Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
LC,
Thank you for highlighting the NIH! The vast significance of Liau’s work and the further development of the DCVax-L cell-based platform technology in combination with other technologies was/is funded in large part by public dollars awarded through NIH peer-reviewed grants. DCVax-L is already supported by peer-reviewed clinical evidence, but there is much more to the story that is yet to unfold.
If you do some research into the NIH and the FDA, you will discover that not every investigational technology receives the same level of support and not every researcher’s work is recognized by consistent support spanning many consecutive years. The NCI division of the NIH develops oncology drugs, and FDA approves drugs. There is some connection, and their historical and continuing support is not insignificant …
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs
Targeted individualized combo therapies are emerging as the defining feature of a new era of oncology. Just look at the impact that PD1 blockade technology has had on the field of oncology in recent years!
https://clinicaltrials.gov/ct2/show/NCT04201873
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.

Mr. Newirth is another DCVax-L survivor well surpassing 10 years following a 2012 diagnosis! Congratulations!
https://www.hawaii.edu/news/2017/03/30/newirth-laker-for-a-day/
June 13, 2023 Interview:
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/












HyGro, Explore Professor Keyoumars Ashkan's passion for neurosurgery and his love for @KingsCollegeNHS below as part of their #BrilliantPeople series🧠👇@KingsNeuro @KingsIoPPN https://t.co/yGKt2681Ie
I hope you’ve been well. Thanks for reposting your opinion again about the ECA, but your post is bs as per usual.
Doctors Liau and Ashkan know how to run a clinical study.
You are never going to see the FDA or MHRA allow trial participants to be used as lab rats.
The crossover design was required to recruit and retain participants. Who would want to join a GBM study without crossover or remain in a placebo group after developing progressive disease without having the option of receiving an efficacious treatment?

Conclusion: The P3 was exemplary—clinically appropriate and compassionate.

Skip to timestamp 8:01. Dr. Liau again confirms that the crossover was mandated by the FDA.
https://edhub.ama-assn.org/jn-learning/audio-player/18738384


https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d

https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato
https://www.fda.gov/media/171667/download
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
Professor Ashkan is a careful and precise brain surgeon—listen to his words.
Skip to timestamp 13:00 of the ASCO video to hear about the ECA and the SAP.
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/


The clinical data in total supporting the DCVax-L platform technology include data beyond the trial with the ECA, and the ECA included over a thousand well-matched contemporaneous patients.
The P3 data were peer reviewed by independent physicians and qualified statisticians—refer to JAMA Oncology.
Safety and efficacy data have been gathered from various clinical settings that include both external controls and placebo controls. DCVax-L has been studied as a monotherapy and in combination with other agents.
If you prefer a randomized placebo design, then you’ll love the pembrolizumab PD1 blockade combo study that is ongoing at UCLA today. The study has a placebo design and the placebo participants are receiving DCVax-L as the best available therapy for rGBM.
The interim PD1 combo data are brilliant, and the trial is significant for a number of reasons relating to FDA guidelines and NIH peer-reviewed grant funding.
https://clinicaltrials.gov/ct2/show/NCT04201873
In the combo trial, patients who receive DCVax-L + placebo are being compared to patients who receive DCVax-L + pembrolizumab.
The control arm is receiving DCVax-L. While DCVax-L is not the current SOC, it is highly significant and notable that DCVax-L is being permitted to fill the role of SOC as the best available therapy within this combo study for rGBM. The combo trial also follows the P3 in sequence which in itself validates the findings of the P3 study—the placebo group in the PD1 study would not be receiving DCVax-L today if the preceding P3 trial had not produced reliable evidence of efficacy.
The U.S. NIH has supported the DCVax-L platform for years, and the NIH renewed its support in 2022 to continue to fund the development of this technology and to fund research into combination therapies.
NIH-funded DCVax-L research is ongoing at UCLA today.
NIH grants are peer-reviewed and the research that NIH funds is highly scrutinized in advance of the award and also intermittently for the term of the various research projects.
https://www.bentley.edu/news/new-study-shows-nih-investment-new-drug-approvals-comparable-investment-pharmaceutical
https://www.bentley.edu/news/nih-funded-research-related-every-new-cancer-drug-approved-2010-2016
https://ashpublications.org/ashclinicalnews/news/3819/NIH-Funding-Tied-to-FDA-Approval-of-210-Drugs

https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials
https://www.uclahealth.org/news/ucla-received-590-million-in-nih-funding-second-highest-total-for-academic-medical-centers-in-2020
https://www.fda.gov/science-research/advancing-regulatory-science/fda-nih-joint-leadership-council-charter
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
learningcurve2020, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh
Thanks for reposting your thoughts about the MAA status and the publisher throughout the day ..everyday 😶
We do however appreciate that you are redirecting the conversation to focus on the regulatory process…
Following the independent peer review, we are entering a new phase for the landmark DCVax-L cell-based platform technology. The regulatory environment also seems to be evolving to embrace a whole new paradigm—we are now in the era of RWD and immunotherapy!
Exciting times … Combo is King!
Some links that seem highly relevant:
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato

https://www.fda.gov/media/171667/download
https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/




https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

I would also recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology.

