to patient selection and treatment planning. Topline results from SUNRISE, a global double-blind Phase III trial of docetaxel + bavituximab (D+B) vs. docetaxel + placebo (D) in previously treated non-squamous NSCLC demonstrated mOS of 10.7mos. in the D+B group and 10.8mos. for the D group, which was unexpectedly different from the assumed 9.1mos. for D+B vs. 7.0mos. used for study powering. VeriStrat, a….[must subscribe]
- - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
Note: this is known Sunrise Biomarker #4 (PD-L1 Expr.)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
…Note (Nov2016):
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
Interesting: 5-1-15/1hr. Webinar Video: Clifford Hoyt (PerkinElmer) & Bernard Fox (Providence CC, Portland) - “Cancer immunology: Charting The Course Forward For Immunoprofiling” (Dr. Hoyt speaks at 31:30)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ABSTRACT - BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela (LEAD), Melba Marie Page, Jenny Kreahling http://www.nilogen.com ”Bringing the complex tumor microenvironment to life with 3D ex vivo assays”
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic and prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.

Known Upcoming Events (incl. ASCO’17: Analysis/Sunrise PD-L1 Expression)
May18-21: ICHC’17, 15th Intl. Congress of Histo/Cyto Chemistry - from Molecules to Diseases, Turkey http://tinyurl.com/kbetmbk
...May19/12pm: Raymond Birge (Rutgers), “Phosphatidylserine Sensing by TAM Receptors Regulates AKT-dependent Chemo-Resistance & PD-L1 Expression”
Jun2-6: ASCO 2017, Chicago https://am.asco.org See: http://tinyurl.com/kkpqvra
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
#1: 6-3-17 1:15-4:45pm, #11603, “Prelim. Correlative Analysis of PD-L1 Expression from the Sunrise Study” (Sunrise Biomarker #4)
...11 Authors: LEAD: Nikoletta Lea Kallinteris (PPHM); SENIOR: Cliff Hoyt (PerkinElmer, "Technology Strategist")
#2: #e23091 (pub.only): “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
...Authors: LEAD is from Nilogen Oncosystems, SENIOR from Moffitt w/Scott Antonio and PPHM co-authors.
**See ASCO 2017 DETAILS BELOW**
Jun18-20: ICDS’17, Annual ICDS Conf. on Cell Death in Development & Disease, Israel http://www.weizmann.ac.il/conferences/CDDD2017
...Jun20 11:30-12:00pm: Raymond Birge (Rutgers), “Regulation of Efforocytosis by Phosphatidyserine Receptor”
Jun19-22/Avid Booth #1411: BIO Intl. Convention, SanDiego http://www.convention.bio.org/2017
~Jul13: FY'17Q4 (fye 4-30-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Aug21-25/Avid Booth #403: CHI's 9th Annual BioProcessing Summit, Boston http://www.bioprocessingsummit.com
~Sep7: FY'18Q1 (qe 7-31-17) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Sep25-28/Avid Exhibiting: Informa’s BPI’17: BioProcess Intl. Conf. & Exhibition, Boston https://lifesciences.knect365.com/bioprocessinternational
~Oct12: Peregrine's Annual Shareholder’s Meeting (2016 attendee reports: http://tinyurl.com/jx7ouay )
.
.
======================ASCO’17 DETAILS:
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
#1: 6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is PerkinElmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
#2: Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17 5:00pmET)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
Note: this is known Sunrise Biomarker #4 (PD-L1 Expr.)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=PerkinElmer)
Amanda Clement (Scientist/PerkinElmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/PerkinElmer)
…Note (Nov2016):
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
Interesting: 5-1-15/1hr. Webinar Video: Clifford Hoyt (PerkinElmer) & Bernard Fox (Providence CC, Portland) - “Cancer immunology: Charting The Course Forward For Immunoprofiling” (Dr. Hoyt speaks at 31:30)
- - - - - - - - - -
**Peregrine: N.Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ABSTRACT - BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela (LEAD), Melba Marie Page, Jenny Kreahling http://www.nilogen.com ”Bringing the complex tumor microenvironment to life with 3D ex vivo assays”
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic and prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: 4 Known Sunrise Biomarkers thru 5-2017(ASCO): B2GPI, Complement+IL10, IFN-y, PD-L1 Expr.

Chopped off again – all that PL said...
