Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
so, any thoughts on the Summit Therapeutics drug trial results? Showing better results than Keytruda? Maybe an opportunity to use PV-10 to help boost Keytruda results when used in combo?
Are they good?
From today’s PR
Third Quarter 2024 Conference Call Plans (September)
Provectus plans to hold a third quarter 2024 conference call, with a Q&A portion, by or before the end of September.
Excellent review! I was disappointed on the timelines, but I think solid progress is being made. I was at the meeting and I think you’re right. The meeting was well received although there is frustration that we’re not moving quicker.
Any thoughts on the annual meeting would be appreciated; including posts from the other boards.
I listened online and thought the overall presentation portion after the formal meeting was good, both Ed and Dom are certainly unique and highly competent investor/officers of the company. Dominic stated the number #1 priority of the company remains the pancreatic/uveal focus though all other clinical applications from other areas were a close number #2; said the company was able to work on multiple fronts without losing sight of overall goal. Wanted the company to clearly become the "expert" resource of all PV10 matters/requests/inquiries. Said progress delay in prior years actually proved to be well spent time as mechanics and capabilities of the molecule are now so much better understood. He was hesitant to give future dates on when initial patients will be dosed in the upcoming trial as he did not want to set false expectations. Made several statements or comments on the need for speed but must be tempered since making uniformed decisions would likely lead to poor outcomes from rushing ahead; something the company can ill afford. Sounds to me they are hearing the shareholders request for progress loud and clear, and have Dominic's support and understanding, but hurdles and unforeseen obstacles are nonetheless in place that make forecasting end dates irresponsible.
Both Dominic and Ed were disappointed about the lack of funding options/pharmaceutical interest available to them at this time, would consider international funding if the right option presented itself. A remark from the audience afterwards even questioned how serious Big Pharma investment would even be for backing a disease "cure" as they were so dependent on selling their maintenance cocktails. Ed believes differently and stated there are good people at these companies and that hopefully the new focus on communications will have a positive impact. Both mentioned a responsibility to the long supporting shareholders so dilution from second tier financing resources fortunately does not seem to a concern. As many of long time shareholders have heard in the past, funding continues to be an ongoing issue. The reverse split is available if needed or would be helpful for the long term growth of the company. They want it as an option on hand but do not have specific plans when/if to use it; NASDAQ listing could eventually be a reason but they are a ways off from accomplishing that.
The Eye company comments were interesting, even though it seems like we are a ways away from having a company established. Awaiting on outcome results from India even though the rose bengal being used is not pharmaceutical grade from PVCT. Then the cost of starting the company will need to be determined. Getting FDA approval for the new company will actually benefit PVCT as the FDA also focuses on clinical grade and the manufacturing environment for approval, and we have the patents in place for our benefit. Additional comments were made on wound healing, canine cancer, anti-infection properties and other such opportunities; all in various stages of initial development.
Dominic listed a multitude of successful achievements in the prior year but it went by to quickly, I will have to view them once posted.
Though I thought the meeting was well received, I can see how some long term shareholders would get frustrated as this was all a bit generic and no time frames were committed to. At the same time, Ed and Dom said they could not be more optimistic, sounds like the industry is starting to take some notice as well. There is no doubt of Ed & Dom's commitment to this company. I will be holding and adding on larger dips in the upcoming year, I think the potential of the molecule warrants that. Hopefully a revenue stream can be forecast at next year's meeting, the company desperately needs an ongoing cash inflow. I remain optimistic but will be back in 'wait and see' mode.
NEWS -- Provectus Biopharmaceuticals Announces Investor Webinar for 2024 Annual Meeting of Stockholders
KNOXVILLE, Tenn., June 13, 2024 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (“Provectus” or the “Company”) (OTCQB: PVCT) announced today that its 2024 annual stockholder meeting (the “2024 Annual Meeting”), including meeting activities and a company update, will be accessible by Zoom Webinar. The meeting will be held on Thursday, June 20, 2024 at the Hilton Knoxville, located at 501 West Church Avenue, Knoxville, Tenn., beginning at 4:00 p.m. Eastern Time.