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

You repost the same nonsense repeatedly…
I believe that you’re drifting off way too far from Earth 🌎️ … You’re misinformed and need better sources of information. It is advisable to seek full-context credible sources and consult licensed oncologists.

Amid all of the misinformation and bad-faith disinformation pollution online, it is crucial that we refocus our attention beyond baseless social media posts to consider better sources…
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.
JAMA Oncology is a peer-reviewed medical journal that us part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers, healthcare professionals, and the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt #dcvax #allsolidtumors $nwbo #gbm 🧠 This weekend, read about Professor Keyoumars Ashkan, Consultant Neurosurgeon at King's, who met with his patient Dagmar Turner after her #BrainSurgery - their experience is showcased at the @HunterianLondon.
Thanks for asking. To be specific, most of your posts sound like sourceless and baseless innuendo.
The CEO once loaned NWBO millions from her own pocket to fund research into the DCVax-L technology. Of course, she was rewarded for loaning her own money, but her actions were a testament to her confidence and her long-term commitment. The leadership have been committed to the company long term and they are shareholders too.
Your vague reference to “ethics” seems defamatory absent of any supporting facts, in my view.
UCLA, King’s College, JAMA Oncology, and Brown University are some excellent sources that you might wish to consider in reference to the reputation of Northwest Biotherapeutics and those who choose to associate with the company, its SAB, and with the novel DCVax® technology.
The world-renowned doctors cited below worked on the P3, and they both were coauthors of a DCVax-L publication along with Dr. Bosch of Northwest Biotherapeutics.
Furthermore, both Liau and Ashkan have had the support of their respective nations’ governments over many years. Ashkan is an advisor to the UK government, and Liau’s work has been supported by NIH peer-reviewed grants for 20 consecutive years.
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html

https://labusinessjournal.com/healthcare/national-academy-medicine-taps-ucla-neurosurgeon/

https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
Great to see, congratulations Linda Liau winning the Charles B. Wilson Excellence Award 2023:
Established by the Brain Tumor Society in 2004, this award recognizes outstanding contributions of a clinician to the field of neuro-oncology.… pic.twitter.com/6L7rhyoNU4
https://www.kch.nhs.uk/news/kings-team-wins-professional-excellence-award/

Read #InsideKings ➡️ https://t.co/mKmO2Hbp4J pic.twitter.com/TZdCDnfXyJ
LC,
Human-generated commentary is great, but AI is an excellent supplement to refute bad-faith short-generated disinformation. Thanks for the tip!😉
“It's important to distinguish between innuendo and factual statements on stock message boards. Factual information is based on evidence and can be verified, while innuendo often involves indirect hints or suggestions without concrete proof. When evaluating stock discussions, focus on reliable sources and verifiable data to make informed decisions.
"Short and distort" refers to a manipulative tactic where individuals short a stock (betting its price will fall) and spread false or misleading information online to drive down the stock's value. This unethical practice can harm investors and the market integrity. Always rely on reputable sources and conduct thorough research before making investment decisions.
The purpose of independent peer-reviewed medical journals is to ensure the quality and credibility of scientific research in the field of medicine. Peer review involves evaluation by impartial experts who check for accuracy, validity, and ethical standards. This rigorous process helps maintain the integrity of medical literature, providing a reliable foundation for medical professionals, policymakers, and the public to make informed decisions about healthcare.” —ChatGPT
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
https://pubmed.ncbi.nlm.nih.gov/33789235/
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
LC,
Thank you for linking us to the Labiotech site—looks like a solid recommendation!

https://www.labiotech.eu/trends-news/nw-bio-reports-positive-top-line-results-from-glioblastoma-trial/
https://www.labiotech.eu/trends-news/northwest-biotherapeutics-cancer-pip-approval/
Thanks also for reminding us that other advances in the research of cancer may elevate the value of DCVax-L! Each existing, emerging, or recently commercialized therapy in the field of oncology represents another potential opportunity to discover an efficacious combo with the DCVax-L platform!
Combo is King!