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
PL: Switching gears now, I’d like to address our plans to preserve our NASDAQ listing. At a backdrop, last April, we were notified by the NASDAQ that our share price did not meet the $1 min. bid price for 30 consecutive days, which is a requirement for continued listing on the NASDAQ market. At that time, we were automatically afforded an initial compliance period of 180 days or until Oct. 10, 2016 to regain compliance. At the end of the initial compliance period, we requested and were granted a 180 day extension to regain compliance. This 2nd extension period will end on April 10, 2017. Let me discuss our strategy and future steps we plan to take over the next few months. First & foremost, we have determined that it is not in the best interest of our stockholders to affect a reverse stock split prior to April 10, 2017. If our share price does not trade above $1 for 10 consecutive trading days by April 10th, we expect to receive a notice of delisting. If we received this notice, we will appeal this decision within the required 7 days and this appeal will stay any delisting actions by NASDAQ. At that point, NASDAQ will schedule a hearing, which is typically held within 45 days, and we will present our plan to regain compliance. We are considering several paths to regain compliance, including having addl. time to continue to allow the market to adjust, to reflect the current value of Avid as evidence with the recent movement in our share price. The hearing panel could afford us up to 180 addl. days to regain compliance, although this addl. time is not guaranteed and this decision is at the discretion of the NASDAQ’s hearing panel. If we are not afforded addl. time from this panel, it is important to note that our stockholders approved the Board’s ability to implement a reverse stock split at a ratio of up to 1:7 as a means to regain compliance and to preserve our listing on NASDAQ. During this entire process, Peregrine will continue to be listed on that NASDAQ capital market as it does today. We will keep you informed on any material developments through our SEC filings on Form 8-K as we execute on this plan.
**I** typed in "(Senior Author)" next to PerkinElmer's Dr. Clifford Hoyt in the author list in my post (http://tinyurl.com/ldt2vp6 ) because I know from Authorship Order Protocol that he's the Senior Author (being the LAST listed author).
= = = = =
Jun2-6: ASCO 2017, Chicago https://am.asco.org See: http://tinyurl.com/ldt2vp6
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
#1: 6-3-17 1:15-4:45pm, #11603, “Prelim. Correlative Analysis of PD-L1 Expression from the Sunrise Study”
...11 Authors: LEAD: Nikoletta Lea Kallinteris (PPHM); SENIOR: Cliff Hoyt (PerkinElmer, "Technology Strategist")
SENIOR AUTHOR:
Cliff Hoyt (Oncology Fellow/PerkinElmer)
…Note (Nov2016):
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
= = = = = = = = = = = = = = = = = = = = =
#2: #e23091 (pub.only): “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
...Authors: LEAD is from Nilogen Oncosystems, SENIOR from Moffitt w/Scott Antonio and PPHM co-authors.
Author List Order: 1st=LEAD(gathered the data together, did the writing, presents the paper); Last=SENIOR(overall responsibility, generated the idea).
“In most cases, the LAST author is the so-called SENIOR AUTHOR, the person serving as the mentor and advisor for the FIRST [LEAD] author, who is responsible for overseeing and guiding the work described in the article. Often the senior author is the source of the original idea that motivated the work in the article, typically with significant input from the first author and often other authors. The senior author is often listed as the corresponding author, indicating that he or she shares responsibility for the integrity of the work as a whole with the first author.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4621797
“The LEAD author will be listed FIRST, followed in order by descending level of contribution to the manuscript. The “SENIOR author,” usually the Principal Investigator of the grant that primarily funded the project, may choose to be LAST author.”
http://catalyst.harvard.edu/pdf/chirp/Appendix%20G%20-%20Dissemination%20Policies.pdf
AK, they don’t just assign you as “Senior-Author” of an ASCO poster. Has to be a reason this latest Sunrise Biomarker Study all of a sudden has a PerkinElmer “Technology Strategist” as its Senior Author. That’s the “main guy in charge of the study”.
ASCO’17: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
**Peregrine: Nikoletta Kallinteris (Lead Author), M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**Precision Medicine, Frederick, MD: Tobias Guennel
**PerkinElmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
- - - - - - - - - -MORE:
SENIOR AUTHOR: Clifford Hoyt (Oncology Fellow/PerkinElmer)
Note (Nov2016):
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
=========================
Known Sunrise Biomarkers thru 4-2017(AACR): B2GPI, Complement+IL10, IFN-y – see http://tinyurl.com/kq7cy5o
The progression of Authors thru the 1st 3 Sunrize Biomakers (ending with aACR’17)...

