Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
10-12-15: Heartware Updates on MVAD Trial; May Not Restart as Expected
Heartware (NASDAQ: HTWR) disclosed the following on Tuesday 10-13-15:
On Oct. 12, 2015, HeartWare Intl., posted the following statement to the Investor Relations section of its corporate website (ir.heartware.com) in connection with its MVAD Ventricular Assist System CE Mark clinical trial:
“As previously disclosed on September 9, 2015, HeartWare paused its MVAD clinical trial to address an MVAD controller manufacturing process issue. HeartWare is in the process of implementing manufacturing improvements as well as software updates, which were also described on September 9, 2015.
“Subsequent to that action and following discussions with the Company’s trial investigators, HeartWare began investigating causes of reported adverse events in certain clinical trial patients. The events being analyzed are typical of those seen in other clinical trials for ventricular assist devices. HeartWare took similar actions successfully during its initial human study for the HVAD® System during the HVAD CE Mark clinical trial in 2007. Although HeartWare may not re-initiate enrollment in the MVAD clinical trial in November as it previously expected, HeartWare remains confident in its MVAD System and the potential for the MVAD design to meaningfully improve outcomes for ventricular assist patients. HeartWare intends to provide additional commentary related to the MVAD clinical trial on its third quarter financial results call and webcast scheduled for October 29, 2015. It has been, and remains, the intention of the Company to present and publish clinical data following the completion of enrollment and follow-up for all trial patients.”
http://www.streetinsider.com/Corporate+News/Heartware+%28HTWR%29+Updates+on+MVAD+Trail%3B+May+Not+Restart+as+Expected/10965359.html
Tradero, 10amPT is 1pmET...
Oct15/10amPT(1pmET): Annual SHM, Irvine Marriott – Final Proxy: http://tinyurl.com/qan2qec Webcast: http://edge.media-server.com/m/p/jghjvabo
Known Upcomings – Oct15 SHM to be webcast...
Oct15/10amPT: Annual SHM, Irvine Marriott – Final Proxy: http://tinyurl.com/qan2qec Webcast: http://edge.media-server.com/m/p/jghjvabo
Oct18-21 2015: “ASTRO's 57th Annual Meeting” San Antonio
“ASTRO's Annual Meeting is the premier radiation oncology scientific event in the world and draws more than 11,000 attendees each year.."
ASTRO = The American Society for Radiation Oncology https://www.astro.org
MTG: https://www.astro.org/Meetings-and-Events/2015-Annual-Meeting/Index.aspx
…CEO Steve King 7-14-15/CC: “We expect interim data from the ongoing Ph1 Rectal adenocarcinoma IST (UTSW/Dr. Jeffrey Meyer) evaluating bavituximab+capecitabine+radiation to be presented at the ASTRO Mtg. in October.” http://tinyurl.com/nw2v5u6
“All abstracts are embargoed and remain confidential until the date & time of presentation at the Annual Meeting. Abstracts are scheduled to go live Saturday, 10-17-15 8amCDT”
Oct26-29 2015: “IBC's BioProcess Intl. Conf. & Exhib”, Boston http://www.ibclifesciences.com/BPI Avid/Booth909
Dec7-10 2015: “IBC's Antibody Eng. & Therapeutics, SanDiego” http://www.ibclifesciences.com/AntibodyEng Avid/Booth307
Dec8-12 2015: “38th Annual San Antonio Breast Cancer Symposium”
http://www.sabcs.org
“...presented by the Cancer Therapy & Research Center at UT Health Science Center San Antonio, AACR, and Baylor College of Medicine. The driving force behind this collaboration is the shared mission of the organizations to advance progress against breast cancer. As exciting strides are made in the field of breast cancer research and treatment our program continues to present essential up-to-the minute information combined with engrossing discussion for basic, translational and clinical cancer research professionals. At SABCS'14, there were 7,362 Symposium attendees + 206 Media + 345 Exhibitors, Non-Exhibiting Sponsors & their Support Staff = 7,913 total attendance. More than 53% came from 94 countries outside the USA”
TRACK: Tumor Cell & Molecular Biology: Immunology & Preclinical Immunotherapy
Poster #P4-04-03 “Targeting of Phosphatidylserine by Monoclonal Antibodies Augments the Activity of Immune Checkpoint Inhibitor PD-1/PD-L1 Therapy in Murine Breast Tumors”
M.Gray, J.Gong, V.Nguyen, Jeff Hutchins, Bruce Freimark - Peregrine Pharmaceuticals
~Dec9: FY'16Q2 (qe 10-31-15) Financials & Conf. Call http://ir.peregrineinc.com/events.cfm
Mar6-11 2016: CHI’s 23rd Molecular Med TRI-CON 2016, SanFran http://www.triconference.com (Speaker: Dr. Jeff Hutchins, VP/Preclin.Res.)
Mar15-17 2016: Immune Checkpoint Inhibitors Conf., Boston http://immune-checkpoint.com (Peregrine is 1 of 4 Corp. Sponsors)
CFH, gotta head out – the answer to your question is probably somewhere in the 6-2013/ASCO FINAL Results PDF...
http://www.peregrineinc.com/images/stories/pdfs/2013_asco_nsclc.pdf
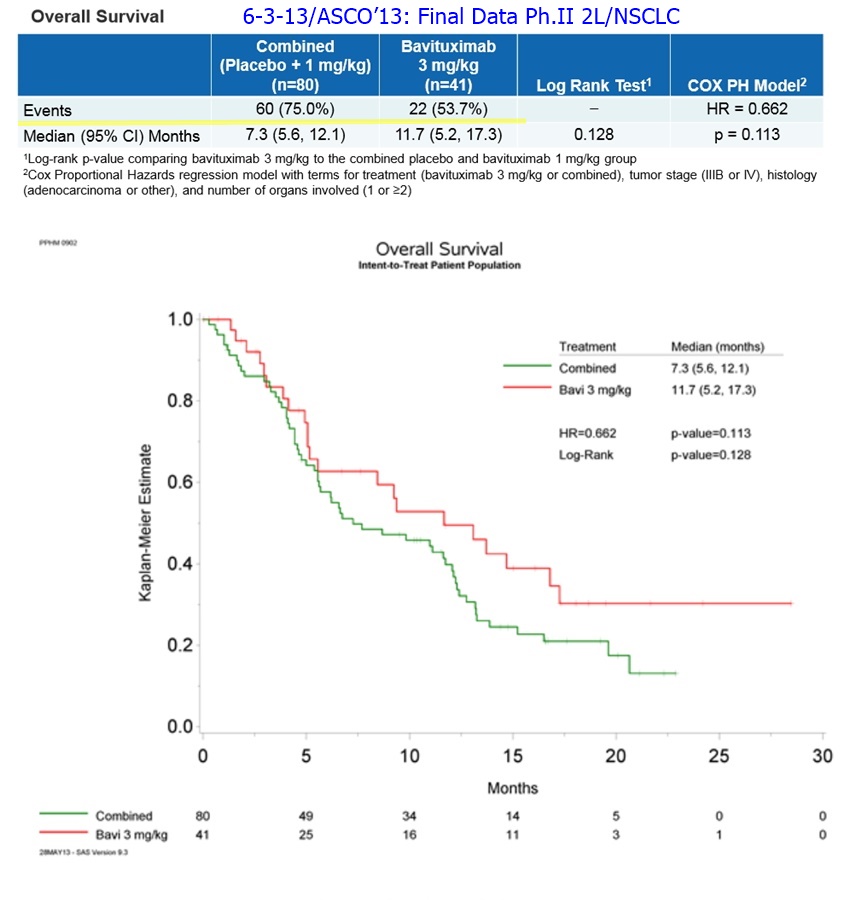
More: 6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
6-3-13: "Immunotherapies have shown significant promise in treating solid tumors and it is encouraging to see these latest results from bavituximab in 2nd-line NSCLC. Bavituximab is a unique targeted immunotherapy with a novel mechanism of action that potentially synergizes well with other compounds currently being developed for oncology," said Martin J. Edelman, M.D., Professor of Medicine at the University of Maryland Greenebaum Cancer Center [ http://medschool.umaryland.edu/facultyresearchprofile/viewprofile.aspx?id=6214 ]. "I look forward to being involved in the upcoming Phase III trial and to seeing the potential of bavituximab in other indications and combinations."
Carboat, I don't think you're intentionally lying this time, just Ignorant. Read ALL of this post again and note the difference between 9-2012/Interim and 6-2013/FINAL.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=117669489
Why didn't you just state, "Adam Feuerstein 9-27-12"? Would have explained it perfectly.
realist, did you get your “49% pts. censored” line that you've been relishing in recently from carboat? The reason I ask is, it's wrong, and way wrong. The correct answer is 32%.
Just so you know, patients are censored from the K-M Survival graph not only if they are lost to the study, but also if THEY ARE STILL ALIVE at the time that the trial was unblinded. These were the Ph2 trial's Final Results, and the protocol required the trial to be stopped when 68% of the patients died. All of the remaining patients must be (and were) 'censored' (ie, are not put in the K-M Survival Graph).
6-3-13/ASCO’13: Final Data Ph.II 2L/NSCLC http://tinyurl.com/my8qxw7
…60% improvement in MOS: Bavi/3mg=11.7mos. vs. 7.3mos. for CTL-arm(combined Bavi/1mg + DoxyOnly arms), HR=.662, P=.113
32% (39) of the 121 pts were censored in the K-M Survival Curve...
= = = = = = = = = = = = = = =
Censoring the Data
“Mathematically removing a patient from the curve at the end of their follow-up time is called "censoring" the patient. This is perhaps an unfortunate choice of words, but you should understand that in this context "censor" is a purely technical term and has nothing to do trying to limit information or prevent expression. The aim is to produce the most accurate possible survival curve taking into account all the of information available. A few conspiracy minded alternative therapy advocates accuse conventional medicine of "censoring their data", presumably to conceal something - if you ever see this argument, one thing you can conclude for sure is that the writer is totally clueless!”
FROM: http://cancerguide.org/scurve_km.html
The names of 3 New Bavi Trials (incl. AZN/Durvalumab & BMS/Opdivo) may well be these once they appear on https://www.clinicaltrials.gov . We'll see!
From: http://adisinsight.springer.com/search – updated 9-9-15:
“Phase I/Ib trial of bavituximab in combination with durvalumab in patients with multiple solid tumors”
PLANNING
Drugs: Bavituximab (Primary)
[Note: Durvalumab = AstraZeneca's anti-PD-L1 mab, aka MEDI4736]
Indications: Solid tumors
Phase of Trial: Phase I
“An open-label multi-center, randomized Phase II trial of the anti-PD-1 monoclonal antibody nivolumab (Opdivo) versus nivolumab plus bavituximab in patients with previously treated locally advanced or metastatic NSCLC”
PLANNING
Drugs: Bavituximab (Primary); Nivolumab
[Note: Opdivo(nivolumab) = Bristol-Myers Squibb's anti-PD-1 mab]
Indications: Non-small cell lung cancer
Phase of Trial: Phase II
“A phase II/III open-label trial of physician's choice of either docetaxel or paclitaxel with or without bavituximab in patients with locally advanced or metastatic HER2 negative breast cancer”
Planning
Drugs: Bavituximab (Primary)
Indications: Breast cancer
Phase of Trial: Phase II/III
= = = = = = = = = = = = = = = = =To The Best of My Knowledge:
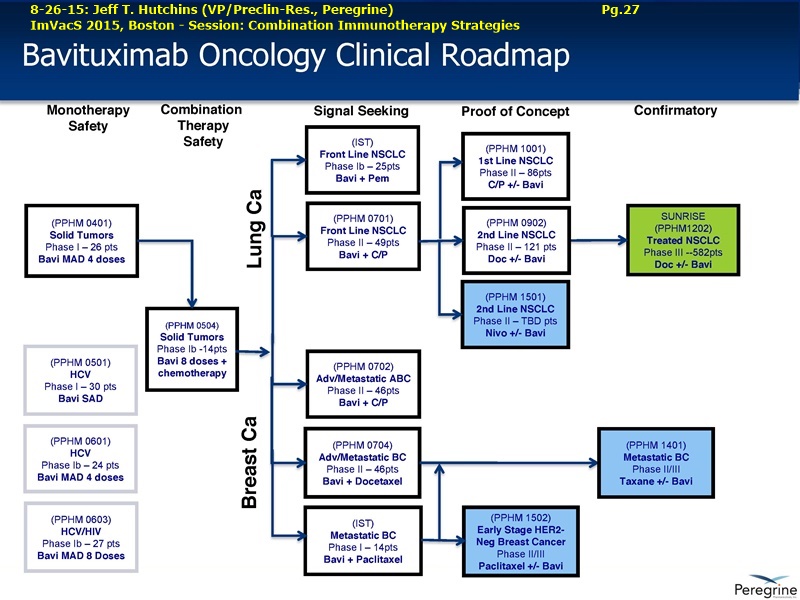
9-2015 UPCOMING NEW BAVI TRIALS (per Dr.Hutchins 8-26-15/ImVacS & 9-9-15/CC) http://tinyurl.com/qz64pzg
...Ph.2 (PPHM 1501), Bavi+Optivo(nivolumab), 2ndLine NSCLC, squam+nonsquam (randomized, open-label)
...Ph.2/3 (PPHM 1502), Bavi+Paclitaxel-or-Docetaxel(Dr’sChoice), Early-Stage Her2- Breast Cancer
...Ph.2/3 (PPHM 1401), Bavi+neoadjuvant-paclitaxel, Her2- Met. Breast Cancer
6-1-15 New Bavi Trials planned: Ph2 Bavi+Opdivo/2L-NSCLC(Squamous+NonSquamous), Ph2/3 Bavi+Doce/PAC/Her2-Breast http://tinyurl.com/qxu4w2x

9-9-15 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/ph22vdn
...CEO S.King: “The Memorial Sloan Kettering & AstraZeneca collaborations are an important part of our announced plans to expand our bavituximab clinical pgm.”
S&P, perusing the iBox might help => http://investorshub.advfn.com/boards/show_ibox.aspx?boardid=2076
BETABODIES: “Potentially the next generation of PS-targeting cancer therapeutics”
BETABODIES (ex: KL15), “potentially the next generation of PS-targeting cancer therapeutics” - generated by fusing domains of the PS-binding the plasma protein, B2-glycoprotein I (B2GPI), to the Fc region of mouse IgG2a. Such ‘betabodies’ potentially have the following advantages: they bind directly to PS and do not require a cofactor protein (B2GPI) for binding; they can be made fully human; they are smaller in size (100 vs. 250 KDa); and they have slower blood clearance rates (half-life of ~5days vs. Bavi’s ~1day).
…See 7-6-13, “Entdoc, here’s a bunch of stuff on Betabodies” http://investorshub.advfn.com/boards/read_msg.aspx?message_id=89680206http://tinyurl.com/khopa3d
MORE in this 9-8-15 post: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=116814508
Opdivo works in only ~20-35% of NSCLC Patients, those that express PD-L1 > 10%. In BMY’s Checkmate-057 NSCLC trial, Opdivo DID NOT increase survival vs. DOCE in the 64% of All Pts that didn’t express PD-L1 > 10% (see BMY’s K-M Charts below). Thus, the window of opportunity for Bavi is essentially 2/3+ of the NSCLC market. Of course, the other 1/3 of the patients where Opdivo works well might live even longer if Bavi were added to the mix, or possibly Bavi will allow Opdivo at reduced dosages to cut back on its considerable side-effects…
- - - - - - - - -
PD-1/PD-L1 blockade therapies like Opdivo+Keytruda benefit 20-25% of patients. Peregrine’s objective with Bavi is to “increase the extent & amplitude” of such therapies…
“Although PD-1/PD-L1 blockade therapy [ex: Opdivo, Keytruda ] provides clinical benefits to approx. 20% of patients with advanced NSCLC, about 80% of patients still remain refractory to this treatment. Therefore, new molecules & combinations are urgently needed to address primary & secondary resistance to these new agents.”