LC, Britain's Medicines and Healthcare products Regulatory Agency (MHRA) will officially join #ProjectOrbis on January 1 and work with regulators in the US, Canada, Australia, Singapore and Switzerland on assessing new treatments. https://t.co/JjaXoq6yfh
Thanks for frequently reposting your thoughts about the MAA status. We appreciate all the posts from LC and ex highlighting the MHRA and FDA—we will soon shift our focus to the regulatory review process…
Following the independent peer review, we are entering a new phase for the landmark DCVax-L cell-based platform technology. The regulatory environment also seems to be evolving to embrace a whole new paradigm—we are now in the era of RWD and immunotherapy!
Exciting times … Combo is King!
Some links that seem highly relevant:
https://www.raps.org/news-and-articles/news-articles/2023/8/fda-finalizes-guidance-on-real-world-evidence-in-d
https://www.raps.org/News-and-Articles/News-Articles/2023/9/FDA%E2%80%99s-Califf-Expect-to-see-more-RWE-based-regulato

https://www.fda.gov/media/171667/download
https://www.uclahealth.org/cancer/researchers/spores/ucla-spore-brain-cancer/research-projects
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.merck.com/news/daiichi-sankyo-and-merck-announce-global-development-and-commercialization-collaboration-for-three-daiichi-sankyo-dxd-adcs/




https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials

https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

I would also recommend that you review the forthcoming NICE appraisal of the DCVax-L cell-based technology.

https://www.nice.org.uk/guidance/indevelopment/gid-ta10143



https://www.fda.gov/about-fda/oncology-center-excellence/project-orbis




https://www.kcl.ac.uk/people/keyoumars-ashkan
The MIA was approved recently and is evidence of a regulatory filing plan that has begun to unfold. Further RA applications have been under construction for a significant period of time and include a voluminous catalog of clinical data and analyses. The announcements of regulatory filings are forthcoming.
The recent MIA approval is an important step in the regulatory process, and this commercial manufacturing license will allow DCVax-L to be exported globally from Sawston. The MIA is a prerequisite of the MAA application.
“Companies applying for a marketing authorisation (also known as a product licence) need to have a manufacturer licence first. The manufacturer licence will be granted first providing the product is in the process of being approved.” —MHRA
https://www.gov.uk/guidance/apply-for-manufacturer-or-wholesaler-of-medicines-licences
https://finance.yahoo.com/news/northwest-biotherapeutics-advent-bioservices-announce-234600744.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-approval-pediatric-133900403.html
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
Grip,

Thanks for reposting your opinions again, but I cannot find any meaningful value in your nonsensical comments. Try to refocus and come back to 🌎
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Combo is King!
https://clinicaltrials.gov/study/NCT04201873

LC,
Thanks for reposting so many times about the MAA PR. The company will likely offer further clarification around the time when the filing is accepted and as other regulators also come into play in other nations…
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
JTORENCE,
Thanks for asking, but I couldn’t possibly know the answer to that—it’s a nonsensical question to ask why they’re not buying more and more while they’re already shareholders, but I’ll offer my speculation since you asked. What I do know is that they have a large number of shares already and seem to be holding for commercialization. Marketing the DCVax-L cell-based platform technology is one of the company’s stated goals.
I would also remind you that the CEO loaned NWBO millions from her own pocket to fund research into the DCVax-L technology. Of course, she was of rewarded for loaning her own money, but her actions were a testament to her confidence and her long-term commitment.
The CEO is holding 130 million shares, if I recall correctly.
I would guess that if large purchases were made by insiders during certain periods of time around the publication of the independent peer review, for example, or during the time preceding the MAA, those insiders could be accused of purchasing based on non-public information.
I also believe that their positions are already very large in proportion to their overall wealth. This inference is drawn from my own personal position in NWBO—I want to add shares at the current price, but my money is already invested. I’m already “maxed out” so to speak. I would buy more shares if I had the opportunity to add to my position. (I am a shareholder because I am interested in wealth and health—I look forward to the regulatory advisory reviews in multiple nations, and I personally hope to have access to an efficacious immunotherapy combo if I ever need treatment for cancer. I am neither an oncologist nor a financial advisor).
Let’s ask ChatGPT for more ideas:
There could be various reasons why a CEO might choose not to purchase stock in their own undervalued company, even if it's expected to rise. Some possible reasons include:
1. **Personal Financial Constraints:** The CEO may have personal financial constraints or commitments that limit their ability to invest in the company at that time.
2. **Regulatory Restrictions:** Legal or regulatory restrictions might prevent the CEO from buying or selling stock during specific periods, such as blackout periods around financial reporting.
3. **Ethical Considerations:** The CEO might want to avoid any perception of impropriety or conflict of interest, even if it's legally permissible, to maintain trust and transparency.
It's important to note that individual decisions can vary, and CEOs may have a combination of these or other reasons influencing their choices.
—ChatGPT
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
LC,
The “financial incentive” for long-term investors is an increase in the value of their investment.
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Sounds like innuendo that is not supported by any real evidence, but I’ll research it …
I don’t really track those data, but I would love to see whatever you’re citing. I only ever saw a “gift” to family from an insider. LP has something like 130 million shares and could have sold many times when the share price was up—she didn’t.
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
https://www.theguardian.com/science/2022/nov/17/im-just-carrying-on-vaccine-gives-brain-cancer-patient-years-of-extra-life
iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

I believe that you’re drifting off way too far from Earth 🌎️ … It sounds like you’re misinformed and need better sources of information, or you’re just reposting baseless mud. It is advisable to seek full-context credible sources and consult licensed oncologists.

