Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
NEWS -- FuelPositive Accelerating Toward Sustainable Solutions
WATERLOO, Ontario, Feb. 06, 2024 (GLOBE NEWSWIRE) -- FuelPositive Corporation, a leading Green Ammonia Company (TSX.V: NHHH) (OTCQB: NHHHF) (the “Company” or “FuelPositive”) provides updates on recent achievements, new initiatives, and completion of financing.
Government Spotlight:
FuelPositive's year got off to a great start with an impactful visit from the Federal Minister of Agriculture, the Honorable Lawrence MacAulay, and two local Members of Parliament, the Honorable Bardish Chagger and MP Tim Louis, at its Northland facility, which was a landmark and celebratory event for the young company. The Minister of Agriculture and the MPs engaged directly with FuelPositive’s team.
Minister MacAulay, an experienced potato farmer from Charlottetown, PEI, understands the challenges Canadian farmers face: the pressure from fluctuating fertilizer prices and supply, the effects of climate change, and the rising demand to sustain a growing population.
Ongoing dialogue and additional minister/government officials' visits are scheduled to explore further avenues of support for the company's rapid growth.
Jeanne Milne, FuelPositive’s Senior Government Relations Advisor, remarked, "Witnessing the FuelPositive team's pride in presenting our groundbreaking technology at our Research and Development Facility at 99 Northland was truly inspiring. Government officials recognized the pivotal role this technology can play in transforming the agricultural sector."
FP300 “On-Farm” Readiness:
FuelPositive remains on track to deliver its first commercial system in early Spring 2024. Due to additional safety verification steps, the updated delivery date is now April 15th.
The company announces the introduction of a Performance Guarantee on operating costs (OPEX) and production output for the trailblazing early adopters of FuelPositive technology.
The first FP300 system has officially been moved to FuelPositive's new 15,000-square-foot manufacturing facility for the Factory Acceptance Test (FAT). The FAT is the final approval step before delivery and on-farm installation.
FuelPositive has invited the Hieberts and other customer-farmers to visit its facilities throughout March 2024. The Hieberts will complete their Factory Acceptance Test and receive training on their initial FP300 system.
Other interested customers will have a firsthand look at FuelPositive’s proprietary technology as they make their final purchase commitments.
Nelson Leite, COO of FuelPositive, announced, "We've relocated the FP300 to our new manufacturing facility. In the next few weeks, we'll focus on finalizing commissioning, optimization, and safety checks for on-farm deployment in Manitoba. We’re looking forward to hosting some select customers to the facility throughout March and welcoming the Hieberts for their Factory Acceptance Testing and related training.”
Marketing and Sales:
FuelPositive is in the final stages of selecting Public and Media Relations firms, with the announcement expected by the end of February.
Ian Clifford, FuelPositive’s CEO, stated, "We are selecting the most strategic media and public relations suppliers with a targeted understanding of our technology and audiences. We will announce our decision shortly."
The company focuses its initial sales efforts in Manitoba, Canada, capitalizing on the green grid, numerous qualifying farmers, and strong government support. Ian Clifford emphasized, "With these advantages, many Manitoba farmers will directly benefit from our fertilizer and fuel solutions."
FuelPositive is also focusing on commercializing the FP1500 system in response to global inquiries from farms 10,000 acres and larger. This system produces 500 metric tonnes of Green Ammonia annually, five times more than the FP300, while maintaining a similar footprint and offering a more cost-effective rate per tonne.
People and Culture:
As part of its strategic expansion, the Company has recruited its first Director of Sales, who will join the team this month. In the last two months, FuelPositive welcomed a Controller, a Senior Purchaser, an Engineer of Record, and a Senior Electrical Engineer to the team. These key hires and promotions from within the organization will support the Company in achieving its ambitious goals.
Cheryl Allen, Director of Human Resources at FuelPositive, remarked, "Leading the crucial task of sourcing and recommending new team members has been an honour and pleasure for me. As we expand into a prominent green technology and manufacturing entity, I look forward to continuing my leadership role in supporting our growth journey."
Sector Recognition:
FuelPositive is proud to announce that it has been selected as an International Industry Innovation Leader and a “Living Lab” by NICCEE (the Global Nitrogen Innovation Center for Clean Energy and the Environment). “NICCEE … involves collaborators from eight countries: academia, NGOs, international organizations, government agencies, and private companies. These partnerships offer complementary expertise and present a diverse portfolio of opportunities to investigate the emerging challenges and possibilities of Green Ammonia technologies.”
FuelPositive's FP300, addressing nitrogen-related pollution, is referenced as a "living lab" for its innovative use of Green Ammonia. This sector recognition aligns with FuelPositive's ongoing efforts to reduce agricultural and fuel-switching nitrogen-related emissions.
Luna Clifford, FuelPositive’s Chief Impact Officer, stated, “NICCEE recognition highlights our innovative technology and commitment to responsible Green Ammonia use, showcasing FuelPositive's holistic approach to mitigating and eliminating all agricultural greenhouse gas emissions. The work that NICCEE is doing is vital to addressing one of the planet's most volatile greenhouse gas emissions. It is a critical and timely undertaking.”
Carbon Credits:
FuelPositive is developing a cutting-edge method to collect carbon credits in real-time while also focusing on the emission-free combustion of Green Ammonia.
This monetization method will be linked to real-time trading platforms, allowing FuelPositive to help offset its customer’s operational and capital costs. The company aims to help farmers profit from producing Green Ammonia on their farms faster.
Andre Mech, FuelPositive’s Advisor for Carbon Credits and Emissions Reduction, stated, "FuelPositive’s Green Ammonia production system is an excellent example of what carbon credit legislation aims to promote. With zero emissions across Scope 1, 2, and 3, a decentralized on-farm system from FuelPositive will maximize carbon credit value within any jurisdiction."
Finances:
FuelPositive has secured an additional $550,000 in the final tranche of its current financing, which is expected to close in the next several days. This brings the total raised in this placement to $1,900,000.
Despite prevailing global challenges in the public markets, FuelPositive remains resilient. This financing will give the company essential funding for final testing, intellectual property advancements, and growth plans.
Ian Clifford, FuelPositive’s CEO, stated, "Our transition to revenue is imminent. We've invested considerable time to reach this point and are deeply grateful for the patience and support of our stakeholders. As we defy skeptics, break new ground, and establish a whole new sector, we recognize the unprecedented position we hold today. Our journey owes much to the trust and confidence of both new and long-standing investors and shareholders. We’re looking forward to our upcoming successes and sharing the rewards with our shareholders."
About FuelPositive Corporation
FuelPositive Corporation is at the forefront of Canadian technology and is committed to delivering sustainable, environmentally responsible Green Ammonia solutions. Our innovative approach includes on-farm/on-site, containerized Green Ammonia production systems, effectively eliminating carbon emissions.
Our commercial Green Ammonia systems are versatile, serving multiple applications, including fertilizer for farming, fuel for grain drying, internal combustion engines, and hydrogen storage for fuel cells and other sectors.
Our main customer base consists of farmers, who currently utilize 80% of the global ammonia production. We address their nitrogen fertilizer needs while advocating for environmental change.
Our Canadian company leverages Canada's expertise in technology and environmental stewardship to empower communities worldwide against food insecurity. We're shaping a more resilient future for generations through collaboration and innovation.
FuelPositive: Fuel for a Mindful World.
See Sales details here: https://fuelpositive.com/sales/.
FuelPositive Corporation is based in Waterloo (Canada) and trades on the TSX Venture Exchange under the symbol NHHH and in the USA on the OTCQB under the symbol NHHHF.
For further information, please contact:
Ian Clifford
Chief Executive Officer and Board Chair
mailto://investors@fuelpositive.com
Tel: 416.535.8395
https://www.fuelpositive.com
Investor Relations United States & International:
RB Milestone Group (RBMG)
mailto://fuelpositive@rbmilestone.com
Investor Relations Canada:
Transcend Capital Inc.
mailto://et@transcendcapitalinc.com
Cautionary Statement
Trading in the securities of the Company should be considered highly speculative. No stock exchange, securities commission or other regulatory authority has approved or disapproved the information contained herein. Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accept responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains certain “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) that are based on expectations, estimates and projections as of the date of this news release. The information in this release about future plans and objectives of the Company, including with respect to further testing and implementation of the FP300 system, are forward-looking statements.
These forward-looking statements are based on assumptions and estimates of management of the Company at the time they were made and involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable by the Company as of the time of such statements, are inherently subject to significant business, economic and competitive uncertainties and contingencies. These estimates and assumptions may prove to be incorrect.
Many of these uncertainties and contingencies can directly or indirectly affect and could cause, actual results to differ materially from those expressed or implied in any forward-looking statements. There can be no assurance that forward-looking information will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements.
Forward-looking information is provided for the purpose of providing information about management’s expectations and plans relating to the future. The Company disclaims any intention or obligation to update or revise any forward-looking information or to explain any material difference between subsequent actual events and such forward-looking information, except to the extent required by applicable law.
NEWS -- NightHawk Biosciences Completes Name Change to Scorpius Holdings; Trading to Commence Under “SCPX” Effective at the Market Open Today
DURHAM, N.C., Feb. 06, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences / Scorpius Holdings, Inc. (NYSE American: NHWK; SCPX), an integrated contract development and manufacturing organization (CDMO), today announced that it had completed its name change from NightHawk Biosciences, Inc. to Scorpius Holdings, Inc., to better reflect the Company’s successful shift into a pure-play, large molecule biomanufacturing CDMO. In connection with the name change, the Company’s ticker will change to “SCPX” effective today, February 6, 2024. The name and symbol changes do not affect the Company's share structure or the rights of the Company's shareholders, and no further action will be required by existing shareholders.
Jeff Wolf, CEO of Scorpius, stated, "Changing our name to Scorpius Holdings reflects our evolution into a revenue-generating biomanufacturing company and expansion of our CDMO services. The biopharmaceutical sector is currently grappling with a severe shortage of clinical-scale biologic manufacturing capacity, a situation exacerbated by the rising demand for large molecule CDMO services. Our cutting-edge San Antonio, Texas facility is strategically positioned to fill this critical void. The feedback from our key biotech and pharmaceutical customers has been overwhelmingly positive, as reflected in our enhanced sales pipeline. We are eager to advance these initiatives under the Scorpius banner."
Scorpius Holdings, Inc.
Scorpius Holdings Inc. is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array of analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the-art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit https://www.scorpiusbiologics.com.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases, forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as the Company’s successful shift into a pure-play, large molecule biomanufacturing CDMO; the rising demand for large molecule CDMO services; the Company’s cutting-edge San Antonio, Texas facility being strategically positioned to fill the critical shortage of clinical-scale biologic manufacturing capacity in the biopharmaceutical sector; the Company’s enhanced sales pipeline; and advancing the Company’s initiatives under the Scorpius banner. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to expand its large molecule biomanufacturing CDMO services and continue to grow revenue; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to leverage fixed costs and achieve long-term profitability; the Company’s ability to obtain regulatory approvals or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://ir@scorpiusbiologics.com
NEWS -- Tokens.com Reports Financial Results for Fiscal Year 2023
TORONTO, January 31, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, reports its financial results for the fiscal year ended September 30, 2023 ("FY2023"). All dollar figures are in United States dollars ("USD"), unless otherwise stated.
Strategic Review Update:
On November 8th, 2023, the Company announced that it had commenced a strategic review. The Company is also exploring acquisitions, sales of its domain names and the sale of its operating businesses and digital assets. As part of this strategic process to enhance shareholder value, on January 29, 2024, the Company entered into a definitive agreement to sell the assets of Metaverse Group and Hulk Labs to StoryFire Inc. ("StoryFire"), which is primarily a transfer of its workforce and contractors. All capital within Metaverse Group will remain at Tokens.com.
Tokens.com will receive a consideration of $4 million for the sale that consists of a 15.3% ownership of StoryFire and $500,000 of its native Blaze cryptocurrency token. The transaction is expected to close on March 1, 2024.
Metaverse Group and Hulk Labs revenue was not forecasted to exceed operating costs and reach profitability for several years. In addition, the funding required by these businesses would require Tokens.com to carry significant cash flow losses. To finance these operations, the Company would need to sell some or all of its cryptocurrency inventory or look to raise dilutive new equity capital. After careful review of all alternatives, management and the board made the decision to de-risk Tokens.com by selling the assets of Metaverse Group and Hulk Labs to a buyer that has a strategic fit and capital to bring the businesses to profitability faster. Tokens.com retains the upside in the combined businesses through our 15.3% equity participation. Tokens.com also is in a position to retain its cryptocurrencies without the requirement to fund the other businesses. Management believes the sale of the businesses results in a win-win scenario and has eliminated an estimated $1.5M of overhead annually. Removing the overhead associated with these assets gives the company additional flexibility in pursuing another acquisition or as an acquisition target as a leaner structure.
Subsequent to the closing of the StoryFire transaction on March 1st, 2024, Tokens.com’s key assets will be its cryptocurrency inventory, cash, a 15.3% ownership in StoryFire, as well as a portfolio of domain names. Operating overhead will be significantly lower, making operations more efficient.
2023 Highlights:

NEWS -- Tokens.com Reports Financial Results for Fiscal Year 2023
TORONTO, January 31, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, reports its financial results for the fiscal year ended September 30, 2023 ("FY2023"). All dollar figures are in United States dollars ("USD"), unless otherwise stated.
Strategic Review Update:
On November 8th, 2023, the Company announced that it had commenced a strategic review. The Company is also exploring acquisitions, sales of its domain names and the sale of its operating businesses and digital assets. As part of this strategic process to enhance shareholder value, on January 29, 2024, the Company entered into a definitive agreement to sell the assets of Metaverse Group and Hulk Labs to StoryFire Inc. ("StoryFire"), which is primarily a transfer of its workforce and contractors. All capital within Metaverse Group will remain at Tokens.com.
Tokens.com will receive a consideration of $4 million for the sale that consists of a 15.3% ownership of StoryFire and $500,000 of its native Blaze cryptocurrency token. The transaction is expected to close on March 1, 2024.
Metaverse Group and Hulk Labs revenue was not forecasted to exceed operating costs and reach profitability for several years. In addition, the funding required by these businesses would require Tokens.com to carry significant cash flow losses. To finance these operations, the Company would need to sell some or all of its cryptocurrency inventory or look to raise dilutive new equity capital. After careful review of all alternatives, management and the board made the decision to de-risk Tokens.com by selling the assets of Metaverse Group and Hulk Labs to a buyer that has a strategic fit and capital to bring the businesses to profitability faster. Tokens.com retains the upside in the combined businesses through our 15.3% equity participation. Tokens.com also is in a position to retain its cryptocurrencies without the requirement to fund the other businesses. Management believes the sale of the businesses results in a win-win scenario and has eliminated an estimated $1.5M of overhead annually. Removing the overhead associated with these assets gives the company additional flexibility in pursuing another acquisition or as an acquisition target as a leaner structure.
Subsequent to the closing of the StoryFire transaction on March 1st, 2024, Tokens.com’s key assets will be its cryptocurrency inventory, cash, a 15.3% ownership in StoryFire, as well as a portfolio of domain names. Operating overhead will be significantly lower, making operations more efficient.
2023 Highlights:

NEWS -- Tokens.com Announces Sale of Metaverse Group and Hulk Labs to StoryFire
TORONTO, January 30, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, is pleased to announce that it has entered into a definitive agreement to sell the assets of Metaverse Group and Hulk Labs to StoryFire, Inc. ("StoryFire"). The transaction is scheduled to close on March 1st, 2024.
Consideration to Tokens.com for the disposition consists of a 15.3% equity interest in StoryFire, valued at US$3.5 million, plus US$500,000 in Blaze tokens, the in-app currency native to StoryFire. In addition, Tokens.com will be granted a board seat on the StoryFire board. Following the completion of the transaction, Tokens.com will no longer have an active business in metaverse or web3 gaming, except through its ownership in StoryFire.
StoryFire is a private online social entertainment and gaming platform that empowers users to create and engage with immersive narratives. With a rapidly growing user base of 2.5 million, StoryFire offers a vibrant and creative community for storytellers and enthusiasts. StoryFire users create social posts akin to Twitter, gain a following and earn an in-app currency known as Blaze. StoryFire is a privately owned entity.