From 3-2015 ClinCancerRes. article, “Immune Checkpoint Modulation for Non–Small Cell Lung Cancer”:
http://clincancerres.aacrjournals.org/content/21/10/2256.abstract
http://www.ncbi.nlm.nih.gov/pubmed/25979932
=> Bavi’s goal is to Extend The Range of patients that would benefit from the anti-PD1 mabs, in addition to helping the 20% that do benefit get Better Results.
= = = = = = = = = = = = = = = = =
BMY’s Ph3 Opdivo NSCLC trial – look at the KM-Curve and OS #’s for those pts that express < 10% PD-L1. The OS separation in the tail just collapses and Opdivo (nivolumab) dead standstill with Doce in MOS => That’s ~64% of ALL the pts in the n=582 trial!
http://investor.bms.com/files/doc_presentations/2015/ASCO-Investor-Presentation-June-1-FINAL_v001_p9iq7v.pdf (pg.25)
= = = = = = = = = = = = = = = = = = = = = = = = = = = = =
5-29-15: Peregrine Pharmaceuticals Enters Into Research Collaboration [with Mem. Sloan Kettering] to Investigate Novel PS-Targeting Immunotherapy Combinations
• Research Collaboration to Focus on Exploring Potential Combinations of PS-Targeting Agents Including Bavituximab With Other Immune Modulators
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=114118480
"The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said (Sloan’s) Dr. Jedd D. Wolchok.
. . .
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
. . .
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said (Sloan’s) Dr. Taha Merghoub.
. . .
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclin. Research at Peregrine. "Our internal & collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."


Carboat, at the time you posted “IBB Up 3-4% today”, I happened to be watching and it was actually up ~1/2%. (it finished down ~1/3% for the day). I asked you this a few weeks back re: something similar - no answer, so I'll ask again: were you intentionally lying or are you just ignorant?
ESMO/Vienna Sept25-29: Peregrine/Booth#405 and Dr. Jeff Hutchins speaking on the 29th...
Sep25–29 2015: 40th ESMO/ECCO European Cancer Congress, Vienna http://www.esmo.org/Conferences/European-Cancer-Congress-2015
ESMO = European Society for Medical Oncology
ECCO = European CanCer Organization
Pgm: http://www.europeancancercongress.org/Scientific-Programme/Searchable-Programme
* Sept29 10:25-10:40am, Dr. Jeff Hutchins (PPHM's Dir./Preclin.Res) speaks: “Targeting Phosphatidylserine-Mediated Immune Suppression Enhances the Efficacy of Immune Checkpoint Blockade in Preclinical Tumor Models of Melanoma & Breast Cancer”
* Peregrine exhibiting at Booth #410.
= = = = = = = = = = = = = = =KNOWN FUTURE:
...Lisa Stepp (PhD/MBA) joined Peregrine 3-2015 as “Sr. Dir./Medical Affairs” - formerly with Gilead, Allos, Genentech, and Wyeth. Here is a summary of her 2 known upcoming presentations (thx BioPh & BamB)...
Sep30-Oct2 2015: “EXL's 17th Medical Science Liaison Best Practices Congress”, SanDiego
“Medical Science Liaisons (MSLs) are a vital part of medical affairs teams and, depending on the size of their company, have many interchangeable functions with other medical affairs professionals. Their main role is to support the dissemination of information to key opinion leaders (KOLs) in a way that not only answers essential medical questions but also strengthens product placement and regulatory compliance.”
http://exlevents.com/17th-medical-science-liaison-best-practices-congress
EXL Events: http://exlevents.com/about-us
Agenda: http://exlevents.com/17th-medical-science-liaison-best-practices-congress/agenda
Oct2 8:45–9am: CHAIRPERSON’S RECAP OF DAY2 - Lisa Stepp, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals
Oct2 9-9:45am: “Strategic MSL Leadership to Improve Operations & Achieve Higher Retention of MSL Teams” - Lisa Stepp, PhD, MBA, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals, Inc.
Oct2 3-3:45pm CLOSING REMARKS - Lisa Stepp, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals, Inc.
Faculty(Lisa Stepp, 1 of 18): http://exlevents.com/17th-medical-science-liaison-best-practices-congress/faculty
Oct7 2015: “Perkin Elmer's Annual Imaging User Group Meeting”, Hopkinton MA
http://go.perkinelmer.com/ImagingUserGroupMeetingTSC2015
Track: “Tissue Imaging – Quantitative Pathology Solutions”
Oct7 9:30-12pm Presentation(1 of 6): "Using Technology to Inform Clinical Trial Design", Lisa Stepp, PhD, Sr.Dir./Medical Affairs, Peregrine Pharmaceuticals
Oct15/10amPT: Annual SHM, Irvine Marriott – Final Proxy: http://tinyurl.com/qan2qec
Oct26-29 2015: IBC's BioProcess Intl. Conf. & Exhib, Boston http://www.ibclifesciences.com/BPI Avid/Booth909
Dec7-10 2015: IBC's Antibody Eng. & Therapeutics, SanDiego http://www.ibclifesciences.com/AntibodyEng Avid/Booth307
~Dec9: FY'16Q2 (qe 10-31-15) Financials & Conf. Call http://ir.peregrineinc.com/events.cfm
Mar6-11 2016: CHI’s 23rd Molecular Med TRI-CON 2016, SanFran http://www.triconference.com (Speaker: Dr. Jeff Hutchins, VP/Preclin.Res.)
Mar15-17 2016: Immune Checkpoint Inhibitors Conf., Boston http://immune-checkpoint.com (Peregrine is 1 of 4 Corp. Sponsors)
BioPh, I sent that obvious Pere+Gilead error to I/R, as I do all typos & little errors I either find myself or somebody else here does & I happen to see it.
Lisa Stepp (PhD/MBA) joined Peregrine 3-2015 as “Sr. Dir./Medical Affairs” - formerly with Gilead, Allos, Genentech, and Wyeth. Here is a summary of her 2 known upcoming presentations (thx BioPh & BamB)...
Sep30-Oct2 2015: “EXL's 17th Medical Science Liaison Best Practices Congress”, SanDiego
“Medical Science Liaisons (MSLs) are a vital part of medical affairs teams and, depending on the size of their company, have many interchangeable functions with other medical affairs professionals. Their main role is to support the dissemination of information to key opinion leaders (KOLs) in a way that not only answers essential medical questions but also strengthens product placement and regulatory compliance.”
http://exlevents.com/17th-medical-science-liaison-best-practices-congress
EXL Events: http://exlevents.com/about-us
Agenda: http://exlevents.com/17th-medical-science-liaison-best-practices-congress/agenda
Oct2 8:45–9am: CHAIRPERSON’S RECAP OF DAY2 - Lisa Stepp, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals
Oct2 9-9:45am: “Strategic MSL Leadership to Improve Operations & Achieve Higher Retention of MSL Teams” - Lisa Stepp, PhD, MBA, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals, Inc.
Oct2 3-3:45pm CLOSING REMARKS - Lisa Stepp, Sr. Dir./Medical Affairs, Peregrine Pharmaceuticals, Inc.
Faculty(Lisa Stepp, 1 of 18): http://exlevents.com/17th-medical-science-liaison-best-practices-congress/faculty
Oct7 2015: “Perkin Elmer's Annual Imaging User Group Meeting”, Hopkinton MA
http://go.perkinelmer.com/ImagingUserGroupMeetingTSC2015
Track: “Tissue Imaging – Quantitative Pathology Solutions”
Oct7 9:30-12pm Presentation(1 of 6): Using Technology to Inform Clinical Trial Design, Lisa Stepp, PhD, Sr.Dir./Medical Affairs, Peregrine Pharmaceuticals
= = = = = = = = = = = = = = = = =
Lisa Stepp: http://www.linkedin.com/pub/lisa-stepp-phd-mba/a/942/345
Peregrine Pharmaceuticals (Mar2015-curr 7mos), Sr. Dir./Medical Affairs
Gilead Sciences (Jul2012–curr <=assuming in error), Dir. Clinical Research, Oncology & Inflammation Therapeutics
Allos Therapeutics (Jun2010–Jul2012 2.2yrs), Senior Dir./Medical Affairs
Genentech (Aug2003–Jun2010 6.9yrs), Sr. Reg. Mgr., BioOncology, MSLs
Genentech BioOncology (Feb2003–Aug2003 7mos), Medical Science Liaison
Wyeth (Aug2000–Nov2002 2.3yrs), Medical Science Liaison II
Bidrite, they didn't conveniently release anything. They PR'd it a couple of days after the article was published - they had zero control over the timing. Why the need for an incorrect statement like that?
4OUR, my heart breaks for you. I went thru basically the same thing beginning 2yrs ago. This disease is so cruel, one of these yrs we're gonna whip it. Be strong My Friend, and know that she's very much Eternally Alive in a much better place, and her love for you and what you did to help her will never diminish.
ExW, I believe you wrote this wrong – pls. check: “So SK does NOT expect the 1st look before mid '16” (or maybe you interpret "1st-Half" as "Mid"? - but that doesn't make sense because he used "mid-yr" w.r.t. the 2nd look)…
SK said in the Q&A to Charles Duncan (Piper Jaffray):
“Charles, what we’ve been projecting is probably 1st-half of next year for the 1st interim data look, and then probably mid-year on for the 2nd interim data look and for the final unblinding - that’s based on sort of just general projections of how we expect the patients to do in the study overall. As we get further along, we’ll be able to get a little bit better guidance as we see more events take place and feel like we’re getting closer to those actual unblinding with interim data looks.”
http://tinyurl.com/ph22vdn
Likewise Robert, I voted YES across the board. I'm with these guys all the way, and I my hat's off to them for lining us up for INEVITABLE SUCCESS, IMO. I actually don't care how many shares it takes - I don't want to give it away before the proper time. That's just ME!!
ATM Sales Summary (3/2009–9/9/2015). Also, PPHM O/S Shares History Table (’06–curr.) at the bottom of this post…
At 9-4-2015, shares O/S = 202,124,031
ATM = “At-The-Market Sales Issuance”
I. WM-SMITH 3-2009:
• $7.5mm ATM/Wm.SMITH 3-26-09: $7,500,000gr. / 2,150,759sh. = $3.49/sh. (commiss: 3%)
• $25mm ATM/Wm.SMITH 7-14-09: $25,000,000gr. / 7,569,314sh. = $3.30/sh. (commiss: 3%/1st$15mm, then 2%)
*Total Raised via WmSmith ATM Sales thru 7-31-10:
. . . . $32,500,000gr. / 9,720,073sh. = $3.34/sh.
II. MLV 6-2010: http://www.mlvco.com
$15mm ATM/MLV 6-22-10 (commiss: 2%) Form424: http://tinyurl.com/24txkxb
• Sold 6/22/10–10/31/10: $6,840,000gr. / 4,031,018sh. = $1.70/sh.
• Sold 11/1/10–11/30/10: $7,407,000gr. / 4,711,611sh. = $1.57/sh.
• Sold 12/1/10–1/31/11: $753,000gr. / 471,744sh. = $1.60/sh.
*Total Raised via MLV June’10 ATM Sales thru 1-31-11:
. . . . $15,000,000gr. / 9,214,373 = $1.63/sh.
III. MLV 12-2010: “Dec’10 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-10 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 12-17-10: http://tinyurl.com/2469b2d )
• Sold 12/29/10-1/31/11: $6,460,000gr. / 2,385,862sh. = $2.71/sh.
• Sold 2/1/11-2/28/11: $2,358,000gr. / 998,142sh. = $2.36/sh.
• Sold 3/1/11-4/30/11: $4,470,000gr. / 1,840,487sh. = $2.43/sh.
• Sold 5/1/11-7/31/11: $3,713,000gr. / 1,912,576sh. = $1.94/sh.
• Sold 8/1/11-10/31/11: $5,582,000gr. / 4,727,840sh. = $1.18/sh.
• Sold 9-2-12 Roth Direct: $6,940,000gr./ 6,252,252sh. = $1.11/sh.
• Sold 11/1/11-1/31/12: $10,961,000gr. / 10,308,025sh. = $1.06/sh.
• Sold 2/1/12-2/29/12: $5,871,000gr. / 5,726,946sh. = $1.03/sh.
• Sold 3/1/12-4/30/12: $1,263,000gr. / 2,198,543sh. = $.57/sh.
• Sold 5/1/12-6/30/12: $1,496,000gr. / 2,752,691sh. = $.54/sh.
• Sold 7/1/12-9/26/12: none**
• Sold 9/27/12-10/31/12: $16,719,000gr./ 18,557,928 = $.90/sh.
• Sold 11/1/12-11/30/12: $7,296,000gr./ 9,220,313 = $.79
• Sold 12/1/12-1/31/13: $1,540,000gr./ 1,131,282 = $1.36
• Sold 2/1/13-3/12/13: $330,000gr./ 201,154 = $1.64
*Total Raised via MLV Dec’10 ATM Sales thru 3-12-2013:
. . . . $75,000,000gr. / 68,214,041 = $1.10sh.
IV. MLV 12-2012: “Dec’12 AMI Agreement” http://www.mlvco.com
$75mm ATM/MLV 12-29-12 (commiss: max=5%) Form8K: http://tinyurl.com/2a6w76g
(pursuant to $75mm S-3 Shelf Reg. filed 3-9-12: http://tinyurl.com/7dl7pjm )
• Sold 2/1/13-3/12/13: $4,475,000gr. / 3,132,402sh. = $1.43/sh.
• Sold 3/13/13-4/30/13: $8,897,000gr. / 6,188,273sh. = $1.44/sh.
• Sold 5/1/13-7/11/13: $12,729,000gr. / 7,927,016sh. = $1.61/sh.
• Sold 7/12/13-7/31/13: $2,468,000gr. / 1,690,864sh. = $1.46/sh.
• Sold 8/1/13-9/9/13: $4,372,000gr. / 3,057,431sh. = $1.43/sh.
• Sold 9/10/13-10/31/13: $4,708,000gr. / 3,262,958sh. = $1.44/sh.
• Sold 11/1/13-12/6/13: NONE – see 10Q note below.
• Sold 12/7/13-1/31/14: $28,130,000gr. / 16,045,717sh. = $1.75/sh.
• Sold 2/1/14-4/30/14: $3,017,000gr. / 1,543,383sh. = $1.95/sh.
• Sold 5/1/14-7/31/14: $425,000gr. / 226,700sh. = $1.92/sh.
• Sold 8/1/14-10/31/14: $3,891,000gr. / 2,494,835sh. = $1.56/sh.
• Sold 11/1/14-1/31/15: $1,878,000gr. / 1,261,825sh. = $1.49/sh.
*Total Raised via MLV Dec’12 ATM Sales thru 10-31-2014:
. . . . $75,000,000gr. / 46,831,404 = $1.60sh.
V. MLV 6-2014: “Jun’14 AMI Agreement – up to $25mm” http://www.mlvco.com
• Sold 11/1/14-1/31/15: $1,193,000gr. / 869,504sh. = $1.43/sh.
• Sold 2/1/15-3/12/15: $6,204,000gr. / 4,354,954sh. = $1.44/sh.
• Sold 3/13/15-4/30/15: $6,147,000gr. / 4,457,299sh. = $1.38/sh.