The 2024 Annual Meeting webinar may be accessed by registering in advance here:
https://us06web.zoom.us/webinar/register/WN_KZTlQulrRCOZ2qQI4C23mQ#/registration.
The webinar is being made available only for informational purposes. The 2024 Annual Meeting is being held in person, and accessing the webinar will neither count as attendance for purposes of meeting quorum requirements nor enable a stockholder to vote. Provectus’s stockholders of record at the close of business on April 24, 2024, the record date for the 2024 Annual Meeting, may vote at the meeting by attending in person or following the instructions in the Company’s proxy materials.
Availability of Proxy Materials
In connection with the 2024 Annual Meeting, Provectus has filed a definitive proxy statement with the Securities and Exchange Commission (the “SEC”) on May 6, 2024, which may be found here:
https://www.sec.gov/Archives/edgar/data/315545/000149315224017882/formdef14a.htm.
The Company mailed a Notice of Internet Availability of Proxy Materials (“Notice”) to its stockholders on or about May 10, 2024. These materials, along with Provectus’s 2023 Annual Report, are available on the Company’s website and may be found here: https://www.provectusbio.com/annual-meeting/.
About Provectus
Provectus Biopharmaceuticals is a clinical-stage biotechnology company developing immunotherapy medicines for different diseases that are based on a class of synthetic bioactive small molecules called halogenated xanthenes. The Company’s lead HX molecule is named Rose Bengal Sodium.
Provectus’s medical science platform includes clinical development programs in oncology, dermatology, and ophthalmology; proof-of-concept in vivo programs in oncology, hematology, full-thickness cutaneous wound healing, and canine cancers; and in vitro discovery programs in infectious diseases, tissue regeneration and repair, and proprietary targets.
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, ClinicalTrials.gov. For additional information about Provectus, please visit the Company’s website at https://www.provectusbio.com.
Forward Looking Statements
The information in this press release may include “forward-looking statements,” within the meaning of the Private Securities Litigation Reform Act of 1995, relating to the business of Provectus and its affiliates, which are based on currently available information and current assumptions, expectations, and projections about future events and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “aim,” “likely,” “outlook,” “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “would,” “project,” “projection,” “predict,” “potential,” “targeting,” “intend,” “can,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the SEC, including those described in Item 1A of:
The Annual Report on Form 10-K for the period ended December 31, 2023, and
The Quarterly Report on Form 10-Q for the period ended March 31, 2024.
Additional Information and Where to Find It
This communication may be deemed to be solicitation material in connection with the proposals to be submitted to the Company’s stockholders at its 2024 Annual Meeting. STOCKHOLDERS ARE URGED TO READ CAREFULLY AND, IN ITS ENTIRETY, THE DEFINITIVE PROXY STATEMENT FILED WITH THE SEC AND OTHER RELEVANT MATERIALS, BECAUSE THEY CONTAIN IMPORTANT INFORMATION ABOUT THE COMPANY AND THE 2024 ANNUAL MEETING PROPOSALS. A Notice with instructions for accessing the definitive proxy statement, 2023 Annual Report, and proxy card was mailed on or about May 10, 2024 to stockholders as of the record date of April 24, 2024. Stockholders may obtain free copies of Provectus’s definitive proxy statement and its other SEC filings electronically by accessing the SEC’s home page at http://www.sec.gov. Copies can also be obtained, free of charge, upon written request to Provectus Biopharmaceuticals, Inc., Attn: Secretary, 800 S. Gay Street, Suite 1610, Knoxville, Tennessee 37929, (866) 594-5999. Copies can also be obtained electronically from the Company’s Annual Meeting webpage, https://www.provectusbio.com/annual-meeting/.