"This transaction provides Tokens.com shareholders with several benefits, including the continued buildout of Metaverse Group by a strategic acquirer. StoryFire is a successful social media brand with over 2 million users and the capital to expand the growth of these businesses," said Andrew Kiguel, CEO of Tokens.com. "Tokens.com retains a significant amount of upside through its equity ownership in StoryFire, while no longer having to cover the overhead required to manage these businesses. Management expects this and other cost cutting measures associated with the transaction to result in savings to Tokens.com of approximately CAD$1.5M annually. The resulting Tokens.com business will own 15.3% of StoryFire, approximately CAD$3.7M in cash, approximately CAD$15.0M in cryptocurrency tokens, as well as a valuable portfolio of domain names that are currently for sale." added Kiguel.
The strategic vision for combining these assets is to leverage Metaverse Group’s capabilities to build content for use by StoryFire's 2.5M users, and expand on the metaverse experiences that Metaverse Group hosts, such as the Metaverse Fashion Week. By virtue of having a larger captive audience and distribution network that is growing by over 100% year over year, Metaverse Group’s 3D design and build capabilities will be able to attract a broader base of clients, and create immersive experiences for StoryFire creators and enterprises. Helix, the underlying metaverse virtual commerce platform provides a powerful engine for StoryFire creators to sell goods and services in the metaverse.
In addition, developers from Hulk Labs and certain IP, including Astraeus Defence, will continue to be developed and shared with StoryFire’s audience. This allows for immediate uptake of the games once developed. The gaming audience of StoryFire creators, as well as streamers, provide a significant pipeline to onboard games, and valuable data for the Yeti.gg platform.
"We are excited to integrate Metaverse Group and Hulk Labs into the StoryFire strategy. We welcome Tokens.com as a key shareholder and contributor to building our business. By leveraging the content creation capabilities, we expect to be able to quickly grow our user base", said Sam Hilder, CEO of StoryFire.
The transaction with StoryFire has been approved by the board of directors and the independent committee of Tokens.com, as well as the board of StoryFire.
In addition, Tokens.com has retained a third party broker to explore the sale of the Tokens.com domain name, initially being listed with an ask of US$8M. Interested parties should contact Tokens.com management at contact@tokens.com.
About Tokens.com
Tokens.com is a web3 technology holding company that owns an inventory of cryptocurrency and a collection of crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
About StoryFire
The StoryFire platform, powered by the BLAZE token, will revolutionize the metaverse paradigm by combining SocialFi, GameFi, and DeFi into one unified experience.
StoryFire simplifies the connections between gaming, social media, and creating three independent platforms within the StoryFire ecosystem covering our three key pillars.
Learn more about StoryFire:
Website: https://www.storyfire.com
X (Twitter): https://www.x.com/storyfireapp
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240130478353/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- Tokens.com Announces Sale of Metaverse Group and Hulk Labs to StoryFire
TORONTO, January 30, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, is pleased to announce that it has entered into a definitive agreement to sell the assets of Metaverse Group and Hulk Labs to StoryFire, Inc. ("StoryFire"). The transaction is scheduled to close on March 1st, 2024.
Consideration to Tokens.com for the disposition consists of a 15.3% equity interest in StoryFire, valued at US$3.5 million, plus US$500,000 in Blaze tokens, the in-app currency native to StoryFire. In addition, Tokens.com will be granted a board seat on the StoryFire board. Following the completion of the transaction, Tokens.com will no longer have an active business in metaverse or web3 gaming, except through its ownership in StoryFire.
StoryFire is a private online social entertainment and gaming platform that empowers users to create and engage with immersive narratives. With a rapidly growing user base of 2.5 million, StoryFire offers a vibrant and creative community for storytellers and enthusiasts. StoryFire users create social posts akin to Twitter, gain a following and earn an in-app currency known as Blaze. StoryFire is a privately owned entity.
"This transaction provides Tokens.com shareholders with several benefits, including the continued buildout of Metaverse Group by a strategic acquirer. StoryFire is a successful social media brand with over 2 million users and the capital to expand the growth of these businesses," said Andrew Kiguel, CEO of Tokens.com. "Tokens.com retains a significant amount of upside through its equity ownership in StoryFire, while no longer having to cover the overhead required to manage these businesses. Management expects this and other cost cutting measures associated with the transaction to result in savings to Tokens.com of approximately CAD$1.5M annually. The resulting Tokens.com business will own 15.3% of StoryFire, approximately CAD$3.7M in cash, approximately CAD$15.0M in cryptocurrency tokens, as well as a valuable portfolio of domain names that are currently for sale." added Kiguel.
The strategic vision for combining these assets is to leverage Metaverse Group’s capabilities to build content for use by StoryFire's 2.5M users, and expand on the metaverse experiences that Metaverse Group hosts, such as the Metaverse Fashion Week. By virtue of having a larger captive audience and distribution network that is growing by over 100% year over year, Metaverse Group’s 3D design and build capabilities will be able to attract a broader base of clients, and create immersive experiences for StoryFire creators and enterprises. Helix, the underlying metaverse virtual commerce platform provides a powerful engine for StoryFire creators to sell goods and services in the metaverse.
In addition, developers from Hulk Labs and certain IP, including Astraeus Defence, will continue to be developed and shared with StoryFire’s audience. This allows for immediate uptake of the games once developed. The gaming audience of StoryFire creators, as well as streamers, provide a significant pipeline to onboard games, and valuable data for the Yeti.gg platform.
"We are excited to integrate Metaverse Group and Hulk Labs into the StoryFire strategy. We welcome Tokens.com as a key shareholder and contributor to building our business. By leveraging the content creation capabilities, we expect to be able to quickly grow our user base", said Sam Hilder, CEO of StoryFire.
The transaction with StoryFire has been approved by the board of directors and the independent committee of Tokens.com, as well as the board of StoryFire.
In addition, Tokens.com has retained a third party broker to explore the sale of the Tokens.com domain name, initially being listed with an ask of US$8M. Interested parties should contact Tokens.com management at contact@tokens.com.
About Tokens.com
Tokens.com is a web3 technology holding company that owns an inventory of cryptocurrency and a collection of crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
About StoryFire
The StoryFire platform, powered by the BLAZE token, will revolutionize the metaverse paradigm by combining SocialFi, GameFi, and DeFi into one unified experience.
StoryFire simplifies the connections between gaming, social media, and creating three independent platforms within the StoryFire ecosystem covering our three key pillars.
Learn more about StoryFire:
Website: https://www.storyfire.com
X (Twitter): https://www.x.com/storyfireapp
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240130478353/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- NightHawk’s Scorpius BioManufacturing Subsidiary Announces the Promotion of Joe Payne to President and Chief Operating Officer
DURHAM, N.C., Feb. 01, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced the promotion of Joe Payne to President and Chief Operating Officer (COO) of its Scorpius BioManufacturing subsidiary.
Payne joined Scorpius in 2023 as VP of Quality and Regulatory Affairs. With a biopharma and pharmaceutical career spanning over 20 years, Payne’s recent experience includes leadership roles at CDMOs Tergus Pharma and Alcami Corporation. He has participated in 40+ regulatory inspections and has extensive knowledge of global regulatory agency requirements. In his role as President and COO, Payne will oversee quality, manufacturing, process sciences, and commercial operations.
Jeff Wolf, CEO of NightHawk Biosciences, commented, “Joe has seen nearly 100 different pharmaceutical facilities and has interacted with thousands of industry professionals at all levels. His expertise and experience will greatly benefit Scorpius during its next phase of growth. Joe has deep experience in developing and implementing quality production processes within large and small CDMO’s. Joe’s appointment reflects our focus on quality biomanufacturing throughout our organization. I’m confident Joe will ensure our clients have the smoothest path from the lab to the clinic to commercialization.”
Joe Payne further noted, “When I joined Scorpius in 2023, I was focused on building a robust quality operation. I was impressed by the team’s commitment, from the leadership all the way to our floor-level operators. I’m truly grateful for the faith that Jeff, the board, and the entire organization have placed in me, and I look forward to working with current and future clients to bring their biologic innovations to patients as efficiently and safely as possible.”
Mr. Wolf, Mr. Payne and the Scorpius commercial team will be at DCAT Week in New York City on March 18-21 to meet with clients and prospects about analytical testing, process development, and manufacturing services.
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic programs to the clinic and beyond. Scorpius offers a broad array of analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the-art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such Mr. Payne’s contribution to Scorpius including his expertise and experience greatly benefitting Scorpius during its next phase of growth and Mr. Payne ensuring clients have the smoothest path from the lab to the clinic to commercialization.. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to derive the contributions to Scorpius anticipated from Mr. Payne, the ability to generate future revenue from manufacturing contracts, the Company’s ability to continue its strategy and to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace, the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- NightHawk Receives Proceeds of $3.25 Million in Connection with Sale of Non-Core Intellectual Property and Issuance of a Low Interest Bearing Note
DURHAM, N.C., Jan. 30, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced it has sold additional none-core IP. Additionally, the Company received proceeds of $2.25 million from a note issued pursuant to the terms of the previously announced divestiture of non-core assets ( the “Note”). The Note bears interest at an annual rate of 1% and, subject to stockholder approval, would be convertible into shares of NightHawk’s common stock at $0.39281 per share, an 8% premium to the closing price of the Company’s common stock on January 29, 2024.
Jeff Wolf, CEO of NightHawk Biosciences, commented, “Receipt of these funds is particularly timely as it strengthens our balance sheet and provides us additional working capital to further accelerate our growth. The sale of this intellectual property is consistent with our strategy to divest non-core assets in order to focus on our Scorpius BioManufacturing subsidiary, which is gaining significant traction in the market.”
Additional details related to the sale of the IP and the Note are included in the Company’s Form 8-K, which has been filed with the Securities and Exchange Commission.
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the-art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as further accelerating the Company’s growth and Scorpius BioManufacturing gaining significant traction in the market . Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate future revenue from manufacturing contracts, the Company’s ability to continue its strategy and to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace, the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- NightHawk’s Scorpius BioManufacturing Subsidiary Awarded Multimillion- Dollar Contract with a Publicly Traded Biotech Company to Support its Phase 2 Clinical Program
Project to benefit from Scorpius’ comprehensive services in process development, bioanalysis, and cGMP manufacturing
DURHAM, N.C., Jan. 25, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced its Scorpius BioManufacturing subsidiary has been awarded a multimillion-dollar contract to provide process development, analytical, and cGMP manufacturing services for a publicly traded biotech company’s Phase 2 clinical program.
Jeff Wolf, CEO of NightHawk Biosciences, commented, “We are proud to have been selected to help advance this potentially lifesaving therapy through the clinic. Scorpius has multiple clients with Orphan Drug Designations from the FDA, and our San Antonio facility is especially well-suited to advance these important programs. This new client, needing biologic manufacturing for a Phase 2 clinical program under cGMP guidelines, will benefit from Scorpius' comprehensive services in process development, bioanalysis, and manufacturing. The client, transitioning from a large CDMO, chose Scorpius to assist in this Phase 2 program based on our adaptability, dedicated service, and commitment to meeting project timelines. The scope of work, which includes producing a monoclonal antibody (mAb) in Scorpius' mammalian facility, is expected to be completed in 2024. We are extremely pleased with the success and traction Scorpius gained in 2023, our first full year in operation, and we are excited to see this momentum continue as we witness rising demand for our mammalian and microbial biomanufacturing services.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the-art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as advancing the potentially lifesaving therapy through the clinic, the San Antonio facility being especially well-suited to advance programs with Orphan Drug Designations from the FDA, the scope of work being completed in 2024 and seeing the momentum continue as demand rises for Scorpius’ mammalian and microbial biomanufacturing services. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to complete the work as planned, the Company’s ability to generate future revenue from manufacturing contracts, the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability,; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace, the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Tokens.com Provides Update on 2023 Annual Filings
TORONTO, January 19, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, today provided an updated on the status of a management cease trade order (the "MCTO") which was granted by the Ontario Securities Commission on January 2, 2024.
The Company reports that the audit is progressing and that the Company and its auditors continue to work diligently toward completing the 2023 Annual Filings as soon as possible. The Company is not aware of any specific accounting or audit concerns at this time. The Company continues to expect that it will be in a position to file the 2023 Annual Filings on or before January 31, 2024. The Company will issue a news release announcing completion of such filings at such time. Further updates on timing will be provided by the Company as necessary.
As previously announced, the Company applied for the MCTO due to a delay in the filing of the Company's annual financial statements and accompanying management’s discussion and analysis for the for the twelve-month period ended September 30, 2023 (the "2023 Annual Filings"), which were due on December 29, 2023.
The MCTO restricts the Chief Executive Officer and Chief Financial Officer from all trading in securities of the Company until such time as the 2023 Annual Filings have been filed by the Company and the MCTO has been lifted. The MCTO does not affect the ability of other shareholders of the Company to trade in securities of the Company.
Until the 2023 Annual Filings are filed, the Company intends to comply with the provisions of the alternative information guidelines as set out in National Policy 12-203 – Management Cease Trade Orders ("NP 12-203") for as long as it remains in default, including the issuance of bi-weekly default status reports in the form of a news release. The Company confirms that, since its prior news release dated December 15, 2023: (a) there has been no material change to the information set out in that press release that has not been generally disclosed; (b) there has not been any other specified default by the Company under NP 12-203; (c) the Company is not subject to any insolvency proceedings; and (d) there is no material information concerning the affairs of the Company that has not been generally disclosed.
About Tokens.com
Tokens.com is a web3 technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240119898640/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- Tokens.com Provides Update on 2023 Annual Filings
TORONTO, January 19, 2024--(BUSINESS WIRE)--Tokens.com Corp. (Cboe Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, today provided an updated on the status of a management cease trade order (the "MCTO") which was granted by the Ontario Securities Commission on January 2, 2024.
The Company reports that the audit is progressing and that the Company and its auditors continue to work diligently toward completing the 2023 Annual Filings as soon as possible. The Company is not aware of any specific accounting or audit concerns at this time. The Company continues to expect that it will be in a position to file the 2023 Annual Filings on or before January 31, 2024. The Company will issue a news release announcing completion of such filings at such time. Further updates on timing will be provided by the Company as necessary.
As previously announced, the Company applied for the MCTO due to a delay in the filing of the Company's annual financial statements and accompanying management’s discussion and analysis for the for the twelve-month period ended September 30, 2023 (the "2023 Annual Filings"), which were due on December 29, 2023.
The MCTO restricts the Chief Executive Officer and Chief Financial Officer from all trading in securities of the Company until such time as the 2023 Annual Filings have been filed by the Company and the MCTO has been lifted. The MCTO does not affect the ability of other shareholders of the Company to trade in securities of the Company.
Until the 2023 Annual Filings are filed, the Company intends to comply with the provisions of the alternative information guidelines as set out in National Policy 12-203 – Management Cease Trade Orders ("NP 12-203") for as long as it remains in default, including the issuance of bi-weekly default status reports in the form of a news release. The Company confirms that, since its prior news release dated December 15, 2023: (a) there has been no material change to the information set out in that press release that has not been generally disclosed; (b) there has not been any other specified default by the Company under NP 12-203; (c) the Company is not subject to any insolvency proceedings; and (d) there is no material information concerning the affairs of the Company that has not been generally disclosed.
About Tokens.com
Tokens.com is a web3 technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240119898640/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- NightHawk’s Scorpius BioManufacturing Subsidiary Commences Work on Preclinical Program for New Client

NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced its Scorpius BioManufacturing subsidiary has been awarded a project to support a preclinical program for a U.S. based biotech company. The initial project value exceeds one million dollars, with the potential to exceed several million dollars over the next two years.
Jeff Wolf, CEO of NightHawk, commented, “We are pleased to have been awarded this competitive bid due to the versatility of our biologics facility, manufacturing readiness, and commitment to quality. This award reflects our growing reputation in the industry, as well as our track record advancing early stage clinical programs. A key element of this project is Scorpius’ specialized analytical and process development laboratories where the client benefits from our expertise in biologics and, in particular, microbial fermentation, which is garnering significant attention in the industry. Although the work outlined in this contract is anticipated to be completed in 2024, our intent is to expand the scope of the project as they prepare for phase 1 clinical trials and beyond.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the-art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as the potential to exceed several million dollars over the life of the project, completing the work outlined in the contract by 2024 and building a long-term relationship with the client in order to expand the scope of the project as they prepare for phase 1 clinical trials and beyond. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to complete the work outlined in 2024 and to expand the scope of the project, the Company’s ability to generate future revenue from manufacturing contracts , the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Tokens.com Provides Crypto Inventory Update
TORONTO,, January 15, 2024--(BUSINESS WIRE)--Tokens.com Corp. (CBOE Exchange Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, is pleased to share an update of its cryptocurrency holdings.
Crypto currency values have seen a steady increase in value since January 1, 2024. The table below shows our key crypto token holdings and corresponding value as of January 15, 2024 based on 1 Eth $2543.75, 1 Dot $7.59, 1 SOL $95.32.