• Sold 5/1/15-7/14/15: $8,896,000gr. / 6,534,400sh. = $1.36/sh.
• Sold 7/15/15-7/31/15: $1,270,000gr. / 1,003,830sh. = $1.27/sh.
• Sold 8/1/15-9/9/15: $1,290,000gr. / 1,091,508sh. = $1.18/sh.
*Total Raised via MLV Jun’14 ATM Sales thru 9-9-2015:
. . . . $25,000,000gr. / 18,311,495 = $1.37/sh.
VI. MLV 8-2015: “Aug’15 AMI Agreement – up to $30mm” http://www.mlvco.com
• Sold 8/7/15-9/9/15: $892,000gr. / 1,091,000sh. = $1.18/sh.
. . . . $892,000gr. / 1,091,000 = $1.18/sh.
TOTAL ALL A-T-M SALES – INCEPTION (3-2009) THRU 9-9-2015:
==> $222,500,000gr. / 152,291,386sh. = $1.46/sh.
- - - - - - - - - -
10-31-11 10Q: “During the 6mos. 10-31-11, we sold 6,440,416 shares of our common stock at mkt-prices for gross proceeds of $9,295,000 under the Dec’10 AMI Agreement before deducting commissions and other issuance costs of $260,000”
1-31-12 10Q: “During the 9mos. ended 1-31-12, we sold 16,948,441 shares of our common stock at mkt-prices for gross proceeds of $20,256,000 under the Dec’10 AMI Agreement before deducting commissions and other issuance costs of $482,000. …During Feb.2012, we sold an addl. 5,726,946 shares of common stock at market prices under the Dec’10 AMI Agreement in exchange for aggregate gross proceeds of $5,871,000. As of 2-29-12, gross proceeds of $38,644,000 remained available under our 2 effective shelf registration statements.”
4-30-12 10K: “Under the Dec. 2010 AMI Agreement with MLV …for aggregate gross proceeds of up to $75,000,000… During FY’s 2011 (5’10-4’11) and 2012 (5’11-4’12), we sold 30,098,421 shares of common stock at market prices under the Dec.2010 AMI for aggregate gross proceeds of $40,678,000 before deducting commissions & other issuance costs of $917,000. As of April 30, 2012, aggregate gross proceeds of up to $27,382,000 remained available under the Dec.2010 AMI… Subsequent to April 30, 2012 and through June 30, 2012, we sold 2,752,691 shares of common stock at mkt prices under the Dec.2010 AMI for aggregate gross proceeds of $1,496,000… Under the registered direct public offering dated Sept. 2, 2011, we entered into separate subscription agreements with 3 institutional investors, pursuant to which we sold an aggregate of 6,252,252 shares of our common stock at a purchase price of $1.11/sh. for gross proceeds of $6,940,000 before deducting placement agent fees and other offering expenses of $525,000.”
10-31-12 10Q: “During the 6mos. 10-31-12, we sold 21,310,619 shares… at varying mkt-prices under the Dec’10 AMI Agreement for gross proceeds of $18,215,000 before deducting commissions and other issuance costs of $620,000. From 11-1-12 thru 11-30-12, we sold 9,220,313 shares gross of $7,296,000. As of 11-30-12, aggregate gross proceeds of up to $1,871,000 remained available under the Dec’10 AMI Agreement. As of 11-30-12, gross proceeds of $151,871,000 remained available under 2 effective shelf registration statements.”
1-31-13 10Q/pg.11: “DEC’10-AMI(max=$75mm): During the 9 mos. ended 1-31-13, we sold 31,662,214 shares at varying mkt-prices for gross proceeds of $27,051,000 before deducting commissions/other-costs of $885,000. As of 1-31-13, gross proceeds of up to $330,000 remained available. From 2-1-13 – 3-12-13, we sold 201,154 shares at mkt prices for gross $330,000. As of 3-12-13, we had raised the full amt of gross proceeds available… DEC’12-AMI(max=$75mm): As of 1-31-13, we had not sold any shares. From 2-1-13 - 3-12-13, we sold 3,132,402 shares at mkt prices for gross proceeds of $4,475,000. As of 3-12-13, gross proceeds of up to $70,525,000 remained available.”
4-30-13 10K/pg.F26: Dec’12-AMI(max=$75mm)Agreement – During FY’13, we sold 9,320,675 shares gross proceeds of $13,372,000 before deducting commissions and other issuance costs of $337,000. As of April 30, 2013, gross proceeds of up to $61,628,000 remained available under the Dec’12-AMI. From 5/1/13 – 7/11/13, we sold 7,927,016 shares for gross proceeds of $12,729,000. As of 7-11-13, gross proceeds of $48,899,000 remained available under the Dec’12-AMI. http://tinyurl.com/p58jcbw
7-31-13 10Q/pg.10: During the 3mos ended 7-31-13, we sold 9,617,880 shares at mkt prices under the Dec’12 AMI Agreement for gross proceeds of $15,197,000 before deducting commissions and other issuance costs of $491,000. As of July 31, 2013, gross proceeds of up to $46,431,000 remained available under the Dec’12-AMI. From 8-1-13 – 9-9-13, we sold 3,057,431 shares for gross proceeds of $4,372,000. As of 9-9-13, gross proceeds of $42,059,000 remained available under the Dec’12-AMI.
10-31-13 10Q/pg.10: During the 6mos ended 10-31-13, we sold 15,938,269 at mkt prices under the Dec’12 AMI for gross proceeds of $24,277,000 before deducting costs of $722,000. As of 10-31-13, aggregate gross proceeds of up to $37,351,000 remained available under the Dec’12 AMI.
***NOTE: There is no stmt in the 10-Q regarding AMI Sales subsequent to 10-31-13 (thru 12-6-13) as has been the case in the 10Q’s for years – the assumption being: NO AMI Sales made 11/1/13-12/6/13, a period where O/S shares only went up by 77,149.
1-31-14 10Q/pg.11: During the 9 mos. ended 1-31-14, we sold 31,983,986 shares under the Dec’12/AMI for gross $52,407,000 before deducting commissions & other iss. costs of $1,427,000. As of 1-31-14, gross proceeds of up to $9,221,000 remained available under the Dec’12/AMI. http://tinyurl.com/pxcjocw
4-30-14 10K pg.F25: “During FY14, we sold 33,527,369 shares under the Dec’12/AMI for gross proceeds of $55,424,000 before deducting commissions & other iss. costs of $1,504,000. As of April 30, 2014, aggregate gross proceeds of up to $6,204,000 remained available under the December 2012 AMI Agreement.” http://tinyurl.com/mhva3k3
7-31-14 10Q/Pg5: During the 3mos. ended 7-31-14, we raised $10,000,000 in aggregate gross proceeds from the sale of our 10.50% SeriesE Convertible Preferred Stock under an At Market Issuance Sales Agreement. Pg.11: During the 3mos. ended 7-31-14, we sold 226,700sh. under the Dec’12-2012 AMI Agreement for gross of $435,000 before deducting commissions & other costs of $14,000. As of 7-31-14, gross of up to $5,769,000 remained available under the Dec’12-2012/AMI. On 6-13-14, we entered into an At Market Issuance Sales Agreement (“June2014/AMI”), with MLV, pursuant to which we may sell shares of our common stock through MLV, for aggregate gross proceeds of up to $25,000,000, in registered transactions from our shelf registration statement on Form S-3. As of 7-31-14, we had not sold any shares of common stock under the June 2014 AMI Agreement. http://tinyurl.com/phw6dkp
10-31-14 10Q/Pg12: COMMON: During 6mos. ended 10-31-14, we sold 2,721,535 sh. at mkt prices under the Dec’12-AMI for gross proceeds of $4,326,000 before deducting commissions and other issuance costs of $113,000. As of 12-31-14, gross proceeds of up to $1,878,000 remained available under the Dec’12-AMI. PREFERRED: During 6mos. ended 10-31-14, we sold 402,858 sh. at mkt prices gross proceeds of $10,070,000 before deducting commission and other issuance costs of $552,000. As of 10-31-14, gross proceeds of up to $19,930,000 remained available under the Series E AMI Agreement. http://tinyurl.com/m6ldhg7
1-31-15 10Q/Pg12: COMMON: During the 9mos. ended 1-31-15, we sold 3,983,360 sh. at mkt prices under the Dec’12-AMI for gross proceeds of $6,204,000 before deducting commissions and other issuance costs of $162,000. As of 1-31-15, we had raised the full-amt of gross proceeds available to us under the Dec’12-AMI. PREFERRED:
Jun’14-AMI: On 6-13-14, we entered into an AMI with MLV…up to $25mm… ,000,000, in registered transactions from our shelf registration statement on Form S-3 (File No. 333-180028). During the 9mos. ended 1-31-15, we sold 869,504 sh. at mkt prices under the Jun’14-AMI for gross of $1,193,000 before deducting commissions and other issuance costs of $31,000. As of 1-31-15, gross proceeds of up to $23,807,000 remained available. Subsequent to 1-15-15 and thru 3-12-15, we sold 4,354,954 sh. of common at mkt prices under the Jun’14-AMI for gross of $6,204,000. As of 3-12-15, 2015, gross proceeds of $17,603,000 remained available under the Jun’14-AMI.
PREFERRED: During the 9mos. ended 1-31-15, we sold 405,004 sh. of our Series E Preferred Stock at mkt prices under the Series E AMI Agreement for gross proceeds of $10,121,000 before deducting commission and other issuance costs of $553,000. As of 1-31-15, 2015, gross proceeds of up to $19,879,000 remained available under the Series E AMI. http://tinyurl.com/mwedt8w
4-30-15 10K pg.F25: During FY’15 we sold 9,681,757sh. under the June’14/AMI for $13,544,000, before deducting costs of $352,000. As of 4-30-15, $11,456,000 remained available. Pg.F35: Subsequent to 4-30-15 & thru 7-14-15, we sold 6,534,400sh. for $8,896,000. A/o 7-14-15, $2,560,000 remain. http://tinyurl.com/ocrtkuj
7-31-15 10Q/Pg12+18: During the 3mos ended 7-31-15, we sold 7,538,230sh. Under the JUNE2014 AMI for gross of $10,166,000 before deducting commiss/iss-costs of $275,000. As of 7-31-15, gross of $1,290,000 remained available under the JUNE2014 AMI, as amended. JUNE2014 AMI: subsequent to 7-31-15 and thru 9-9-15, we sold 1,091,508sh. gross of $1,290,000. As of 9-9-15, 2015, we had raised the full amount available under the June2014 AMI. AUG2015 AMI: on 8-7-15, we entered into an At Market Issuance Sales Agreement with MLV, for gross proceeds of up to $30,000,000, 2.5% commission; as of 9-9-15, we sold 752,760sh. under the AUG2015 AMI for gross of $892,000. http://tinyurl.com/pemub47
PREFERRED SHARES:
4-30-15 10K/p.F26: On 6-13-14, we entered into an At Market Issuance Sales Agreement with MLV, pursuant to which we may issue & sell shares of our Series E Preferred Stock through MLV, as agent, for gross proceeds of up to $30,000,000, in registered transactions from our Jan2014 Shelf. During FY’2015, we sold 799,764 shares of our Series E Preferred Stock at mkt prices for gross $19,205,000 before deducting commissions/costs of $1,002,000. As of 4-30-15, gross proceeds of up to $10,795,000 remained available under the Series E AMI Agreement.
NOTE: One of the holders of Preferred is KENNETH DART (EASTERN CAPITAL), who in a 2-13-15 SG14G ( http://tinyurl.com/k4nsfuu ) reported their 240,000sh. Preferred holdings as 2,000,000 common shares (SHARED VOTING POWER: 9,921,760,% OF CLASS REPRESENTED: 5.4%).
“which has a liquidation preference of $25.00/sh. and a conversion price of $3.00/sh.” 2,000,000 / 240,000 = 8.333
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-15 iss. 7-14-15 http://tinyurl.com/ocrtkuj PR: http://tinyurl.com/nw2v5u6 (Cash 4-30-15=$68.0mm)
Latest 10Q 7-31-15 iss. 9-9-15 http://tinyurl.com/pemub47 PR: http://tinyurl.com/ph22vdn (Cash 7-31-15=$59.0mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
- - - - - - - - - - - - - - - - - -
PPHM’S ATM PHILOSOPHY, CFO PAUL LYTLE, 12-9-10 CC:
“Beyond these 2 sources of capital ([Avid & Gov’t], we have raised addl. capital through the equity markets and it’s important to note that over the past 3 years we have sold every share at market prices [“ATM”], without warrants, without discounts. We continue to be active in the investment community and we have had strong interest from institutional investors intrigued by our clinical data, by our multiple trials to evaluate Bavituximab’s broad therapeutic potential, and by the interim survival data we have seen from our novel brain cancer therapy Cotara. Our goal is to maintain a balanced financial approach with multiple sources of capital and to carefully manage our cash burn as we continue to advance these programs.” http://tinyurl.com/24xmcsn
= = = = = = = = = = = = = = = = = =
PPHM - O/S Shares History (’06–curr.)
4-30-06 35,876,438
1-31-07 39,222,440
4-30-07 39,222,440
7-6-07 45,233,123
7-31-07 45,242,123
10-31-07 45,242,123
1-31-08 45,242,123
4-30-08 45,242,123
7-31-08 45,242,123
10-31-08 45,242,123
1-31-09 45,242,123
4-30-09 45,537,711
7-10-09 47,392,883
7-31-09 47,393,783
10-31-09 48,869,563 +1,475,780
1-31-10 50,903,404 +2,033,841
4-30-10 53,094,894 +2,191,490
6-21-10 54,388,917 +1,294,023 (6-22-10 ATM/mlv Form424)
7-9-10 55,069,449 +475,987 (4-30-10 10K iss. 7-14-10)
7-31-10 55,784,955 +715,506
10-31-10 59,220,742 +3,435,787
11-30-10 63,932,353 +4,711,611 (10-31-10 10Q iss. 12-9-10)
12-15-10 64,404,097 +471,744 (12-17-10 S-3: $75M Shelf Reg.)
1-31-11 66,813,419 +2,409,322
2-28-11 67,885,811 +1,072,392 (1-31-11 10Q iss. 3-11-11)
4-30-11 69,837,142 +1,951,331
7-8-11 71,069,858 +1,232,716 (4-30-11 10K iss. 7-14-11)
8-22-11 72,704,647 +1,634,789 (Proxy iss. 8-26-11)
8-31-11 73,284,016 +579,369 (424B5 iss. 9-2-11)
9-8-11 79,536,268 +6,252,252 (Roth Sale to 3 Inst’s @ $1.11/sh.)