.
.

N40k, take a look at the summary-of-Authors I made for PPHM’s ASCO’17 poster #11603 ( http://tinyurl.com/ldt2vp6 ), esp. the distinctions between their actual workplaces/functions and their disclosures. I think I got it right – part of what was in the “Authors Disclosures” screen was confusing and in error.
For example, it says, "Ronald B. Natale, Employment=AstraZeneca” - that’s not true – he is Med.Dir of the /Lung-Cancer-Pgm at Cedars-Sinai Medical Center and merely receives funding from AZN and advises them.
Another example – it says, "Amanda Clement, Employment – Abbvie”. Again, this is not true. She is a scientist with Perkin Elmer and clearly has some sort of research/advisory relationship with Abbvie.
See the full picture below...
= = = = = = =
Jun2-6: ASCO 2017, Chicago https://am.asco.org See: http://tinyurl.com/ldt2vp6
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=Perkin Elmer)
Amanda Clement (Scientist/Perkin Elmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/Perkin Elmer)
…Note (Nov2016): Clifford Hoyt, Perkin Elmer
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
- - - - - - - - - -
**Peregrine: N.Kallinteris, M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**Perkin Elmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel

RHW, the 3 prior Sunrise Biomarker revelations (B2GPI, Complement+IL10, IFN-y), plus now this new one coming 6-3-17 at ASCO’17, are all part of Peregrine’s “Comprehensive SUNRISE Biomarker Program” which has been advancing for a year or more now… See Joe Shan’s 3-13-17 comments below.
3-13-17/CC: JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
During the quarter, we continued to wrap-up patient treatment and follow-up with our SUNRISE trial. While the study was discontinued in Feb.2016, follow-up data collection continues, and patients assigned to bavituximab containing arm were allowed to continue receiving study drugs, if the investigators determined it was in the patients’ best interest. This has been a valuable effort and we are very pleased with the amount of data that has been collected over the past year. It is important to mention that we are now working diligently to transition the remaining patients still receiving bavituximab to compassionate-use programs. We have recently concluded data collection and are now working towards day-to-day slots and beginning our final analyses, and hope to present results later in the year. We’re in the meantime evaluating ways to advance bavituximab with immune stimulating therapies as resources permit, while supporting the stated corporate profitability goals. Concurrently, we’re completing patient sample testing from the comprehensive SUNRISE Biomarker Program [Biomarkers: http://tinyurl.com/jrenezs ]. Biomarkers play an increasingly important role in helping identify specific patient characteristics that correlate with or even potentially predict response to a treatment. For this reason, our SUNRISE study protocol pre-specified the collection of thousands of patient samples for exploratory analyses over a wide range of possible biomarkers. Through this effort, our team identified a correlation between pre-treatment levels of beta-2 glycoprotein-1 [B2GPI] and overall survival, which was presented at ESMO last October. In early April, at the upcoming AACR Annual Meeting, we will present data from an analysis of pre-treatment interferon gamma [IFN-y]. As the remaining SUNRISE patient samples are tested, we plan to correlate these data with the final clinical data, and plan to share these results as they become available later this year. http://tinyurl.com/grhwjvy
= = = = = = = = = = = = =
Jun2-6: ASCO 2017, Chicago https://am.asco.org See: http://tinyurl.com/ldt2vp6
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
#1: 6-3-17 1:15-4:45pm, #11603, “Prelim. Correlative Analysis of PD-L1 Expression from the Sunrise Study”
...11 Authors: LEAD: Nikoletta Lea Kallinteris (Lead/PPHM); SENIOR: Cliff Hoyt (Senior/Perkin-Elmer, "Technology Strategist")
#2: #e23091 (pub.only): “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”
...Lead author is from Nilogen Oncosystems http://www.nilogen.com , Senior Author from Moffitt w/Scott Antonio https://moffitt.org , and PPHM co-authors.
ASCO’17: PPHM’s 2 Abstracts (11603/June3, e23091/pub.only)
6-3-17 poster #11603 “Prelim. Corr. Analysis of PD-L1/Expression from SUNRISE” has 11 authors: Lead author is PPHM’s Nikoletta L.Kallinteris, and Senior Author is Perkin Elmer’s Technology Strategist Cliff Hoyt, whose function at PKI is to "help identify clinical opportunities that can be addressed through new technologies & capabilities, including forming collaborations & partnerships with academic & industrial institutions.”
