Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
thanks buddy youre the best
$1+ coming this is the right place for those who wants to make quick money because a lot of news is on the way and the stock is brutally undervalued
hey Kei ,Bob or anybody can you please do me a favor and post the Infos below on $heff board ? Im not a premium member so i cant post there i cant even send you a private email . THANK YOU VERY MUCH IN ADVANCE .Oh and please remove this post then .
--------
IGXT (MC $37 M) 2 FDA approvals coming + 2 new NDA Filings in 4Q = 1000% Potential
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there are another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .Their unique Thin-film version of the Blockbuster Drug Cialis (Erectile Dysfunction) alone is worth $2-3 per share , NDA filing for the Drug coming before Year end .
IGXT has a ridiculous Market Cap of $35 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT) (IGX.V)
Market Cap: $37 Million
Cash: $2.1 Million
Price: $0.59
Burn-Rate: ~$300k per Quarter
Shares Out : 63.6 Million
Upcoming Milestones :
Big Partnership imminent :
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
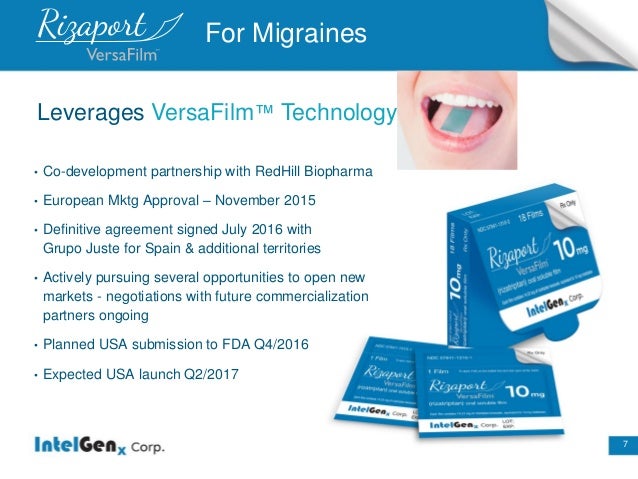
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
IntelGenx Corp., today announced the recent initiation of a phase 1 clinical trial of montelukast, a unique drug repurposing opportunity for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016.


IGXT (MC $37 M) 10 BAGGER GEM HERE EASILY ...........
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there are another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .Their unique Thin-film version of the Blockbuster Drug Cialis (Erectile Dysfunction) alone is worth $2-3 per share , NDA filing for the Drug coming before Year end .
IGXT has a ridiculous Market Cap of $35 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT) (IGX.V)
Market Cap: $35.8 Million
Cash: $2.1 Million
Price: $0.56
Burn-Rate: ~$400k per Quarter
Shares Out : 63.6 Million
Upcoming Milestones :
Big Partnership imminent :
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
IntelGenx Corp., today announced the recent initiation of a phase 1 clinical trial of montelukast, a unique drug repurposing opportunity for the treatment of degenerative diseases of the brain, such as: mild cognitive impairment and Alzheimers disease, the most prominent form of dementia. IntelGenx expects results from the phase 1 trial to be available in September 2016.



IGXT on the run again
Partnerships + New NDA filings + FDA approval around the corner ..Market cap of $36 M is still a very BIG JOKE
UP SHE GOES and still brutally underpriced and undiscovered Goldmine
IGXT-DD for Newbies ..A Once in a lifetime here guys .
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there are another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .Their unique Thin-film version of the Blockbuster Drug Cialis (Erectile Dysfunction) alone is worth $2-3 per share , NDA filing for the Drug coming before Year end .
IGXT has a ridiculous Market Cap of $33 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT)
Market Cap: $33 Million
Cash: $2.1 Million
Price: $0.52
Burn-Rate: $400k per Quarter
Shares Out : 63.6 Million
UPcoming Milestone :
Big Partnership imminent :
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
Presentation July 2016
http://s2.q4cdn.com/790425727/files/doc_presentations/2016/INTELGENX_July-6_2016-(1).pdf



IPCI & IGXT most attractive Bio stocks to own NOW both are close to Big News . Love them
IGXT start moving get some before she explodes higher ,Big News like Partnership(s),NDA submissions and new approvals in EU on the way . Market cap of $35 Million is more than ridiculous .Fair Value is minimum $3+
IGXT start moving get some before she explodes higher ,Big News like Partnership(s),NDA submissions and new approvals in EU on the way . Market cap of $35 Million is more than ridiculous .