Amid all of the misinformation and bad-faith disinformation pollution online, it is crucial that we refocus our attention beyond baseless social media posts to consider better sources…
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.
JAMA Oncology is a peer-reviewed medical journal that us part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers, healthcare professionals, and the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


Fact ✅
The company leadership are holding more shares than any other individual shareholders. They never sell. They seem to be holding for regulatory approval(s) and commercialization worldwide, and they stand to benefit more than anyone from achieving those objectives. A rise in the share price will benefit all shareholders.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
LC,
Thanks for reposting about the MAA PR again. I think there will be updates as the regulatory process unfolds—plenty of opportunities to clarify each step as the filing is accepted and as other regulators also come into play in other nations…
It is important to appreciate that the first filing was not only an enormous undertaking, but its significance is monumental and its voluminous catalog of documents must be presented in good order.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
Thanks for reposting about that again—always a great reminder! The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!


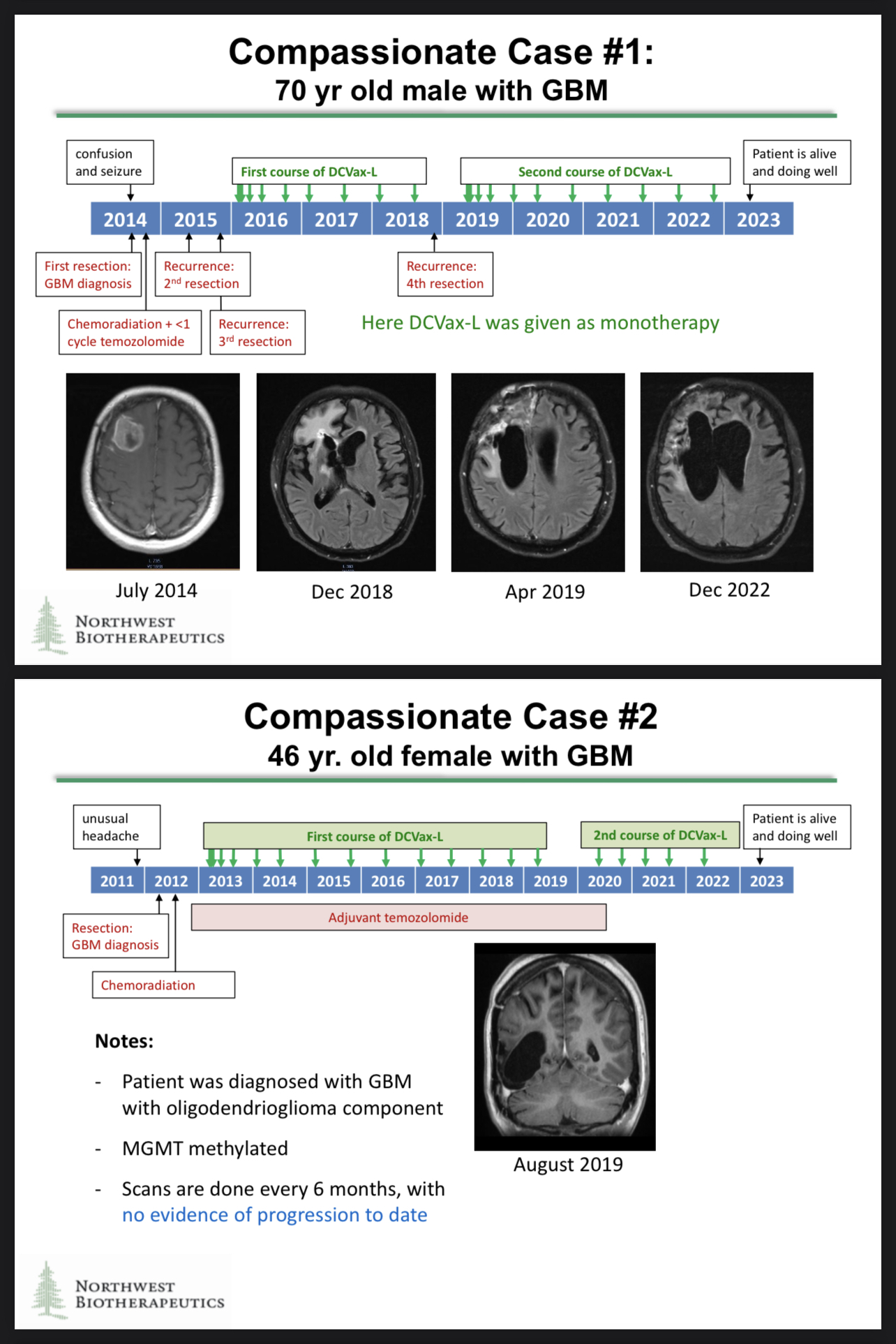
https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
Your suggestion of using AI to supplement responses to postings on messages boards was actually a good idea!
“Yes, compassionate use data, also known as expanded access or named patient programs, can be included in regulatory submissions for drug approval. Compassionate use refers to the use of an investigational drug outside of a clinical trial to treat a patient with a serious or life-threatening illness for whom no other treatment options are available.
Regulatory agencies, such as the U.S. Food and Drug Administration (FDA), recognize that compassionate use programs can provide valuable information about a drug's safety and potential efficacy. However, the inclusion of compassionate use data in regulatory submissions is subject to certain conditions:
1. **Data Quality and Relevance:**
- The compassionate use data must meet the standards of quality, relevance, and reliability expected in clinical trials. Robust data collection and reporting practices are crucial.
2. **Consistency with Regulatory Requirements:**
- The data collected under compassionate use should align with the regulatory requirements for the type of approval sought (e.g., New Drug Application, Biologics License Application).
3. **Ethical Considerations:**
- Ethical considerations surrounding patient safety and informed consent are essential. The compassionate use program should adhere to ethical standards, and patients should be well-informed about the risks and benefits.
4. **Corroboration with Controlled Clinical Trial Data:**
- While compassionate use data can be informative, regulatory agencies typically prioritize data from well-designed, controlled clinical trials. Compassionate use data may complement such trial data but is not a substitute for it.
5. **Regulatory Agency Consultation:**
- It's advisable to consult with the relevant regulatory agency early in the drug development process to discuss the potential inclusion of compassionate use data in regulatory submissions. Agencies may provide guidance on how this data can be integrated appropriately.” —ChatGPT
learningcurve2020,
If anyone strings together dozens of your published posts and looks at them all lined up in a row, they kind of appear to be part of a coordinated campaign to intentionally defame certain individuals.
—Just an observation.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847