The 2nd Abstract is Pub. Only (Jrnl Clin. Oncology), “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients”. Senior author is Moffitt’s Soner Altiok (with co-author Moffitt’s Scott Antonia), and Lead author is Nilogen Oncosystems’ Melanie Mediavilla-Varela (see http://www.nilogen.com ).
Jun2-6: “ASCO 2017”, Chicago https://am.asco.org
...PPHM Exhibiting – booth #13124 (next to Moffitt CC)
Abstracts: http://abstracts.asco.org (rel. 5-17-17 5:00pmET)
PPHM #1: 6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study” (poster board #303)
http://abstracts.asco.org/199/AbstView_199_190902.html
AUTHORS:
LEAD: Nikoletta Lea Kallinteris (PPHM)
Rachel E. Sanborn (Co-Dir., Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland; Honoraria=AstraZeneca; Consulting=Amgen/Ariad/DNA/PPHM/SeaGen; Funding=BMS/MedImmune/Merck)
Leora Horn (Oncologist/Vanderbilt-Ingram CC; Honoraria=Biodesix; Funding=AZN)
David E. Gerber (UTSW)
Ronald B. Natale (Med.Dir/Lung-Cancer-Pgm/Cedars-Sinai Medical Center; consult/funding=AstraZeneca)
Min Tang (PPHM)
Sean Downing (Scientist/Molecular Pathology, Foundation Medicine Inc., Cambridge MA) http://www.foundationmedicine.com ; disclosure=Perkin Elmer)
Amanda Clement (Scientist/Perkin Elmer; disclosure=Abbvie)
Tobias Guennel (Sen.Dir., Translational Informatics & Biometrics, Precision for Medicine, Frederick MD https://www.precisionmedicinegrp.com/pfm )
Joseph Shan (PPHM, VP/Clin+Reg)
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/Perkin Elmer)
…Note (Nov2016): Clifford Hoyt, Perkin Elmer
“Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations & partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
- - - - - - - - - -
**Peregrine: N.Kallinteris, M.Tang, Joe Shan
**Robert W. Franz CC, Earle A. Chiles Res. Inst., Providence CC, Portland: Rachel E. Sanborn
**Vanderbilt Univ. MC, Nashville: Leora Horn
**UTSW-MC/Dallas: David Gerber
**Cedars-Sinai Outpatient CC, Los Angeles: Ronald B. Natale
**Perkin Elmer, Inc., Hopkinton, MA: Amanda Clement, Clifford Hoyt (Senior Author)
**Precision Medicine, Frederick, MD: Tobias Guennel
- - - - - - - - - - - - - -
ABSTRACT - BACKGROUND:
SUNRISE, a global, double-bind, Phase III trial of docetaxel (D) + bavituximab (B) or D plus placebo (P) in previously treated non-squamous NSCLC, demonstrated similar overall survival (OS) in both treatment arms. Biomarkers including pre-treatment PD-L1 expression are being retrospectively assessed in on-going exploratory analyses.
METHODS:
Archival tissue obtained at the time of diagnosis was requested but not required in the SUNRISE trial. FFPE slides were stained with a panel of lymphoid cell markers: CD3+, CD8+, FoxP3+, PD-L1+, CD163+, CK+ and DAPI using a 6-plex quantitative immunohistochemistry (IHC) assay (OPAL, PerkinElmer, Hopkinton, MA). Baseline PD-L1 expression was retrospectively scored on tumor cells (TC) as a percentage of PD-L1 expressing tumor cells: TC3>=50%, TC2>=5% and < 50%, TC1>=1% and < 5%, and TC0 < 1%. Cox regression models for PD-L1 IHC subgroup populations were used for correlation with OS.
RESULTS:
In the subset of patients with available diagnostic biopsies (110 out of 597 randomized patients), the prevalence of PD-L1 expression was 5% for TC3, 18% for TC2/3, 35% for TC1/2/3, 65% for TC0. Median OS (mOS) of the D+B arm is 11.5 months (TC0, < 1%) and 6.0 months (TC1/2/3, >=1%) with HR 0.38 (95% CI, 0.19-0.76); p-value = 0.004. mOS of the D+P arm is 11.1 months (TC0, < 1%) and 10.4 months (TC1/2/3, >=1%) with HR 0.93 (95% CI, 0.47-1.87); p value = 0.844.