Participants in Solicitation
The Company and its directors, executive officers, and advisors may be deemed to be participants in the solicitation of proxies from the holders of Provectus’s Common Stock, Series D Convertible Preferred Stock, and Series D-1 Convertible Preferred Stock in respect of the 2024 Annual Meeting proposals.
Investors may obtain additional information regarding the interest of those participants by reading the Company’s definitive proxy statement and other relevant proxy materials, and Provectus’s annual reports on Form 10-K and quarterly reports on Form 10-Q, as filed with the SEC.
Contacts:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
(866) 594-5999
Investor Relations & Media
IR Labs Inc. (irlabs)
Alyssa Barry
mailto://alyssa@irlabs.ca
(833) 947-5227
thx Trimble ... I've not yet re-joined IVillage.
here's a post I put up a few hours ago on the IVillage PVCT semi-private and Group Boards, reconstituted here if you don't have access to those boards:
Block trading today
Interesting consecutive block trading this morning between 9:52:11 and 9:53:16 ET, 245,000 then 250,000 then 476,923 then 143,100 then 250,000 shares traded, all at $.19, a total of 1,365,023 shares out of a day total of 1,883,184 shares traded. That left 518,161 shares traded the rest of the day, which would be a fairly heavy day of trading for PVCT outside of the fact that there were 66,000 shares traded pre-market at $.165, $.1711 and $.1721, and then 105,000 shares traded within 1 second after the opening at $.165 and then another 80,000 shares traded at 9:48:22 at $.1899. Throw in a 50,000 share trade at 10:32:16 for $$.196 and then 2 31,322 share trades at $.1901 and $.1902, and you have a grand total of 1,598,667 shares block traded out of that total of 1,883,184 total shares traded.
That left 284,517 shares traded in smaller lots, a more usual pattern for PVCT.
Is someone saying something to us, with this unusual activity?
It happened early today, so somebody definitely took a position.
Interesting pop on volume today. Maybe somebody out there likes what they see. Only $342k transacted or so, but a bit more than lunch money.
Down 3 cents earlier this morning on no news. Just wanted to check in again before lunch, and it is back to almost flat. I understand the need for the MM to have some latitude on price, to make a market, but this is getting ridiculous. Take the price higher for a change.
totally agree... the minute there is some monetization of PV 10 , this stock will be a gold mine
Interesting approach for directed radiation. I like your thought about a possible application for PV-10. Hopefully a clinician can weigh in on the merits. PV-10 seeking out solid tumors throughout the body seems like a potential candidate if it can somehow carry the targeting particle.
Looking forward to the start of an ocular revenue stream next year. This company finally starting to inch forward. Great potential.
take a look at this article - https://www.cnbc.com/2024/05/23/this-up-and-coming-cancer-treatment-could-be-a-25-billion-market-opportunity.html Tell me PV-10 isnt in this potential category?
NEWS -- Provectus Biopharmaceuticals Announces Poster Presentation of Rose Bengal Sodium for Treatment of Full-Thickness Cutaneous Wounds at SID 2024 Annual Meeting
Ed on TV
Ed Pershing talks about being CEO of $PVCT #RoseBengalSodium #RoseBengal @knoxnews https://t.co/rCcuznYI7z
— Provectus Biopharmaceuticals, Inc. (@ProvectusBio) May 18, 2024
From Provectus's most recent substack posting.

this seems so astonishing to me ... we've just got to find a way to get this to the people it can benefit.
Full substack post here.
Can’t seem to break past that .20 point.
NEWS -- Provectus Biopharmaceuticals Announces Eight Presentations of Rose Bengal Photodynamic Antimicrobial Therapy at ARVO 2024 Annual Meeting
PDAT clinical outcomes for infectious keratitis from Brazil and Mexico
KNOXVILLE, TN, May 14, 2024 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (“Provectus” or the “Company”) (OTCQB: PVCT) today announced that clinical and non-clinical data on rose bengal photodynamic antimicrobial therapy (“RB PDAT”) were presented at the annual meeting of the Association for Research in Vision and Ophthalmology (“ARVO”), held May 5-9, 2024 in Seattle, Washington. The ARVO annual meeting brings together approximately 10,000 vision researchers, clinicians, and scientists from around the world for scientific sessions, poster presentations, and invited speaker sessions.