IGXT (MC $33 M) 2 FDA approvals coming + 2 New NDA Submissions in 4Q = 10+ BAGGER EASILY
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there are another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .Their unique Thin-film version of the Blockbuster Drug Cialis (Erectile Dysfunction) alone is worth $2-3 per share , NDA filing for the Drug coming before Year end .
IGXT has a ridiculous Market Cap of $33 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT)
Market Cap: $33 Million
Cash: $2.1 Million
Price: $0.52
Burn-Rate: $400k per Quarter
Shares Out : 63.6 Million
UPcoming Milestone :
Big Partnership imminent :
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
Presentation July 2016
http://s2.q4cdn.com/790425727/files/doc_presentations/2016/INTELGENX_July-6_2016-(1).pdf



Here is one of the reason why i like this stock so much is because of this thin-flim version of the Blockbuster Cialis which works faster than tablet . There are NO generics available on the Markets so IGXT could capture a significant market share with their better version . NDA submission expected in 4Q and FDA approval late 2017 .This Drug alone is worth more than $2-3 per share and they have a full Pipeline of Drugs which are close to Market entry .
Product :Tadalafil (Erectile Dysfunction)
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Wafer Viagra For Instant Results One of the big challenges for erectile dysfunction medicine manufacturers is to quicken the time between the application of the medicine and its response. Men want instant results when they take Viagra, Cialis or Levitra and complain that having to wait as much as thirty minutes before the active ingredient is effective can burst the bubble of spontaneity and passion which are often essential requirements of good sex. Well a solution could be at hand following research done by a Canadian research company called IntelGenx who have developed wafers [like the common breath freshness ones] that are designed to melt on your tongue which they say will get the medicine into your bloodstream around 30% faster than by using conventional tablets. IntelGenx say that because the mouth is lined with tiny blood vessels applying the wafer Viagra here can give a very fast application of the drug directly into the bloodstream. IntelGenx said that they would be conducting more clinical trials to corroborate this and they hoped to develop a whole range of fast acting wafer medications to treat not only erectile dysfunction but also pain relief, depression and hypertension. Sometimes men complain that if they have eaten a large meal it can take an even longer time for the Viagra, Cialis and Levitra to take effect. If on the other hand they made use of erectile dysfunction medication in a wafer form applied directly to the mouth and therefore bypassing their digestive system this problem would disappear. Using a wafer type of Viagra would also mean that you wouldn't have to go and get a glass of water in the middle of a passionate encounter in order to swallow your pill. Wafer Viagra would be a much more discreet application of impotence medication and Ukmedix News will be looking out for updates on the IntelGenx clinical testing done on this new idea
Strong Insider & Institutional Ownership on this Goldmine . Big Jump to new highs could come anytime now .GL
Insider Ownership :
Horst G. Zerbe 4,670,243.5 7.34%
Ingrid Zerbe 5,471,356.5 8.60%
Bernard J. Boudreau 287,500
Bernd Melcher 232,500
Andre Godin 214,500
Ian Troup 187,500
John Marinucc 175,000
Nadine Paiement 135,000
Clemens Mayr 68,750
John Durham 50,000
Robert Bechard 10,000
All directors and officers as a group (11 persons) 11,502,351 18.08%
Institutional Holders :
Alpha Capital ...3.2M
Roadmap Capital Inc. 3.1M
Insider activity
BECHARD ROBERT J. Officer Purchase at $0.47 - $0.51 per share. Direct 4,900 Jan 22, 2016 10,000
BOUDREAU BERNARD Director Purchase at $0.51 - $0.51 per share. Indirect 6,120 Jan 22, 2016 12,000
BOUDREAU BERNARD Director Purchase at $0.51 - $0.51 per share. Direct 19,000 Jan 22, 2016 38,000
BOUDREAU BERNARD Director Purchase at $0.45 - $0.45 per share. Indirect 3,375 Jan 21, 2016 7,500
GODIN ANDRE Officer Purchase at $0.40 - $0.4 per share. Direct 8,000 Jan 20, 2016 20,000
BOUDREAU BERNARD Director Purchase at $0.41 - $0.41 per share. Indirect 3,075 Jan 20, 2016 7,500
GODIN ANDRE Officer Purchase at $0.52 - $0.52 per share. Direct 4,940 Dec 30, 2015 9,500
GODIN ANDRE Officer Purchase at $0.51 - $0.51 per share. Direct 7,828 Dec 23, 2015 15,350
GODIN ANDRE Officer Purchase at $0.50 - $0.51 per share. Direct 6,640 Dec 22, 2015 13,150
GODIN ANDRE Officer Purchase at $0.50 - $0.5 per share. Direct 3,250 Dec 21, 2015 6,500
IR Reply regarding the ThermSheet .. Partnerdeal news could come anytime now .