LC,
I agree 100% —so much bs!
Thanks for posting and reposting your opinions, but it is advisable to consult financial professionals and licensed oncologists while avoiding baseless message board disinformation.
Thanks also for suggesting that we utilize AI—I thought I would try an AI app in reference to the NWBO conversation:
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.” —ChatGPT
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
learningcurve2020, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

I tried to read your 20 posts today, but you’re only reposting random bs and you’re really not making much sense. I believe that you’re drifting off way too far from Earth 🌎️ … It sounds like you’re misinformed and need better sources of information. It is advisable to seek full-context credible sources and consult licensed oncologists.

Amid all of the misinformation and bad-faith disinformation pollution online, it is crucial that we refocus our attention beyond baseless social media posts to consider better sources…
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.
JAMA Oncology is a peer-reviewed medical journal that is part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers and other professionals in the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


LC,
DCVax-L needs to reach patients worldwide—I agree 100% … NWBO needs regulatory approval(s) to achieve that objective.
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well. NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion topics online was a good idea! Here is some further commentary from an AI app about the importance of having filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
Yes! “Long term” effects! That’s an awesome point! Anything “long term” would be a very welcome side effect!
https://www.theguardian.com/science/2022/nov/17/im-just-carrying-on-vaccine-gives-brain-cancer-patient-years-of-extra-life
LC,
Thanks for the prediction, but it is advisable to consult financial professionals and licensed oncologists while avoiding message board disinformation and baseless innuendo.
Thanks also for suggesting that we utilize AI —not a bad idea… I thought I would try an AI app in reference to the NWBO conversation:
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.” —ChatGPT
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
aesop1,

You’re slipping way too far off track—try to refocus and come back to 🌎
https://braintumourresearch.org/en-us/blogs/research-campaigning-news/astonishing-results-for-brain-tumour-vaccine-trial
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
Combo is King!
https://clinicaltrials.gov/study/NCT04201873

iclight, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru

I believe that you’re drifting off way too far from Earth 🌎️ … It sounds like you’re misinformed and need better sources of information. It is advisable to seek full-context credible sources and consult licensed oncologists.

Amid all of the misinformation and bad-faith disinformation pollution online, it is crucial that we refocus our attention beyond baseless social media posts to consider better sources…
For a reliable appraisal of the DCVax-L P3 trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.
JAMA Oncology is a peer-reviewed medical journal that is part of the Journal of the American Medical Association (JAMA) network, which is one of the most widely respected and influential medical journal publishers globally.
Being a peer-reviewed journal means that research articles submitted to JAMA Oncology undergo a thorough evaluation process by independent experts in the field before they are accepted for publication. This process helps ensure the quality, validity, and scientific rigor of the research presented in the journal.
Researchers and other professionals in the medical community rely on publications like JAMA Oncology for up-to-date and credible information in the field of oncology.
https://ceoworld.biz/2022/04/05/top-5-medical-journals-in-the-world-everyone-should-know-about/