Provectus entered into an exclusive worldwide license agreement with the University of Miami (the “University”) in April 2024 for its intellectual property related to RB PDAT for treating bacterial, fungal, and parasitic (acanthamoeba) eye infections. The Company and University plan to form a start-up biotechnology company (e.g., “Eyecare NewCo”), potentially in the third quarter of 2024, aimed at commercializing the University’s PDAT light source medical device in combination with a drug formulation of Provectus’s proprietary pharmaceutical-grade rose bengal active pharmaceutical ingredient (“RB API”). The Company would contribute the RB PDAT license to and have an exclusive RB API supply arrangement with Eyecare NewCo.
RB PDAT emerged under the leadership of Jean-Marie Parel, IngETS-G, Ph.D., FARVO, Director of Bascom Palmer Eye Institute’s (“BPEI’s”) Ophthalmic Biophysics Center at the University of Miami Miller School of Medicine. Dr. Parel and the OBC team have spent more than ten years advancing RB PDAT technology against different types of infectious keratitis.
ARVO 2024 RB PDAT presentations included:
@kiddlesnmore2
3 weeks ago
1) Powdered eggs
2) Powdered milk
3) Powdered whey protein
4) Powdered soups
5) Powdered Cheese
6) Powdered peanut butter
7) Powdered fruits
8) Powdered vegetables
Bascon Palmer has 4 papers related to PV-10 at the ARVO conference this week
Corneal epithelium
B0448: Evaluating the Safety of Rose Bengal Photodynamic Therapy 227
Jordan Huang, Esdras A. Arrieta, Andrea Naranjo, Pedro Monsalve, Keenan Mintz, Jeffrey C. Peterson, Alejandro Arboleda, Heather Ann Durkee, Mariela C. Aguilar, Sander R. Dubovy, Darlene Miller, Roger Leblanc, Guillermo Amescua, Jean-Marie A. Parel, Jaime D. Martinez-Martinez
Keratitis
B0580: Title: Enhanced Fungal Inhibition with High-Dose Rose Bengal Photodynamic Antimiocrobial Therapy 372
Salomon Merikansky, Heather Ann Durkee, Juan Carlos Navia, Felipe Echeverri Tribin, Katherine Krishna, Anam Ahmed, Alex Gonzalez, Cornelis Rowaan, Harry W. Flynn, Jean-Marie A. Parel, Darlene Miller, Guillermo Amescua
Keratitis
Inhibition of Fungal Isolates via Singlet Oxygen Generation from Erythrosin B and Rose Bengal Photodynamic Antimicrobial Therapy 432
Anam Ahmed, Braulio Carrera Loureiro B Ferreira, Heather Ann Durkee, Lillian Aston, Leonardo Gonzalez, Jessica Mar, Jeffrey C. Peterson, Katherine Krishna, Brandon Chou, Marco Ruggeri, Fabrice Manns, Harry W. Flynn, Guillermo Amescua, Darlene Miller, Roger Leblanc, Jean-Marie A. Parel
Pathobiology of microbial infections
B0010: Exploring the Combination of Rose Bengal Photodynamic Antimicrobial Therapy and Existing Antifungals 503
Katherine Krishna, Brandon Chou, Heather Ann Durkee, Felipe Echeverri Tribin, Anam Ahmed, Jessica Mar, Braulio Carrera Loureiro B Ferreira, James Lai, Roger Leblanc, Harry W. Flynn, Guillermo Amescua, Darlene Miller, Jean-Marie A. Parel
NEWS -- Provectus Biopharmaceuticals Announces Notice of Allowance for U.S. Patent of Oral Administration of Rose Bengal Sodium in Hematology (Leukemia)
KNOXVILLE, TN, May 02, 2024 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (“Provectus” or the “Company”) (OTCQB: PVCT) today announced that the United States Patent and Trademark Office has allowed patent application 17/232,393, titled “Halogenated Xanthene Composition and Method for Treating Hematologic Cancers” (formerly “Composition and Method for Oral Treatment of Leukemia”). The application covers the use of Provectus’s pharmaceutical grade rose bengal sodium (“RBS”) active pharmaceutical ingredient for the single agent or combination therapy treatment of pediatric and adult leukemias. Earlier this year, Provectus announced notices of allowance and award for U.S. patents of RBS in virology, pediatric solid tumor cancers, and vaccines.