Dear xxxxxx,
Thank you for your email and support as a shareholder. IntelGenx has gone through an extensive due diligence process which we have completed on our side all the work required. We are working hard with the major pharmaceutical company to finalize a definitive agreement. As soon as the agreement is finalized we will announce. Going forward we will not be announcing term sheets, only definitive agreements. We just closed Grupo Juste and instead of people being pleased by IntelGenx closing its first commercialization deal, we were reminded that we were a month late. Agreements take considerable amount of time. We have to make sure they are closed properly that IntelGenx is protected with a long term plan in mind.
Please do not hesitate to contact me.
Best regards,
Edward
Yes it was stagnat but now we are very close to Big News which will change the company .
IGXT is light years better than PTX which is close to Bankruptcy . PTX has over $300 Million Debt and burning around $40 Million per Quarter i wouldnt touch the stock not even for free .
IGXT start moving still brutally undervalued gem
Very thin ASK = Good News like FDA approval and/or Partnerdeals which are imminent could push this low float stock to $1 and higher very fast .
Real-Time Best Bid & Ask
http://www.otcmarkets.com/stock/IGXT/quote
IGXT (MC $32 M) 2 FDA approvals coming + 2 New NDA Submissions in 4Q = 10+ BAGGER EASILY
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there is another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .
IGXT has a ridiculous Market Cap of less than $32 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT)
Market Cap: $31.8 Million
Cash: $2.1 Million
Price: $0.49
Burn-Rate: $400k per Quarter
Shares Out : 63.6 Million
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
Presentation July 2016
http://s2.q4cdn.com/790425727/files/doc_presentations/2016/INTELGENX_July-6_2016-(1).pdf



10 BAGGER GEM HERE EASILY ...........
Undiscovered Intelgenx (IGXT) is definitely one of the most attractive and undervalued stock in the Biotech Sector . The Company has already 2 Drugs approved and another 2 Big Drugs close to FDA approval .BIG Partnership News could come any day for up to 3 Drugs for which IGXT got a Therm Sheet from a Global Pharma Company and there is another partnering discussion ongoing for 2 Drugs (see pipeline chart below) .
IGXT has a ridiculous Market Cap of less than $32 Million which is a Big Joke for a Company with such a MEGA Product Pipeline , this Co has everything to be the next 10 Bagger minimum .GL
Intelgenx (IGXT)
Market Cap: $31.8 Million
Cash: $2.1 Million
Price: $0.49
Burn-Rate: $400k per Quarter
Shares Out : 63.6 Million
•Announced a development and commercialization term sheet with a global pharmaceutical company for up to three products. If entered into, IntelGenx expects the definitive agreement to be finalized in the second quarter of 2016
Product :Rizaport (Migraine)
•European Mktg Approval–November 2015
•Planned USA submission to FDA Q4/2016
•Expected USA launch Q2/2017
Product :Tadalafil (Erectile Dysfunction) a better Oral thin-film version of Blockbuster Drug Cialis
•505(b)(2) USA NDA submission in Q4/2016
•Expected USA launch Q4/2017
Product :Indicated for Opioid Dependence
•Awaiting FDA approval
•According to IMS data, the oral film market for opioid dependence was worth more than $1.4B US in 2014.