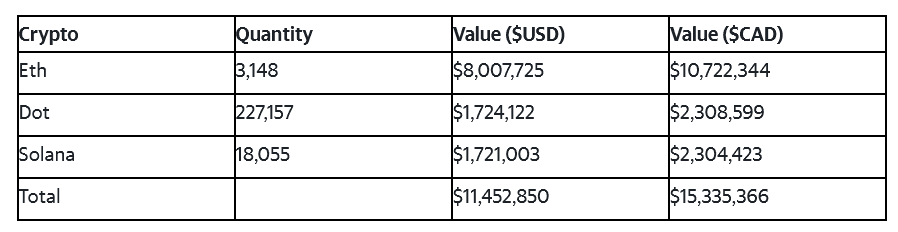
NEWS -- Tokens.com Provides Crypto Inventory Update
TORONTO,, January 15, 2024--(BUSINESS WIRE)--Tokens.com Corp. (CBOE Exchange Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a web3 technology company that owns an inventory of cryptocurrencies, is pleased to share an update of its cryptocurrency holdings.
Crypto currency values have seen a steady increase in value since January 1, 2024. The table below shows our key crypto token holdings and corresponding value as of January 15, 2024 based on 1 Eth $2543.75, 1 Dot $7.59, 1 SOL $95.32.

RG6501 (OpRegen®) Phase 1/2a Results Will Be Featured at 2024 Angiogenesis Exudation and Degeneration Meeting in Presentation by Allen Ho, MD, FACS, FASRS
CARLSBAD, Calif., January 16, 2024--(BUSINESS WIRE)--Lineage Cell Therapeutics, Inc. (NYSE American and TASE: LCTX), a clinical-stage biotechnology company developing allogeneic cell therapies for unmet medical needs, today announced that results showing retinal structure improvements with RG6501(OpRegen) from a Phase 1/2a clinical study (ClinicalTrials.gov Identifier: NCT02286089) in geographic atrophy (GA) secondary to advanced age-related macular degeneration (AMD), will be presented at the 2024 Angiogenesis, Exudation, and Degeneration Meeting. The virtual meeting will be held February 3, 2024, and is sponsored by the University of Miami Health System Bascom Palmer Eye Institute. The presentation, "Retinal Structure Improvements with OpRegen RPE Cell Therapy in a Phase I/IIa Study in Geographic Atrophy," will be presented by Allen Ho, MD, FACS, FASRS, Co-Director, Wills Eye Retina Service and Director, Retina Research, Wills Eye Hospital on behalf of Roche and Genentech, a member of the Roche Group.
About OpRegen
RG6501 (OpRegen) is a suspension of human allogeneic retinal pigment epithelial (RPE) cells currently in development for the treatment of GA secondary to AMD. OpRegen subretinal delivery has the potential to counteract RPE cell loss in areas of GA lesions by supporting retinal cell health and improving retinal structure and function. It is being developed under an exclusive worldwide collaboration between Lineage, and Roche and Genentech, a member of the Roche Group, and is currently being evaluated in a Phase 2a clinical study in patients with GA secondary to AMD (ClinicalTrials.gov Identifier: NCT05626114).
About Angiogenesis
The 21st Annual Angiogenesis meeting, entitled Angiogenesis, Exudation, and Degeneration 2024, will be held virtually on February 3, 2024. The program will feature an exceptional group of basic scientists, clinicians, and healthcare experts, all focused on understanding and treating neovascular, exudative, and degenerative diseases of the eye. The program will highlight the revolutionary pharmacotherapies now in development and clinical practice for the management of neovascular AMD, macular edema, diabetic retinopathy, retinopathy of prematurity, and inherited retinal degenerations with a special emphasis on the present and future financial impact of these drugs on clinical practices and Medicare. Angiogenesis 2024 follows the tradition of excellence established by Bascom Palmer’s widely acclaimed Angiogenesis programs between 2004 and 2023. Designed for retina specialists, general ophthalmologists and researchers, the current program will review the latest in imaging, translational research, and clinical trials with an emphasis on how these results will impact clinical ophthalmology. For more information visit: https://umiamihealth.org/bascom-palmer-eye-institute/healthcare-professionals/continuing-medical-education/angiogenesis.
About the OpRegen Phase 1/2a Study
The Phase 1/2a study is an open-label, single-arm, multi-center, dose-escalation trial evaluating a single administration of OpRegen delivered subretinally in patients with bilateral GA secondary to AMD. Twenty-four patients were enrolled into 4 cohorts. The first 3 cohorts enrolled only legally blind patients with a best corrected visual acuity (BCVA) of 20/200 or worse. The fourth cohort enrolled 12 patients with impaired vision (BCVA from 20/65 to 20/250 with smaller mean areas of GA). Cohort 4 also included patients treated with a new "thaw-and-inject" formulation of OpRegen, which can be shipped directly to sites and used immediately upon thawing, removing the complications and logistics of having to use a dose preparation facility. The primary objective of the study was to evaluate the safety and tolerability of OpRegen as assessed by the incidence and frequency of treatment-emergent adverse events. Secondary objectives include evaluating the preliminary activity of OpRegen treatment by assessing the changes in ophthalmological parameters measured by various methods of primary clinical relevance.
About Geographic Atrophy
Geographic atrophy (GA) is an advanced form of age-related macular degeneration (AMD) characterized by severe loss of visual function. GA is a leading cause of adult blindness in the developed world, affecting at least 5 million people globally. There are two forms of advanced AMD: neovascular AMD and GA. GA and neovascular AMD can occur simultaneously in the same eye, and patients treated for neovascular AMD may still go on to develop GA. GA typically affects both eyes.
About Lineage Cell Therapeutics, Inc.
Lineage Cell Therapeutics is a clinical-stage biotechnology company developing novel cell therapies for unmet medical needs. Lineage’s programs are based on its robust proprietary cell-based therapy platform and associated in-house development and manufacturing capabilities. With this platform Lineage develops and manufactures specialized, terminally differentiated human cells from its pluripotent and progenitor cell starting materials. These differentiated cells are developed to either replace or support cells that are dysfunctional or absent due to degenerative disease or traumatic injury or administered as a means of helping the body mount an effective immune response to cancer. Lineage’s clinical and preclinical programs are in markets with billion dollar opportunities and include five allogeneic ("off-the-shelf") product candidates: (i) OpRegen, a retinal pigment epithelial cell therapy in Phase 2a development for the treatment of geographic atrophy secondary to age-related macular degeneration, is being developed under a worldwide collaboration with Roche and Genentech, a member of the Roche Group; (ii) OPC1, an oligodendrocyte progenitor cell therapy in Phase 1/2a development for the treatment of acute spinal cord injuries; (iii) VAC2, a dendritic cell therapy produced from Lineage’s VAC technology platform for immuno-oncology and infectious disease, currently in Phase 1 clinical development for the treatment of non-small cell lung cancer; (iv) ANP1, an auditory neuronal progenitor cell therapy for the potential treatment of auditory neuropathy; and (v) PNC1, a photoreceptor neural cell therapy for the potential treatment of vision loss due to photoreceptor dysfunction or damage. For more information, please visit https://www.lineagecell.com or follow the company on Twitter @LineageCell.
Forward-Looking Statements
Lineage cautions you that all statements, other than statements of historical facts, contained in this press release, are forward-looking statements. Forward-looking statements, in some cases, can be identified by terms such as "believe," "aim," "may," "will," "estimate," "continue," "anticipate," "design," "intend," "expect," "could," "can," "plan," "potential," "predict," "seek," "should," "would," "contemplate," "project," "target," "tend to," or the negative version of these words and similar expressions. Such statements include, but are not limited to, statements relating to: the potential therapeutic benefits of OpRegen in patients with GA secondary to AMD. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause Lineage’s actual results, performance or achievements to be materially different from future results, performance or achievements expressed or implied by the forward-looking statements in this press release, including, but not limited to, the following risks: that positive findings in early clinical studies of a product candidate may not be predictive of success in subsequent clinical and/or nonclinical studies of that candidate; that Roche and Genentech may not successfully advance OpRegen or be successful in completing further clinical trials for OpRegen and/or obtaining regulatory approval for OpRegen in any particular jurisdiction; and those risks and uncertainties inherent in Lineage’s business and other risks discussed in Lineage’s filings with the Securities and Exchange Commission (SEC). Lineage’s forward-looking statements are based upon its current expectations and involve assumptions that may never materialize or may prove to be incorrect. All forward-looking statements are expressly qualified in their entirety by these cautionary statements. Further information regarding these and other risks is included under the heading "Risk Factors" in Lineage’s periodic reports with the SEC, including Lineage’s most recent Annual Report on Form 10-K filed with the SEC and its other reports, which are available from the SEC’s website. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date on which they were made. Lineage undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240116211178/en/
Contacts
Lineage Cell Therapeutics, Inc. IR
Ioana C. Hone
(mailto://ir@lineagecell.com)
(442) 287-8963
LifeSci Advisors
Daniel Ferry
(mailto://daniel@lifesciadvisors.com)
(617) 430-7576
Russo Partners – Media Relations
Nic Johnson or David Schull
(mailto://mailto://Nic.johnson@russopartnersllc.com)
(David.schull@russopartnersllc.com)
(212) 845-4242
NEWS -- Theriva Biologics to Participate in the B. Riley Securities Annual Oncology Conference
ROCKVILLE, Md., Jan. 16, 2024 (GLOBE NEWSWIRE) -- Theriva Biologics, Inc. (NYSE American: TOVX), a diversified clinical-stage company developing therapeutics designed to treat cancer and related diseases in areas of high unmet need, today announced that Company’s Management will participate in a fireside chat at the B. Riley Securities Oncology Conference to be held virtually on Thursday, January 18, 2024 at 9:30 a.m. ET.
To register for this event, please reach out to your B. Riley Securities sale representative. The replay of the presentation will be accessible in the “Investors” section of the company’s website, https://www.therivabio.com, under “Events.”
About Theriva Biologics, Inc.
Theriva™ Biologics (NYSE American: TOVX), is a diversified clinical-stage company developing therapeutics designed to treat cancer and related diseases in areas of high unmet need. The Company is advancing a new oncolytic adenovirus platform designed for intravenous (IV), intravitreal and antitumoral delivery to trigger tumor cell death, improve access of co-administered cancer therapies to the tumor, and promote a robust and sustained anti-tumor response by the patient’s immune system. The Company’s lead candidates are: (1) VCN-01, an oncolytic adenovirus designed to replicate selectively and aggressively within tumor cells, and to degrade the tumor stroma barrier that serves as a significant physical and immunosuppressive barrier to cancer treatment; (2) SYN-004 (ribaxamase) which is designed to degrade certain commonly used IV beta-lactam antibiotics within the gastrointestinal (GI) tract to prevent microbiome damage, thereby limiting overgrowth of pathogenic organisms such as VRE (vancomycin resistant Enterococci) and reducing the incidence and severity of acute graft-versus-host-disease (aGVHD) in allogeneic hematopoietic cell transplant (HCT) recipients; and (3) SYN-020, a recombinant oral formulation of the enzyme intestinal alkaline phosphatase (IAP) produced under cGMP conditions and intended to treat both local GI and systemic diseases. For more information, please visit Theriva Biologics’ website at https://www.therivabio.com.
For further information, please contact:
Investor Relations:
Chris Calabrese
LifeSci Advisors, LLC
mailto://ccalabrese@lifesciadvisors.com
917-680-5608
Source: Theriva Biologics, Inc.
NEWS -- Oncolytics Biotech® Appoints Patricia S. Andrews to its Board of Directors

SAN DIEGO and CALGARY, AB, Jan. 9, 2024 /CNW/ -- Oncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, today announced the appointment of Patricia S. Andrews to its Board of Directors (the "Board").
"I am pleased to welcome Pat Andrews to our Board of Directors," said Dr. Matt Coffey, President and Chief Executive Officer of Oncolytics. "Pat's extensive board and executive leadership experience navigating first registrational trials in oncology and completing transformational business development agreements with global pharmaceutical companies makes her a natural fit for Oncolytics. As we begin preparations to initiate pivotal studies with pelareorep in breast and pancreatic cancers and progress partnering discussions, we look forward to benefitting from her insights and strategic and operational experience."
"I am honored to join the Oncolytics board at this exciting time for the organization as it advances pelareorep toward registrational studies," said Ms. Andrews. "Promising recent results from the BRACELET-1 and GOBLET studies suggest that pelareorep has the potential to make an important contribution to cancer care, and I look forward to working with my fellow board members in addition to Dr. Coffey and the Executive Team to execute Oncolytics' clinical and corporate development plans."
Ms. Andrews is an accomplished biopharmaceutical executive and public company board member with a track record of success in corporate strategy, first-in-class and first-for-the-company new product commercializations, and business development. Ms. Andrews currently serves as a Director and Member of the Audit Committee at GlycoMimetics. During her tenure, the company planned and initiated its first Phase 3 study and is preparing for commercialization.
Ms. Andrews previously served as Chief Executive Officer for Sumitomo Pharma Oncology, Inc. (SMP Oncology), where she led the organization through the integration of its multiple predecessor companies. Under her leadership, the organization completed three Phase 3 trials and expanded the clinical pipeline from two programs to eight. Prior to SMP Oncology, Ms. Andrews served as Chief Commercial Officer for Incyte and led the organization through the launch preparations and introduction of Jakafi®, the company's first commercial product, a novel, first-in-class, first-in-disease agent for the treatment of myelofibrosis. While with Incyte, she also steered landmark licensing arrangements with Novartis and Eli Lilly that were critically important to fund the company's long-term trajectory.
Prior to Incyte, Ms. Andrews spent 17 years with Pfizer, ultimately serving as Vice President and General Manager of the U.S. Oncology Business Unit, a $900 million portfolio at the time. Under her leadership, Sutent® was launched, becoming the market leader in renal cell carcinoma, and a refreshed positioning of Camptosar® led to robust revenue growth after years of declining sales in colorectal cancer. Ms. Andrews is a member of the Boston chapter of the Board of Advisors for Life Science Cares, a collective impact organization dedicated to fighting poverty in local communities. She earned an MBA from the University of Michigan and her BA from Brown University.
About Oncolytics Biotech Inc.
Oncolytics is a clinical-stage biotechnology company developing pelareorep, an intravenously delivered immunotherapeutic agent. Pelareorep has demonstrated promising results in Phase 2 studies in breast and pancreatic cancers. It acts by inducing anti-cancer immune responses and promotes an inflamed tumor phenotype -- turning "cold" tumors "hot" -- through innate and adaptive immune responses to treat a variety of cancers.
Pelareorep has demonstrated synergies with multiple approved oncology treatments. Oncolytics is currently conducting and planning combination clinical trials with pelareorep in solid and hematological malignancies as it advances towards registrational studies in metastatic breast cancer and pancreatic cancer. For further information, please visit: https://www.oncolyticsbiotech.com.
This press release contains forward-looking statements, within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended and forward-looking information under applicable Canadian securities laws (such forward-looking statements and forward-looking information are collectively referred to herein as "forward-looking statements"). Forward-looking statements contained in this press release include Oncolytics' belief as to the potential, mechanism of action and benefits of pelareorep as a cancer therapeutic; our plans to advance towards registrational studies in metastatic breast cancer and pancreatic cancer; our plans to progress partnering discussions; and other statements related to anticipated developments in Oncolytics' business and technologies. In any forward-looking statement in which Oncolytics expresses an expectation or belief as to future results, such expectations or beliefs are expressed in good faith and are believed to have a reasonable basis, but there can be no assurance that the statement or expectation or belief will be achieved. Such forward-looking statements involve known and unknown risks and uncertainties, which could cause Oncolytics' actual results to differ materially from those in the forward-looking statements. Such risks and uncertainties include, among others, the availability of funds and resources to pursue research and development projects, the efficacy of pelareorep as a cancer treatment, the success and timely completion of clinical studies and trials, Oncolytics' ability to successfully commercialize pelareorep, uncertainties related to the research and development of pharmaceuticals, uncertainties related to the regulatory process and general changes to the economic environment. In particular, we may be impacted by business interruptions resulting from COVID-19 coronavirus, including operating, manufacturing supply chain, clinical trial and project development delays and disruptions, labour shortages, travel and shipping disruption, and shutdowns (including as a result of government regulation and prevention measures). We may incur expenses or delays relating to such events outside of our control, which could have a material adverse impact on our business, operating results and financial condition. Investors should consult Oncolytics' quarterly and annual filings with the Canadian and U.S. securities commissions for additional information on risks and uncertainties relating to the forward-looking statements. Investors are cautioned against placing undue reliance on forward-looking statements. The Company does not undertake any obligation to update these forward-looking statements, except as required by applicable laws.