Innovate Calgary, the innovation company of the University of Calgary in Alberta, Canada, is a co-assignee and Aru Narendran, MD, PhD, Professor of Pediatrics, Oncology, Biochemistry and Molecular Biology and Physiology and Pharmacology at the University’s Cumming School of Medicine is a co-inventor on all four patent awards.
Dominic Rodrigues, President and Vice Chairman of Provectus’s Board of Directors said, “As we remain focused on advancing intratumoral administered cancer immunotherapy PV-10 towards initial drug approval for metastatic pancreatic cancer, our evidence-based confidence in the potential and possibilities of oral administration of rose bengal sodium for the treatment of disease continues to grow.”
About Provectus
Provectus Biopharmaceuticals, Inc. is a clinical-stage biotechnology company developing immunotherapy medicines for different diseases that are based on a class of synthetic small molecule immuno-catalysts called halogenated xanthenes. Provectus’s lead HX molecule is named rose bengal sodium.
Provectus’s medical science platform includes clinical development programs in oncology, dermatology, and ophthalmology; proof-of-concept in vivo development programs in oncology, hematology, full-thickness cutaneous wound healing, and canine cancers; and in vitro discovery programs in infectious diseases, tissue regeneration and repair, and proprietary targets.
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://ClinicalTrials.gov. For additional information about Provectus, please visit the Company’s website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of:
The Company’s Annual Report on Form 10-K for the period ended December 31, 2023.
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
It’s the same as before except no stock price posted.
I know several who where there that are no longer sure what exists there (IV) as you can no longer see anything but a sign in page. Not going to join something I dont even know what it is anymore... Are you a member there? Has it changed? what is going on there? How the heck are they supposed to get new members when no one knows what they are getting?
interesting substack post for PV
Intralesional Path to Cancer Drug Approval
Fill Factor: the ratio of PV-10 volume to tumor volume to deliver a constant dose of rose bengal sodium per quantity of tumor
IV is subscription based service only now.
Do you need to pay 10 dollars a month to get back on IV now or is it still like it was?
Blue got right back to me and I now have access restored! I'm much impressed, to say the least...
So I'll see everybody going forward on InvestorVillage!
Ralph "Blue" Kidd
Chief Customer Liaison blue@investorvillage.com
504-250-0021
Unfortunately I don't have Blue's email address or other means of making contact since I can't log-in on the site! You got an email address for Blue that you can provide me? Quite frustrating, I must say...
I'm gonna try using Blue@InvestorVillage.com to see if that works...
Send Blue a message
It won't let me sign in using my long time password on my Mac when I use either Safari or Brave browsers. It did allow me to sign in the last time the site was up for a day or two, more than a week ago...
I was able to sign in on my computer and iPhone.
I have been able to sign on using my old password on a Win 10 desktop using the Brave browser. Can not sign on with my Android phone, have not tried my Android tablet.
It looks like IVillage is online again but won't allow sign in with old passwords, anybody have any insight/opinion on what to do to best protect ourselves in this situation? Has anyone gotten any correspondence from them about the situation?