https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials


Fact ✅
I don’t know anything about the doctor that you cited and I’m not commenting about him, but your assertion that he “has more than twice the publications” is verifiably false. You seem to just make stuff up and post it.
Both of these excerpts are from the exact same source—The Society of Neurological Surgeons:
“Dr. Liau has authored over 170 peer-reviewed research articles, along with several book chapters, and a textbook entitled Brain Tumor Immunotherapy. She is on the editorial boards of several scientific/medical journals and was the Editor-in-Chief of the Journal of Neuro-Oncology (2007-2017).”
https://www.societyns.org/about/officers-detail/linda-m-liau-md-phd-mba-4
“He holds several patents on gene transfer techniques and is author or co-authors of more than 200 peer-reviewed publications. He has been elected to the American Society for Clinical Investigation (ASCI), is a fellow of the American Association for the Advancement of Science (AAAS), and a member of the American Academy of Neurosurgery.”
https://www.societyns.org/about/officers-detail/president-e-antonio-chiocca-md-phd

To clarify, you’re citing Liau’s spoken words during an oral podcast interview in conversational format.
In contrast, the printed JAMA Oncology publication was proofed for errors and is accurate.
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847

LC,
Just as the independent peer review process required a lot of groundwork, preparation, and coordination, the MAA does as well.
The company has provided timeline forecasts and updates, and they have also advised us appropriately that some of the MAA work is independent of the company—this was also the circumstance while working though the independent peer review process, as you may recall.
NWBO is a small pre-commercial biotech. The executive team includes well-educated and experienced leaders whose business acumen is formidable. They have enlisted the support of consultants to ensure that regulatory applications are complete, accurate, and fully compliant with filing guidelines.
Your suggestion of using AI to research and supplement responses to discussion board topics online was a good idea! Here is some further commentary from an AI app about the importance of enlisting expert filing support:
“A Marketing Authorization Application (MAA) filing with the Medicines and Healthcare products Regulatory Agency (MHRA) or any regulatory agency involves submitting comprehensive documentation to gain approval for marketing a medicinal product. Both a publisher and a quality control team play crucial roles in ensuring the success and compliance of the MAA filing. Here's why:
1. **Complexity of Documentation:**
- MAA submissions are highly complex and involve a vast amount of documentation, including clinical trial data, manufacturing processes, quality control measures, safety data, and more. A dedicated publisher helps organize, format, and compile these documents into a coherent and compliant submission.
2. **Regulatory Compliance:**
- Regulatory agencies like the MHRA have strict requirements regarding the format, structure, and content of submissions. A skilled publisher ensures that the MAA complies with these regulatory guidelines, increasing the likelihood of acceptance and speeding up the regulatory review process.
3. **Accuracy and Consistency:**
- Quality control teams are crucial for ensuring the accuracy and consistency of the information presented in the MAA. Any inconsistencies, errors, or inaccuracies could lead to delays in the review process or rejection of the application.
4. **Adherence to Standards:**
- Quality control teams ensure that the submission adheres to the highest quality and data integrity standards. This is particularly important for critical sections such as clinical trial data and manufacturing processes, where precision and reliability are paramount.
5. **Risk Mitigation:**
- Thorough quality control processes help mitigate the risk of regulatory issues or deficiencies. Identifying and addressing potential problems before submission increases the chances of a successful review and approval.
6. **Efficient Communication:**
- A well-organized submission facilitated by a publisher enables efficient communication between the applicant and the regulatory agency. Clear and organized documentation helps regulatory reviewers navigate the submission, leading to more effective communication and a smoother review process.
In summary, the collaboration of a skilled publisher and a robust quality control team is essential for preparing a high-quality, compliant MAA filing with the MHRA. This collaborative effort increases the chances of a successful regulatory review and approval, allowing the medicinal product to be marketed and distributed to patients.”
—ChatGPT
https://www.prnewswire.com/news-releases/northwest-biotherapeutics-provides-update-on-final-preparations-for-marketing-authorization-application-301995932.html
https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
LC,
The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!