Company Contact
Jon Patton
Director of IR & Communication
+1-858-886-7813
mailto://jpatton@oncolytics.ca
Investor Relations for Oncolytics
Timothy McCarthy
LifeSci Advisors
+1-917-679-9282
mailto://tim@lifesciadvisors.com
View original content: https://www.prnewswire.com/news-releases/oncolytics-biotech-appoints-patricia-s-andrews-to-its-board-of-directors-302029215.html
SOURCE Oncolytics Biotech® Inc.
View original content: http://www.newswire.ca/en/releases/archive/January2024/09/c2494.html
NEWS -- FuelPositive Joins the Canadian Hydrogen and Fuel Cell Association and Sets Completion Date for Non-Brokered Private Placement
WATERLOO, Ontario, Jan. 08, 2024 (GLOBE NEWSWIRE) -- FuelPositive Corporation, a leading Green Ammonia Company (TSX.V: NHHH) (OTCQB: NHHHF) (the “Company” or “FuelPositive”) announces that it has joined the Canadian Hydrogen and Fuel Cell Association (CHFCA).
“We are pleased to welcome Fuel Positive to the CHFCA as a start-up member. With deep knowledge of the green ammonia and agricultural space, FuelPositive’s initiatives will be a key driver in the industry. We eagerly anticipate supporting FuelPositive’s industry growth and influence,” said Ivette Vera-Perez, CEO of CHFCA.
Nelson Leite, Chief Operating Officer and Board Member of FuelPositive, stated, “Farmers use 80% of the traditional, highly polluting, grey ammonia produced today, and ammonia is a critical chemical in many other sectors. The possibility of producing Green Ammonia on-site for nitrogen fertilizers, fuel-switching, and water purification – with no carbon emissions – is drawing attention globally. It can replace the grey ammonia in virtually all the traditional sectors, significantly reducing carbon emissions worldwide.”
Leite continued, “On-site Green Hydrogen production is a key component of our system. FuelPositive is excited to join the CHFCA to bring us closer to the greatest minds focused on Canadian Green Hydrogen technologies!”
Ian Clifford, FuelPositive’s Chief Executive Officer and Chair, stated, “We are very excited to partner with Canadian Green Hydrogen manufacturers and innovators through the CHFCA. FuelPositive is testing and accessing Green Hydrogen technologies worldwide for our systems. Teaming up with Canadian technology companies and supporting the Canadian economy as we build our Green Ammonia solutions aligns with our mission as a Company.”
About the Canadian Hydrogen and Fuel Cell Association (CHFCA)
The Canadian Hydrogen and Fuel Cell Association (CHFCA) is a national association that supports industry, academia, government agencies, financial organizations, and other stakeholders focused on hydrogen and fuel cell technologies and products. As the collective voice of Canada’s world-leading hydrogen and fuel cell sector, the CHFCA’s mission is to strengthen Canadian leadership, raise awareness of the benefits of the technology, and accelerate the adoption of its members’ products and services in Canada and abroad. The CHFCA currently has more than 170 members across Canada and regional branches in British Columbia, Quebec and Ontario. You can visit www.chfca.ca for more information.
FuelPositive Sets Completion Date for Non-Brokered Private Placement
The Company has set the deadline for completing its ongoing non-brokered private placement (the “Offering”) for January 31, 2024. The Offering consists of up to 90,909,090 units (each a “Unit”) at a price of $0.055 per Unit. Each Unit consists of one common share of the Company and one common share purchase warrant allowing holders to purchase an additional common share at a price of $0.07 for a period of sixty months.
On December 15, 2023, the Company completed an initial tranche of the Offering and issued 24,545,454 Units for aggregate gross proceeds of $1,350,000.
All securities issued in connection with the Offering are subject to restrictions on resale for a period of four months and one day in accordance with applicable securities laws. The Company will pay finders’ fees to eligible third parties who have assisted in introducing subscribers to the Offering. The net proceeds from the Offering will be utilized for general working capital purposes.
About FuelPositive Corporation
FuelPositive is a Canadian technology company that delivers economical and sustainable clean technology solutions. FuelPositive strives to ensure that the lifecycle of our products is environmentally friendly. This includes an on-farm/onsite, containerized Green Ammonia (NH3) production system that effectively eliminates carbon emissions during the production process.
By focusing on technologies that are clean, sustainable, economically advantageous and realizable, the Company aims to help mitigate climate change, addressing unsustainable agricultural practices through innovative technology and practical solutions that can be implemented now. The FuelPositive on-farm/onsite, containerized Green Ammonia production system is designed to produce pure, anhydrous ammonia for multiple applications, including fertilizer for farming, fuel for grain drying and internal combustion engines, a practical alternative for fuel cells and a solution for grid storage. Green Ammonia is also considered a key enabler of the hydrogen economy.
FuelPositive systems are designed to provide for Green Ammonia production on-farm/onsite, where and when needed. This eliminates wildly fluctuating supply chains and offers end-users clean fertilizer, energy and Green Ammonia supply security while eliminating carbon emissions from the production process. The first customers will be farmers. Farmers use 80% of the traditional grey ammonia produced today as fertilizer.
See pre-sale details here: https://fuelpositive.com/pre-sales/.
FuelPositive Corporation is based in Waterloo (Canada) and trades on the TSX Venture Exchange under the symbol NHHH and in the USA on the OTCQB under the symbol NHHHF.
For further information, please contact:
Ian Clifford
Chief Executive Officer and Board Chair
mailto://ian@fuelpositive.com
Tel: 416.535.8395
https://www.fuelpositive.com
Investor Relations United States & International:
RB Milestone Group (RBMG)
mailto://fuelpositive@rbmilestone.com
Investor Relations Canada:
Transcend Capital Inc.
mailto://et@transcendcapitalinc.com
Cautionary Statement
Trading in the securities of the Company should be considered highly speculative. No stock exchange, securities commission or other regulatory authority has approved or disapproved the information contained herein. Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accept responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains certain “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) that are based on expectations, estimates and projections as of the date of this news release. The information in this release about future plans and objectives of the Company, including with respect to further testing and implementation of the FP300 system, are forward-looking statements.
These forward-looking statements are based on assumptions and estimates of management of the Company at the time they were made and involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable by the Company as of the time of such statements, are inherently subject to significant business, economic and competitive uncertainties and contingencies. These estimates and assumptions may prove to be incorrect.
Many of these uncertainties and contingencies can directly or indirectly affect and could cause, actual results to differ materially from those expressed or implied in any forward-looking statements. There can be no assurance that forward-looking information will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements.
Forward-looking information is provided for the purpose of providing information about management’s expectations and plans relating to the future. The Company disclaims any intention or obligation to update or revise any forward-looking information or to explain any material difference between subsequent actual events and such forward-looking information, except to the extent required by applicable law.
NEWS -- NightHawk Biosciences Announces Planned Name Change to Scorpius Holdings, Reflecting Successful Transition to a Large Molecule Biomanufacturing CDMO
Announces senior leadership appointments to further support client programs and growing sales pipeline
DURHAM, N.C., Jan. 08, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK; NHWK), an integrated contract development and manufacturing organization (CDMO), today announced that it is changing the name of the Company to Scorpius Holdings, Inc. (“Scorpius”) to better reflect the Company’s successful shift into a pure-play, large molecule biomanufacturing CDMO. The Company will continue to operate its CDMO within the Scorpius BioManufacturing subsidiary.
In connection with the name change, the Company’s ticker will change to SCPX as well. The name and symbol changes will not affect the Company's share structure or the rights of the Company's shareholders, and no further action will be required by existing shareholders.
In addition, the Company announced that it has bolstered its leadership team with the promotions of Brian O’Mara to VP of Process Sciences and Steve Lavezoli to VP of Commercial Operations, as well as the additions of Juan Lagos as Senior Director of Cell-Based Technologies and Ania Szymanska as Site Head of Quality.
Jeff Wolf, CEO of Nighthawk, commented, “Our rebranding to Scorpius Holdings is a testament to our successful transition and the growth of our CDMO operations. The industry faces a critical shortage of clinical-scale biologic manufacturing capacity, driven by increasing demand for large molecule CDMO services. Our state-of-the-art facility in San Antonio, Texas ideally positions us to address this gap in CDMO manufacturing capacity and services. The response from major biotech companies and leading research institutions to our CDMO capabilities has been positive, as evidenced by the expansion of our sales pipeline."
Mr. Wolf further noted, “To better service increasing demand for our specialized CDMO services, we have expanded our team with key hires to support our growing client base. Each of these team members brings extensive industry experience and will help ensure that we continue to provide our clients with exceptional service. Given the investments in both our facility and operations, we now have significant capacity to scale our operations, which we expect will generate high incremental margins and attractive returns for our shareholders in the years ahead."
Senior Promotions and Appointments
Brian O’Mara - VP of Process Sciences. Mr. O’Mara joined Scorpius in June 2022 as the Senior Director, Manufacturing Science & Technology. He has more than 20 years of industrial biotechnology experience in downstream process development of early- and late-stage protein therapeutics from mammalian and microbial expression systems. He also has extensive experience in the development and scale-up of protein conjugates, including antibody-drug conjugates (ADCs), bi-specifics, and PEGylated molecules, as well as experience in technology transfer, CDMO management, process characterization, preparation and oversight of PPQ campaigns, and associated CMC regulatory filings. Brian earned a BS in Biology from Binghamton University and an MS in Chemistry from Lehigh University.
Steve Lavezoli - VP of Commercial Operations. Mr. Lavezoli joined Scorpius in December 2022 as the Regional VP of Business Development for the central region. He now oversees Scorpius’ business development, proposals, and marketing strategy. He brings extensive experience in various roles throughout business development and marketing. He spent 12 years in the industrial gasses industry with Linde Gas, as well as W.L. Gore in their Startup Biopharmaceutical division working on a Commercial Business/Market Development role for Bulk Drug Substance Single-Use items. He also spent 3 years with Catalent Biologics, focused on Drug Substance Business Development in the US for early-stage clinical programs with clients to bring life-changing therapies to the market. Steve holds a BS in Chemical Engineering from Pennsylvania State University and an MBA in Marketing from Robert Morris University.
Juan Lagos - Senior Director of Cell-Based Technologies. Mr. Lagos brings more than 20 years of experience in cell culture and upstream process development from lab bench to cGMP manufacturing and leads Scorpius’ cell-based technologies team, which is responsible for analytical and cell therapy processes. Before joining Scorpius, he was the Associate Director MS&T / LVV Suspension Process Development at Rocket Pharmaceuticals. His industry experience includes director-level and senior engineering roles at Allakos and Bristol Myers Squibb, as well as experience at biopharma service providers like WuXi Apptec and Patheon. He holds a dual B.S. in Computer Science and Chemical Engineering from Rutgers University, where he continues to pursue a Ph.D. in Biochemical Engineering.
Ania Szymanska - Site Head of Quality. Ms. Szymanska brings over 19 years of leadership and management experience in Quality Control, Quality Assurance, and Compliance at pharmaceutical and biotech companies. She joined Scorpius from Marker Therapeutics, Inc. where she was Vice President of Quality and built the Quality Management Systems. She played a critical role in the design, construction, and qualification of their state-of-the-art cell therapy GMP manufacturing facility and Quality Control laboratories. Prior to Marker, she served as Director of Quality Control for Bellicum Pharmaceuticals, Inc. where she developed its Quality Control department and was responsible for aseptic facility qualification. She also served as Quality Validation Specialists of Opexa Therapeutics, Inc. and in roles of increasing responsibility at Woodfield Pharmaceutical, LLC (formerly Pernix Manufacturing, LLC), most recently as Director of Microbiology. Ms. Szymanska earned an M.S. in Microbiology from the University of Warsaw.
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward-Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as generating high incremental margins and attractive returns for our shareholders in the years ahead. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate, high incremental margins and attractive returns for our shareholders in the years ahead, the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure-play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Tokens.com Provides Update on 2023 Annual Filings
TORONTO, January 05, 2024--(BUSINESS WIRE)--Tokens.com Corp. (NEO Exchange Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a technology company that builds immersive experiences and games in 3D internet environments and owns an inventory of cryptocurrencies, today announced that, further to its news release dated December 15, 2023, its principal regulator, the Ontario Securities Commission, has accepted the Company’s application for, and has granted, a management cease trade order (the "MCTO"). As previously announced, the Company applied for the MCTO due to a delay in the filing of the Company's annual financial statements and accompanying management’s discussion and analysis for the for the twelve-month period ended September 30, 2023 (the "2023 Annual Filings"), which were due on December 29, 2023.
The MCTO restricts the Chief Executive Officer and Chief Financial Officer from all trading in securities of the Company until such time as the 2023 Annual Filings have been filed by the Company and the MCTO has been lifted. The MCTO does not affect the ability of other shareholders of the Company to trade in securities of the Company.
The Company and its auditors continue to work diligently toward completing the 2023 Annual Filings as soon as possible. The Company is not aware of any specific accounting or audit concerns at this time. The Company expects that it will be in a position to file the 2023 Annual Filings on or before January 31, 2024.
Until the 2023 Annual Filings are filed, the Company intends to comply with the provisions of the alternative information guidelines as set out in National Policy 12-203 – Management Cease Trade Orders ("NP 12-203") for as long as it remains in default, including the issuance of bi-weekly default status reports in the form of a news release. The Company confirms that, since its prior news release dated December 15, 2023: (a) there has been no material change to the information set out in that press release that has not been generally disclosed; (b) there has not been any other specified default by the Company under NP 12-203; (c) the Company is not subject to any insolvency proceedings; and (d) there is no material information concerning the affairs of the Company that has not been generally disclosed.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies.
Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite.
Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms.
Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240105538122/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- Tokens.com Provides Update on 2023 Annual Filings
TORONTO, January 05, 2024--(BUSINESS WIRE)--Tokens.com Corp. (NEO Exchange Canada: COIN)(Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a technology company that builds immersive experiences and games in 3D internet environments and owns an inventory of cryptocurrencies, today announced that, further to its news release dated December 15, 2023, its principal regulator, the Ontario Securities Commission, has accepted the Company’s application for, and has granted, a management cease trade order (the "MCTO"). As previously announced, the Company applied for the MCTO due to a delay in the filing of the Company's annual financial statements and accompanying management’s discussion and analysis for the for the twelve-month period ended September 30, 2023 (the "2023 Annual Filings"), which were due on December 29, 2023.
The MCTO restricts the Chief Executive Officer and Chief Financial Officer from all trading in securities of the Company until such time as the 2023 Annual Filings have been filed by the Company and the MCTO has been lifted. The MCTO does not affect the ability of other shareholders of the Company to trade in securities of the Company.
The Company and its auditors continue to work diligently toward completing the 2023 Annual Filings as soon as possible. The Company is not aware of any specific accounting or audit concerns at this time. The Company expects that it will be in a position to file the 2023 Annual Filings on or before January 31, 2024.
Until the 2023 Annual Filings are filed, the Company intends to comply with the provisions of the alternative information guidelines as set out in National Policy 12-203 – Management Cease Trade Orders ("NP 12-203") for as long as it remains in default, including the issuance of bi-weekly default status reports in the form of a news release. The Company confirms that, since its prior news release dated December 15, 2023: (a) there has been no material change to the information set out in that press release that has not been generally disclosed; (b) there has not been any other specified default by the Company under NP 12-203; (c) the Company is not subject to any insolvency proceedings; and (d) there is no material information concerning the affairs of the Company that has not been generally disclosed.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies.
Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite.
Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms.
Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to the strategic review process and the work of the Committee; whether a strategic change, transaction or any outcome will result from or be consummated or implemented as a result of the strategic review process; and whether any transaction resulting from the strategic review process, if any, will ultimately enhance shareholder or stakeholder value in the long term.
Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240105538122/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- Oncolytics Biotech® Recaps 2023 Accomplishments, Provides Outlook for 2024

SAN DIEGO, CA and CALGARY, AB, Jan. 4, 2024 /CNW/ -- Oncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, reported on significant 2023 accomplishments and announced corporate priorities and anticipated clinical development milestones for 2024.
Overview: "2023 was an outstanding year for Oncolytics and the development of pelareorep as a potential cancer therapy. To date, we have studied pelareorep in more than 1,100 patients, and it has been shown to be well-tolerated with mild side effects. Promising clinical and translational results from our Phase 2 programs and mechanism of action learnings from a wide range of tumor types, as detailed below, provided consistent data and efficacy signals with durable responses that support the use of pelareorep as an immunotherapeutic agent, either as a monotherapy or in combination with other agents. These data readouts from multiple clinical studies at numerous sites and from diverse cancer indications that are clinically important make a compelling argument for pelareorep as a true backbone immunotherapy with the potential to help countless patients across a range of tumors," said Dr. Matt Coffey, President and Chief Executive Officer of Oncolytics. "We believe our cash balance and strong data, supported by our active business development program, position us well for 2024."