CONCLUSIONS:
Baseline PD-L1 expression in a subset of SUNRISE patients demonstrated that PD-L1 expression (TC0) was associated with a significantly prolonged OS compared to positive PD-L1 expression (TC1/2/3) in patients receiving D+B. No difference in OS was observed in the D+P group by PD-L1 expression. These observations are consistent with the hypothesis that bavituximab may demonstrate more effect in PD-L1 negative or low expressing “immune cold” tumors.
Clinical trial info: https://www.clinicaltrials.gov/ct2/show/NCT01999673

= = = = = = = = = = = = = =
PPHM #2 (PUB. ONLY, NOT PRESENTED AT MEETING - J Clin Oncol 35/2017/Suppl)
Category: Tumor Biology, Sub-cat: Immunobiology
#e23091: “Effect Of Bavituximab In Combination With Nivolumab on Tumor Immune Response in a 3D Ex Vivo System of Lung Cancer Patients” (poster board #303)
http://abstracts.asco.org/199/AbstView_199_193973.html
AUTHORS:
Nilogen Oncosystems: Melanie Mediavilla-Varela (LEAD), Melba Marie Page, Jenny Kreahling http://www.nilogen.com
Moffitt Cancer Center: Scott J. Antonia, Soner Altiok (SENIOR AUTHOR) https://moffitt.org
Peregrine: Bruce D Freimark, Joseph Shan, Nikoletta Lea Kallinteris
- - - - - - - - - -
Dr. Scott J. Antonia, MD/PhD serves as Medical & Scientific Advisor of Peregrine Pharmaceuticals. He advises Cellular Biomedicine Group on immuno-oncology. Dr. Antonia serves as Chair of Thoracic Oncology Dept. and Leader of the Immunology Pgm. at the H. Lee Moffitt Cancer & Research Institute in Tampa. Using his molecular biology & cellular background in the dev. of immunotherapeutic strategies for the treatment of cancer patients, he has developed strategies designed to thwart the immunosuppressive mechanisms used by tumors to evade T-cell mediated rejection...” Honoraria/Advisor=AstraZeneca/Boehringer Ingelheim/BMS/Merck http://tinyurl.com/klnbfkh
Dr. Soner Altiok, MD/PhD - ”As a Cytopathologist at Moffitt CC, my focus is on applying techniques of cell biology & proteomics to the field of cytopathology to identify novel diagnostic and prognostic biomarkers.” Honoraria/Advisor/ExpertTestamony=Genentech https://moffitt.org/research-science/researchers/soner-altiok/
ABSTRACT
BACKGROUND:
Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Our previous studies showed that bavituximab enhances the activation of CD8+ TILs that correlates with increased cytokine production by lymphoid and myeloid cells in lung cancer with low PD-L1 expression suggesting that the interruption of the PD-1/PD-L1 axis by nivolumab may enhance the bavituximab effect in tumors.
METHODS:
Fresh tumor tissues obtained from consented patients with NSCLC at the time of surgical resection were utilized in a 3D ex vivo tumor miscrosphere assay, where 3D tumor microspheres were treated with bavituximab or nivolumab alone or in combination at 10 mg/ml for 36 hours. At the end of the treatment, a multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines, including human IFNg, in culture media as a surrogate of TIL activation. In addition, a gene expression analysis using a NanoString platform containing probes to quantitate 770 immune function genes.
RESULTS:
Preliminary results indicate the combination treatment with bavituximab & nivolumab led to increased expression of genes involved in M1 polarization of tumor associated macrophages in a subpopulation of lung tumors that closely correlated with release of cytokines such as MIP1b (CCL4) which is a chemoattractant for natural killer cells, monocytes and a variety of other cells involved in tumor immune response.
CONCLUSIONS:
This lung patient derived ex-vivo approach indicates that bavituximab in combination with nivolumab may enhance immune response. This response likely involves M1 polarization of tumor associated macrophages and suggests potential clinical implications in the treatment of lung cancer.

=========================
NOTE: Known Sunrise Biomarkers thru 4-2017(AACR): B2GPI, Complement+IL10, IFN-y – see http://tinyurl.com/kq7cy5o
.

