Sunday, November 26, 2023 9:31:22 AM
The compassionate use cases generated some astonishing data, and those data may be included in the regulatory filings!


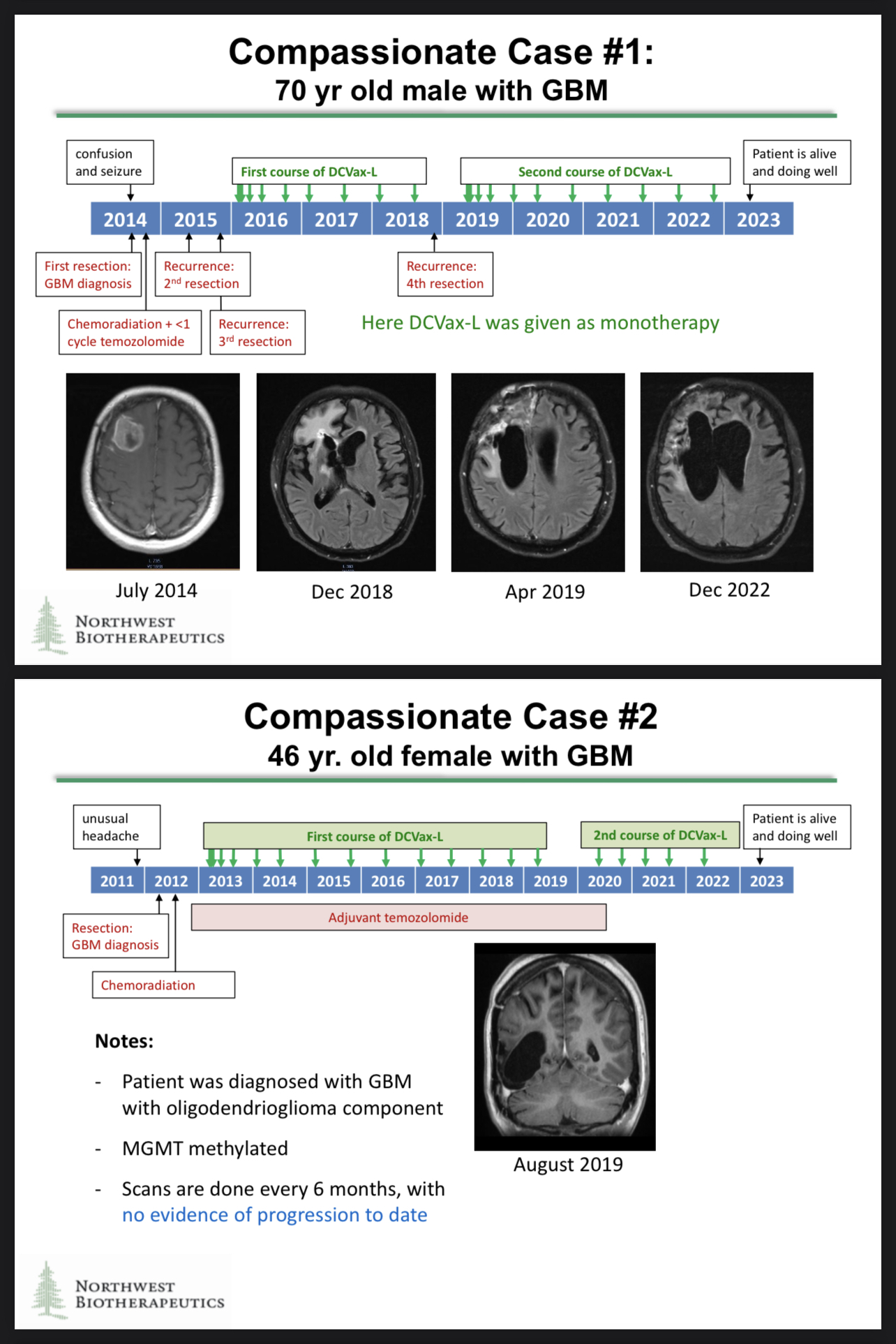
https://t.co/082urSTQ0s

https://www.braintumourresearch.org/media/news/news-item/2022/11/18/astonishing-results-for-brain-tumour-vaccine-trial
LC’s suggestion of using AI to supplement responses to postings on message boards was actually a good idea!