Presentation July 2016
http://s2.q4cdn.com/790425727/files/doc_presentations/2016/INTELGENX_July-6_2016-(1).pdf



Great News
JULY 15, 2016 Knight Acquires Minor Stake in Pediapharm MONTREAL, QUEBEC--(Marketwired - July 15, 2016) - Knight Therapeutics Inc. ("Knight") (TSX:GUD) today announced that it has acquired 11,470,920 common shares (the "Shares") of Pediapharm Inc. ("Pediapharm") in exchange for 221,126 common shares of Knight and 221,126 four-year warrants of Knight, with an exercise price of $10.00. The Shares represent approximately 15.8% of the outstsanding common shares of Pediapharm. Knight owns an aggregate of 13,413,420 common shares of Pediapharm representing approximately 18.5% of the outstanding common shares of Pediapharm. The closing price on the TSX on the last trading day immediately preceding the purchase of the Shares was $8.35, representing an aggregate dollar value in respect of the Knight shares issued of $1,846,402. Using the closing price, the Black Scholes model renders a value of $2.82 for each warrant issued, or $623,575 in the aggregate. Knight holds 757,500 warrants, which are convertible into 757,500 common shares of Pediapharm at an exercise price of $0.33 per common share at any time until March 30, 2019. Should Knight exercise the warrant, Knight would acquire an additional, 757,500 common shares of Pediapharm, representing 1.0% of the issued and outstanding of common shares of Pediapharm, after taking effect of the exercise of the warrant. Knight acquired the Shares for investment purposes. Knight may in the future purchase or sell Shares of Pediapharm or otherwise trade in securities of or engage in other transactions with respect to Pediapharm depending on a number of factors, including but not limited to, Pediapharm's financial position, the price levels of the common shares of Pediapharm, conditions in the securities markets and general economic and industry conditions, Pediapharm's business or financial condition, and other factors and conditions Knight deems appropriate.
PDP.V (Mkap 12 M€) 6 Produkte am Markt + 4 warten auf Zulassung | wallstreet-online.de - Vollständige Diskussion unter:
http://www.wallstreet-online.de/diskussion/1210274-371-380/pdp-v-mkap-12-m-6-produkte-markt-4-warten-zulassung#neuster_beitrag
2 Approvals around the corner = 300-500% upside Potential here
Rupafin approval in August will push this unknown low float gem 100% and higher .Another Approval (Otixal) is expected in November . Market Cap of $10.8 Million is ridiculous for this great Company especially when the Cash blance is over $7 Million .Get some before they get the Approval .GL
Pediapharm (PDP.V)
Market Cap: c$10.8 Million
Cash: c$7.5 Million
Price 0.15
Presentation
http://pedia-pharm.com/wp-content/themes/ifeature-tpl/PDF/Pediapharm-Corporate-Presentation-MARCH-2016-v4-WEBSITE-PDF.pdf
2 Health Canada approval:Rupafin(Aug-Sept 2016) and Otixal (Nov-Dec 2016)
Rupatadine’s estimated launch is now estimated to be in the period of July-September 2016 vs October-December 2016 as previously stated due to the fact the Company now estimates it will receive Health Canada’s approval by August 2016.
PRODUCT INDICATION MARKET SIZE (CDN $) EST. ANNUAL PEAK SALES (CDN$) EST. LAUNCH DATE
Rupatadine(Rupafin) Antihistamine (RX Indication) 120M 6M Q-3 2016
Cetraxal-Plus (Otixal) Ear Infection, Swimmer’s Ear 25M 4M Q-4 2016
you forget the ADCOM meeting which happens a few weeks before FDA deicion .
There are a lot of potential investors on the sideline waiting for the NDA Submission to get in .
IPCI (MC $43 M) 8 Drugs awaiting FDA approval= 10 Bagger Potential ...A great stock for near and long term .