10-31-11 82,638,201 +3,101,933
12-9-11 86,788,817 +4,150,616 (10-31-11 10Q iss. 12-12-11)
1-31-12 93,146,226 +6,357,409
2-29-12 98,873,172 +5,726,946 (1-31-12 10Q iss. 3-9-12)
4-30-12 101,421,365 +2,548,193
7-13-12 104,174,056 +2,752,691 (4-30-12 10K iss. 7-16-12)
7-31-12 104,178,431 +4,375 (7-31-12 10Q iss. 9-10-12)
8-16-12 104,191,176 +12,745 (prelim. proxy 14A http://tinyurl.com/c48bvof )
9-7-12 104,191,176 nochg (7-31-12 10Q iss. 9-10-12)
10-31-12 123,310,188 +19,119,012
12-7-12 132,539,783 +9,229,595 (10-31-12 10Q iss. 12-10-12)
1-31-13 133,770,614 +1,230,831
3-12-13 137,110,758 +3,340,144 (1-31-13 10Q iss. 3-12-13)
4-30-13 143,768,946 +6,658,188 (4-30-13 10K iss. 7-11-13)
7-5-13 151,602,765 +7,833,819 (4-30-13 10K iss. 7-11-13)
7-31-13 153,506,811 +1,904,046
9-5-13 156,461,114 +2,954,303 (7-31-13 10K iss. 9-9-13)
10-31-13 160,248,742 +3,781,628
12-6-13 160,325,891 +77,149 (10-31-13 10K iss. 12-10-13)
1-31-14 176,453,261 +16,127,370
3-3-14 176,481,054 +27,793 (1-31-14 10Q iss. 3-7-14)
4-30-14: 178,871,164 +2,390,110
7-7-14: 179,209,458 +338,294 (4-30-14 10-K cover page, iss. 7-14-14)
7-31-14: 179,216,032 +6,574 (7-31-14 10Q iss. 9-9-14)
8-22-14: 179,226,424 +10,392 (8-28-14 Proxy/Def14A)
9-5-14: 179,505,424 +279,000 (7-31-14 10Q iss. 9-9-14)
10-31-14: 182,000,583 +2,495,159 (10-31-14 10Q iss. 12-10-14)
12-5-14: 182,081,234 +80,651 (10-31-14 10Q cover pg., iss. 12-10-14)
12-19-14: 182,081,234 -0- (12-23-14 S-3)
1-31-15: 184,244,698 +2,163,464 (1-31-15 10Q iss. 3-12-15)
3-12-15: 188,332,872 +4,088,174 (1-31-15 10Q iss. 3-12-15)
4-30-15: 193,346,627 +5,013,755 (4-30-15 10-K iss. 7-14-15)
7-10-15: 199,934,918 +6,588,291 (4-30-15 10-K/cover-pg, iss. 7-14-15)
7-31-15: 200,983,948 +1,049,030 (7-31-15 10Q iss. 9-9-15)
9-4-15: 202,124,031 +1,140,083 (7-31-15 10Q iss. 9-9-15)
O/S WARRANTS & STOCK-OPTIONS A/O 7-31-2015 (10-Q pg.15):
• WARRANTS: As of 7-31-2015, warrants to purchase 273,280 shares at an exercise price of $2.47 were outstanding and are exercisable through Aug30 2018. These warrants were issued in FY’13 in connection with the Aug.2012 [Oxford] loan agreement, which was paid in full during Sept.2012.
• STOCK OPTIONS OUSTANDING A/O 7-31-2015 (10-Q pg.14): 23,867,253 shares at a wgt.avg. exercise price of $1.51. (during 3mos. ended 7-31-15, 99,091sh. pur. at avg=.94)
= = = = = = = = = = =PREFERRED STOCK
...2-5-14: PPHM Announces Public Offering of 10.5% Series E Convertible Preferred Stock http://tinyurl.com/lkxsna6
...2-11-14: PPHM Raises net $16.2mm selling 700k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/jwmsnsk (8-K)
...6/14/14-7/14/13: PPHM Raises net $9.5mm selling 400k Preferred Shares with 10.5% div. at $25/sh, convertible to common at $3/sh http://tinyurl.com/mhva3k3 (4-30-14 10-K pg. F-34; $20mm gross remaining)
9-9-15 Qtly CC-Transcript, PR(Fins/Devs Q1FY16/qe7-31-15), updated Avid Revenues History Table By Quarter…
=> Total Revs May06-Jul15: $138.6mm/Avid + $24.1mm/Govt + $2.4mm/Lic. = $165.2mm
This large post has 3 sections:
I. 9-9-15 Q1/FY16 Qtly. Earnings Conf. Call TRANSCRIPT (q/e 7-31-15)
II. 9-9-15 PPHM Press Release: Q1/FY16 Earnings & Developments
III. Updated Table of Avid Revenues By Quarter (May’06-Current)
…Recall: Peregrine’s FY runs May-Apr, so FY’16 = May’15-Apr’16.
((( Orig. transcript from SeekingAlpha.com [ http://tinyurl.com/q8bgmcz ], with numerous corrections made. )))
Link to webcast replay:
http://ir.peregrineinc.com/events.cfm => http://edge.media-server.com/m/p/2h8985bu
FULL TRANSCRIPT…
9-9-2015 FY’16/Q1 Earnings Conf. Call (q/e 7-31-15)
WELCOME & FWD-LOOKING STATEMENTS: Tim Brons, Vida Strategic Partners (IR) http://www.peregrineinc.com
Speakers: Steve King, Joe Shan, Stephen Worsley, Paul Lytle; Q&A session.
CEO STEVE KING – OPENING COMMENTS:
Thanks to all of you who have dialed in, and to all of you who are participating via webcast today. FY’2016 [May’15-Apr’16] is off to a great and busy start at Peregrine. In May, we announced a research collaboration with leading immuno-oncology researchers at the Memorial Sloan Kettering Cancer Center. In June, we presented important translational data at ASCO, showing the potential of bavi to enhance immune responses in PD-L1 negative tumors. In July, we reported that our Phase III SUNRISE trial in NSCLC remained on track for normal completion by year-end, and as we draw closer to December, we are increasingly confident that we will hit this milestone. And most recently we announced collaboration with AstraZeneca to expand our immuno-oncology combination clinical research program.
The Memorial Sloan Kettering & AstraZeneca collaborations are an important part of our announced plans to expand our bavituximab clinical program. The initial efforts is to initiative new Lung & Breast cancer clinical trials by year-end, and then to expand to new addl. indications by early 2016. Taken together, these clinical efforts are meant to expand bavituximab's eventual market opportunity in NSCLC by providing important data supporting the combination of bavituximab with both chemotherapy and PD-1 or PD-L1 inhibitors, while simultaneously pursuing equally large market opportunities in Breast cancer and eventually other indications.
Fueling these efforts, our research group remains busy generating clinical, translational, and preclinical study results that consistently demonstrate bavituximab's activity in a range of tumor types. In recent weeks, we've received very positive responses to findings presented at the Intl. Association for the Study of Lung Cancer (IASLC) World's Congress [WCLC] and at the Immunotherapy & Vaccine Summit [ImVacS]. At these meetings, we presented results from multiple studies demonstrating bavituximab's ability to promote anti-tumor T-Cell-mediated activity and its potential mechanistic synergies with chemotherapy & checkpoint inhibitors targeting the PD-1 & PD-L1 pathways.
Finally our contract mfg. business, Avid Bioservices, hit a record high for revenues for any quarter [$9.4mm, GP%=51%] during Q1/FY’16, putting us on track for another record revenue year. I'll continue my comments later, but first the other members for our team will give a detailed overview of our clinical, business development, and operational achievements. We will begin with Joe Shan, VP/Clinical&Regulatory.
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
I'd like to start with an update on the company's ongoing Phase III SUNRISE trial, which is evaluating the use of bavituximab & docetaxel in patients with previously treated locally advanced or metastatic non-squamous NSCLC. This global study remains our top clinical priority and I'm happy to say this trial is proceeding according to plan and we continue to stay on target to complete accrual by the end of the calendar year. As a reminder, SUNRISE is designed as a blinded registration trial with 2 planned interim analyses. The 1st will be conducted when 33% of the targeted survival events are reached, and the 2nd will be conducted at 50% of events. As these events are patient deaths, we are at this time unable to accurately predict when the interim analyses will occur, but we plan to provide updates as soon as possible.
Meanwhile as SUNRISE nears the completion of enrollment and we await the trial results, we have begun to expand the bavituximab clinical program and are planning to initiate several new trials in the coming months to identify other clinical settings where the addition of bavituximab might help improved patient outcomes. Our strategy behind this decision is to continue building on our significant clinical experience, combining Bavituximab with chemotherapy, while simultaneously exploring new opportunities with immunotherapy combinations.
Let me first address the expansion of our NSCLC program beyond the combination with docetaxel, which we believe will remain a key treatment option in a number of cancers, including Lung cancer. The decision to initiate another Lung cancer trial is supported by the considerable amount of preclinical data demonstrating that treatment effects of CTLA-4 or PD-1 inhibitors is greatly enhanced when combined with Bavituximab. This is very timely as anti-PD-1 agents such as nivolumab ( Opdivo) & pembrolizumab ( Keytruda) are starting to enter the market as single agent therapies. Thus, we're planning to initiate around the end of this year an open-label, randomized Phase II trial of nivolumab vs. nivolumab+bavituximab in patients with previously treated metastatic NSCLC who have not received a prior PD-1 or PD-L1 inhibitor. The primary endpoint of this trial is expected to be overall response rates with secondary endpoints to include duration of response, progression-free survival, overall survival, and safety. Importantly, as translational studies have demonstrated that bavituximab enhances multiple markers of immune activation even in tumors that express low levels of PD-L1, we plan to look at patient outcomes by pre-treatment PD-L1 expression levels to better understand which patients benefit most from bavituximab's effects.
Now beyond Lung cancer, we are also compelled to initiate addl. clinical trials in Breast cancer based on the totality of our clinical experience in advanced Breast cancer to-date. Data from our Phase I IST of bavituximab+paclitaxel in patients with HER2- metastatic Breast cancer published in Cancer Medicine earlier this year [3-31-15/A.Stopeck http://tinyurl.com/nm5oog4 ] demonstrated an impressive 85% response rate. Data from this IST, together with 2 prior Peregrine sponsored trials of bavituximab with taxane-based chemotherapy which yielded between 61% to 74% overall response rates and a MOS of over 20mos. in advanced or metastatic Breast cancer patients provides strong rationale to advance this indication. Additionally, taxanes continue to be a key std. treatment option for many stages of Breast cancer. Accordingly, we plan to initiate a seamless Phase II/III trial in patients with HER2- negative metastatic Breast cancer, with all patients receiving physician's choice of paclitaxel or docetaxel, either alone or in combination with bavituximab. The primary end point for Phase II is overall response rate, while the Phase III part of the trial has a primary end point of progression-free survival.
Furthermore we are planning a trial evaluating neoadjuvant paclitaxel with or without bavituximab, in the hopes of further elucidating bavituximab's immune modulating mechanism and look for clinical signal in early stage HER2- Breast cancer. As with the Phase II lung cancer trial described earlier, we plan to initiate the Phase II/III Breast trial before the end of this year. The open-label nature of these trials may provide us the opportunities for data updates, and our goal is to generate as much clinical data as possible prior to SUNRISE unblinding.
Now last but not least, I'd like to finish up with a few words about our recent collaboration announcement with AstraZeneca [8-24-15: http://tinyurl.com/owlxpsf ]. Under this clinical collaboration agreement, we will combining bavituximab with chemotherapy and AZ's durvalumab [aka “MEDI4736”, an anti-PD-L1 inhibitor] in a Phase I/Ib trial in multiple solid tumors. The Phase I part of the trial will confirm the tolerability of the 2 IO agents and establish a recommended dose regiment for the Phase Ib part of the trial, which will assess the safety & activity of the triple investigational combination, which includes std. chemotherapy. We are particularly excited about the collaboration because we believe that these 3 drugs have different and potentially complementary mechanisms. Chemotherapy is known to kill tumor cells, increase phosphatidylserine exposure and generate tumor antigens. Bavituximab, by targeting exposed PS, a highly immune-suppressive molecule exposed on the surface of cells in the tumor micro environment, has been shown to trigger macrophage re-polarization and tumor specific T-Cell activation. Durvalumab is a monoclonal antibody directed against programmed cell death ligand 1 PD-1 and signals from PD-1 help tumors avoid detection by the immune system. We believe that by combining these 3 approaches the potential exists for a more complete and lasting anti-tumor immune response and look forward to testing this hypothesis in the clinic. And to provide more context regarding the AZ collaboration and Peregrine’s ongoing business development efforts, I'll turn the call over to Steve Worsley.
STEPHEN WORSLEY (VP/Business Dev.):
All of our foundational of science and positive clinical results consistently point us to bavituximab's potential as a high value, next generation anti-cancer agent, and in the last 3mos. these achievements have compelled others to align with us as we continue to develop bavituximab.
In May, Peregrine announced an exciting collaboration with Memorial Sloan Kettering Cancer Center [5-29-15: http://tinyurl.com/qxu4w2x ] to evaluate combinations of bavituximab with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new & increasingly effective anti-cancer treatments. Only 3mos. months later, we announced a new collaboration with AstraZeneca [8-24-15: http://tinyurl.com/owlxpsf ] to evaluate bavituximab in combination with AstraZeneca's investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab also known as MEDI4736, in multiple solid tumors.
By aligning with these world leaders to develop novel immuno-oncology combination therapies, we are positioning ourselves to maximize the potential role that bavituximab can play in this new era of innovative immuno-oncology [I-O] treatments. We believe these collaborations validate the promise of bavituximab and are indicative of the well-validated science we are conducting at Peregrine. We are in the fortunate position to have full rights to this valuable Phase III asset. We believe with the substantial scientific support and compelling data, bavituximab will continue to bring world leading organizations to the partnering table. I will now turn the call over to CFO Paul Lytle, who will discuss the company’s financial performance and our Avid Bioservices business.
CFO Paul Lytle:
Let me shift gears now and give you a brief overview of our financials. Peregrine continues to carefully weigh its opportunities & expenditures with its current cash and financing options. Our strategic investment in the Avid Bioservices business is already starting to pay dividends. Our clients are reserving mfg. slots in the new facility, which has increased our revenue backlog to approx. $42mm. In addition to our backlog, current qtr revenue hit an all-time high at $9.4mm, representing a 71% increase in revenue compared to the same prior year qtr. For the full FY2016, we expect mfg. revenues to increase to a range of $30-35mm. We also believe this business has more opportunity to grow, as our 2nd mfg. facility has the capacity to generate approx. $40mm in new revenue. The new mfg. suite is fully built and the 1st internal pilot run is currently underway to verify all systems & equipment are properly functioning. We plan to announce the official launch of the facility in the near-term, which meets both our internal mfg. timelines and those of our clients. As mentioned on previous calls, the non-dilutive income generated from our mfg. operations continues to offset the amount of capital we need to raise by other means. Plus, it’s important to note that preparing bavituximab for commercial production is a significant financial endeavor and this strategic asset saves us millions of dollars each year in manufacturing cost.
Now turning to expenses, R&D expenses for the qtr increased as we continue to invest in our Phase III SUNRISE trial, while SG&A expenses remained flat qtr-over-qtr. A more detailed analysis of our statement of operations is included in our Form 10-Q that was filed today [ 9-9-15 10-Q: http://tinyurl.com/pemub47 ]. This concludes my financial overview. Now I’ll turn the call back over to Steve to discuss some important upcoming milestones.
CEO STEVE KING – MILESTONES:
It’s only been 8 weeks since we last reported our financial results and development progress, yet in this short time much has been done. We remain on track to complete enrollment in our cornerstone SUNRISE trial, and based on the compelling data we’ve seen to-date from other clinical, translational, and pre-clinical research we are deep into planning for our next set of trials in NSCLC & Breast cancers. Our pre-clinical work continues to provide an ever expanding understanding about the expense mechanism of action, and importantly it is leading us to identify the most promising therapeutic combinations with other immune stimulatory agents, ensuring that bavituximab will play a critical role in the treatment of cancer, regardless of which protocols are in use. These study results, particularly as they relate to the potential synergies between bavituximab and checkpoint inhibitors, create great excitement for us as we begin and continue work with our new collaborators at Memorial Sloan Kettering Cancer Center and AstraZeneca, while we continue our long standing relationship with the Univ. of Texas SW Medical Center, where this technology was originally developed. We look forward to initiating new collaborations, completing enrollment in our SUNRISE trial, beginning dosing in our new NSCLC cancer and Breast cancer studies, and announcing the opening of Avid’s new mfg. site, all in the next few months. These are exciting times for Peregrine and we will continue to keep you posted on all these projects as we make progress.