I agree, the current SP is down right silly, and when this takes off it will be amazing for all long term suffering investors lol
When you read thru the last two posts on this board and the latest Provectus Substack posting, you can not help being impressed with the potential of this company. Combine that with the `shareholder value' theme that Dom and Ed constantly align themselves with, I just can not see why we are trading for a couple dimes per share. Collaborations with Moffet Cancer, Univ of Miami/Bascom Palmer, Univ of Calgary, Univ of TN, HD Anderson and others sure think we are worth their time and effort. It feel like something will break in our favor soon. Yes much of this is all pre-trial, but progress is being made.
Identification and In Vivo Validation of Unique Anti-Oncogenic Mechanisms Involving Protein Kinase Signaling and Autophagy Mediated by the Investigational Agent PV-10
Simple Summary
Novel therapeutics are urgently needed for high-risk and refractory solid tumors. Clinical studies have demonstrated the safety and efficacy of PV-10 (10% rose bengal sodium) by intralesional injection in skin cancer. However, this agent has not yet been evaluated for the treatment of various adult solid tumors. The aim of our study was to test PV-10 in breast, colorectal, head and neck, and testicular cancers. Using a combination of in vitro and in vivo experiments, we found that PV-10 exhibits anti-cancer activity against a panel of human cell lines derived from these tumors. Our results support further clinical development of PV-10 for the treatment of solid tumors in adults.
link to webpage with paper
NEWS -- Provectus Biopharmaceuticals Announces Publication of Preclinical Data of Oral Administration of Provectus Rose Bengal for Solid Tumor Cancers
KNOXVILLE, TN, April 18, 2024 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (“Provectus” or the “Company”) (OTCQB: PVCT) today announced that data from preclinical research by the University of Calgary on oral administration of Provectus’s pharmaceutical-grade rose bengal for the treatments of solid tumor cancers were published in the open access journal of oncology Cancers.
Titled Identification and In Vivo Validation of Unique Anti-Oncogenic Mechanisms Involving Protein Kinase Signaling and Autophagy Mediated by the Investigational Agent PV-10, a copy of the University of Calgary’s journal article is available here: https://www.mdpi.com/2072-6694/16/8/1520.
This work has been part of research conducted by Aru Narendran, M.D., Ph.D., professor in the departments of Pediatrics, Oncology, and Biochemistry & Molecular Biology, and members of the Narendran Lab at the University of Calgary’s Cumming School of Medicine in Calgary, Alberta, Canada.
The Narendran Lab provided preclinical proof-of-concept data supporting the efficacy of Provectus’s rose bengal in a panel of adult solid tumors. The Lab identified that the Company’s rose bengal downregulated WNK1 and Wnt signaling. In mice, the Narendran Lab also confirmed the clinical utility of Provectus’s rose bengal by intratumoral (aka intralesional) administration and demonstrated potential utility by oral administration.
Dominic Rodrigues, the Company’s President and Vice Chairman of its Board of Directors stated, “Provectus is focused on initiating an FDA-cleared, lead clinical development program for intratumoral cancer immunotherapy PV-10 to treat hepatic metastatic pancreatic cancer, and pursuing a path to an initial drug approval for PV-10. The Company previously demonstrated its systemic response and systemic immune signaling and activation in clinical settings for metastatic liver cancers.”
Mr. Rodrigues added, “Building on PV-10’s clinical safety, efficacy, and immunotherapeutic profile, the Narendran Lab’s preclinical data suggest that Provectus’s pharmaceutical-grade rose bengal delivered by oral administration may have the potential to treat a larger pool of solid tumor cancers.”
About Provectus
Provectus Biopharmaceuticals, Inc. is a clinical-stage biotechnology company developing immunotherapy medicines for different diseases that are based on a class of synthetic small molecule immuno-catalysts called halogenated xanthenes. Provectus’s lead HX molecule is named rose bengal sodium.