IPCI has a very large and attractive Product Pipeline with 8 Drugs awaiting FDA approval which could they could approved anytime now . Their Key Drug is Rexista a potential Best in Class Abuse Deterrent Pain Drug with HUGE Market Potential . NDA Filing for the Drug is planned within Weeks (June or July) .We could see $6++ easily with NDA filing and Runup to FDA decision (4Q16 or early 1Q2017) and much higher if there is any approval of the Drugs under review by FDA . This ultra Low float Stock is a lifetime opportunity here close at ATL . GL
Intellipharmaceutics (Nasdaq: IPCI)
Market Cap: $43 Million
Cash: $6 Million
Price: $1.59
Shares Out: 28 Million
Slide Presentation
http://files.shareholder.com/downloads/ABEA-43EQSZ/2133989055x0x881307/0AA81A60-E645-4481-8DED-AD17516D579E/Roth_Investor_Conference_March_2016_upload_version.pdf
Intellipharmaceutics(IPCI-NASDAQ, Speculative Buy, US$12 target)
Intellipharmaceutics International Inc.: Investors should focus on Rexista Oxycodone XR (pain) targeting a US$2.3 billion market. On May 21, FDA gave guidance to IPCI that the Rexista NDAcould be filed without a Phase 3 trial if they could demonstrate bioequivalence to Oxycontin. By IPCI only having to do a bioequivalence trial for Rexista Oxycodone XR, compared to Purdue’s Oxycontin, the company will now run a smaller trial of only 24-40 patients. IPCI should be able to file for FDA approval in H1 2016.
There are several catalysts for IPCI expected: (i) clinical results for the Rexista oxycodone
pharmacokinetic studies (fed and fasting) likely in November 2015 (ii) an NDA filing for Rexista Oxycodone in H1 2015 (iii) 7 ANDA approvals could come at any point as the generic arm of the FDA clears its backlog and (iv) management has been actively working on licensing its products (generics and brand) such a transaction could be forged in H1 2016. On May 26, FDA granted Rexista Oxycodone XR fast track designation. With a “fast track designation” for Rexista Oxycodone XR, it is possible the NDA could be reviewed in 6 months or less and the drug could be launched at the end of 2016/H1 2017 (we have modeled in a mid 2017 to H2 2017 launch which is now very conservative).
$2 coming guys
Wall Street analysts polled by Zacks Research have set a consensus target price of $8.666 on company shares. These target prices fall in the range of 6 to $12. These same analysts have also given stock ratings.
http://www.bibeypost.com/intellipharmaceutics-international-inc-nasdaqipci-average-stock-rating-recap/
once the stock retake the MA(50) at 1.86 then quick jump back over $2 . IPCI is still one of the most undervalued Biotechs out there
IPCi is massive Oversold and brutally undervalued its a gift at this price . Rebound to $2+ could start anytime now
Any FDA approval of the Drugs under review and this will be back around $3 within minutes like in February .
For those who bought below the offering price like me will make big money here .
hey doog r u a premium member on ihub ?
http://www.fdanews.com/articles/150808-par-acquires-seroquel-anda-expects-2016-first-filer-launch?v=preview
Par Pharmaceutical can launch a generic version of AstraZeneca’s extended-release antipsychotic Seroquel XR by November 2016 following its acquisition of Handa Pharmaceuticals’ ANDA.
i did my dd very well and for me its a fantastic opportunity especially at current level .
They say June or July so June is still Q2 and if July then 3Q
So he could be right about $30 because he knows what the real value of the company is . The Company would be in much much better Shape if the corrupt FDA would not play dirt games with them.
Dr. Isa Odidi of Intellipharmaceutics (Nasdaq:IPCI) (TSX:I) is easily one of the sector's genius drug delivery innovators. While he was at Biovail, Odidi was performing drug alchemy with formulations that mimicked several hard-to-manufacture generic versions of expensive drugs- among them, Pfizer’s (NYSE:PFE) Procardia XL, Bayer’s (BAYRY.PK) Adalat CC, and Novartis’ (NYSE:NVS) Voltaren XR. While serving as the company’s Vice President of Research of Drug Development and New Technologies he helped grow Biovail through its adolescence.