Q&A: [17:20 mark]
1. Joe Pantginis – Roth Capital Partners: [ http://www.roth.com & https://roth2.bluematrix.com/docs/pdf/BLUE.pdf ]
JP: ” I’d like to focus 1st on the Lung cancer area. With SUNRISE, have you seen any impact with regard to the recent approval of Opdivo for enrollment in this study?”
Steve King: I can take a first stab at that and then Joe can jump in. Basically, we haven't really seen any impact of it in the enrollment itself. Obviously these drugs are just now coming on the market or even aren't yet available for reimbursement, so at this point it hasn’t been a much of an issue. On the other side of it, it opens up in our new studies the combination potential in those indications where now insurance can help pay for those drugs as a standard of care. So, based on the timing of when we anticipate completing the study and the fact that this is the worldwide study including not just sites in the U.S. but actually the majority of our sites are in Europe and in the Asia-Pacific region, we don't anticipate this will be a hindrance for us completing enrollment on the scheduled timeframe by the end of the year.
JP: “The lung cancer treatment landscape with the Opdivo approval is evolving pretty quickly. So how do you see Bavi fitting into that landscape?”
Steve King: That’s a great question. We view that chemotherapy is still going to be a major part of the way cancer patients are treated, and that's in NSCLC, as well as other indications. So, I think while the order in which drugs are used may change over time, eventually the immuno-oncology agents may end up in a frontline setting. We really feel like bavituximab is in an excellent position because we will have the data coming from the SUNRISE study, supporting the effectiveness with docetaxel, which is one of the std. treatment paradigms in 2nd-line NSCLC. But also, the new study we're initiating by year end, in which we're combining with Opdivo, will then give us we think a nice set of data coming in the immuno-oncology combination. So, as everything sorts itself out and new agents come into the marketplace, then we'll be in a good position for combining with either one of those treatment modalities. Joe, do you want to expand?
Joe Shan: Much like in the melanoma space, the initial approvals of immuno-oncology agents are as single agent therapies. But, you can see there is obviously a lot of combination work being done, and I think that's where bavituximab is really uniquely positioned in our mechanism can really complement a host of different other mechanisms. The tolerability of the compound in our trials to-date lends itself to be combined with different therapies. So, I think there are going to be a lot of opportunities for making other products work better.
JP: ”In addition to AstraZeneca, what addl. types of partnerships or collaborations are you targeting at this point?”
Steve Worsley: What we're looking to do is to do fundamentally more of the collaborations like we formed with AstraZeneca - partnering, which we cannot speculate on right now due to forward projection – it’s certainly within the wheelhouse of the organization. We will and have seen quite an uptick with regards to the activities in companies looking at us because of these initial collaborations that we’ve set up this year. So we anticipate more of the same.
Steve King: Just to expand on that a little bit, our stated goal has been ex-U.S. partnering, to primarily keep as much of the U.S. rights as we can, and that remains intact. The reason obviously at this point of partnering is to allow us to expand what we are doing with bavituximab program. We’ve obviously got a number of new clinical studies we’ve identified as being important to start over the coming months, in order so that we can have the data from those studies by the time we unblind the SUNRISE study, but also to be able to run addl. studies in other combinations and other indications, because clearly that’s where the value of the program can be driven upward is through addl. indications and utilization of bavituximab across many different treatment paradigms.
2. Charles Duncan – Piper Jaffray [http://www.piperjaffray.com – 3-5-13 Initiates PPHM: http://tinyurl.com/bxhntk3 ]
CD: ”Congratulations on some of the recent research collaborations. Re: SUNRISE, thanks for the update that enrollment is on track to complete by yr-end. Joe, could you give us now, or plan to in the future, any sense of kind of blinded patient characteristics and geographic backdrop in terms of where those patients came from, any sense of how the enrollment is going with regard to previous lines of therapy et cetera?”
Joe Shan: I think we’re going to probably wait till at least after the 1st interim analysis and completion of our enrollment before announcing any kind of demographics on the trial.
CD: ”I know that it’s an event driven trial, but can you gauge at all when your sense is that you might get to that 1st interim of 33% of the events?”
Joe Shan: No, we can’t really guess right now.
Steve King: Charles, what we’ve been projecting is probably 1st-half of next year for the 1st interim data look, and then probably mid-year on for the 2nd interim data look and for the final unblinding - that’s based on sort of just general projections of how we expect the patients to do in the study overall. As we get further along, we’ll be able to get a little bit better guidance as we see more events take place and feel like we’re getting closer to those actual unblinding with interim data looks.
CD: ”That make sense to me and I appreciate the added color - good execution thus far. Re: the recent AstraZeneca deal, that level or types of diligence did they conduct and was the motivation the publication that came out roughly mid-year, or earlier this year, a key driver to that collaboration?”
Steve King: Steve & Joe can talk bit more about it, but in general it’s great to have a partner like AstraZeneca, who is essentially associating with us and wants to see how bavituximab may be able to impact the treatment paradigm with their particular PD-L1 inhibitor. And so I think that before any big company enters into those sort of collaborations, there is a significant amount of work that they do on their side to pick the right partners also, because there are lots & lots of opportunities for these big companies to collaborate with people, and so they want to make sure that they utilize their resources wisely as well. So, I think it was a great process; I think that it was really driven by a combination of some of the publications that have come out, but also by some of the data we’ve been presenting since last fall showing the real potential of bavituximab to enhance the potential number of patients that will respond to PD-1 or PD-L1 inhibitors. So, it’s really been a combination of the constant news flow we’ve had thats really driven that interest. Joe or Steve, do you want to add to that?
Steve Worsley: The thing with regard to due diligence is they [AstraZeneca] take these types of collaborations very seriously. There is a serious significant investment that they make in terms of investigating combination therapies out there, and again, don’t think that there is anything that’s out of the ordinary, but it was definitely a very exhaustive process they have conducted.
CD: ”Re: that current interaction, does that in anyway put them in a pole position for further interactions with you or is it simply, ‘let's do this experiment together?’”
Steve Worsley: I think it positions them [AstraZeneca] as an organization that is going to get used to working with our compound. By no means are they in a pole position for future partnering activities.
CD: ”But certainly they know you folks, they know what bavituximab could do and its mechanism and then how it interacts with their compound.”
Steve King: Yeah, I think that's the main advantage from their standpoint. We'll work very closely with them in this clinical trial as well as also discussing other potential types of collaborations and other trials and what have you, so it’s clear that proximity creates something of an advantage, but it's a wide open race.
CD: ”Re: Avid, you spoke about increasing capacity substantially here soon. Could you project any new collaborations partnerships, customer signings in the next 12mos. or so?”
Paul Lytle: Right now what we've said publicly is that our current guidance is $30-35mm for this FY. Obviously, Halozyme is one of our key anchor customers that represent a good portion of our revenue, and that supports both their Baxter & Roche collaborations, so it has opportunity to grow as they're successful too. The new facility will support the Bavituximab commercial launch in addition to providing growth for Avid's business, so we're excited about having that business there. We’ve had number of customers come through and inspect the operations and inspect the facility, and everybody believes it's a world-class facility, so we're excited to launch that here shortly.
Steve King: To add just a little more color on that, on the new facility, a lot of the interest comes from the existing client base, even as much as we've had new potential customers coming through. We’re very happy with the response to the new facility, and I think it really opens the door even for future additional expansion, if that's the direction we want to move. So, it's exciting, it's a real nice showpiece and it's really showing in the interest that it's generated from the existing client base.
3. George Zavoico – Jones Trading http://jonestrading.com
GZ: ”Good qtr certainly for Avid and the partnership and the collaboration. Re: the rapidly changing landscape with Opdivo & Krytruda in lung cancer which certainly probably multiplies the possibilities for combinations. But that also begs the question of the patients that are enrolled in SUNRISE, will you know for patients that are still being enrolled, what their PD-1/PD-L1 status may be, if that's part of the plan? Is that something you’re now starting to do now that those assays are becoming available?”
Joe Shan: Yes, that's something we're looking at in an exploratory fashion, George. We’re not requiring pretreatment biopsy specimens to get onto the study. It's on a volunteer basis, and we are collecting a significant proportion of patients' samples. So but we'll see at the end of the day is this enough to give us a hint that how predictive the primary assessments are at predicting outcomes.
GZ: ”How about in terms of going forward, like for example, when the patients begin these progressions and the subsequent therapies that the patients will then undergo after they’ve quit Bavi & docetaxel. Some of those patients might be tested for PD-L1 and go onto an immuno-oncology drug, Opdivo especially, and they may end up skewing or perhaps biasing somehow those sets of patients in survival post-SUNRISE. How do you look at that potential possibility?”
Joe Shan: We definitely collect what [???] patients go on, that’s standard for a survival, primary endpoint trial and you plan a multivariate analysis based on variables like that.
Steve King: A lot of this bias should be handled by the fact that it is a placebo-controlled study, so you would think those patients would equally fall into either of the arms of the study post being on the treatment in the SUNRISE study. As we go forward I think there are a lot of interesting things that we want to do with regard to the PD-L1 status, because from this translation data we were able to show at ASCO and around that time period this year really highlights the fact it may be actually PD-L1 negative tumors that we can have a significant impact in. And so, as we continue to go forward with the plan studies as well as other studies that are sort of more in the development phase, the ability to look at PD-L1 negative tumors and actually select for those patients, take a look at how they do on treatment with bavituximab, where we think we can enhance the effectiveness of a PD-1 or PD-L1 inhibitor is one trial design that I know has gotten a lot of interest. In addition, there is potential of, kind of the other way around, of eventually taking PD-1 refractory patients and actually then adding bavituximab to treatment regimen and seeing if we could convert those into responding patients. We’ll certainly be doing as much analysis as we can in the SUNRISE trial, sort of retrospectively, and looking for imbalances from what we can tell in the PD-1 & PD-L1 status.
GZ: ”Re: the [AZN] collaboration – you’re planning to increase the number of programs & trials you have going. This adds cost. Do any of these collaborations, AstraZeneca or MSK, providing any funds or is it going to be mainly funded by Peregrine? How much of non-dilutive cash might come in to help you with the AZ and the other trials that you are starting?”
Steve King: As part of the AZ collaboration, they are providing the drug itself, which is a huge cost benefit in this sort of trial, because it allows us, #1, full flexibility to start study as quickly as we can, and it takes the need to purchase any of the PD-1, PD-L1 inhibitors off the table. So, that's a significant benefit for their participation in the current collaboration. For us, again, one of the key drivers was the ability to get these studies started as soon as we can, because our bigger goal here is to be able to generate data by the time that we unblind the SUNRISE study, and the reason for that is, ‘planning for success’ - with good SUNRISE data and with addl. combination immunotherapy data in hand, it gives our regulatory group a lot more to work with when it comes to totally working with the FDA toward eventual label claims and what have you, as we look toward approval. For us right now, time is of the essence. As we go forward, we will be wanting to find probably more partners that can bring addl. funding to the table and help us to run studies, either from an operational standpoint, because again we have a relatively small internal group so we’re somewhat limited in the number of studies we can run simultaneously, but also some of the funding to take care of patient costs and what have you. At this early stage the most important thing for us was to have control of the study, be able to get it up and running because we feel the real value is getting this data in a timely fashion because of the value it can a add on the back end.
GZ: ”Re: the AZ study, have you guided at all when the first AZ study might start?”
Joe Shan: Not yet. We are working with the teams very closely to finalize the protocols, internal design right now. When we have a better handle on the operational timelines we’ll provide that.
GZ: ”The Memorial Sloan Kettering, those are mainly translation studies, correct?”
Steve King: Correct. That's the recent [MSK] collaboration that was announced. They are very active in the clinical space and they’ve got lots of good ideas for clinical studies. They've also got good relationships with a lot of the key players in the immuno-oncology space, within the PD-1/PD-L1 space specifically. So, we think that down the road they will be very active in both our clinical & preclinical research programs.
GZ: ”Great. Look forward the start of those trials and some of the data in upcoming conferences. Thank you.”
4. Rahul Jasuja - Noble Life Science Partners http://noblelsp.com/research
RJ: ”Thanks for taking my question and glad to know that PS is now being recognized by big pharma and the immunology mafia so to say. So couple of questions pertaining to how PS with checkpoint inhibitors is differentiated from the conventional checkpoints inhibitors, like the CTLA-4, PD-1, PD-L1 pathways. From the data that's been represented at ImVacS, and also in discussions they’ve had with you guys, besides blocking just PS binding to its receptors on immune cells, there is an FC- FC gamma -based effector function leading to ADCC and so on and so forth - an immune effector function that other checkpoint inhibitors do not have blocking PD-1 or blocking CTLA-4. So, you’ve got a component here that is beyond other checkpoint inhibitors, that brings in an effector function that others do not have. Looking at that aspect or looking at some of the translational data, do you think that beyond the PD-1, PD-L1 non-immunogenic tumors, there is a possibility that PS blockade will have far more effect potentially driving [ph??] antigen presentation. Any comments on that?”
Steve King: That’s a very important point you're bringing up. Because we're blocking an inhibitor, you still need an activating function to get an immune response started. It's a little bit different with PD-L1/PD-1 inhibitors, because there, you’ve basically brought up a marker to stop an active immune response. So, taking away the stopping of that immune response actually allows that continuing immune response to take place. But for the more upstream checkpoints, it's important to have that activating event, and in fact it's taking away the negative PS signal and then activating this T-Cell response which we think is such a great fit with PD-1 & PD-L1 inhibitors. Absolutely you're right, you need that activating event when you're taking off the breaks, if you will, upstream, because you still now need that trigger point for getting the immune response started, and so we're providing that with, as you said, the FC gamma receptor interaction.
RJ: ”And also the other question sort of stems from the fact that there is much said about connecting Adaptive with Innate immunity and there is a bunch of NK cell strategies in play as well. From the cytokine profiles and looking at the translational work that you guys presented at ImVacS in lung cancer [8-15-15 Dr. Jeff Hutchins http://tinyurl.com/qz64pzg ] , you're seeing interferon [???], IL-12, and other cytokines, and also converting M2 to M1 macrophages. How significant do you think your approach would be, because you could be connecting Innate to Adaptive, where other strategies do not really do that – some of the other strategies like [???], but no other checkpoint inhibitor does that. Any color on that aspect, where you could be connecting both the arms of the immune system, and deciding which tumors you're going to attack nd how the combination approaches may pan out?”
Steve King: I think you've made a great point, because what we're doing to this activating events in starting this immune response is basically taking away that natural defense the tumor has for stopping all immune responses, so that's both a native & adaptive killing of the tumor. We're really basically reversing that, probably equally on both sides of the equation, and we have good data that supports that. As you're pointing out, it really brings up a lot of new different types of combinations that we can pursue, and it's one of the reasons over the long run, our goal is, outside of PD-1, PD-L1, to combine with other immune-modulating technologies, and that could even include dendritic cell vaccines, other types of vaccine approaches, as well as the again the litany of different checkpoints that play a role downstream of our target. So, it really opens lot of doors for us, and, as Joe Shan alluded to earlier, one of the great things about bavituximab has been that it seems really very combinable so far with other agents. By having this good safety profile really allows us lot of opportunity on the combination front.
RJ: ”Great. On the SUNRISE trial, are you guys actually measuring all the cytokines that were measured in the translation work with the microspheres like IL-12, interferon gamma, IL-10, or all those not being measured for each patient?”
Joe Shan: Yes, that’s part of our exploratory endpoint, so we have a number of immune correlate testing, most of those are blood-based of course. So, yes, we do plan to look at changes in the cytokine levels. We've been working with some labs to develop ultra-sensitive assays.