Provectus’s medical science platform includes clinical development programs in oncology, dermatology, and ophthalmology; proof-of-concept in vivo development programs in oncology, hematology, full-thickness cutaneous wound healing, and canine cancers; and in vitro discovery programs in infectious diseases, tissue regeneration and repair, and proprietary targets.
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://ClinicalTrials.gov. For additional information about Provectus, please visit the Company’s website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information provided in this press release may include forward-looking statements, within the meaning of the Private Securities Litigation Reform Act of 1995, relating to the business of Provectus and its affiliates, which are based on currently available information and current assumptions, expectations, and projections about future events and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Such statements are made in reliance on the safe harbor provisions of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements are often, but not always, identified by the use of words such as “aim,” “likely,” “outlook,” “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “would,” “project,” “projection,” “predict,” “potential,” “targeting,” “intend,” “can,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of Provectus’s drug agents and/or their uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and the Company undertakes no obligation to update or revise any forward-looking statements, whether because of new information, future events, or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission, including those described in Item 1A of:
The Company’s Annual Report on Form 10-K for the period ended December 31, 2023.
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
LTG, sure hope you're right about a rocket to da moon soon. And glad to see some IVillagers posting here, let's use this site until IV can recover their site and once again provide a safe posting place for us. I'm not a facebooker but my wife is and asked to join that group, but we see no posts there today so we wonder how active it is anyway. Not that IVillage was all that active, outside of BM who I've generally kept on ignore the past few years!
My best to everybody in PVCT land!!
For all the PVCT people that can not get into IV. We would love to have you join us on our PCVT Facebook chat if you are interested.
Thanks for the advice, getting any IV info is sketchy. Hope you and Wehalls keep posting here. Eye company organization should be disclosed soon, will be nice to have that $ commitment known . . . and behind us. A revenue stream in 12 - 16 months would be awesome.
That site is all the way hacked. I doubt the owners have any control over the site as of now.
I would HIGHLY recommend NOT giving them any payment information and I wouldn't even log into the site using a Username and Password that most have across numerous websites.
We can all assume our UID and passwords have been compromised. However, I believe the payments we've sent to the site IN THE PAST are still secured because it was through a 3rd party. Do NOT PUT NEW PAYMENT INTO INTO THE SITE. I also believe IV owes us all some money for being offline for so long.
Also, PVCT is about to rocket to the moon!!!!!
NEWS -- Provectus Biopharmaceuticals Announces Management Additions and Reiterates Commitment to Shareholder Value Creation
KNOXVILLE, TN, April 16, 2024 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (“Provectus” or the “Company”) (OTCQB: PVCT) today announced additions to its executive leadership team, reflecting Provectus's dedication to enhancing shareholder value. The Company’s Board of Directors (the “Board”) has appointed Ed Pershing as Chief Executive Officer (“CEO”) and Dominic Rodrigues as President.
New Executive Appointments
Mr. Pershing has served as Board chairman since 2018 and was a Board observer and chairman of the Company’s Strategic Advisory Board from 2017 to 2018. Mr. Rodrigues has served as Provectus’s chief operating consultant in 2024 and vice chairman since 2018 and was chairman from 2017 to 2018. They will continue to serve in their respective Board roles.
Mr. Pershing co-founded Pershing Yoakley & Associates (“PYA”) in 1983 and was its President and CEO until his retirement from the firm in 2019. PYA is a top 20 healthcare consulting and top 100 accounting firm in the U.S., growing from a three-employee office to more than 350 employees and four affiliate companies serving more than 3,500 clients in all 50 states. Mr. Pershing’s healthcare experience and expertise include turnaround and performance improvement initiatives, long-range planning studies, development of numerous hospital and medical office projects, restructuring of healthcare organizations, liaison between boards of directors and management, mergers, acquisitions, divestitures, and leasing arrangements. He also served as an expert witness on healthcare industry matters and represented healthcare organizations before federal and state regulatory agencies.