Positive Clinical and Translational Data: "Positive, early results from the Phase 1/2 GOBLET study in three gastrointestinal cancers - pancreatic, anal, and colorectal - showed that the combination of pelareorep and atezolizumab produced clinically meaningful improvements in response rates and survival timelines that are notably improved compared to historical results with no toxicity concerns. Translational data from studies in solid tumors, breast, pancreatic, and colorectal cancers showed that treatment with pelareorep remodels the tumor microenvironment and stimulates tumor-directed immune responses, affirming its mechanism of action as an immunotherapeutic agent. Together, these data provide a strong foundation to support advancing pelareorep into registrational-track studies, starting in 2024," continued Dr Coffey.
2024 Outlook: Dr. Coffey concluded by saying, "Looking ahead to 2024, we expect to initiate the first Phase 3 study for pelareorep in pancreatic cancer. The transition to a late-stage biopharmaceutical company will mark an important inflection point for investors, clinical collaborators and potential partners by providing a line of sight towards the path to regulatory approval and achieving our mission of developing pelareorep as an immunotherapeutic agent for cancer. We are excited to launch a new Phase 1/2 pancreatic cancer study investigating, for the first time, pelareorep in combination with modified FOLFIRINOX (mFOLFIRINOX). Finally, our discussions with regulators on a pivotal Phase 3 trial have been productive to date, and we expect to provide guidance on the registration path for metastatic breast cancer in the first half of 2024. We look forward to updating our stakeholders on our progress as the year unfolds."
Immunotherapeutic Mechanism of Action: Thomas Heineman, M.D., Ph.D., Chief Medical Officer at Oncolytics, said, "Translational data from multiple studies in 2023 have helped define pelareorep's role in shaping the tumor microenvironment and have provided a better understanding of its ability to induce the expansion of T cell populations. Notably, the positive association between tumor response and TIL clone expansion could become a useful marker of clinical outcomes in future studies and during patient care. Moreover, these findings further distinguish pelareorep's mechanism of action from that of other immunotherapeutic agents and provide support for its immunologic effects, which are largely driven by the introduction of its double-stranded RNA genome into cancer cells."
Financial Strength and Partnering Outlook: Kirk Look, Oncolytics' Chief Financial Officer, commented, "Our $40 million cash balance as of September 30, 2023, and the grant funding from The Pancreatic Cancer Action Network (PanCAN) provides us with over 12 months of runway to support our operations, including the initiation of our first Phase 3 study and the BRACELET-1 survival data. We continue to have active conversations with potential partners and work diligently with clinical collaborators, including Roche and Pfizer. We believe the positive, extended dataset, including continued and durable responses, survival results, and translational observations, will provide potential partners with a well-defined and unique target product profile that will add to our discussions."
Anticipated Milestones are expected to include:
NEWS -- NightHawk Biosciences Commences Work on Multi-Million-Dollar Development and Manufacturing Agreement For a Leading National University
Scorpius subsidiary to manufacture novel biologic targeting substance use disorder
DURHAM, N.C., Jan. 03, 2024 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced its Scorpius BioManufacturing subsidiary (“Scorpius”) has commenced work on a contract that it was awarded by a leading university to develop and manufacture a novel biologic targeting substance use disorder, which includes drug, alcohol and opioid addiction.
Jeff Wolf, CEO of NightHawk, commented, “We are honored to have been awarded this multi-million dollar contract by one of the foremost research universities in the United States. Scorpius was selected due to its process development and large molecule manufacturing expertise, as well as its flexibility and dedication to customer service. We are off to a strong start and look forward to expanding this relationship into additional programs in 2024 and beyond. Overall, we could not be more excited by the traction and growing pipeline within Scorpius.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as expanding the relationship with the university into additional programs in 2024 and beyond Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate future revenue from manufacturing contracts, the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure-play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
Go to 36 minutes and 5 seconds before it starts, Go to stocks wizard on YouTube.
NEWS -- NightHawk Biosciences Announces Completion of Demonstration Run for a Top-Tier NIH and DTRA Funded Research University
Next phase of multi-million-dollar project to include scaling up cGMP manufacturing for phase 1 trial targeting infectious disease
CDMO services attracting considerable interest as state-of-the-art San Antonio facility helps address industry-wide scarcity for large molecule manufacturing
DURHAM, N.C., Dec. 21, 2023 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced its Scorpius BioManufacturing subsidiary (“Scorpius”) completed a demonstration run in the Company’s process development lab for a microbial vaccine program developed by a top-tier research university that is NIH and DTRA (Defense Threat Reduction Agency) funded.
Jeff Wolf, CEO of NightHawk, commented, “This first phase of a multi-million-dollar project was particularly noteworthy, as we achieved a successful demonstration run following a prior failed attempt by a competitor CDMO. The complexity of the project led this top-tier university to choose Scorpius, drawn by our more engaged management approach and accelerated development timeline. The successful completion of this demonstration run showcases our team's dedication and expertise, and represents an important step in advancing our relationship with this nationally renowned university. The next phase of this project will include scaling up to cGMP manufacturing to support a phase 1 infectious disease trial. Our CDMO services continue to attract considerable interest, thanks to our extraordinary team, state-of-the-art facilities, and the industry-wide scarcity of capacity for large molecule manufacturing.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as recognizing revenue from the agreement during 2024, expanding our scope of work and revenue potential of this agreement and the CDMO activities holding the potential to generate meaningful revenue and cash flow Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate future revenue from manufacturing contracts booked in 2023 the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO, he continued maintenance and growth of the Company’s and its subsidiaries’ patent estates the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Radiogel™ Precision Radionuclide Therapy™ Receives FDA Breakthrough Device Designation
Richland WA, Dec. 20, 2023 (GLOBE NEWSWIRE) -- Vivos Inc. (OTCQB: RDGL), Vivos Inc is pleased to announce that the Food and Drug Administration has granted RadioGel Precision Radionuclide Therapy the designation as a Breakthrough Device pursuant to the FDA’s Breakthrough Devices Program.
Dr. Mike Korenko stated “We are pleased with the FDA’s decision to grant Breakthrough Device Designation following our recent data submissions. The FDA did a very thorough review. The analysis in our Risk Management Report concluded that RadioGel™ is intrinsically safe and our animal therapy data verified its effectiveness. We are currently working closely with Mayo Clinic for our initial indication for use of Radiogel for delivering “therapeutic radiation to solid metastatic tumors in lymph nodes associated with papillary thyroid cancer, accessible by percutaneous direct needle injection, in patients who are not surgical candidates or have declined surgery, had non-radioiodine avid disease and had limited burden regional nodal disease.” The Breakthrough Device classification will enhance the schedule priority for our upcoming IDE submission seeking authorization to begin human clinical trials.
The FDA’s Breakthrough Devices program facilitates accelerated development and expedites the review of breakthrough technologies to help patients potentially gain timely access to technologies that can provide more effective treatment options.
Contact for journalists:
Mike Korenko
Email: mailto://MKorenko@RadioGel.com
Follow Vivos Inc @VivosIncUSA., Radiogel™ @RadioGel and Isopet® on X (fka Twitter):
About Vivos Inc. (OTCQB: RDGL)
Vivos Inc. has developed an Yttrium-90-based injectable Precision Radionuclide Therapy brachytherapy device to treat tumors in animals (IsoPet®) and humans (RadioGel™). Using the company's proprietary hydrogel technology, Brachytherapy uses highly localized radiation to destroy cancerous tumors by placing a radioactive isotope directly inside the treatment area. The injection delivers therapeutic radiation from within the tumor without the entrance skin dose and associated side effects of treatment that characterize external-beam radiation therapy. This feature allows the safe delivery of higher doses needed for treating non-resectable and radiation-resistant cancers.
RadioGel™ is a hydrogel liquid containing tiny yttrium-90 phosphate microparticles that may be administered directly into a tumor. The hydrogel is a yttrium-90 carrier at room temperature that gels within the tumor interstitial spaces after injection to keep the radiation sources safely in place. The short-range beta radiation from yttrium-90 localizes the dose within the treatment area so that normal organs and tissues are not adversely affected.
RadioGel™ also has a short half-life – delivering more than 90% of its therapeutic radiation within 10 days. This compares favorably to other available treatment options requiring up to six weeks or more to deliver a full course of radiation therapy. Therapy can be safely administered as an outpatient procedure, and the patient may return home without subsequent concern for radiation dose to family members.
University veterinary hospitals use the IsoPet® Solutions division to demonstrate animal cancers' safety and therapeutic effectiveness. Testing on feline sarcoma at Washington State University was completed in 2018, and testing on canine soft tissue sarcomas at the University of Missouri was completed in 2019. The Company has obtained confirmation from the FDA Center for Veterinary Medicine that IsoPet® is classified as a medical device according to its intended use and means by which it achieves its intended purpose. The FDA also reviewed the product labeling, which included canine and feline sarcomas as the initial indications for use. The FDA does not require pre-market approval for veterinary devices, so no additional approval was required to generate revenue through the sale of IsoPet® to University animal hospitals and private veterinary clinics.
IsoPet® for treating animals uses the same technology as RadioGel™ for treating humans. The Food and Drug Administration advised using different product names to avoid confusion and cross-use.
Safe Harbor Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. You can identify these statements by the use of the words "may," "will," "should," "plans," "expects," "anticipates," "continue," "estimates," "projects," "intends," and similar expressions. Forward-looking statements involve risks and uncertainties that could cause results to differ materially from those projected or anticipated. These risks and uncertainties include, but are not limited to, the Company's ability to successfully execute its expanded business strategy, including by entering into definitive agreements with suppliers, commercial partners, and customers; general economic and business conditions, effects of continued geopolitical unrest and regional conflicts, competition, changes in technology and methods of marketing, delays in completing various engineering and manufacturing programs, changes in customer order patterns, changes in product mix, continued success in technical advances and delivering technological innovations, shortages in components, production delays due to performance quality issues with outsourced components, regulatory requirements and the ability to meet them, government agency rules and changes, and various other factors beyond the Company's control.
CONTACT:
Vivos Inc.
Michael K. Korenko, Sc.D.
President & CEO
mailto://MKorenko@VivosInc.co
NEWS -- Tokens.com Announces Launch of Ecommerce App on Google Play and Apple iOS Stores
TORONTO, December 19, 2023--(BUSINESS WIRE)--Tokens.com Corp. (NEO Exchange Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a publicly-traded company that builds web3 businesses and owns an inventory of digital assets, is pleased to announce the launch of Valor, a proprietary ecommerce platform within Metaverse Group that allows product placement and advertising within virtual gaming worlds including Roblox and Fortnite.
Metaverse Group has spent the last year developing Helix Worlds, the technology that powers the Valor app. Valor is designed as a loyalty program that gives its clients the ability to place branded product placement in games like Roblox and Fortnite. Players in these environments use the app by scanning branded items and are redirected to the brands page to claim offers and earn points that are redeemable for rewards.
Valor is available for download on the Google Play Store, and iOS App Store.
"The Valor app was developed to solve a key problem for brands who were looking to advertise in popular gaming platforms like Roblox and Fortnite where links and QR codes are not allowed," said Andrew Kiguel, CEO of Tokens.com. "Valor provides a solution by giving brands the ability to offer a shopping experience to players, without taking them out of the game flow and disrupting the environment with links and QR codes."
Imagine a gamer walking through a virtual city and encountering your brand’s latest sneaker collection or wearable tech gadget right within the metaverse. With just 2-3 taps on their mobile device or Helix Worlds compatible apps and or via VR Goggles, players can interact with the product, purchase it in the physical world, and earn exclusive in-game rewards.
The consumer ecommerce experience has not changed substantially in the last 20 years. However, with the advent and popularity of new 3D platforms such as Roblox and Fortnite, brands are seeking innovative ways to evolve their ecommerce solutions. Through Helix Worlds, brands have the unprecedented opportunity to place their products into games and 3D environments without breaking the immersive experience and breaking platform guidelines. Valor using Helix Worlds technology was innovated for brands to market their products to a new audience and for consumers to enjoy an improved shopping experience. Helix Worlds can be implemented into any app and will be available across multiple platforms, and popular games.
Brands and businesses interested in learning more about Valor can contact mailto://info@metaversegroup.com. For a demo of how Valor works click here or visit https://getvalor.gg.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies.
Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite.
Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms.
Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to completion of the acquisition and closing date thereof and the benefits to be realized from the transaction, including the potential synergies between Metaverse Group and Tokens.com (including Hulk Labs, the gaming unit of Tokens.com). Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231219150315/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- Tokens.com Announces Launch of Ecommerce App on Google Play and Apple iOS Stores
TORONTO, December 19, 2023--(BUSINESS WIRE)--Tokens.com Corp. (NEO Exchange Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a publicly-traded company that builds web3 businesses and owns an inventory of digital assets, is pleased to announce the launch of Valor, a proprietary ecommerce platform within Metaverse Group that allows product placement and advertising within virtual gaming worlds including Roblox and Fortnite.
Metaverse Group has spent the last year developing Helix Worlds, the technology that powers the Valor app. Valor is designed as a loyalty program that gives its clients the ability to place branded product placement in games like Roblox and Fortnite. Players in these environments use the app by scanning branded items and are redirected to the brands page to claim offers and earn points that are redeemable for rewards.
Valor is available for download on the Google Play Store, and iOS App Store.
"The Valor app was developed to solve a key problem for brands who were looking to advertise in popular gaming platforms like Roblox and Fortnite where links and QR codes are not allowed," said Andrew Kiguel, CEO of Tokens.com. "Valor provides a solution by giving brands the ability to offer a shopping experience to players, without taking them out of the game flow and disrupting the environment with links and QR codes."
Imagine a gamer walking through a virtual city and encountering your brand’s latest sneaker collection or wearable tech gadget right within the metaverse. With just 2-3 taps on their mobile device or Helix Worlds compatible apps and or via VR Goggles, players can interact with the product, purchase it in the physical world, and earn exclusive in-game rewards.
The consumer ecommerce experience has not changed substantially in the last 20 years. However, with the advent and popularity of new 3D platforms such as Roblox and Fortnite, brands are seeking innovative ways to evolve their ecommerce solutions. Through Helix Worlds, brands have the unprecedented opportunity to place their products into games and 3D environments without breaking the immersive experience and breaking platform guidelines. Valor using Helix Worlds technology was innovated for brands to market their products to a new audience and for consumers to enjoy an improved shopping experience. Helix Worlds can be implemented into any app and will be available across multiple platforms, and popular games.
Brands and businesses interested in learning more about Valor can contact mailto://info@metaversegroup.com. For a demo of how Valor works click here or visit https://getvalor.gg.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies.
Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite.
Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms.
Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names.
Visit https://Tokens.com to learn more.
Keep up-to-date on Tokens.com developments and join our online communities on Twitter, LinkedIn, Facebook, Instagram and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identified by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements in this news release include statements relating to completion of the acquisition and closing date thereof and the benefits to be realized from the transaction, including the potential synergies between Metaverse Group and Tokens.com (including Hulk Labs, the gaming unit of Tokens.com). Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities filings available at www.sedar.com. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231219150315/en/
Contacts
Tokens.com Corp.
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
NEWS -- NightHawk Biosciences Announces Major Milestone with Completion of Manufacturing Run for a Premier U.S.-Based Biopharmaceutical Company
Manufacturing run at Scorpius’ state-of-the-art San Antonio microbial facility sets the stage for commercial manufacturing
Advances multi-million dollar biomanufacturing contract awarded earlier this year; set to expand upon client’s regulatory filing and start of commercial manufacturing
DURHAM, N.C., Dec. 18, 2023 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) (“NightHawk” or the “Company”), an integrated contract development and manufacturing organization (CDMO), today announced its Scorpius BioManufacturing subsidiary initiated its first manufacturing run for a premier U.S.-based biopharmaceutical company in the Company’s new, state-of-the-art microbial manufacturing facility.
Jeff Wolf, CEO of NightHawk, commented, “We are generating strong interest in our CDMO services, given our state-of-the-art facility and the industry-wide lack of available capacity for large molecule manufacturing. This demonstration run in our new microbial facility marks a major milestone, as it represents the first at-scale production run with a premier biopharmaceutical company and reflects positively upon our manufacturing readiness, as well as our ability to meet their stringent product requirements. Importantly, this milestone enables us to build upon the initial multi-million-dollar contract with this client, which we expect to recognize as revenue during 2024. We also look forward to further expanding our scope of work and revenue potential of this agreement, as we work with our client to submit the necessary regulatory filings to commence commercial-scale production. We appreciate their trust in Scorpius to ensure this important product gets to patients as safely and quickly as possible.”