Steve King: And it will also be a major part of lot of the new studies we are starting as well, because as we’ve learned more, even since we started the SUNRISE trial, we’ve now got the ability, with all the new translational data in hand, to really design some of these new studies with some of these addl. endpoints in mind – we’re looking at cytokine profiles, changes in macrophage profile, MDSE levels, all those things.
RJ: ”Great, thank you. That’s all I had guys.”
Steven W. King - President and CEO
5. Thomas Yip (MLV & Co.): http://www.mlvco.com
TY: ”…Congrats on a nice quarter, good to see Avid continues to grow. What would be considered a successful outcome for the Phase I/Ib trial that is in partnership with AstraZeneca?”
Joe Shan: I think first, we will need to establish the tolerability of the two investigational agents. So success would mean we can move into the triple combo setting and then just continue with kind of a basket design, where we look at several different tumor types, combining with the std. chemotherapy as well. They’re Phase I, so they’re uncontrolled studies, but these likely will be heavily pretreated patients. So, if we see some good responses, then that would be considered a signal and then we can always expand each cohort of the trial to other addl. tumor types.
Steve King: As in any of the basket studies, one of the nice things here is that we’ll have the ability to take a look at the PD-L1 status of the tumors as they go into treatment - do we see differences between PD-L1 negative, PD-L1 positive? So, alot of exploratory work can be done in this sort of trial design that we are looking at. And, I think, particularly for me, it’s exciting because we’ve gotten a lot of good clinical data combining bavituximab with chemotherapy, and this is a great opportunity to now expand that into that nice combination with a nice novel PD-L1 inhibitor to take a look at what a triple combination can do. I think that’s another huge benefit here, because particularly as we think about how the treatment landscape is going to change in various indications, still the majority of patients in most indications are not getting cured. So, the more we can figure out how to fit bavituximab in the different treatment regiments, we think the more value it will have overall in the program.
TY: ”Re: Avid, I see that the backlog is now ballooned to $42mm. Just wondering, what is the accounting treatment of that backlog increase, does it go to customer deposits or does it increase deferred revenue?”
Paul Lytle: Our typical contract requires an upfront deposit to secure that mfg. slot - that would actually go into customer deposits, and then once we kick off that mfg. run it gets booked into deferred revenue until that run is shipped, and then it moves into revenue. The $42mm in backlog right now covers work to be completed during FY’16 [May’15-Apr’16] and also into FY’17, so it's a combination of both years in terms of timing.
TY: ”Again, great to see Avid continue to grow and also while expanding bavituximab’s clinical footprint at the same time.”
MR. KING’S CLOSING COMMENTS:
I'd like to thank all of you again for participating in today's phone call. In closing, I would like to again express our enthusiasm around the bavituximab program and our growing biomanufacturing business. We are already helping other companies treat patients worldwide through our mfg. services, and we look forward to the potential that our own product, bavituximab, can play in helping the countless patients battling cancer worldwide by helping them to overcome the immune suppression so commonly found in the tumor environment. In closing, as always, I want to thank our stockholders for their continued support, and I’d like to especially thank our patients and their families that are participating in our bavituximab clinical trials. With that we will conclude the call.
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = == = = =
9-9-15 PR: “Peregrine Pharmaceuticals Reports Financial Results for Q1/FY2016 and Recent Developments”
• Peregrine and AstraZeneca to Collaborate on Immuno-Oncology Combination Clinical Trial
• Phase III SUNRISE Clinical Trial on Track to Complete Patient Enrollment by Calendar Year-End 2015
• Avid Bioservices Reports $9.4 Million in First Quarter Revenue
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=930887
TUSTIN, Sept. 9, 2015: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM) (NASDAQ:PPHMP), a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer, today announced financial results for the first quarter of fiscal year (FY) 2016 ended July 31, 2015, and provided an update on its advancing clinical pipeline and other corporate developments.
HIGHLIGHTS SINCE APRIL 30, 2015:
"Over the years, Peregrine's foundational science and positive clinical results have consistently pointed to bavituximab's potential as a high-value, next-generation anti-cancer agent," said Steven W. King, President and CEO of Peregrine. "In the last three months, these achievements have compelled others to align with us as we continue to develop bavituximab. In May, Peregrine announced an exciting collaboration with Memorial Sloan Kettering Cancer Center [5-29-15: http://tinyurl.com/qxu4w2x ] to evaluate combinations of bavituximab with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anti-cancer treatments. Only three months later, we announced a collaboration with AstraZeneca [8-24-15: http://tinyurl.com/owlxpsf ] to clinically evaluate bavituximab in combination with AstraZeneca's investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab (MEDI4736) in multiple solid tumors. These collaborations with world leaders in immuno-oncology speak to the promise of bavituximab and validate our ever-growing enthusiasm for the investigational product. We look forward to advancing both of these programs and completing enrollment of our SUNRISE trial in the next few months."
CLINICAL DEVELOPMENT HIGHLIGHTS
Peregrine and AstraZeneca entered into a cancer immunotherapy clinical trial collaboration to evaluate bavituximab in combination with AstraZeneca's investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab (MEDI4736). The planned Phase I/Ib trial will evaluate the safety and efficacy of bavituximab in combination with durvalumab in multiple solid tumors. Peregrine is working closely with AstraZeneca to finalize the trial design.
Phase III SUNRISE clinical trial in non-small cell lung cancer (NSCLC) continues to enroll patients and remains on track to complete patient enrollment by end of calendar year 2015.
Peregrine announced plans to expand the bavituximab clinical development program to include a Phase II trial to evaluate the combination of bavituximab and Opdivo® (nivolumab), an anti-PD-1 antibody, in previously treated, metastatic NSCLC [6-1-15: http://tinyurl.com/qxu4w2x ]. This trial is expected to be initiated by the end of calendar year 2015.
Peregrine announced plans to expand the bavituximab clinical development program to include a Phase II/III trial to evaluate bavituximab with chemotherapy combinations in HER2-negative metastatic breast cancer [6-1-15: http://tinyurl.com/qxu4w2x ]. This trial is expected to be initiated by the end of calendar year 2015.
SUPPORTIVE RESEARCH HIGHLIGHTS
Peregrine and Memorial Sloan Kettering Cancer Center entered into a research agreement [5-29-15: http://tinyurl.com/qxu4w2x ] to explore the potential of Peregrine's proprietary PS-targeting antibody platform. The goal of the research is to identify effective treatments combining bavituximab with other checkpoint inhibitors or immune stimulating agents.
New data presented at the International Association for the Study of Lung Cancer's (IASLC's) World Conference on Lung Cancer (WCLC) [9-8-15: http://tinyurl.com/p9eduac ] from a translational study of bavituximab demonstrated the ability of bavituximab, alone or in combination with docetaxel, to induce signs of immune activation in non-small cell lung cancer (NSCLC) patient-derived tumor samples, particularly when there was negative PD-L1 expression in the tumor sample. These data further support the potential mechanistic synergies for bavituximab with chemotherapy and checkpoint inhibitors targeting the PD-1/PD-L1 pathway.
Summary data presented at the Combination Immunotherapy Strategies session at the 10th Annual Immunotherapy and Vaccine Summit (ImVacS) [ 8-15-15/Dr.J.Hutchins: http://tinyurl.com/qz64pzg ], highlighted key findings from several recent bavituximab-focused studies including: the potential of bavituximab to shift the tumor microenvironment from immuno-suppressive in which tumors evade immune detection to a state of immune activation in which the immune system recognizes and fights the tumor; bavituximab's potential to increase the number of activated CD8+ cells in the tumor, which stimulates PD-1 expression, potentially increasing the number of patients able to respond to PD-1 and PD-L1 targeting immunotherapies; and, results from several clinical and preclinical studies in a range of tumor types showing that bavituximab and bavituximab-like antibodies, in combination with conventional therapy, have consistently demonstrated estimated survival curves that plateau.
Data from preclinical studies presented at the 2015 ASCO annual meeting [6-1-15: http://tinyurl.com/qxu4w2x ] demonstrated the ability of the company's PS-targeting antibodies to significantly increase the prevalence of tumor infiltrating CD8+ T-cells and immune-activating cytokines, while decreasing tumor-promoting macrophages and myeloid cells. These findings highlight the ability of the antibodies to enhance the anti-tumor effects of both chemotherapy and immune checkpoint inhibitors.
AVID BIOSERVICES HIGHLIGHTS
Avid's new manufacturing suite is fully constructed and the first internal pilot run is currently underway to verify all systems and equipment are properly functioning. Company plans to announce the launch of the new facility in the near term, allowing us to meet our internal manufacturing timelines as well as those of our third-party clients.
Contract manufacturing committed backlog reached $42 million from existing customers covering services to be completed in FY 2016 and into FY 2017.
CORPORATE HIGHLIGHTS
The European Patent Office (EPO) granted Patent Number 2,269,656, licensed to Peregrine titled "Selected Antibodies Binding to Aminophospholipids and their Use in Treatment, Such as Cancer." The patent covers bavituximab as a composition of matter and for use in therapy, such as for treating cancer including in combination with radiotherapy or chemotherapy, e.g., with docetaxel. This important patent expands upon the company's intellectual property portfolio, which now numbers more than 140 worldwide issued patents and pending applications for the bavituximab oncology program.
[ http://worldwide.espacenet.com/publicationDetails/biblio?DB=EPODOC&II=0&ND=3&FT=D&date=20141202&CC=PT&NR=2269656E&KC=E ]
FINANCIAL RESULTS
Total revenues for the first quarter of FY 2016 were $9,671,000, compared to $5,496,000 for the same quarter of the prior fiscal year. The increase was primarily attributed to an increase in contract manufacturing revenue generated from Avid Bioservices.
Contract manufacturing revenue from Avid's clinical and commercial biomanufacturing services provided to its third-party clients for the first quarter FY 2016 were $9,379,000, compared to $5,496,000 for the same quarter of the prior fiscal year. Peregrine expects third-party contract manufacturing revenue for FY 2016 to be between $30 and $35 million. In addition to providing biomanufacturing services to its third-party clients, Avid will continue to support the clinical and potential commercialization of bavituximab.
Total costs and expenses in the first quarter of FY 2016 were $23,425,000, compared to $18,667,000 in the first quarter of FY 2015. This increase was primarily attributable to current quarter increases in research and development expenses associated with the SUNRISE Phase III trial and increases in the cost of contract manufacturing associated with higher reported revenue. For the first quarter of FY 2016, research and development expenses were $13,918,000, compared to $10,201,000 for the first quarter of FY 2015. For the first quarter of FY 2016, cost of contract manufacturing was $4,608,000, compared to $3,583,000 for the first quarter of FY 2015.
Peregrine's consolidated net loss attributable to common stockholders was $15,101,000, or $0.08 per share, for the first quarter of FY 2016, compared to a net loss attributable to common stockholders of $14,157,000, or $0.08 per share, for the same prior year quarter.
Peregrine reported $59,016,000 in cash and cash equivalents as of July 31, 2015 compared to $68,001,000 at fiscal year ended April 30, 2015.
More detailed financial information and analysis may be found in Peregrine's Quarterly Report on Form 10-Q, which will be filed with the Securities and Exchange Commission today. [ 9-9-15: http://tinyurl.com/pemub47 ]
CONFERENCE CALL
Peregrine will host a conference call and webcast this afternoon, September 9, 2015, at 4:30 PM EDT (1:30 PM PDT). To listen to the conference call, please dial (877) 312-5443 or (253) 237-1126 and request the Peregrine Pharmaceuticals conference call. To listen to the live webcast, or access the archived webcast, please visit: http://ir.peregrineinc.com/events.cfm .
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials focused on the treatment of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small cell lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
THREE MONTHS ENDED
July 31, 2015 July 31, 2014
Unaudited Unaudited
REVENUES:
Contract manufacturing revenue $ 9,379,000 $ 5,496,000
License revenue 292,000 -
Total revenues 9,671,000 5,496,000
COSTS AND EXPENSES:
Cost of contract manufacturing 4,608,000 3,583,000
Research and development 13,918,000 10,201,000
Selling, general and administrative 4,899,000 4,883,000
Total costs and expenses 23,425,000 18,667,000
LOSS FROM OPERATIONS (13,754,000 ) (13,171,000 )
Interest and other income 31,000 42,000
NET LOSS $ (13,723,000 ) $ (13,129,000 )
COMPREHENSIVE LOSS $ (13,723,000 ) $ (13,129,000 )
Series E preferred stock accumulated dividends (1,378,000 ) (1,028,000 )
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS $ (15,101,000 ) $ (14,157,000 )
WEIGHTED AVERAGE COMMON SHARES OUTSTANDING
Basic and diluted 197,317,374 179,118,255
BASIC AND DILUTED LOSS PER COMMON SHARE $ (0.08 ) $ (0.08 )
PEREGRINE PHARMACEUTICALS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
JULY 31,
2015 APRIL 30,
2015
Unaudited
ASSETS
CURRENT ASSETS:
Cash and cash equivalents $ 59,016,000 $ 68,001,000
Trade and other receivables, net 1,805,000 3,813,000
Inventories 10,457,000 7,354,000
Prepaid expenses and other current assets, net 1,052,000 1,355,000
Total current assets 72,330,000 80,523,000
Property and equipment, net 18,395,000 15,124,000
Other assets 1,307,000 1,817,000
TOTAL ASSETS $ 92,032,000 $ 97,464,000
LIABILITIES AND STOCKHOLDERS' EQUITY
CURRENT LIABILITIES:
Accounts payable $ 9,840,000 $ 10,385,000
Accrued clinical trial and related fees 4,106,000 3,910,000
Accrued payroll and related costs 3,094,000 4,606,000
Deferred revenue 8,291,000 6,630,000
Customer deposits 9,599,000 11,363,000
Other current liabilities 620,000 437,000
Total current liabilities 35,550,000 37,331,000
Deferred rent, less current portion 1,036,000 1,098,000
Commitments and contingencies
STOCKHOLDERS' EQUITY:
Preferred stock - $0.001 par value; authorized 5,000,000 shares; issued and outstanding - 1,574,764 and 1,574,764, respectively 2,000 2,000
Common stock-$0.001 par value; authorized 325,000,000 shares; issued and outstanding - 200,983,948 and 193,346,627, respectively 201,000 193,000
Additional paid-in capital 522,590,000 512,464,000
Accumulated deficit (467,347,000 ) (453,624,000 )
Total stockholders' equity 55,446,000 59,035,000
TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY $ 92,032,000 $ 97,464,000
• Jay Carlson Peregrine Pharmaceuticals, Inc. 800-987-8256 info@peregrineinc.com
• Stephanie Diaz (Investors) Vida Strategic Partners 415-675-7401 sdiaz@vidasp.com
• Tim Brons (Media) Vida Strategic Partners 415-675-7402 tbrons@vidasp.com
- - - - - - - - -
[ From 10-Q header: “As of Sept 4, 2015, there were 202,124,031 shares outstanding.”