Mr. Rodrigues worked in management consulting and corporate development in science and technology-driven industries prior to Provectus. He also was a finance professor at the University of Nevada, Las Vegas, a venture capitalist at defense contractor SAIC, a currency derivatives trader at Bank of Montreal, and a project manager and engineer at Jacques Whitford. Mr. Rodrigues holds business, economics, engineering, and public policy degrees from The Wharton School, the London School of Economics, MIT, and the University of Toronto.
Mr. Pershing stated, “Since 2017, Mr. Rodrigues has managed clinical development, research collaborations, key vendors, and other Provectus activities in all biotechnology business functions, supported our finance and accounting team, and been a contributing member of the board of directors.”
Mr. Pershing added, “Mr. Rodrigues has secured existing molecular biochemical, medical scientific, and manufacturing process knowledge from Provectus’s former founders and contributed to the creation of new insights in these areas. His efforts ensure that all corporate understandings can be efficiently conveyed to new team members and development partners in the future.”
Mr. Pershing concluded, “Mr. Rodrigues and I first met as fellow shareholders at the 2010 annual meeting of the American Society of Clinical Oncology where Provectus clinical data were being presented. We have long shared a supreme confidence in the singular distinctiveness of the Company’s rose bengal sodium molecule. In asking him to join me in 2017 to save Provectus and its truly unique science, Mr. Rodrigues unequivocally embraced my vision of pushing the molecule to achieve its true potential as an expansive therapeutic platform capable of transforming the way the healthcare industry treats disease. With our team of dedicated, perseverant executives, employees, consultants, and clinical and research collaborators, we will continue to execute on our mission to develop and commercialize innovative, broad-spectrum, immunotherapy medicines that are safe, effective, accessible, and affordable, and that can revolutionize the healthcare industry as we know it.”
Commitment to Shareholder Value Creation
Provectus remains committed to delivering long-term value to its shareholders. On the Company’s First Quarter 2024 Conference Call, Provectus outlined its key areas of focus for 2024:
I purchased the lifetime membership several years ago when Blue was trying to raise money, but I think nine dollars is a reasonable access fee and helps support the site. As they say, nothing is free.
I just looked at the website, and the free access is no longer available.
I keep getting a message that says no more ad supported free service. $9 a month to use now.
|
Followers
|
186
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9951
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
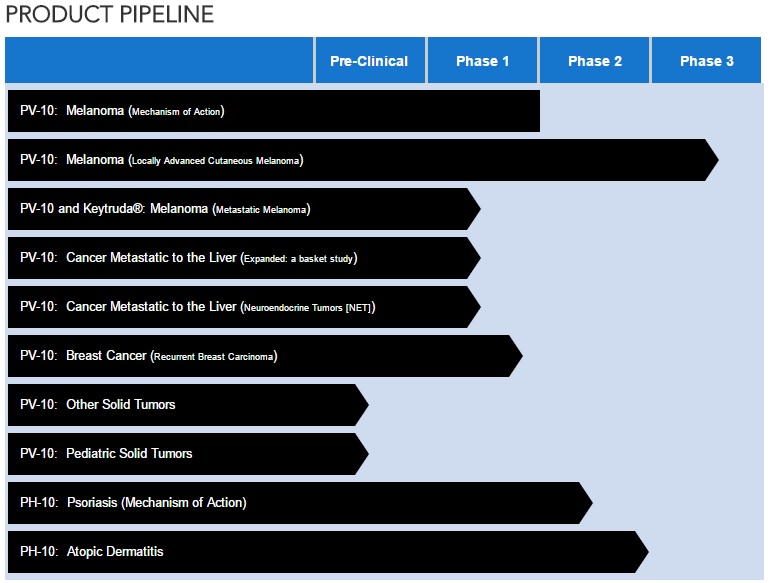
CURRENT PVCT PIPELINE (as of May 23, 2017)
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