“Yes, compassionate use data, also known as expanded access or named patient programs, can be included in regulatory submissions for drug approval. Compassionate use refers to the use of an investigational drug outside of a clinical trial to treat a patient with a serious or life-threatening illness for whom no other treatment options are available.
Regulatory agencies, such as the U.S. Food and Drug Administration (FDA), recognize that compassionate use programs can provide valuable information about a drug's safety and potential efficacy. However, the inclusion of compassionate use data in regulatory submissions is subject to certain conditions:
1. **Data Quality and Relevance:**
- The compassionate use data must meet the standards of quality, relevance, and reliability expected in clinical trials. Robust data collection and reporting practices are crucial.
2. **Consistency with Regulatory Requirements:**
- The data collected under compassionate use should align with the regulatory requirements for the type of approval sought (e.g., New Drug Application, Biologics License Application).
3. **Ethical Considerations:**
- Ethical considerations surrounding patient safety and informed consent are essential. The compassionate use program should adhere to ethical standards, and patients should be well-informed about the risks and benefits.
4. **Corroboration with Controlled Clinical Trial Data:**
- While compassionate use data can be informative, regulatory agencies typically prioritize data from well-designed, controlled clinical trials. Compassionate use data may complement such trial data but is not a substitute for it.
5. **Regulatory Agency Consultation:**
- It's advisable to consult with the relevant regulatory agency early in the drug development process to discuss the potential inclusion of compassionate use data in regulatory submissions. Agencies may provide guidance on how this data can be integrated appropriately.”
—ChatGPT
Believe carefully. This is the greatest and most powerful lesson that I have learned since arriving on Earth. Examine what you believe about yourself most importantly, and then believe carefully as you interact with the world.
Recent NWBO News
- Biophma Announces Exclusive In License for Dendritic Cell Technology, Sending Shares Higher • AllPennyStocks.com • 06/17/2024 04:40:00 PM
- Form 8-K - Current report • Edgar (US Regulatory) • 06/04/2024 09:11:16 PM
- Form DEF 14A - Other definitive proxy statements • Edgar (US Regulatory) • 06/03/2024 09:22:55 PM
- Form PRE 14A - Other preliminary proxy statements • Edgar (US Regulatory) • 05/22/2024 08:13:36 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 05/10/2024 09:04:57 PM
- Form NT 10-K - Notification of inability to timely file Form 10-K 405, 10-K, 10-KSB 405, 10-KSB, 10-KT, or 10-KT405 • Edgar (US Regulatory) • 03/01/2024 10:04:38 PM
- Form 4 - Statement of changes in beneficial ownership of securities • Edgar (US Regulatory) • 12/02/2023 01:31:35 AM
- Form 8-K - Current report • Edgar (US Regulatory) • 11/16/2023 10:11:54 PM
- Epazz, Inc. (OTC Pink: EPAZ) ZenaDrone Demonstration to Defense Departments of UAE and Saudi Arabia • InvestorsHub NewsWire • 11/15/2023 12:19:31 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 11/09/2023 09:30:39 PM
- Epazz, Inc. (OTC Pink: EPAZ) US Navy Collaboration ZenaDrone 1000 • InvestorsHub NewsWire • 11/09/2023 01:00:34 PM
- Epazz, Inc. (OTC Pink: EPAZ) US Navy Collaboration ZenaDrone 1000 Extreme Weather Demo • InvestorsHub NewsWire • 11/07/2023 12:29:43 PM
- Form 10-Q - Quarterly report [Sections 13 or 15(d)] • Edgar (US Regulatory) • 08/09/2023 08:36:14 PM
FEATURED POET Wins "Best Optical AI Solution" in 2024 AI Breakthrough Awards Program • Jun 26, 2024 10:09 AM
HealthLynked Promotes Bill Crupi to Chief Operating Officer • HLYK • Jun 26, 2024 8:00 AM
Bantec's Howco Short Term Department of Defense Contract Wins Will Exceed $1,100,000 for the current Quarter • BANT • Jun 25, 2024 10:00 AM
ECGI Holdings Targets $9.7 Billion Equestrian Apparel Market with Allon Brand Launch • ECGI • Jun 25, 2024 8:36 AM
Avant Technologies Addresses Progress on AI Supercomputer-Driven Data Centers • AVAI • Jun 25, 2024 8:00 AM
Green Leaf Innovations, Inc. Expands International Presence with New Partnership in Dubai • GRLF • Jun 24, 2024 8:30 AM