7-31-15 10-Q/pg.17/OTHER LEGAL MATTERS – CMS SETTLEMENT EXECUTED 9-8-13:
“On 9-24-12, we filed a lawsuit, captioned Peregrine Pharmaceuticals v. Clinical Supplies Management (CSM), Case# 8:12-cv-01608, against CSM, in the U.S. District Court for the Central District of California. In 2010, we had contracted with CSM as our 3rd-party vendor responsible for distribution of the blinded investigational product used in our bavituximab Phase IIb 2nd-line NSCLC trial. As part of the routine collection of data in advance of an end-of-Phase II meeting with regulatory authorities, we discovered major discrepancies between some patient sample test results and patient treatment code assignments. Consequently, we filed this lawsuit against CSM alleging, among other causes of action, breach of contract, negligence, negligence per se, constructive fraud and negligent misrepresentation arising from CSM’s performance of its contracted services. We are seeking monetary damages. On 6-5-14, CSM filed with the court a Notice of Motion and Motion for Partial Summary Judgment seeking partial summary judgment on our claims for damages on the grounds that the limitation of liability clauses contained in our master services agreement with CSM are valid and enforceable. Our opposition to CSM’s motion was filed with the court on 6-23-14, and the hearing on the motion was held on 7-28-14. On 7-30-14, the court issued its order holding that the limitation of liability clause did not apply to our claims for active negligence, negligent misrepresentation and constructive fraud, but did apply to our causes of action for breach of contract, passive negligence and negligence per se. On 3-27-15, CSM filed with the court a Notice of Motion and Motion for Partial Summary Judgment seeking partial summary judgment on our claims for damages on the grounds that the causes of action for negligence, negligence per se, negligent misrepresentation, and constructive fraud are barred by the economic loss doctrine, as well as that the causes of action for negligent misrepresentation and constructive fraud cannot be established as a matter of law. Our opposition to CSM’s motion was filed with the court on 4-13-15 and CSM’s reply to our motion was filed on 4-20-15. On 6-22-15, the court issued its order granting CSM’s Motion for Partial Summary Judgment. On 9-8-15, we and CSM entered into a confidential settlement and release agreement to resolve all claims related to the complaint we filed on 9-24-12 against CSM. Pursuant to the terms of the Settlement Agreement, (i) all claims asserted in the litigation by us will be dismissed with prejudice, (ii) each of the parties to the litigation will receive a full release of all claims, of any nature whatsoever, whether known or unknown, and (iii) CSM will pay to us the sum of $600,000 within 30 days. We will record the settlement amount when payment is received.”
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-15 iss. 7-14-15 http://tinyurl.com/ocrtkuj PR: http://tinyurl.com/nw2v5u6 (Cash 4-30-15=$68.0mm)
Latest 10Q 7-31-15 iss. 9-9-15 http://tinyurl.com/pemub47 PR: http://tinyurl.com/ph22vdn (Cash 7-31-15=$59.0mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
.
.
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY16/Q1 (q/e 7-31-15), per the 7-31-15 10-Q (http://tinyurl.com/pemub47 ) issued 9-9-15.
• Total Revs since May’06: ($138.6mm/Avid + $24.1mm/Govt + $2.4mm/Lic.) = $165.2mm
• Deferred-Revs at 7-31-15, going fwd into FY’16/Q2 (q/e 10-31-15), total $8.3mm, UP from the $6.6mm of Deferred-Revs at 4-30-15 that drove into FY’16/Q1.
• Avid’s Gross-Profit over last 3 qtrs: $12.5mm on revs of $24.4mm (GP% = 49%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
Totals: 138591 24149 2416 165156 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
AVID “Total Services”:
AVID OUTPUT$ 3rd-PARTY + PEREGRINE = TOTAL-OUTPUT$
FY09 4-30-09 13mm 10mm $23mm #
FY10 4-30-10 13mm 17mm $30mm #
FY11 4-30-11 9mm 11mm $20mm @
FY12 4-30-12 15mm 11mm $26mm @
FY13 4-30-13 21mm ~10mm ~$31mm ^
LTM ended 1/2010 3rd/$15.3mm + Govt/$8.3mm + PPHM/$8.8mm = $32.4mm *
@SKing 3-18-2013 RothOC/DanaPT (Slide21) http://tinyurl.com/cebtwen
#SKing 7-12-2012 JMP/NYC Conf. (Slide27) http://tinyurl.com/csdclwb
*SKing 3-17-2010 RothOC/DanaPT Conf. (Slide18) http://tinyurl.com/ye9v7jq
^PLytle 7-11-2013 Qtly-CC “Avid did ~$10mm in equivalent services for Peregrine in FY13, which doesn’t get reflected into the fin. statements, it's eliminated in consolidation.”
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.2x)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
StoneR, this was definitely a huge error - SK did not say "opening new bavi mfg. sites". He said, "announcing the opening of Avid’s new mfg. site". See: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=116907891
CEO STEVE KING – MILESTONES: 9-9-15/CC
It’s only been 8 weeks since we last reported our financial results and development progress, yet in this short time much has been done. We remain on track to complete enrollment in our cornerstone SUNRISE trial, and based on the compelling data we’ve seen to-date from other clinical, translational, and pre-clinical research we are deep into planning for our next set of trials in NSCLC & Breast cancers. Our pre-clinical work continues to provide an ever expanding understanding about the expense mechanism of action, and importantly it is leading us to identify the most promising therapeutic combinations with other immune stimulatory agents, ensuring that bavituximab will play a critical role in the treatment of cancer, regardless of which protocols are in use. These study results, particularly as they relate to the potential synergies between bavituximab and checkpoint inhibitors, create great excitement for us as we begin and continue work with our new collaborators at Memorial Sloan Kettering Cancer Center and AstraZeneca, while we continue our long standing relationship with the Univ. of Texas SW Medical Center, where this technology was originally developed. We look forward to initiating new collaborations, completing enrollment in our SUNRISE trial, beginning dosing in our new NSCLC cancer and Breast cancer studies, and announcing the opening of Avid’s new mfg. site, all in the next few months. These are exciting times for Peregrine and we will continue to keep you posted on all these projects as we make progress.
Avid's Gross Profit=$4.8mm for qe7/31/15, 51% GP Margin
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
9-10-15/OutsourcingPharma: “Avid Revs Likely to Grow Substantially”
9-10-15: “Peregrine Up on CMO Q1 Sales & Backlog, as New Plant Set to go Online”
By Dan Stanton, Outsourcing-Pharma
http://www.outsourcing-pharma.com/Contract-Manufacturing/Peregrine-up-on-CMO-sales-and-backlog-as-new-plant-set-to-go-online
Manufacturing backlog at Avid Bioservices has reached $42mm as the firm books up space at a new facility currently undergoing its first internal pilot run. For Q1/FY2016, Peregrine Pharmaceuticals reported record revenue from its contact manufacturing business Avid Bioservices of $9.4mm, up 71% year-on-year. But revenues are likely to grow substantially, the firm said, as there is a $42mm committed backlog from existing customers which will be carried-out in part once a new mammalian cell culture manufacturing facility in Tustin, California comes online.
“The new manufacturing suite is fully built and the first internal pilot run is currently underway to verify all systems and equipment are properly functioning,” Peregrine CFO Paul Lytle said during an investor call yesterday. ”Our strategic investment in the Avid Bioservices business is already starting to pay dividends. Our clients are reserving manufacturing slots in the new facility which has increased our revenue backlog to approximately $42mm.”
A large proportion of the firm’s revenues come from its major client, Halozyme Therapeutics, servicing monoclonal antibody development projects with Roche and Baxter. While the company hopes the new facility will attract new customers, it is the current customer base showing the most interest.
“In the new facility, a lot of the interest comes from the existing client base, even as much as we've had new potential customers coming through,” said CEO Steven King. “It's exciting, it's a real nice showpiece and it's really showing in the interest that it's generated from again the existing client base.”
The site, first announced last year, more than doubles Avid’s manufacturing capacity, though some of the space has been reserved to service its parent company’s lead product bavituximab, a chimeric mAb in Phase III trials for non-squamous non-small cell lung cancer. END

• 6-17-15: Avid’s John Haney (ex-Genentech/Pfizer) speaking at BIO-INTL’5/Philly http://tinyurl.com/pnlquu3 & http://tinyurl.com/nl4vbgk
...”Designing & Implementing Avid’s New State-of-the-Art Single-Use Facility for Late Ph.3 & Commercial Prod.” - SK: "We've seen tremendous interest for production in the new facility, both from new & existing clients"
• 12-10-14: Avid to Double Mfg. Capacity (“expanding client roster; potential commercial launch of bavituximab”) http://tinyurl.com/mmc3qgy & http://tinyurl.com/kmdgq8t
• 3-24-15: Avid Receives CMO Leadership Awards for Its Commitment to Innovation & Reliability http://tinyurl.com/psep47f
• 3-12-13: Avid Q3'FY13 GP=$3.3mm; S.King 3-2012, "We have a profitable CMO, Avid Bioservices" http://tinyurl.com/l97rzm8
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
Updated PPHM REVS-BY-QTR TABLE, now thru FY16/Q1 (q/e 7-31-15), per the 7-31-15 10-Q ( http://tinyurl.com/pemub47 ) issued 9-9-15.
• Total Revs since May’06: ($138.6mm/Avid + $24.1mm/Govt + $2.4mm/Lic.) = $165.2mm
• Deferred-Revs at 7-31-15, going fwd into FY’16/Q2 (q/e 10-31-15), total $8.3mm, UP from the $6.6mm of Deferred-Revs at 4-30-15 that drove into FY’16/Q1.
• Avid’s Gross-Profit over last 3 qtrs: $12.5mm on revs of $24.4mm (GP% = 49%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
Totals: 129212 24149 2416 165156 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
Q1FY16 Avid Revs=$9.4MM(record), GP=$4.8mm, GP%=51% (Licensing-Revs=$292k)
Updated PPHM REVS-BY-QTR TABLE, now thru FY16/Q1 (q/e 7-31-15), per the 7-31-15 10-Q (http://tinyurl.com/pemub47 ) issued 9-9-15.
• Total Revs since May’06: ($138.6mm/Avid + $24.1mm/Govt + $2.4mm/Lic.) = $165.2mm
• ***Deferred-Revs at 7-31-15, going fwd into FY’16/Q2 (q/e 10-31-15), total $8.3mm, UP from the $6.6mm of Deferred-Revs at 4-30-15 that drove into FY’16/Q1(qe 7-31-15).
• Avid’s Gross-Profit over last 3 qtrs: $12.5mm on revs of $24.4mm (GP% = 49%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%*
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%*
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY15 TOTAL: 26,744,000 15,393,000 11,151,000 42%*
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 6630 0 6148
FY16Q1 7-31-15 9379 0 292 9671 8291 0 10457
Totals: 129212 24149 2416 165156 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
.
AVID “Total Services”:
AVID OUTPUT$ 3rd-PARTY + PEREGRINE = TOTAL-OUTPUT$
FY09 4-30-09 13mm 10mm $23mm #
FY10 4-30-10 13mm 17mm $30mm #
FY11 4-30-11 9mm 11mm $20mm @
FY12 4-30-12 15mm 11mm $26mm @
FY13 4-30-13 21mm ~10mm ~$31mm ^
LTM ended 1/2010 3rd/$15.3mm + Govt/$8.3mm + PPHM/$8.8mm = $32.4mm *
@SKing 3-18-2013 RothOC/DanaPT (Slide21) http://tinyurl.com/cebtwen
#SKing 7-12-2012 JMP/NYC Conf. (Slide27) http://tinyurl.com/csdclwb
*SKing 3-17-2010 RothOC/DanaPT Conf. (Slide18) http://tinyurl.com/ye9v7jq
^PLytle 7-11-2013 Qtly-CC “Avid did ~$10mm in equivalent services for Peregrine in FY13, which doesn’t get reflected into the fin. statements, it's eliminated in consolidation.”
.
PPHM’S QTLY. NET LOSS BY QTR:
FY08Q1 7-31-07 4,656,000
FY08Q2 10-31-07 6,207,000
FY08Q3 1-31-08 6,154,000
FY08Q4 4-30-08 6,159,000
FY09Q1 7-31-08 5,086,000
FY09Q2 10-31-08 4,497,000
FY09Q3 1-31-09 3,332,000
FY09Q4 4-30-09 3,609,000
FY10Q1 7-31-09 2,428,000
FY10Q2 10-31-09 2,787,000
FY10Q3 1-31-10 1,538,000
FY10Q4 4-30-10 7,741,000
FY11Q1 7-31-10 7,695,000
FY11Q2 10-31-10 7,513,000
FY11Q3 1-31-11 8,929,000
FY11Q4 4-30-11 10,014,000
FY12Q1 7-31-11 8,092,000
FY12Q2 10-31-11 12,055,000
FY12Q3 1-31-12 11,090,000
FY12Q4 4-30-12 10,882,000
FY13Q1 7-31-12 7,664,000
FY13Q2 10-31-12 8,753,000
FY13Q3 1-31-13 4,914,000
FY13Q4 4-30-13 8,449,000
FY14Q1 7-31-13 7,600,000
FY14Q2 10-31-13 7,790,000
FY14Q3 1-31-14 9,724,000
FY14Q4 4-30-14 10,248,000
FY15Q1 7-31-14 13,129,000
FY15Q2 10-31-14 12,100,000
FY15Q3 1-31-15 12,994,000
FY15Q4 4-30-15 12,135,000
FY16Q1 7-31-15 13,723,000
= = = = = = = =
OPER. CASH BURNS* BY QTR(FROM THE 10-Q/K’S):
FY10Q1 7-31-09 2,024,000 (from 10Q pg.25)
FY10Q2 10-31-09 2,351,000 (Q1+Q2: 4,375,000 pg.28)
FY10Q3 1-31-10 1,158,000 (Q1+Q2+Q3: 5,533,000 pg.30)
FY10Q4 4-30-10 6,375,000 (FY’10: 11,908,000 10K pg.58)
FY11Q1 7-31-10 6,567,000 (from 10Q pg.24)
FY11Q2 10-31-10 6,167,000 (Q1+Q2: $12,734,000 pg.25)
FY11Q3 1-31-11 7,736,000 (Q1+Q2+Q3: $20,470,000 pg.26)
FY11Q4 4-30-11 8,961,000 (FY’11: 29,431,000 10K pg.54)
FY12Q1 7-31-11 6,984,000 (from 10Q pg.25)
FY12Q2 10-31-11 11,668,000 (Q1+Q2: 18,652,000 pg.25)
FY12Q3 1-31-12 8,490,000 (Q1+Q2+Q3: 27,142,000 pg.25)
FY12Q4 4-30-12 11,265,000 (FY’12: 38,407,000 10K pg.55)
FY13Q1 7-31-12 6,742,000 (from 10Q pg.21)
FY13Q2 10-31-12 6,162,000 (Q1+Q2: 12,904,000 pg.23)
FY13Q3 1-31-13 3,597,000 (Q1+Q2+Q3: 16,501,000 pg.23)
FY13Q4 4-30-13 7,053,000 (FY’13: 23,554,000 10K pg.60)
FY14Q1 7-31-13 5,750,000 (from 10Q pg.23)
FY14Q2 10-31-13 5,834,000 (Q1+Q2: 11,584,000 10Q pg.24)
FY14Q3 1-31-14 7,875,000 (Q1+Q2+Q3: 19,459,000 10Q pg.26)
FY14Q4 4-30-14 8,706,000 (FY’14: 28,165,000 10K pg.55)
FY15Q1 7-31-14 11,076,000 (from 10Q pg.23)
FY15Q2 10-31-14 9,947,000 (Q1+Q2: 21,023,000 10Q pg.25)
FY15Q3 1-31-15 11,116,000 (Q1+Q2+Q3: 32,139,000 10Q pg.26)
FY15Q4 4-30-15 10,474,000 (FY’15: 42,613,000 10K pg.54)
FY16Q1 7-31-15 12,306,000 (from 10Q pg.2x)
FY’09 total Op-Burn: $14,715,000
FY’10 total Op-Burn: $11,908,000
FY’11 total Op-Burn: $29,431,000
FY’12 total Op-Burn: $38,407,000
FY’13 total Op-Burn: $23,554,000
FY’14 total Op-Burn: $28,165,000
FY’15 total Op-Burn: $42,613,000
8-18-15: “In July2015, Avid DEBUTED its new state-of-the-art biomanufacturing facility in Tustin, CA”
8-18-15: “Avid Bioservices Adds a New State-of-the-Art Biomanufacturing Facility”
Jon S. Gingrich, Manager Bus. Dev., Avid Bioservices
“In July 2015, Avid Bioservices debuted its new state-of-the-art biomanufacturing facility in Tustin, CA. Designed to be one of the first U.S. late Phase3 and commercial production facilities for the exclusive operation of single-use equipment to produce biologics, the 40,000-ft2 build-out of an industrial 110,000-ft2 warehouse provides the perfect setting to install state-of-the-art modular cleanroom bioproduction suites. This new facility incorporates the relatively recent concept of converting an existing building in to a new bioproduction facility, known as a “brownfield” design. The end result is an innovative and flexible design that will support the transition of your program from late Phase3 into commercial production.”