As difficult as the scientific process was, the business plan was simple. Odidi and his team worked out the chemical composition to develop the drugs and pushed their versions through the regulatory process at the U.S. Food and Drug Administration. Once approved, the company licensed the drugs to different companies and collected royalties. While Odidi perfected his science and learned to navigate the FDA process, Biovail grew into a 10+billion market cap company
US-NDA Submission within 6 Weeks / 7 Drugs under review by FDA / Dirt low Valuation of $42 Million / Ultra Low float Stock /
This Stock is so brutally undervalued that it hurts wouldnt be surprised if this Company gets bought .Any FDA approval and this low float rocket is back over $3 like in February .
IPCI is definitely one of the most attractive Biotechs out there at current level .
IPCI is a must have for those who loving Biotechs .
Intellipharmaceutics (Nasdaq: IPCI)
Market Cap: $44 Million
Cash: $6 Million
Price: $1.59
Shares Out: 28 Million
Slide Presentation
http://files.shareholder.com/downloads/ABEA-43EQSZ/2133989055x0x881307/0AA81A60-E645-4481-8DED-AD17516D579E/Roth_Investor_Conference_March_2016_upload_version.pdf
Product Pipeline
http://www.intellipharmaceutics.com/common/download/download.cfm?companyid=ABEA-43EQSZ&fileid=359879&filekey=91A3B0AE-43EA-4788-9F96-81DD2007A44C&filename=pipeline_table.JPG
Dialing Back Euphoria: The Intellipharmaceutics Prescription for Opioid Abuse
https://www.streetwisereports.com/pub/na/dialing-back-euphoria-the-intellipharmaceutics-solution-to-opioid-abuse
James bond’-like tech is the end to overdoses, says formulation firm
http://www.in-pharmatechnologist.com/Drug-Delivery/James-bond-like-tech-is-the-end-to-overdoses-says-formulation-firm
Intellipharmaceutics(IPCI-NASDAQ, Speculative Buy, US$12 target)
Intellipharmaceutics International Inc.: Investors should focus on Rexista Oxycodone XR (pain)
targeting a US$2.3 billion market. On May 21, FDA gave guidance to IPCI that the Rexista NDA
could be filed without a Phase 3 trial if they could demonstrate bioequivalence to Oxycontin. By
IPCI only having to do a bioequivalence trial for Rexista Oxycodone XR, compared to Purdue’s Oxycontin, the company will now run a smaller trial of only 24-40 patients. IPCI should be able to file for FDA approval in H1 2016.
There are several catalysts for IPCI expected: (i) clinical results for the Rexista oxycodone
pharmacokinetic studies (fed and fasting) likely in November 2015 (ii) an NDA filing for Rexista
Oxycodone in H1 2015 (iii) 7 ANDA approvals could come at any point as the generic arm of the
FDA clears its backlog and (iv) management has been actively working on licensing its products
(generics and brand) such a transaction could be forged in H1 2016.
On May 26, FDA granted Rexista Oxycodone XR fast track designation. With a “fast track designation” for Rexista Oxycodone XR, it is possible the NDA could be reviewed in 6 months or less and the drug could be launched at the end of 2016/H1 2017 (we have modeled
in a mid 2017 to H2 2017 launch which is now very conservative).
Additionally, multiple ANDA (generic) approvals are expected including: Glucophage XR ANDA filed on August 2010, Effexor XR ANDA filed January 2010, Protonix ANDA filed June 2010, Seroquel XR ANDA filed February 2011, Lamictal XR ANDA filed September 2011, Keppra XR ANDA filed June 2012 and Pristiq ANDA filed September 2012. The timing of ANDA approvals cannot be predicted, but
they are long overdue based on historical precedents. It now appears the FDA is attempting to clear its backlog of ANDAs, as the Office of Generic Drugs (OGD) broke its old post-GDUFA approvals record of 57 by one, issuing 58 full ANDA approvals in August 2015.The company has the option to license all of its products which are not partnered – this would provide non-dilutive financing.
AYTU still trading under radar and is dirt cheap with a market cap of laughable $19 million .
Fantastic buying opp here at ATL once the Share offering is closed then Stock will run back over $2 .
NDA filing of Rexista this or next month will attract new investors and then we will see a move like TTNP did .