https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
LC’s suggestion of using AI to supplement responses to postings on message boards was actually a good idea!
“Yes, compassionate use data, also known as expanded access or named patient programs, can be included in regulatory submissions for drug approval. Compassionate use refers to the use of an investigational drug outside of a clinical trial to treat a patient with a serious or life-threatening illness for whom no other treatment options are available.
Regulatory agencies, such as the U.S. Food and Drug Administration (FDA), recognize that compassionate use programs can provide valuable information about a drug's safety and potential efficacy. However, the inclusion of compassionate use data in regulatory submissions is subject to certain conditions:
1. **Data Quality and Relevance:**
- The compassionate use data must meet the standards of quality, relevance, and reliability expected in clinical trials. Robust data collection and reporting practices are crucial.
2. **Consistency with Regulatory Requirements:**
- The data collected under compassionate use should align with the regulatory requirements for the type of approval sought (e.g., New Drug Application, Biologics License Application).
3. **Ethical Considerations:**
- Ethical considerations surrounding patient safety and informed consent are essential. The compassionate use program should adhere to ethical standards, and patients should be well-informed about the risks and benefits.
4. **Corroboration with Controlled Clinical Trial Data:**
- While compassionate use data can be informative, regulatory agencies typically prioritize data from well-designed, controlled clinical trials. Compassionate use data may complement such trial data but is not a substitute for it.
5. **Regulatory Agency Consultation:**
- It's advisable to consult with the relevant regulatory agency early in the drug development process to discuss the potential inclusion of compassionate use data in regulatory submissions. Agencies may provide guidance on how this data can be integrated appropriately.”
—ChatGPT
LC,
Why not consider a quiet period of your own?
…Unless you have another gem💎to share 😉
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173294572
BTW, I really liked your AI suggestion! Here is a bit more from ChatGPT:
”Investors can take several steps to avoid falling victim to stock disinformation:
**Verify Information from Reliable Sources:**
- Cross-reference information from multiple reputable sources. Avoid relying solely on one channel, especially social media or anonymous message boards.
**Understand the Company:**
- Conduct thorough research on the companies you're interested in. Understand their financials, business model, and industry. Reliable information often comes from official reports, financial statements, and reputable news sources.
**Use Caution with Social Media:**
- While social media platforms can provide information, they can also be a source of misinformation. Be critical of information shared on these platforms and verify it through reliable sources.
**Stay Informed about Market Regulations:**
- Understand the regulations governing financial markets. Authorities such as the SEC (U.S. Securities and Exchange Commission) take actions against those engaged in fraudulent activities.
**Consider Professional Advice:**
- Consult with financial advisors or professionals who can provide guidance based on expertise and experience.
**Participate in Reputable Forums:**
- If you engage in online discussions, consider reputable financial forums where discussions are moderated, and misinformation is less likely to thrive.
**Be Skeptical of Unsubstantiated Claims:**
- If an investment opportunity sounds too good to be true or is based on unverifiable claims, approach it with skepticism.
By staying informed and exercising caution, investors can better protect themselves from falling prey to stock disinformation.” —ChatGPT
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173294873
LC, This study found that autologous tumor lystate-loaded dendritic cell vaccination plus standard of care (SOC) was associated with improved survival in both newly diagnosed & recurrent glioblastoma compared with matched controls who received SOC treatment. https://t.co/Uso7Pkh1Kt
Thanks for the tip! Consulting AI is not a bad idea, so I thought I would try an AI app in reference to the NWBO conversation:
"Short and distort refers to a manipulative practice in financial markets. It involves individuals or entities taking short positions in a stock (betting that its price will fall) and then spreading false or misleading information about the company on stock message boards or other media platforms. The goal is to drive down the stock price, allowing those behind the scheme to profit from their short positions.
This unethical tactic can harm investors, create market volatility based on false information, and potentially damage the reputation of the targeted company. Securities regulators actively monitor and investigate such activities to maintain the integrity of financial markets.” —ChatGPT
Perhaps consider looking beyond social media to seek trustworthy full-context sources…
For a reliable appraisal of the trial, its design, and the significance of the resulting survival data, it is advisable to refer to the analyses of qualified physicians. One such analysis can be found on the JAMA Oncology website where the link to the independent peer review and supplementary materials are published.


https://finance.yahoo.com/news/northwest-biotherapeutics-announces-dr-linda-140000962.html
https://www.jci.org/articles/view/169314
https://www.uclahealth.org/news/fda-approval-brain-cancer-alzheimers
https://hotb.org/wp-content/uploads/2022/05/spore-article-for-donors-4.18.22.pdf
https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials



 [color=red][/color]
[color=red][/color]
iclight,
The fact that those patients were alive to have additional resections is significant. Patients who enjoy extended survival have the chance to try any number of additional medical treatments or wellness therapies.
https://brownneurosurgery.com/breakthrough-brain-cancer-vaccine/
https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847
LC,
Thanks for asking about the clinical data! There are safety and efficacy data beyond the P3. If you’re looking for high percentages, you might want to take a look at the PD1 combo study at UCLA!
https://clinicaltrials.gov/study/NCT04201873
Dr. Toms briefly mentions the interim survival data for the small but mighty pembrolizumab combo in this press conference. If you never saw this video before, I highly recommend watching it all the way through!
https://www.jci.org/articles/view/169314
Combo is King!
learningcurve2020,
You might consider posting less frequently and more thoughtfully. Your sophomoric approach is transparent and counterproductive to your objectives—you’re only demonstrating that the prospective value of NWBO is likely vast. The incessant random innuendo spaghetti that you throw at the wall signals value! If a sharp rise in the price were not on the horizon, you would not commit so many hours to publishing thoughtless mud on a message board throughout a holiday weekend. Thanks for the indicator!! 😉
Ironically however, you do sometimes post a real gem💎! The JCI publication set me on a path of exploration that I had previously overlooked. Now, I can appreciate more fully how the combo implications are potentially significant and game-changing. I am not an oncologist, but I find this technology fascinating as a platform…
Soon after LC posted the link to JCI, the Merck PR was published about the Daiichi Sankyo deal, and then I went back to reread the article again and I watched some videos of Dr. Liau’s discussions on the topic of combo therapy…
✅ DCVax-L is efficacious (see JAMA Oncology).
✅ DCVax-L + PD1 is perhaps far more efficacious than DCVax-L as a monotherapy (see interim survival data).
⭐️ DCVax-L technology + PD1 technology + CFS-1R technology or ADC technology could be yet another breakthrough … The P3 that was published in JAMA Oncology looks like only the tip of an enormous iceberg!
Perhaps without realizing it, I think LC may have actually dropped the mic after announcing the JCI publication!