Mr. Wolf added, “This demonstration run and expansion of this agreement is further validation of our recent decision to streamline the organization through the divestiture of non-core assets to double-down on our CDMO activities, which we believe hold the potential to generate meaningful revenue and cash flow. We are witnessing growing interest in the industry as we build our track record and enhance our reputation. This is reflected in our rapidly expanding sales pipeline that now includes a number of premier biopharma companies, as well as leading government and academic research institutes.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as recognizing revenue from the agreement during 2024, expanding our scope of work and revenue potential of this agreement and the CDMO activities holding the potential to generate meaningful revenue and cash flow Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate future revenue from manufacturing contracts booked in 2023 the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO, he continued maintenance and growth of the Company’s and its subsidiaries’ patent estates the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Lineage Announces Submission of OPC1 Investigational New Drug Amendment for Treatment of Chronic and Subacute Spinal Cord Injury
CARLSBAD, Calif., December 18, 2023--(BUSINESS WIRE)--Lineage Cell Therapeutics, Inc. (NYSE American and TASE: LCTX), a clinical-stage biotechnology company developing allogeneic cell therapies for unmet medical needs, announced today that the Company has submitted an Investigational New Drug amendment (INDa) to the U.S. Food and Drug Administration (FDA), for OPC1, its investigational allogeneic oligodendrocyte progenitor cell transplant, for the treatment of spinal cord injury (SCI). Receipt of FDA clearance of the INDa would enable the Company to initiate its DOSED (Delivery of Oligodendrocyte Progenitor Cells for Spinal Cord Injury: Evaluation of a Novel Device) clinical study, to evaluate the safety and utility of a novel spinal cord delivery device in both subacute and chronic SCI patients. The OPC1 cell transplant is designed to replace or support cells that are absent or dysfunctional due to traumatic injury, with a goal to help improve the quality of life and restore or augment functional activity in persons suffering from traumatic cervical or thoracic SCIs.
"The submission of our IND amendment for OPC1 and its return to clinical testing represents a significant milestone for this program and a diligent effort by our team," stated Brian M. Culley, Lineage CEO. "With so few opportunities for SCI patients to participate in clinical trials, it is a privilege to be able to re-engage with the community as part of our efforts to improve outcomes for individuals with a debilitating condition for which there currently are no FDA-approved treatments. Our team has made a significant effort to introduce improvements to the OPC1 program, including enhancements to the production process and quality of our product candidate and an improved delivery system compatible with our immediate use, thaw-and-inject formulation. We are encouraged by the meaningful quality of life and safety results observed with OPC1 in previous trials, and we look forward to building on that promising work. We are excited by the opportunity to initiate the DOSED study and the opening of the first clinical sites in early 2024, assuming no further comments are received from the FDA in the 30 days following our INDa submission."
OPC1 has been tested in two clinical trials to date; a five patient Phase 1 clinical safety trial in acute thoracic SCI, where all subjects have been followed for at least 10 years; as well as a 25 patient Phase 1/2a multicenter dose-escalation clinical trial in subacute cervical SCI, where all subjects were evaluated for at least two years, one of the first cell therapy clinical trials supported by the California Institute for Regenerative Medicine under Proposition 71. Results from both studies have been published in the Journal of Neurosurgery: Spine: the data from the Phase 1/2a clinical study of OPC1 in subacute cervical SCI is available here and the data from the Phase 1 clinical study of OPC1 in acute thoracic SCI is available here.
About the DOSED Study
The Delivery of Oligodendrocyte Progenitor Cells for Spinal Cord Injury: Evaluation of a Novel Device (DOSED) clinical study is an open label, multi-center, device safety study, in approximately 3-5 subacute and 3-5 stable chronic subjects with complete (ASIA Impairment Scale A) or incomplete (ASIA Impairment Scale B), traumatic, focal SCI affecting either cervical (C4-C7) or thoracic (T1-T10) vertebrae. The primary objective of this study is to evaluate the safety of a novel spinal cord delivery device to administer OPC1 to the spinal parenchyma. The primary endpoint is safety, as measured by the frequency and severity of adverse events (AEs) through 30 days following OPC1 injection that are related to the injection procedure. Secondary endpoints are safety and tolerability, as measured by the frequency and severity of AEs, including AEs of special interest, through 90 days following OPC1 injection, that are related to OPC1 and/or the concomitant short-term immunosuppression. Safety parameters will be evaluated by magnetic resonance imaging (MRI) data evaluating evidence of deterioration or changes in the following: intramedullary hemorrhage, cerebral spinal fluid (CSF) leak, epidural abscess, infection; evidence of an expanding cyst or mass at the injection site or elsewhere in the central nervous system (CNS); evidence of inflammatory lesion(s) at injection site or elsewhere in CNS and evidence of CSF flow obstruction. Exploratory endpoints include: (1) potential improvements in neurological impairment, function, and pain, evaluated by changes from baseline on the following endpoints: changes in neurological function as measured by sensory and motor scores and motor level on International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI) examinations; changes in post-injection pain, defined as a worsening of pain or neuropathic pain of greater than 7 days duration from baseline levels, as assessed by the International Spinal Cord Injury Pain Basic Data Set or occurrence of allodynia; (2) changes from baseline at 30, 90 and 365 days post-OPC1 injection in: ISNCSCI, SCIM, International Spinal Cord Injury Pain Questionnaire; and (3) patient and clinician impressions of changes in quality of life as reported by changes from baseline at 30, 90, and 365 days post-OPC1 injection.
About OPC1
OPC1 is an oligodendrocyte progenitor cell (OPC) transplant therapy designed to provide clinically meaningful improvements to motor recovery in individuals with spinal cord injuries (SCI). OPCs are naturally occurring precursors to the cells which provide electrical insulation for nerve axons in the form of a myelin sheath. SCI most often occurs when the spinal cord is subjected to a severe crush or contusion injury and typically results in severe functional impairment, including limb paralysis, aberrant pain signaling, and loss of bladder control and other body functions. There are approximately 18,000 new spinal cord injuries annually in the U.S. and there currently are no FDA-approved drugs or interventions specifically for the treatment of SCI. The OPC1 program has been partially funded by a $14.3 million grant from the California Institute for Regenerative Medicine. OPC1 has received Regenerative Medicine Advanced Therapy (RMAT) designation and Orphan Drug designation from the U.S. Food and Drug Administration (FDA).
About the California Institute for Regenerative Medicine (CIRM)
CIRM, California’s Stem Cell Agency, was created by the voters of California in 2004 with the passing of Proposition 71, which authorized $3 billion in funding for stem cell research in California. The agency funds stem cell research at institutions and companies throughout California (as well as institutions and companies outside of the state that conduct a portion of their research in California) with the goal of accelerating treatments to patients with unmet medical needs. In 2020, California voters approved to continue funding California’s Stem Cell Agency through the passage of Proposition 14. CIRM’s mission is to accelerate world class science to deliver transformative regenerative medicine treatments in an equitable manner to a diverse California and world as well as to accelerate stem cell treatments to patients with unmet medical needs. By promoting and encouraging the growth of the stem cell biotechnology sector, the agency is also helping attract the best scientists to the state and establishing California as a global leader in stem cell research. For more information, please visit https://www.cirm.ca.gov/ and follow the agency on Twitter: @CIRMnews.
About Lineage Cell Therapeutics, Inc.
Lineage Cell Therapeutics is a clinical-stage biotechnology company developing novel cell therapies for unmet medical needs. Lineage’s programs are based on its robust proprietary cell-based therapy platform and associated in-house development and manufacturing capabilities. With this platform Lineage develops and manufactures specialized, terminally differentiated human cells from its pluripotent and progenitor cell starting materials. These differentiated cells are developed to either replace or support cells that are dysfunctional or absent due to degenerative disease or traumatic injury or administered as a means of helping the body mount an effective immune response to cancer. Lineage’s clinical and preclinical programs are in markets with billion dollar opportunities and include five allogeneic ("off-the-shelf") product candidates: (i) OpRegen®, a retinal pigment epithelial cell therapy in Phase 2a development for the treatment of geographic atrophy secondary to age-related macular degeneration, is being developed under a worldwide collaboration with Roche and Genentech, a member of the Roche Group; (ii) OPC1, an oligodendrocyte progenitor cell therapy in Phase 1/2a development for the treatment of acute spinal cord injuries; (iii) VAC2, a dendritic cell therapy produced from Lineage’s VAC technology platform for immuno-oncology and infectious disease, currently in Phase 1 clinical development for the treatment of non-small cell lung cancer; (iv) ANP1, an auditory neuronal progenitor cell therapy for the potential treatment of auditory neuropathy; and (v) PNC1, a photoreceptor neural cell therapy for the potential treatment of vision loss due to photoreceptor dysfunction or damage. For more information, please visit https://www.lineagecell.com or follow the company on Twitter @LineageCell.
Forward-Looking Statements
Lineage cautions you that all statements, other than statements of historical facts, contained in this press release, are forward-looking statements. Forward-looking statements, in some cases, can be identified by terms such as "believe," "aim," "may," "will," "estimate," "continue," "anticipate," "design," "intend," "expect," "could," "can," "plan," "potential," "predict," "seek," "should," "would," "contemplate," "project," "target," "tend to," or the negative version of these words and similar expressions. Such statements include, but are not limited to, statements relating to: the ability of cell transplant therapies, including OPC1, to improve recovery, allow a patient to regain more function than what could otherwise be expected, or improve a patient’s quality of life; and the potential future achievements of our clinical, preclinical and development programs, the planned initiation of clinical trials, including the DOSED study in early 2024, and the timing and availability of clinical data updates related to our programs. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause Lineage’s actual results, performance or achievements to be materially different from future results, performance or achievements expressed or implied by the forward-looking statements in this press release, including, but not limited to, the following risks: that positive findings in early clinical and/or nonclinical studies of a product candidate may not be predictive of success in subsequent clinical and/or nonclinical studies of that candidate; that the feedback received from the FDA for OPC1 may not enable further clinical development; and those risks and uncertainties inherent in Lineage’s business and other risks discussed in Lineage’s filings with the Securities and Exchange Commission (SEC). Lineage’s forward-looking statements are based upon its current expectations and involve assumptions that may never materialize or may prove to be incorrect. All forward-looking statements are expressly qualified in their entirety by these cautionary statements. Further information regarding these and other risks is included under the heading "Risk Factors" in Lineage’s periodic reports with the SEC, including Lineage’s most recent Annual Report on Form 10-K filed with the SEC and its other reports, which are available from the SEC’s website. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date on which they were made. Lineage undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231218883834/en/
Contacts
Lineage Cell Therapeutics, Inc. IR
Ioana C. Hone
(mailto://ir@lineagecell.com)
(442) 287-8963
LifeSci Advisors
Daniel Ferry
(mailto://daniel@lifesciadvisors.com)
(617) 430-7576
Russo Partners – Media Relations
Nic Johnson or David Schull
(mailto://Nic.johnson@russopartnersllc.com)
(mailto://David.schull@russopartnersllc.com)
(212) 845-4242
NEWS -- Provectus Biopharmaceuticals Announces Notice of Allowance of First U.S. Patent of Rose Bengal Sodium for Use as Anticancer and Antiviral Vaccine Adjuvant
KNOXVILLE, TN, Dec. 18, 2023 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed patent application 17/488,430, titled “Halogenated Xanthenes as Vaccine Adjuvants.” The allowed patent application covers the use of Provectus’s pharmaceutical-grade rose bengal sodium (RBS) drug substance as an adjuvant in anticancer, antiviral, and possibly other vaccines to potentially make them work better by enhancing T-cell response.
The allowed application would be Provectus’s first patent award in the field of vaccines from the USPTO. Innovate Calgary, the innovation company of the University of Calgary in Alberta, Canada, is a co-assignee. Aru Narendran, MD, PhD, Professor of Pediatrics, Oncology, Biochemistry and Molecular Biology and Physiology and Pharmacology at the University’s Cumming School of Medicine is a co-inventor. The research underlying the allowed application was led by Dr. Narendran and his lab team (the Narendran Lab).
Preclinical data from ongoing research on the potential use of investigational cancer immunotherapy PV-10 (rose bengal sodium) as a vaccine adjuvant was the subject of a poster presentation by the Narendran Lab at the Society for Immunotherapy of Cancer (SITC) 2023 annual meeting. A copy of the SITC 2023 poster, titled “The iodinated fluorescein derivative PV-10 enhances the antiviral activity of CD8+ T-Cells by inducing STING dimerization: Implications for enhanced vaccine applications,” is available on Provectus’s website at https://www.provectusbio.com/media/docs/2023-SITC-poster.pdf.
The Narendran Lab previously discovered that PV-10 activated stimulator of interferon (IFN) genes (STING), demonstrating its potential as a vaccine adjuvant in PV-10-mediated systemic anti-tumor immune responses. This work, titled “Association of Heat Shock Proteins as Chaperone for STING: A potential link in a key immune activation mechanism revealed by a novel anticancer agent PV-10” was the subject of a poster presentation at the American Association for Cancer Research (AACR) 2020 Virtual Annual Meeting II. A copy of the AACR 2020 poster is available on Provectus’s website at: https://www.provectusbio.com/media/docs/publications/AACR-2020_meeting_posterV3.pdf.
In its SITC 2023 work, the Narendran Lab showed that PV-10 treatment induced STING activation, upregulated cytokines and chemokines, and increased IFN-? secretion by CD8+ T-cells. Dr. Narendran and his colleagues demonstrated PV-10’s ability to function as an effective adjuvant to enhance T-cell responses and concluded that PV-10’s unique modulation of the STING pathway was a potential mechanism of this activity. This work also portends the potential benefit of PV-10 in combination with targeted immunotherapies and antibody-drug conjugates for cancer treatment.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different diseases that are based on a class of synthetic small molecule immuno-catalysts called halogenated xanthenes (HXs). Provectus’s lead HX molecule is named rose bengal sodium (RBS).
The Company’s proprietary, patented, pharmaceutical-grade RBS is the active pharmaceutical ingredient (API) in the drug product candidates of Provectus’s clinical development programs and preclinical formulations of the Company’s drug discovery programs. Provectus’s pharmaceutical-grade RBS displays different therapeutic effects at different concentrations and can be formulated for delivery by different routes of administration. The International Nonproprietary Names Expert Committee of the World Health Organization selected “rose bengal sodium” for the nonproprietary name of the Company’s API.
RBS may target disease in a bifunctional manner. Direct contact may lead to cell death or repair depending on the disease being treated and the concentration of Provectus’s RBS utilized in the treatment. Multivariate immune signaling, activation, and response may follow that may manifest as stimulatory, inhibitory, or both.
The Company believes that it is the first entity to advance an RBS formulation into clinical trials for the treatment of a disease. Provectus believes that it is the first and only entity to date to make pharmaceutical-grade RBS successfully, reproducibly, and consistently at a purity of nearly 100%.
Provectus’s small molecule HX medical science platform includes clinical development programs in oncology, dermatology, and ophthalmology; proof-of-concept in vivo development programs in oncology, hematology, wound healing, and animal health; and in vitro drug discovery programs in infectious diseases and tissue regeneration and repair.
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://ClinicalTrials.gov. For additional information about Provectus, please visit the Company’s website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of:
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Plus Updates Financial and Cash Guidance for 2024
Plus Receives $3.3M in grant revenue in Q4 2023
Forecasts additional $6.9M of grant revenue for 2024
Enters into debt restructuring deal with Pershing Capital to reduce 2024 cash burn by $3.7M
AUSTIN, Texas, Dec. 18, 2023 (GLOBE NEWSWIRE) -- Plus Therapeutics, Inc. (Nasdaq: PSTV) (the “Company”), a clinical-stage pharmaceutical company developing targeted radiotherapeutics with advanced platform technologies for central nervous system (CNS) cancers, today announced financial updates and guidance for 2024.
Plus is currently in the second year of a Cancer Prevention and Research Institute of Texas (CPRIT) award totaling $17.8 million. The grant funds are advances on future spend and received based on hitting certain milestones. The $3.3 million payment is the first advance for the second year of the award. The Company forecasts an additional $6.9 million in grant revenue during the 2024 calendar year.
Furthermore, the Company currently has a debt facility with Oxford Finance LLC. The remaining balance of the facility is $0.7 million as of YE 2023 with a $3.3 million deferred interest payment due in June 2024. The new facility carries a lower interest rate and principal payment flexibility allowing for principal payments to be deferred into 2025 and beyond and is expected to close in January 2024.