Senior Author is Perkin Elmer’s Cliff Hoyt. Check out what his function is there at Perkin:
SENIOR AUTHOR: Cliff Hoyt (Oncology Fellow/Perkin Elmer)
Nov2016:
Clifford Hoyt, Perkin Elmer - “Mr. Hoyt is a Technology Strategist in the area of oncology, helping to identify clinical opportunities that can be addressed through new technologies & capabilities. This includes forming collaborations and partnerships with academic & industrial institutions.”
http://www.sciencemag.org/custom-publishing/webinars/multiplexed-immunohistochemistry-illuminating-tumor-microenvironment
For one thing, June6=Last day of ASCO...
Jun2-6: ASCO 2017, Chicago https://am.asco.org (Abstracts: Titles/Apr20, Full=May17 5pmET)
6-3-17 1:15-4:45pm, Abstract #11603, Track: Tumor Biology
“Preliminary Correlative Analysis of PD-L1 Expression from the Sunrise Study”, Nikoletta Lea Kallinteris (1st Author), Peregrine Pharmaceuticals
…...NOTE: Known Sunrise Biomarkers thru 4-2017(AACR): B2GPI, Complement+IL10, IFN-y – see http://tinyurl.com/ktzr78
PPHM Exhibiting – booth #13124 (next to Moffitt CC)
I got TWO Mobius’ from CEO Steve King/3-13-17. The fact that he added, “within the next few months”, tells me TWO is the number. Of course, Two=Multiple. I do ack I could be wrong.
3-13-17 CC, CEO Steve King: “...An important component of our Avid growth strategy is capacity expansion within our Myford facility. We are currently on track to install two 2,000L bioreactors [Mobius 2000L single-use] in the facility within the next few months, with a book of business for the reactors already in place. We believe the total capacity potential of the facility, when operating in campaign mode, can exceed more than $75mm annually, bringing us to well over $100mm in total potential revenue between our 2 mfg. facilities, providing us with adequate capacity to continue Avid revenue growth through FY’18. As we look beyond this FY and into the future, based on current operations & projected demand from our clients, we have also recently secured addl. space adjacent to our Myford facility, which we already have the use for as part of our growing operations. And additionally, would also allow us to further expand mfg. capacity based on committed business. While we will only begin converting space into mfg. capacity once client commitments and other necessary financing is in place, this puts us in an excellent position for continuing to grow the business beyond the coming FY.” http://tinyurl.com/grhwjvy
= = = = = = = = = = =
5-3-17 BioPharma-Reporter: “Latest Avid investment to turn bioreactor capacity up to 11,000L”
Avid Bioservices will add multiple MilliporeSigma 2,000L bioreactors at its Californian single-use production plant in an effort to its increase annual revenue to over $100m.
The Myford facility [Avid II] came online in Dec.2015, but continued demand for clinical & commercial biomanufacturing has led to a capacity upgrade.
While an Avid spokesperson told Biopharma-Reporter the firm has experience with a “wide number of different single-use systems across multiple vendors,” this is the CMO’s first foray into the 2,000L scale...
The investment will “bring total capacity within the Myford facility to over 9,000L of capacity at the 1,000 & 2,000L scale and the total capacity at our 2 facilities to over 11,000L,” we were told.
Avid’s other facility is the Franklin plant, also located in Tustin, CA.
“With the installation of the new bioreactors at Myford, we expect that facility, when operating in campaign mode, can exceed more than $75m annually. This would bring our total potential annual revenue across both of manufacturing facilities to well over $100m.”
http://www.biopharma-reporter.com/Upstream-Processing/Avid-selects-MilliporeSigma-2-000L-single-use-bioreactors-at-CA-plant
CP, not home now, but not sure if we can infer from the HALO 10Q commentary issued 5-9-17 about Avid II (Myford) that the 2 new 2000L Miobus(sp?) single-use bioreactors will be fully validated during Q2/2017 or not. Recall that prior to this 4000L expansion, Myford capacity was already at "greater than 5000L" ($40M+/yr if I recall), and the 2 new Miobus's will take Myford to > 9000L. Just not sure about that part of Halozyme's commentary.
Halozyme IS talking about Avid.II(Myford) in their 3-31-17/10Q pub. 5-9-17: “work associated with bringing on-line a 2nd contract mfg. facility. The validation of the new facility is scheduled to end in Q2/2017… Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20.”