Combo is King!
The field of cell-based therapy is evolving rapidly and the DCVax-L platform might be a key component to unlocking the efficacy of more than one class of drugs!
Thanks in part to all of LC’s posts on this message board, I have arrived at the view that each existing, emerging, or recently commercialized drug class in the field of oncology represents another potential opportunity to discover an efficacious combo with the DCVax-L platform! PD1 blockade is only one excellent example that bodes well for further successful combos.
https://www.cell.com/trends/cancer/fulltext/S2405-8033(23)00005-5
https://thehealthcaretechnologyreport.com/merck-and-daiichi-sankyo-enter-landmark-5-5-billion-partnership/
https://www.jci.org/articles/view/169314

The Nobel Prize was a harbinger of the novel DCVax-L cell-based platform technology!
https://www.livescience.com/16354-nobel-prize-medicine-2011-immune-research.html


Dr. Steinman’s work lives on at UCLA!


https://www.uclahealth.org/news/brain-cancer-discovery-clinical-trials

learningcurve2020, Our team just met with the @NorthwestBio team to learn about #DCVax and had the pleasure of speaking with longterm DCVax patients. An honor and a privilege. #endbraincancer #sno2023 pic.twitter.com/dw3X76DBru
You raise a valid point: The DCVax-L cell-based platform technology deserves to be celebrated and recognized for the landmark innovation that it is! I agree 100% … There are more important things for us to discuss. Did you catch Dr. Bosch’s presentation in Vancouver last week?



https://www.soc-neuro-onc.org/
learningcurve2020,
Beautiful story about Mr. Bienvenu who has not suffered a recurrence following surgery, chemo, and radiation… now six years from diagnosis! Thank you for sharing such a hopeful story!
More treatment options are on the horizon for nGBM, rGBM, and other cancers too!
A wise man once likened the accelerating speed of the evolution of cell-based immunotherapy technology to the speed of chocolates on Lucy’s conveyor belt! I quite agree!
Each existing, emerging, or recently commercialized drug class in the field of oncology represents another potential opportunity to discover an efficacious combo with the DCVax-L platform!
Combo is King!
https://www.fda.gov/drugs/news-events-human-drugs/50-years-progress-treating-patients-cancer

https://www.jci.org/articles/view/169314
https://clinicaltrials.gov/study/NCT04201873
Wow—thanks!
I have been so focused on the amazing interim data from the DCVax-L PD-1 combo study at UCLA that I have not paid much attention to all of the other emerging possibilities …
The JCI article was enlightening, and then the Daiichi Sankyo news was intriguing as well!
Around the time that the DCVax-L P3 JAMA peer review was published, I had never heard of CSF-1R inhibitors or ADCs.
The field of cell-based therapy is evolving rapidly and the DCVax-L platform might be a key component for unlocking the efficacy of more than one class of drugs.
Adding CSF-1R to PD-1 blockade and DCVax-L is a combo that UCLA plans to study with the support of the NIH, and Merck seems to be on the cutting edge with their recently announced landmark partnership with Daiichi Sankyo. I would love to see Dr. Liau and Northwest Bio partner with Merck—the pembrolizumab combo study interim results bode well for the possibility of other efficacious combos with DCVax-L as a platform technology.
https://www.cell.com/trends/cancer/fulltext/S2405-8033(23)00005-5
https://www.jci.org/articles/view/169314
Replying to myself to expand on the topic of combo therapies…
Just to clarify: I did not mean to suggest that pexidartinib (PLX3397) is an ADC. CSF-1R inhibitors and Antibody Drug Conjugates (ADCs) are distinct types of therapies.
I’m just beginning to research this topic and I am interested in learning more about the potential for combining DCVax-L with other immunotherapy agents beyond only PD-1 blockade. The Merck deal with Daiichi Sankyo has cast some light on fascinating new possibilities…
From my research:
“CSF-1R inhibitors target the Colony-Stimulating Factor 1 Receptor, often involved in regulating the immune system. On the other hand, ADCs are a type of targeted therapy that combines an antibody with a drug payload.”
https://www.daiichisankyo.com/products/
LC,
Have you considered trying a quiet period yourself?
… Unless you have more gems to share 💎😉
https://investorshub.advfn.com/boards/read_msg.aspx?message_id=173289965