“This combination of forecasted non-dilutive grant revenue and debt restructuring significantly strengthens our balance sheet and reduces our burn,” said Andrew Sims, Chief Financial Officer of Plus Therapeutics. “Following closure of the debt restructure, the Company should have capital sufficient to extend the runway to the second half of 2025.”
To date, the Company has received $7 million from CPRIT and, in 2024, anticipates receiving an additional $6.9 million, $3.3 million in Q2 and $3.6 million in Q4, as part of the planned cadence of award payments. The Company reported $11.0 million in cash as of September 30, 2023. The Company believes that the award payment together with its cash on hand provide sufficient runway to fund operations to the second half of 2025.
Plus’ grant from CPRIT provides for funding over the three-year grant period which began on August 31, 2022, and covers the majority of the development costs of the Company’s lead investigational targeted radiotherapeutic, rhenium (186Re) obisbemeda, for the treatment of patients with leptomeningeal metastases (LM).
About Plus Therapeutics
Plus Therapeutics, Inc. is a clinical-stage pharmaceutical company developing targeted radiotherapeutics for difficult-to-treat cancers of the central nervous system with the potential to enhance clinical outcomes for patients. Combining image-guided local beta radiation and targeted drug delivery approaches, the Company is advancing a pipeline of product candidates with lead programs in recurrent glioblastoma (GBM) and leptomeningeal metastases (LM). The Company has built a robust supply chain through strategic partnerships that enable the development, manufacturing and future potential commercialization of its products. Plus Therapeutics is led by an experienced and dedicated leadership team and has operations in key cancer clinical development hubs including Austin and San Antonio, Texas. For more information, visit https://plustherapeutics.com/.
About the Cancer Prevention & Research Institute of Texas (CPRIT)
CPRIT was created by the Texas Legislature and approved by a statewide vote in 2007 to lead the Lone Star State’s fight against cancer. In 2019, Texas voters again voted overwhelmingly to continue CPRIT with an additional $3 billion for a total $6 billion investment in cancer research and prevention.
To date, CPRIT has awarded over $3 billion in grants to Texas research institutions and organizations through its academic research, prevention and product development research programs. CPRIT has recruited 281 distinguished researchers, supported the establishment, expansion or relocation of 52 companies to Texas and generated over $7.66 billion in additional public and private investment. CPRIT funding has advanced scientific and clinical knowledge and provided 8.2 million life-saving cancer prevention and early detection services reaching Texans from all 254 counties. Learn more at cprit.state.tx.us.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains statements that may be deemed “forward-looking statements” within the meaning of U.S. securities laws. All statements in this press release other than statements of historical fact are forward-looking statements. These forward-looking statements may be identified by future verbs, as well as terms such as “designed to,”, “forecast”, “believes”, “will,” “can,” “potential,” “focus,” “preparing,” “next steps,” “possibly,” and similar expressions or the negatives thereof. Such statements are based upon certain assumptions and assessments made by management in light of their experience and their perception of historical trends, current conditions, expected future developments and other factors they believe to be appropriate. These statements include, without limitation, statements regarding the following: the potential promise of 186Re including the ability of 186Re to safely and effectively deliver radiation directly to the tumor at high doses; expectations as to the Company’s future performance including the next steps in developing the Company’s current assets; the Company’s clinical trials including statements regarding the timing and characteristics of the ReSPECT-GBM and ReSPECT-LM clinical trials; the anticipated completion of the ReSPECT-GBM Phase 2 enrollment; the continuation of the ReSPECT-GBM phase 1 trial to maximum tolerated dose and the next phase of the program; the continued evaluation of rhenium (186Re) obisbemeda including through evaluations in additional patient cohorts; and the intended functions of the Company’s platform and expected benefits from such functions.
The forward-looking statements included in this press release are subject to a number of risks and uncertainties that may cause actual results to differ materially from those discussed in such forward-looking statements. These risks and uncertainties include, but are not limited to: the Company’s actual results may differ, including materially, from those anticipated in these forward-looking statements as a result of various factors, including, but not limited to, the following: the early stage of the Company’s product candidates and therapies, the results of the Company’s research and development activities, including uncertainties relating to the clinical trials of its product candidates and therapies; the Company’s liquidity and capital resources and its ability to raise additional cash, the outcome of the Company’s partnering/licensing efforts, risks associated with laws or regulatory requirements applicable to it, market conditions, product performance, litigation or potential litigation, and competition within the cancer diagnostics and therapeutics field, among others; and additional risks described under the heading “Risk Factors” in the Company’s Securities and Exchange Commission filings, including in the Company’s annual and quarterly reports. There may be events in the future that the Company is unable to predict, or over which it has no control, and its business, financial condition, results of operations and prospects may change in the future. The Company assumes no responsibility to update or revise any forward-looking statements to reflect events, trends or circumstances after the date they are made unless the Company has an obligation under U.S. federal securities laws to do so.
Investor Contact
Corey Davis, Ph.D.
LifeSci Advisors
(212) 215-2577
mailto://cdavis@lifesciadvisors.com
NEWS -- Tokens.com Announces Filing Timeline for 2023 Annual Financial Statements
TORONTO, December 15, 2023--(BUSINESS WIRE)--Tokens.com Corp. (CBOE Exchange Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a technology company that builds immersive experiences in 3D internet environments and owns an inventory of cryptocurrencies, today announced that it expects to complete and file its 2023 Annual Filings (as defined below) on or before January 31, 2024. Because the 2023 Annual Filings will be filed following the requisite filing date of December 29, 2023, the Company has applied to the Ontario Securities Commission (the "OSC") for a management cease trade order that will prohibit the Chief Executive Officer and the Chief Financial Officer of the Company from trading in the securities of the Company until such time as the 2023 Annual Filings have been filed. The OSC has yet to make a decision on the aforementioned application.
The default is due primarily to delays on account of the Company’s recent change in auditor (which change was made effective as of August 23, 2023). The change in auditor has resulted in additional procedures and enhanced quality controls which has necessitated a request by the auditor for more time to complete the audit. The proposed filing timeline will provide the Company's recently appointed auditor, Davidson & Company LLP, with sufficient time to complete the audit. Management notes that there are no disagreements or major unresolved issues with the auditor on any matter of the audit scope or procedures, accounting principles or policies, or financial statement disclosure.
The annual filings consist of: (a) the audited financial statements of the Company for the twelve-month period ended September 30, 2023; (b) the management's discussion & analysis for the twelve-month period ended September 30, 2023; and (c) the chief executive officer and chief financial officer certification of filings under Section 5.1 of National Instrument 52-109 – Certification of Disclosure in Issuers' Annual and Interim Filings (collectively, the "2023 Annual Filings").
During the period of default and until filing of the 2023 Annual Filings, the Company intends to satisfy the provisions of the alternative information guidelines as required by National Policy 12-203 – Management Cease Trade Orders. The Company’s management and other insiders are subject to a trading blackout reflecting the principles contained in Section 9 of National Policy 11-207 – Failure-to-File Cease Trade Orders and Revocations in Multiple Jurisdictions until the 2023 Annual Filings have been filed.
The Company is not subject to any insolvency proceedings, and, other than as disclosed in the Company’s press releases, there has been no material information that has not been disclosed.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies. Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite. Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms. Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names. All our businesses are tied together by the utilization of blockchain technology and are linked to high-growth macro trends within web3. Through sharing resources and infrastructure across these business segments, Tokens.com is able to ef?ciently incubate these businesses from inception to revenue. Visit Tokens.com to learn more. Keep up-to-date on Tokens.com developments and join our online communities on X, LinkedIn, and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identi?ed by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities ?lings available at www.sedarplus.ca. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231215400259/en/
Contacts
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
Media Contact: Ali Clarke – Talk Shop Media
Email: mailto://ali@talkshopmedia.com
NEWS -- Tokens.com Announces Filing Timeline for 2023 Annual Financial Statements
TORONTO, December 15, 2023--(BUSINESS WIRE)--Tokens.com Corp. (CBOE Exchange Canada: COIN) (Frankfurt Stock Exchange: 76M) (OTCQB US: SMURF) ("Tokens.com" or the "Company"), a technology company that builds immersive experiences in 3D internet environments and owns an inventory of cryptocurrencies, today announced that it expects to complete and file its 2023 Annual Filings (as defined below) on or before January 31, 2024. Because the 2023 Annual Filings will be filed following the requisite filing date of December 29, 2023, the Company has applied to the Ontario Securities Commission (the "OSC") for a management cease trade order that will prohibit the Chief Executive Officer and the Chief Financial Officer of the Company from trading in the securities of the Company until such time as the 2023 Annual Filings have been filed. The OSC has yet to make a decision on the aforementioned application.
The default is due primarily to delays on account of the Company’s recent change in auditor (which change was made effective as of August 23, 2023). The change in auditor has resulted in additional procedures and enhanced quality controls which has necessitated a request by the auditor for more time to complete the audit. The proposed filing timeline will provide the Company's recently appointed auditor, Davidson & Company LLP, with sufficient time to complete the audit. Management notes that there are no disagreements or major unresolved issues with the auditor on any matter of the audit scope or procedures, accounting principles or policies, or financial statement disclosure.
The annual filings consist of: (a) the audited financial statements of the Company for the twelve-month period ended September 30, 2023; (b) the management's discussion & analysis for the twelve-month period ended September 30, 2023; and (c) the chief executive officer and chief financial officer certification of filings under Section 5.1 of National Instrument 52-109 – Certification of Disclosure in Issuers' Annual and Interim Filings (collectively, the "2023 Annual Filings").
During the period of default and until filing of the 2023 Annual Filings, the Company intends to satisfy the provisions of the alternative information guidelines as required by National Policy 12-203 – Management Cease Trade Orders. The Company’s management and other insiders are subject to a trading blackout reflecting the principles contained in Section 9 of National Policy 11-207 – Failure-to-File Cease Trade Orders and Revocations in Multiple Jurisdictions until the 2023 Annual Filings have been filed.
The Company is not subject to any insolvency proceedings, and, other than as disclosed in the Company’s press releases, there has been no material information that has not been disclosed.
About Tokens.com
Tokens.com is a technology company that builds products and services that connects brands to consumers in 3D internet environments, including popular gaming and metaverse platforms. The Company also earns revenues through staking an inventory of owned cryptocurrencies. Our solutions give our clients a more engaging way to connect with their existing clients, and access to a new generation of consumers who spend time on platforms such as Roblox and Fortnite. Our key areas of focus are: i) branded virtual stores, sales centres, and 3D branded content, ii) web2 and web3 game development and game analytics, and ii) innovative e-commerce solutions that integrate shopping into existing popular gaming platforms. Tokens.com also manages an inventory of valuable cryptocurrency, digital real estate, and a collection of top ranked crypto related domain names. All our businesses are tied together by the utilization of blockchain technology and are linked to high-growth macro trends within web3. Through sharing resources and infrastructure across these business segments, Tokens.com is able to ef?ciently incubate these businesses from inception to revenue. Visit Tokens.com to learn more. Keep up-to-date on Tokens.com developments and join our online communities on X, LinkedIn, and YouTube.
Forward-Looking Statements
This news release includes certain forward-looking statements as well as management’s objectives, strategies, beliefs and intentions. Forward looking statements are frequently identi?ed by such words as "may", "will", "plan", "expect", "anticipate", "estimate", "intend" and similar words referring to future events and results. Forward-looking statements are based on the current opinions and expectations of management. All forward-looking information is inherently uncertain and subject to a variety of assumptions, risks and uncertainties, including the speculative nature of cryptocurrencies, as described in more detail in our securities ?lings available at www.sedarplus.ca. Actual events or results may differ materially from those projected in the forward-looking statements and we caution against placing undue reliance thereon. We assume no obligation to revise or update these forward-looking statements except as required by applicable law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231215400259/en/
Contacts
Andrew Kiguel, CEO
Telephone: +1-647-578-7490
Email: mailto://contact@tokens.com
Jennifer Karkula, Head of Communications
Email: mailto://contact@tokens.com
Media Contact: Ali Clarke – Talk Shop Media
Email: mailto://ali@talkshopmedia.com
NEWS -- FuelPositive Update: 2023's Milestones Achieved - 2024 Path Forward. Additional Capital Secured
WATERLOO, Ontario, Dec. 15, 2023 (GLOBE NEWSWIRE) -- FuelPositive Corporation, a leading Green Ammonia company, (TSX.V: NHHH) (OTCQB: NHHHF) (the “Company” or “FuelPositive”) provides a recap of 2023 Milestones Achieved, Highlights for 2024, as well as details on securing further financing and the closing of the First Tranche of $1,350,000, in an active private placement.
The ammonia industry is currently one of the world's most polluting sectors. Grey ammonia production depends on fossil fuels and follows the oil industry as one of the most carbon-intensive processes.
Grey ammonia is produced in massive, complicated, centralized plants and must be transported worldwide to the end-user through complex and polluting distribution networks. 80% of ammonia is used today as essential nitrogen fertilizer on farms. In light of global conflict, climate change and shifting politics, this antiquated mode of grey ammonia production and distribution is unsustainable, which creates uncertainty in supply and price for the end-user, mainly farmers.
FuelPositive's patent-pending, 3rd party validated breakthrough technology supports farmers' independence and needed change. The Company’s modular, containerized, Green Ammonia on-site system enables farmers to produce their own fertilizer and fuel, simply powered by green electricity. This decentralized solution is scalable and provides farmers with price and supply certainty.
By producing Green Ammonia on site, FuelPositive customers eliminate up to 4 tonnes of CO2e emissions per tonne of Green Ammonia produced!
In short, FuelPositive technology has the potential to create a lasting positive impact on farmer's health and finances and ultimately, on global food security.
2023 Summary:
First System Milestones:
NEWS -- NightHawk Biosciences Announces Strategic Shift into a Pure-Play Large Molecule Biomanufacturing CDMO to Capitalize on Rapid Growth in Sales Pipeline along with Divestiture of Non-Core Assets
DURHAM, N.C., Dec. 12, 2023 (GLOBE NEWSWIRE) -- NightHawk Biosciences (NYSE American: NHWK) today (“NightHawk” or the “Company”) announced its strategic shift into a pure-play biomanufacturing Contract Development & Manufacturing Organization (CDMO), as well as the divestiture of certain non-core assets, in order to focus on the growing sales within its Scorpius BioManufacturing subsidiary (“Scorpius”). The Company has booked contracts thus far in 2023, Scorpius’ first full year of operation, that are expected to generate over $20 million in revenue, a substantial portion of which will be recognized in 2024. To facilitate this strategic shift, the Company has divested its Elusys subsidiary and related assets, and has eliminated most of its R&D and associated expenses to focus its resources on growing and expanding Scorpius.
The assets will be acquired by a private company established by Jeff Wolf, CEO of NightHawk, which will assume Elusys’ contracted financial commitments, currently estimated at over $40 million. Under the terms of the transaction, the private company will also provide an upfront payment of $500,000, a note for $2.25 million that is convertible into shares of NightHawk’s common stock subject to receipt of stockholder approval, and royalties of 3% on all of Elusys’ gross sales until June 30, 2031. The royalties are subject to a mandatory minimum payment of $5 million during the first five years of the royalty term. The divestiture of Elusys and R&D cuts are expected to reduce the Company’s annual operating expenses by over $13 million and fully eliminate the need for NightHawk to raise capital to support Elusys’ programs.
Jeff Wolf, Chief Executive Officer of NightHawk and Elusys following the closing, commented, “Given the strength of our CDMO operations at Scorpius, we have made the strategic decision to refocus our efforts around those activities that hold the potential to generate meaningful cash flow while substantially reducing non-core costs and associated overhead. Scorpius booked $3 million in contracted sales in 2022, which has grown to over $20 million in contracted sales thus far in 2023. As contracted sales are generally recognized as revenue as work is performed, we expect to recognize substantial revenue on these booked contracts in 2024. We also have a very significant and growing sales pipeline, which includes contracts from premier pharma and biotech companies, as well as leading research institutes. We expect to close a number of new contracts before year-end and believe the growth in our sales pipeline is strong validation of the meaningful investments we have made in our Scorpius subsidiary. As we continue to grow our revenue and leverage our fixed costs, we expect our CDMO business to generate high margins and long-term profitability.”
Mr. Wolf added, “There is a significant shortage of clinical scale biologic manufacturing capacity within the industry as a result of growing demand for large molecule CDMO services. In turn, this has led to significant M&A activity within the sector. We believe we are extremely well positioned to capitalize on these trends given our state-of-the-art facility, experienced and dedicated team, broad service offering, and robust sales pipeline.”