HALO’s 3-31-17 10-Q filed 5-9-17: http://tinyurl.com/k6zb9uy (Halozyme)
Pg.41: "Product sales, net decreased in the 3mos ended 3-31-17 compared to the same period in 2016, due to a decrease in the sales of bulk rHuPH20 to Roche and a decrease in the sales of Hylenex. We expect that product sales of bulk rHuPH20 will fluctuate in future periods based on the needs of our collaborators. In 2016, we performed services for Roche to bring on-line a 2nd contract mfg. facility for bulk rHuPH20. Once this new facility is approved, it will become the primary source for Roche of bulk rHuPH20. As a result, we anticipate Roche will deplete their existing inventory of rHuPH20 ahead of the transition to the new facility, which will result in lower bulk product sales during 2017 & 2018. We expect that future product sales of Hylenex to be flat or experience modest growth, although there may be periods with declining revenue as we experience competition for market share.”
Pg.42: “Revenue from reimbursements for R&D services increased in the 3mos ended 3-31-17, 2017, compared to the same period in 2016 mainly due to an increase in services provided to Roche related to work associated with bringing on-line a 2nd contract mfg. facility. The validation of the new facility is scheduled to end in Q2/2017 and, therefore, we expect to see a decrease in R&D service revenue from Roche going forward. R&D services rendered by us on behalf of our collaborators are at the request of the collaborators; therefore, the amount and timing of future revenues related to reimbursable R&D services is uncertain.”
= = = = = = = = = = = = = = =All HALO Filings: http://tinyurl.com/k6zb9uy
PROOF:
HALO’s 12-31-16 10-K, iss. 2-28-17 (pg.12):
“In addition, we are working to scale-up, validate, and qualify a new facility operated by Avid [Avid II, “MYFORD”] as a manufacturer of bulk rHuPH20 for use in the products & product candidates under the Roche Collaboration.”
Pg.9: ROCHE COLLABORATION:
“In Dec. 2006, we and Roche entered into a collaboration & license agreement under which Roche obtained a worldwide, license to develop & commercialize product combinations of rHuPH20 and up to 13 Roche target compounds (the Roche Collaboration). Roche initially had the exclusive right to apply rHuPH20 to 3 pre-defined Roche biologic targets with the option to develop & commercialize rHuPH20 with 10 addl. targets. Roche had the right to exercise this option to identify addl. targets for 10 years. As of the 10 year anniversary of the Roche Collaboration in Dec. 2016, Roche had elected a total of 8 targets, 2 of which are exclusive.”
= = = = = = = = = = = = = = = = = = = = = = = =
3-13-17: PPHM's Revs & Burns By Qtr Table, FY'07/Q1 thru FY'17/Q3 (q/e 1-31-17): http://tinyurl.com/grhwjvy (since 5-2006: Avid=$213.3mm, Total=$239.9mm, incl.Govt)
......Avid FY17 (fye 4-30-17) revs guidance upped: $60-65mm; committed backlog=$70mm. Recently Leased +42,000sf in same bldg. as MYFORD for future expansion. Expect future sustainable profitability for the co. in 15mos.”
5-2017: Avid II (Myford) adds 2 MilliporeSigma Mobius 2,000L single-use bioreactors; total mfg. capacity now ">11,000L". http://tinyurl.com/ky7bmu4
2-28-17: Avid & Cook remain Halozyme's 2 CMO's (“working to scale-up/validate/qualify Avid II/Myford for Roche collab.”): http://tinyurl.com/h75teta

There we go again, leaving out the rest of what he said, to try to make it...
3-13-17 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/grhwjvy
PL: Switching gears now, I’d like to address our plans to preserve our NASDAQ listing. At a backdrop, last April, we were notified by the NASDAQ that our share price did not meet the $1 min. bid price for 30 consecutive days, which is a requirement for continued listing on the NASDAQ market. At that time, we were automatically afforded an initial compliance period of 180 days or until Oct. 10, 2016 to regain compliance. At the end of the initial compliance period, we requested and were granted a 180 day extension to regain compliance. This 2nd extension period will end on April 10, 2017. Let me discuss our strategy and future steps we plan to take over the next few months. First & foremost, we have determined that it is not in the best interest of our stockholders to affect a reverse stock split prior to April 10, 2017. If our share price does not trade above $1 for 10 consecutive trading days by April 10th, we expect to receive a notice of delisting. If we received this notice, we will appeal this decision within the required 7 days and this appeal will stay any delisting actions by NASDAQ. At that point, NASDAQ will schedule a hearing, which is typically held within 45 days, and we will present our plan to regain compliance. We are considering several paths to regain compliance, including having addl. time to continue to allow the market to adjust, to reflect the current value of Avid as evidence with the recent movement in our share price. The hearing panel could afford us up to 180 addl. days to regain compliance, although this addl. time is not guaranteed and this decision is at the discretion of the NASDAQ’s hearing panel. If we are not afforded addl. time from this panel, it is important to note that our stockholders approved the Board’s ability to implement a reverse stock split at a ratio of up to 1:7 as a means to regain compliance and to preserve our listing on NASDAQ. During this entire process, Peregrine will continue to be listed on that NASDAQ capital market as it does today. We will keep you informed on any material developments through our SEC filings on Form 8-K as we execute on this plan.
3-13-17/CC: “There are patients still still receiving bavituximab.”
3-13-17/CC: JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
“During the quarter, we continued to wrap-up patient treatment and follow-up with our SUNRISE trial. While the study was discontinued in Feb.2016, follow-up data collection continues, and patients assigned to bavituximab containing arm were allowed to continue receiving study drugs, if the investigators determined it was in the patients’ best interest. This has been a valuable effort and we are very pleased with the amount of data that has been collected over the past year. It is important to mention that we are now working diligently to transition the remaining patients still receiving bavituximab to compassionate-use programs...”
3-13-17 Q&A: [beg. 20:30]
1. Kumara Raja - Noble Life Science Partners http://noblelsp.com/research
KR: ”...In terms of the SUNRISE trials, what are you seeing in patients who are continuing on bavituximab? What kind of benefits are you seeing there? And you’ve talked about Compassionate Use Program, what needs to be done for that in different countries?”
STEVE KING: …For the question regarding patients that are still on bavi and how are they doing, and the Compassionate Use, I’ll turn it over to Joe.
JOE SHAN: ...There are of course patients still as we mentioned still receiving bavituximab, so that gives you an indication that there are patients that are not only, what we call long-term survivors but there are diseases that stabilized. That’s why it's was important that the trial is formally concluded that we allow the patients the opportunity to continue receiving bavituximab under compassionate use...Regarding the compassionate use, the procedure varies from country-to-country, but basically, investigators that have patients still receiving bavituximab need to file an IND or expanded access to application in the U.S. It’s a single patient IND, and in Europe it's under the expanded access. It’s a bit of paper work, but we’re assisting where we can, but basically the investigator takes responsibility from that point on, and the approvals are pretty quick.
STEVE KING: That’s one of the things that we see with bavituximab is, while we’re all disappointed that the overall trial results weren’t what we expected, certainly, you see these anecdotal signs where we obviously see a number of patients that are still on therapy who are then coming in for quite now very long extended period of time, we feel like they’re getting benefit. You see these anecdotal signs activity in those patients that have been on therapy, and for us that’s one of the reasons we’re still very happy and actually excited about the collaborations at Sloan Kettering and UTSW, in particular, where there’s not just an interest in studying bavituximab in preclinical models, but actually taking some of that data and eventually turning that into clinical indications where the drug excels.
3-13-17/CC Transcript: http://tinyurl.com/grhwjvy
3-13-17/S.King: “...currently on track to install TWO 2000L-bioreactors [Mobius 2000L single-use, from MilliporeSigma, the "life science business of Merck KGaA, Darmstadt, Germany"] in the facility within the next few months, with a book of business for the reactors already in place. We believe the total capacity potential of the facility, when operating in campaign mode, can exceed more than $75mm annually, bringing us to well over $100mm in total potential revenue between our 2 mfg. facilities, providing us with adequate capacity to continue Avid revenue growth through FY’18. As we look beyond this FY and into the future, based on current operations & projected demand from our clients, we have also recently secured addl. space adjacent to our Myford facility, which we already have the use for as part of our growing operations. And additionally, would also allow us to further expand mfg. capacity based on committed business. While we will only begin converting space into mfg. capacity once client commitments and other necessary financing is in place, this puts us in an excellent position for continuing to grow the business beyond the coming FY.”
5-3-17/BioPharmaReporter article: “the investment will bring total capacity within the Myford facility to over 9,000L of capacity at the 1,000 & 2,000L scale and the total capacity at our 2 facilities to over 11,000L, we were told."
More: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=131098036