John Prendergast, Ph.D., NightHawk’s lead director stated, “The divestiture of our non-core assets enables NightHawk to focus its resources on growing its Scorpius subsidiary, and will result in the immediate elimination of approximately $40 million of contractual obligations and reduction of over $13 million in operating expenses per year. Moreover, NightHawk will receive an upfront cash payment, as well as a $2.25 million convertible loan and at least $5 million in royalties on future Elusys gross payment receipts for approximately 7.5 years. This was vetted and approved by an independent committee of the board to be in the best long-term interests of NightHawk and its shareholders. As a pure-play CDMO, the board of NightHawk believes that NightHawk now has the opportunity to create substantial shareholder value in an underserved marketplace.”
Mr. Wolf further noted, “The senior management team and board of directors are laser-focused on transforming our company into a revenue-generating CDMO. We have shuttered most all of our research, substantially cut costs and divested our non-core assets to achieve this. Our goals are clear; provide the highest quality manufacturing services to our CDMO customers and focus on driving near and long-term revenue. We believe this strategic shift and divestiture of non-core assets will not only enhance our balance sheet and cashflow, but also drive meaningful returns to our shareholders for years to come.”
NightHawk Biosciences, Inc.
NightHawk Biosciences, through its Scorpius BioManufacturing subsidiary, is an integrated contract development and manufacturing organization (CDMO) focused on rapidly advancing biologic and cell therapy programs to the clinic and beyond. Scorpius offers a broad array analytical testing, process development, and manufacturing services to pharmaceutical and biotech companies at its state-of-the art facilities in San Antonio, TX. With an experienced team and new, purpose-built U.S. facilities, Scorpius is dedicated to transparent collaboration and flexible, high-quality biologics biomanufacturing. For more information, please visit: https://www.nighthawkbio.com or https://www.scorpiusbiologics.com, and also follow us on Twitter.
Forward Looking Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. In some cases forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions, and include statements such as booking over $20 million in booked contracts in 2023 alone; the Elusys divestiture and research and development cuts to eliminating over $40M of commitments and reducing annual operating expenses by more than $13 million; recognizing substantial revenue on contracts next year; closing a number of new contracts before year-end; the growth in the Company’s sales pipeline being strong validation of the meaningful investments it has made in its Scorpius subsidiary; the CDMO business generating high margins and achieving long-term profitability; being extremely well positioned to capitalize on trends given the Company’s state-of-the-art facility, broad service offering, and robust sales pipeline; the the reduction in personnel and other expenses substantially reducing the Company’s operating expenses; transforming the Company into a revenue-generating CDMO; providing the highest quality manufacturing services to CDMO customers and driving near and long-term revenue; and the strategic shift into a pure-play, large molecule CDMO and divestiture of non-core assets, enhancing the Company’s balance sheet and cashflow and also helping to drive meaningful returns for shareholders for years to come. Important factors that could cause actual results to differ materially from current expectations include, among others, the Company’s ability to generate over $20 million in future revenue from manufacturing contracts booked in 2023; the Company’s ability to close on a number of new CDMO contracts before year-end; the Company’s ability to continue to grow revenue, leverage fixed costs and achieve long-term profitability; the Company’s ability to create substantial shareholder value as a pure-play CDMO in an underserved marketplace; the Company’s financing needs, its cash balance being sufficient to sustain operations and its ability to raise capital when needed, the Company’s ability to successfully operate as a CDMO, he continued maintenance and growth of the Company’s and its subsidiaries’ patent estates the ability to obtain regulatory approval or to comply with ongoing regulatory requirements, regulatory limitations relating to the Company’s ability to successfully promote its services and compete as a pure- play CDMO, and other factors described in the Company’s annual report on Form 10-K for the year ended December 31, 2022, subsequent quarterly reports on Form 10-Qs and any other filings the Company makes with the SEC. The information in this presentation is provided only as of the date presented, and the Company undertakes no obligation to update any forward-looking statements contained in this presentation on account of new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
mailto://investorrelations@nighthawkbio.com
NEWS -- Plus Therapeutics Partners With K2bio for Development of Novel Tests for Cerebrospinal Fluid (CSF) Tumor Cell and Molecular Biomarker Analyses
Collaboration to explore novel CSF cancer testing modalities and initial testing panel to be available in Q1 2024 for ReSPECT-LM patients
AUSTIN, Texas, Dec. 12, 2023 (GLOBE NEWSWIRE) -- Plus Therapeutics, Inc. (Nasdaq: PSTV) (the “Company”), a clinical-stage pharmaceutical company developing targeted radiotherapeutics with advanced platform technologies for central nervous system (CNS) cancers, today announced that it has partnered with K2bio (Houston, Texas) to implement novel analysis for cerebrospinal fluid (CSF) tumor and molecular biomarkers for CNS cancers. Initial clinical specimen processing and testing will begin in Q1 2024 in the Company’s ongoing Phase 1 ReSPECT-LM trial of rhenium (186Re) obisbemeda in patients with leptomeningeal metastases (LM). This trial is currently receiving grant funding through The Cancer Prevention and Research Institute of Texas (CPRIT). The Company expects testing costs to be partially covered under this grant.
“The initial diagnosis, therapeutic selection and monitoring of patients with CNS cancers such as leptomeningeal metastases are significant problems in everyday clinical practice,” said Marc H. Hedrick, M.D., President & Chief Executive Officer of Plus Therapeutics. “To turn these very lethal CNS cancers into treatable diseases, we must ideally address both the diagnostic as well as the therapeutic needs of caregivers and patients. This partnership with K2bio is the next part of our overall strategy to address both needs in parallel.”
K2bio is a hybrid contract research organization (CRO) enabling life science companies to develop the next generation of innovative therapies. K2bio is based in Houston and is part of the Texas Medical Center life sciences ecosystem specializing in all aspects of translational cancer diagnostic and therapeutic research and development.
“K2bio is a leader in enabling rapid diagnostic and therapeutic progress for innovative companies such as Plus,” said Colby Suire, PhD, acting President and CEO of K2bio. “We have all the necessary capabilities and expertise to accelerate and support Plus’ mission to be a leader in the development of targeted radiotherapeutics and related diagnostics for CNS cancers.”
Plus’ tumor cell and molecular biomarker analysis is an exploratory endpoint in the ReSPECT-LM Phase 1 trial that has shown promise in early cohorts. In Phase 1/part A of the ReSPECT-LM trial presented at the 2023 SNO/ASCO Meeting in San Francisco, Plus data showed an average 53% reduction in CSF tumor cells 28 days after a single intrathecal administration of rhenium (186Re) obisbemeda in patients with LM.
The FDA has granted Fast Track designation to rhenium (186Re) obisbemeda for the treatment of LM, and the ReSPECT-LM Phase 1 program continues to be funded in part by a 3-year $17.6 million grant from the Cancer Prevention & Research Institute of Texas (CPRIT). Patients interested in learning more about the ReSPECT-LM trial can visit ClinicalTrials.gov (NCT05034497).
About Plus Therapeutics
Plus Therapeutics, Inc. is a clinical-stage pharmaceutical company developing targeted radiotherapeutics for difficult-to-treat cancers of the central nervous system with the potential to enhance clinical outcomes for patients. Combining image-guided local beta radiation and targeted drug delivery approaches, the Company is advancing a pipeline of product candidates with lead programs in recurrent glioblastoma (GBM) and leptomeningeal metastases (LM). The Company has built a robust supply chain through strategic partnerships that enable the development, manufacturing and future potential commercialization of its products. Plus Therapeutics is led by an experienced and dedicated leadership team and has operations in key cancer clinical development hubs including Austin and San Antonio, Texas. For more information, visit https://plustherapeutics.com/.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains statements that may be deemed “forward-looking statements” within the meaning of U.S. securities laws. All statements in this press release other than statements of historical fact are forward-looking statements. These forward-looking statements may be identified by future verbs, as well as terms such as “designed to,” “will,” “can,” “potential,” “focus,” “preparing,” “next steps,” “possibly,” and similar expressions or the negatives thereof. Such statements are based upon certain assumptions and assessments made by management in light of their experience and their perception of historical trends, current conditions, expected future developments and other factors they believe to be appropriate. These statements include, without limitation, statements regarding the following: the potential promise of 186Re including the ability of 186Re to safely and effectively deliver radiation directly to the tumor at high doses; expectations as to the Company’s future performance including the next steps in developing the Company’s current assets; the Company’s clinical trials including statements regarding the timing and characteristics of the ReSPECT-GBM and ReSPECT-LM clinical trials; the anticipated completion of the ReSPECT-GBM Phase 2 enrollment; the continuation of the ReSPECT-GBM phase 1 trial to maximum tolerated dose and the next phase of the program; the continued evaluation of rhenium (186Re) obisbemeda including through evaluations in additional patient cohorts; and the intended functions of the Company’s platform and expected benefits from such functions.
The forward-looking statements included in this press release are subject to a number of risks and uncertainties that may cause actual results to differ materially from those discussed in such forward-looking statements. These risks and uncertainties include, but are not limited to: the Company’s actual results may differ, including materially, from those anticipated in these forward-looking statements as a result of various factors, including, but not limited to, the following: the early stage of the Company’s product candidates and therapies, the results of the Company’s research and development activities, including uncertainties relating to the clinical trials of its product candidates and therapies; the Company’s liquidity and capital resources and its ability to raise additional cash, the outcome of the Company’s partnering/licensing efforts, risks associated with laws or regulatory requirements applicable to it, market conditions, product performance, litigation or potential litigation, and competition within the cancer diagnostics and therapeutics field, among others; and additional risks described under the heading “Risk Factors” in the Company’s Securities and Exchange Commission filings, including in the Company’s annual and quarterly reports. There may be events in the future that the Company is unable to predict, or over which it has no control, and its business, financial condition, results of operations and prospects may change in the future. The Company assumes no responsibility to update or revise any forward-looking statements to reflect events, trends or circumstances after the date they are made unless the Company has an obligation under U.S. federal securities laws to do so.
Investor Contact
Corey Davis, Ph.D.
LifeSci Advisors
(212) 215-2577
mailto://cdavis@lifesciadvisors.com
NEWS -- Oncolytics Biotech® and SOLTI Present Positive Translational Data at SABCS
SAN DIEGO and CALGARY, AB, Dec. 7, 2023 /CNW/ -- Oncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, today announced the presentation of additional translational data from the AWARE-1 breast cancer window-of-opportunity study conducted in combination with SOLTI-Innovative Cancer Research at The San Antonio Breast Cancer Symposium (SABCS).
"Translational data presented from the AWARE-1 study at SABCS showed that pelareorep induced the expansion of tumor-infiltrating lymphocytes, or TILs, in matched tumor biopsy and peripheral blood samples of newly diagnosed breast cancer patients. In addition, sequencing of the T cell receptors (TCRs) showed a more prominent expansion of existing TIL clones in the blood. We consider these results to be positive and important because they build on additional translational results reported this fall at two other medical meetings, The Society for Immunotherapy of Cancer (SITC) and The European Society for Medical Oncology (ESMO), from the AWARE-1 and GOBLET studies respectively, and provide further support for pelareorep's unique immunologic mechanism of action," said Dr. Matt Coffey, President and Chief Executive Officer of Oncolytics. "Taken together, these translational data affirm pelareorep's ability to enhance T cell infiltration into tumors and expand TILs in the peripheral blood, which have been correlated with tumor response. We intend to incorporate these learnings into the designs of our registrational studies in metastatic breast cancer and pancreatic cancer."
Thomas Heineman, M.D., Ph.D., Chief Medical Officer at Oncolytics, commented, "A review of the AWARE-1 study translational data presented at this year's SABCS and previous scientific conferences demonstrates that pelareorep treatment, especially in combination with atezolizumab, results in:
NEWS -- FuelPositive Update: The First On-Farm Green Ammonia System
WATERLOO, Ontario, Dec. 05, 2023 (GLOBE NEWSWIRE) -- FuelPositive Corporation (TSX.V: NHHH) (OTCQB: NHHHF) (the “Company” or “FuelPositive”) is excited to announce the official delivery date for its first on-farm, containerized, Green Ammonia demonstration system, the FP300. On March 31st2024 the Company anticipates the world’s first, on-demand, modular, Green Ammonia fertilizer and fuel plant to be delivered and operational on the Hiebert’s farm in Manitoba, Canada.
The FP300 is destined to become the first farmer-owned Green Ammonia plant in the world. The first system will support optimizing of future versions of this transformative technology. This new approach to farming has the potential of offering reliable supply and cost in addition to eliminating up to 4 tonnes of atmospheric polluting CO2e emissions for every tonne of Green Ammonia produced. Ammonia supply and cost are the second most challenging risk factors for many farmers after the weather.
FuelPositive recently signed a lease in Waterloo, Ontario, for a twelve thousand square foot manufacturing site to accommodate the production of the following 30 systems. The delivery of these systems is expected throughout 2024 and 2025.
Nelson Leite, COO and Board Member, commented, “We have entered the final stage of the pre-shipment preparation. Our team have witnessed the system running at full rate and can’t wait to see it installed and producing Green Ammonia on farm. We are extremely proud of our incredible team efforts and accomplishments.”
Leite continued, “The breakthrough FP300 is a pinnacle of engineering and technological innovation, and our stakeholders and customers who have already seen our system are excited for the positive change that it represents for people and industries across many different sectors.”
Ian Clifford, CEO and Board Chair, concluded, “This is a dream come true for all of us, a potential game changer for global food security, and we’re just getting started!”
About FuelPositive Corporation
FuelPositive is a Canadian technology company dedicated to delivering commercially feasible and sustainable clean technology solutions that follow a circular approach, ensuring the entire lifecycle of our products is environmentally friendly. This includes an on-farm/onsite, containerized Green Ammonia (NH3) production system that effectively eliminates carbon emissions during the production process.
By focusing on technologies that are clean, sustainable, economically advantageous and realizable, the Company aims to help mitigate climate change, addressing unsustainable agricultural practices through innovative technology and practical solutions that can be implemented now. The FuelPositive on-farm/onsite, containerized Green Ammonia production system is designed to produce pure, anhydrous ammonia for multiple applications, including fertilizer for farming, fuel for grain drying and internal combustion engines, a practical alternative for fuel cells and a solution for grid storage. Green Ammonia is also considered a key enabler of the hydrogen economy.
FuelPositive systems are designed to provide for Green Ammonia production on-farm/onsite, where and when needed. This eliminates wildly fluctuating supply chains and offers end-users clean fertilizer, energy and Green Ammonia supply security while eliminating carbon emissions from the production process. The first customers will be farmers. Farmers use 80% of the traditional grey ammonia produced today as fertilizer.
See pre-sale details here: https://fuelpositive.com/pre-sales/.
FuelPositive Corporation is based in Waterloo (Canada) and trades on the TSX Venture Exchange under the symbol NHHH and in the USA on the OTCQB under the symbol NHHHF.
For further information, please contact:
Ian Clifford
Chief Executive Officer and Board Chair
mailto://Ian@fuelpositive.com
Tel: 416.535.8395
https://www.fuelpositive.com
Investor Relations United States & International:
RB Milestone Group (RBMG)
mailto://fuelpositive@rbmilestone.com
Investor Relations Canada:
Transcend Capital
mailto://et@transcendcapitalinc.com
Cautionary Statement
Trading in the securities of the Company should be considered highly speculative. No stock exchange, securities commission or other regulatory authority has approved or disapproved the information contained herein. Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accept responsibility for the adequacy or accuracy of this release.
Forward-Looking Statements
This news release contains certain “forward-looking information” and “forward-looking statements” (collectively, “forward-looking statements”) that are based on expectations, estimates and projections as of the date of this news release. The information in this release about future plans and objectives of the Company, including with respect to further testing and implementation of the FP300 system, are forward-looking statements.
These forward-looking statements are based on assumptions and estimates of management of the Company at the time they were made and involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Forward-looking statements are necessarily based upon a number of estimates and assumptions that, while considered reasonable by the Company as of the time of such statements, are inherently subject to significant business, economic and competitive uncertainties and contingencies. These estimates and assumptions may prove to be incorrect.
Many of these uncertainties and contingencies can directly or indirectly affect and could cause, actual results to differ materially from those expressed or implied in any forward-looking statements. There can be no assurance that forward-looking information will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements.
Forward-looking information is provided for the purpose of providing information about management’s expectations and plans relating to the future. The Company disclaims any intention or obligation to update or revise any forward-looking information or to explain any material difference between subsequent actual events and such forward-looking information, except to the extent required by applicable law.
Insiders buying stock like crazy!!!