http://www.bioprocessintl.com/sponsored-content/avid-bioservices-adds-a-new-state-of-the-art-biomanufacturing-facility

Avid Bioservices, Inc. (wholly-owned mfg. subsidiary): http://www.avidbio.com
Avid ad in '13-'14 BPI Industry Yearbook: http://tinyurl.com/mhhs4e2
Avid Linkedin: http://www.linkedin.com/company/avid-bioservices
Avid BioProcessIntl: http://www.bioprocessintl.com/?s=avid+bioservices
• 6-17-15: Avid’s John Haney (ex-Genentech/Pfizer) speaking at BIO-INTL’5/Philly http://tinyurl.com/pnlquu3 & http://tinyurl.com/nl4vbgk
...”Designing & Implementing Avid’s New State-of-the-Art Single-Use Facility for Late Ph.3 & Commercial Prod.” - SK: "We've seen tremendous interest for production in the new facility, both from new & existing clients"
• 12-10-14: Avid to Double Mfg. Capacity (“expanding client roster; potential commercial launch of bavituximab”) http://tinyurl.com/mmc3qgy & http://tinyurl.com/kmdgq8t
• 3-24-15: Avid Receives CMO Leadership Awards for Its Commitment to Innovation & Reliability http://tinyurl.com/psep47f
• 3-12-13: Avid Q3'FY13 GP=$3.3mm; S.King 3-2012, "We have a profitable CMO, Avid Bioservices" http://tinyurl.com/l97rzm8
= = = = = = = = = = = = = = = = = = = = = =
9-6-15: 7-2015 Visiongain Report/CMO’s interviews 2 CMO’s: Avid & Fujifilm/Diosynt. “Our report predicts the world market for producing those therapeutic agents will reach $5.78B in 2019, expanding fast from 2015-2025.”
…You also discover interviews with 2 companies in the industry – FUJIFILM Diosynth Biotechnologies and Avid Bioservices Inc.
LONDON, 9-3-15: “Outsourced Biopharma Production – New Analysis Showing You Revenue Forecasts”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=116781652
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
PPHM REVS-BY-QTR TABLE thru FY15/Q4 (fye 4-30-15), per the 4-30-15 10-K (http://tinyurl.com/ocrtkuj ) issued 7-14-15. Deferred-Revs at 4-30-15, going fwd into FY’16/Q1 (q/e 7-31-15), total $6.6mm, up from the $5.8mm of Deferred-Revs at 1-31-15 that drove into FY’15/Q4.
Total Revs since May’06: ($129.2mm/Avid + $24.1mm/Govt + $2.1mm/Lic.) = $155.5mm
Avid’s Gross-Profit over last 3 qtrs: $9.4mm on revs of $21.2mm (GM% = 44%)
=> Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GM%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 5752 0 6148
Totals: 129212 24149 2124 155485 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
BETABODIES: “Potentially the next generation of PS-targeting cancer therapeutics”
BETABODIES (ex: KL15), “potentially the next generation of PS-targeting cancer therapeutics” - generated by fusing domains of the PS-binding the plasma protein, B2-glycoprotein I (B2GPI), to the Fc region of mouse IgG2a. Such ‘betabodies’ potentially have the following advantages: they bind directly to PS and do not require a cofactor protein (B2GPI) for binding; they can be made fully human; they are smaller in size (100 vs. 250 KDa); and they have slower blood clearance rates (half-life of ~5days vs. Bavi’s ~1day).
…See 7-6-13, “Entdoc, here’s a bunch of stuff on Betabodies” http://investorshub.advfn.com/boards/read_msg.aspx?message_id=89680206http://tinyurl.com/khopa3d
- - - - - - - - - - - - - - - -
UTSW/PPHM’s BetaBodies patent AWARDED(GRANTED) 2-17-15, U.S. Patent #8,956,616
‘BETABODIES’ patent app #20060228299, filed 1-24-2006, pub 10-12-2006, AWARDED 2-17-2015…
"Constructs Binding to Phosphatidylserine and Their Use in Disease Treatment" ("Betabodies & Receptorbodies”)
Inventors: Philip E. Thorpe, Troy A. Luster, Steven W. King
Assignee1: BOARD OF REGENTS, UNIV. OF TEXAS SYSTEM, 201 W. 7TH ST, AUSTIN, TX
Assignee2: PEREGRINE PHARMACEUTICALS, INC., 14272 FRANKLIN AVE, TUSTIN, CA
USPTO Patent #8,956,616: http://tinyurl.com/nj4jpry (Granted 2-17-15)
ABSTRACT: “Disclosed are new phosphatidylserine binding constructs with surprising combinations of properties, and a range of diagnostic and therapeutic conjugates thereof. The new constructs effectively bind phosphatidylserine targets in disease and enhance their destruction, and can also specifically deliver attached imaging or therapeutic agents to the disease site. Also disclosed are methods of using the new construct compositions, therapeutic conjugates and combinations thereof in tumor vasculature targeting, cancer diagnosis and treatment, and for treating viral infections and other diseases.”
1. A construct comprising an antibody Fc region operatively attached to two .beta.2-glycoprotein I (.beta.2GPI) polypeptides, wherein said .beta.2GPI polypeptides each comprise at least an intact domain V of .beta.2GPI, wherein said intact domain V binds to phosphatidylserine, wherein said .beta.2GPI polypeptides form a dimer when attached to said antibody Fc region and wherein said construct retains the property of binding to phosphatidylserine.
[0015] ReceptorBodies & BetaBodies: The invention first provides a range of phosphatidylserine binding construct compositions, in which the constructs comprise at least a first phosphatidylserine binding protein, polypeptide or receptor operatively attached to at least a first antibody Fc region. Joining a phosphatidylserine binding protein, polypeptide or "receptor" to an "antibody" Fc region gives rise to the terms "receptorbody" and "receptorbodies", which are used herein to refer to the phosphatidylserine-binding Fc constructs of the invention.
- - - - - - - - - - - - - - - - -
• Betabodies (Clipped/Nicked B2GPI - ex: KL15, “2nd-gen. PS-Targeting”) - bind to PS directly, are smaller in size (100 vs. 250KDa) and have a longer serum half-life (~5days) than natural antibodies (Bavi=~1day) – see 7-6-13, “Entdoc, here’s a bunch of stuff on Betabodies” http://investorshub.advfn.com/boards/read_msg.aspx?message_id=89680206
11-6-14 Edcpf/iHub#196158, “Such Betabodies potentially have advantages over bavituximab”: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=107891618
= = = = = = = = = = = = = = =
From Peregrine’s IASCL’15 9-8-15 PR: http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=930479
”Peregrine's PS signaling pathway inhibitor candidates, including bavituximab, reverse the immunosuppressive environment that many tumors establish in order to proliferate, while also fighting cancer by activating macrophages and cytotoxic T cells in tumors. “
Scott Antonia’s (Moffitt CC) 9-8-15 IASLC’15 Mini-Oral Presentation Slideshow (9pgs: Bavi Lung Translational Data) now on PeregrineInc.com, as is UTSW’s Dr. David Gerber’s poster overviewing the ongoing Ph.3 Bavi+Doce 2L/NSCLC “SUNRISE” trial – see below…
(recall, UTSW’s Dr. Rolf Brekken gives an Oral Pres. tomorrow, Sept9, discussing, “previously announced study findings demonstrating that a combination of bavituximab and an anti-PD-1 antibody was more effective at reducing tumor burden in preclinical lung cancer models than either treatment alone.”)
Sept6-9 2015: “IASLC’s 16th World Conf. on Lung Cancer”, Denver
http://wclc2015.iaslc.org/
“The World Conference on Lung Cancer (WCLC) is the world’s largest meeting dedicated to lung cancer and other thoracic malignancies. More than 7,000 delegates come from more than 100 countries to discuss the latest developments in thoracic malignancy research. Attendees include surgeons, medical oncologists, radiation oncologists, pulmonologists, radiologists, pathologists, epidemiologists, basic research scientists, nurses, allied health professionals, advocates, and patients.”
http://www.iaslc.org/events/16th-world-conference-lung-cancer
2015 Exhibitor: Peregrine Pharm. (1 of 39) - booth #1724 (adjacent to Abbvie)
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: Treatment of Advanced Diseases - NSCLC
9/7/15 9:30am-5:00pm, Exhibit Hall (Hall B+C)
POSTER #P1.01-075: “Phase III, Randomized, Double-Blind Trial of Bavituximab Plus Docetaxel in Previously Treated Stage IIIb/IV Non-Squamous NSCLC (SUNRISE) (ID 1581)
David R. Spigel, Joseph Shan, Alberto Chiappori, Ulrich Keilholz, Martin Reck, Martin Edelman, Manuel Domine, Keunchil Park, Tae Won Jang, Wu-Chou Su, Rachel E. Sanborn, Leora Horn, Rebecca Heist, Paul Mainwaring, David E. Gerber (UTSW)
ABSTRACT…
BACKGROUND: Exposed phosphatidylserine (PS) in the tumor microenvironment is highly immunosuppressive. PS binding to PS receptors on myeloid derived suppressor cells (MDSC) and M2 macrophages leads to production of anti-inflammatory cytokines such as TGF-B and IL-10. Bavituximab, a first-in-class PS-targeting monoclonal antibody, counters these effects, resulting in production of pro-inflammatory cytokines such as TNF-a and IL-12, maturation of dendritic cells and induction of tumor specific cytotoxic T lymphocyte (CTL) immunity. Docetaxel has also been shown to suppress MDSCs while increasing tumor antigens and T-cell mediated cytotoxicity, thereby enhancing bavituximab’s immunomodulatory effects. In a prior double-blind Phase II trial in 2nd line non-squamous NSCLC, bavituximab 3 mg/kg + docetaxel was well-tolerated and demonstrated 60% improvement (11.7 vs 7.3 mos) in median overall survival (OS) compared to control.
METHODS: SUNRISE is a Phase III, double-blind trial where patients with previously treated Stage IIIb/IV non-squamous, non-small cell lung cancer are randomized in a 1:1 ratio to receive up to six 21-day cycles of docetaxel in combination with either weekly 3 mg/kg bavituximab or placebo, followed by maintenance with weekly bavituximab or placebo until progression or toxicity. Patients will be stratified by region (North America, Europe, or Rest of World), disease stage (IIIb or IV), and previous maintenance/targeted therapy (yes or no). This trial was initiated in Dec.2013 and accrual of 582 patients across 160+ sites in 14 countries is planned over 24 months. The primary endpoint is OS and 2 interim analyses are planned. Secondary endpoints include progression-free survival (PFS), overall response rate (ORR) and safety. Radiographic tumor response is centrally assessed every 2 cycles during combination therapy and every nine weeks during maintenance. Exploratory analysis will include the assessment of changes in circulating immune cells and cytokines to better understand the immunotherapeutic mechanism.
RESULTS: Trial in progress
CONCLUSION: Trial in progress
POSTER: http://www.peregrineinc.com/images/stories/pdfs/iaslc_2015_sunrise.pdf
From Peregrine’s 9-8-15 PR:
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=930479
"A large and growing body of research continues to support our belief that, as a fundamental checkpoint inhibitor, bavituximab may have the potential to broadly enhance the therapeutic activity of a range of cancer treatments including chemotherapy, radiation and immunotherapies. These latest data being presented at the World Conference on Lung Cancer specifically highlight this synergistic potential in the area of checkpoint inhibitors by clearly illustrating the manner in which bavituximab is able to induce the appropriate immune responses required for a successful therapeutic response to anti-PD-1 & anti-PD-L1 agents," said Steven W. King, President and CEO of Peregrine. "We look forward to continuing our investigation of bavituximab in combination with other immuno-oncology agents through our ongoing collaborations with AstraZeneca, the Memorial Sloan Kettering Cancer Center and UT Southwestern."
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: MINI ORAL #14 - Pre-Clinical Therapy (ID 119)
Moderators: Lynnette Fernandez-Cuesta Adi F. Gazdar
9/8/15, 10:45am-12:15pm, Mile High Ballroom 2c-3c
MINI-ORAL #14.07: “Bavituximab Activates CD8+ TILs in a 3D Ex Vivo System of Lung Cancer Patients Derived Tumors With Negative PD-L1 Expression”
Soner Altiok, Melanie Mediavilla Valera, Jenny Kreahling, Nikoletta L. Kallinteris, David Noyes, Tiffany N. Razabdouski, Joseph Shan, Jeff Hutchins, Kerstin Menander, Scott J. Antonia (Moffitt CC)
ABSTRACT…
BACKGROUND: Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Bavituximab blocks PS-mediated immune suppression and activates cytotoxic T lymphocyte anti-tumor responses.
METHODS: Tissues from consented patients with adenocarcinoma of the lung were extracted at the time of surgical resection and disaggregated to characterize expression of immune checkpoint proteins such as PD-1, CTLA-4, LAG3, TIM3, BTLA and adenosine A2A receptor on both CD4+ and CD8+ tumor infiltrating cells by flow cytometry (FACS) and stained for PD-L1, CD68, and CD163 via immunohistochemistry (IHC). 3D tumor microspheres were prepared and treated ex vivo with an IgG control, F(ab)’2 version of bavituximab, bavituximab, docetaxel, anti-PD-1 or PD-L1 and combinations of bavituximab, anti-PD-1 or PD-L1 and docetaxel for 36 hours within an intact tumor microenvironment. A multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines. Additionally, a NanoString platform containing probes to quantitate 770 immune function genes was used to determine potential positive or negative associations between expression of immune function genes and TIL activation by treatment. In a few cases, the expanded TILs were tested in PDX models using the consented patient’s tumor.
RESULTS: Bavituximab induces activation of TILs in 3D ex vivo tumor microsphere models of lung cancer, as demonstrated by a significant increase in IFN-y, TNF-a, and GM-CSF secretion. FACS, IHC, and NanoString gene function analysis read out assays revealed that this effect was associated with low PD-1 expression on CD8+ cells, negative PD-L1 expression in the stating biopsy tissue, and a conversion of the M2 to M1 macrophage phenotype.
CONCLUSION: These data support the use of bavituximab as an immunomodulatory treatment in adenocarcinoma of the lung by enhancing the activation of CD8+ TIL derived from patients' tumors with negative PD-L1 expression; correlating with increased cytokine production by lymphoid cells and repolarization of myeloid cells from an immunosuppressive to an immune active state.
SLIDESHOW (9 pgs.): http://www.peregrineinc.com/images/stories/pdfs/iaslc_2015_altiok.pdf

From Peregrine’s 9-8-15 PR:
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=930479
”For the study presented at the WCLC, researchers from Nilogen Oncosystems [<=Moffitt Startup: http://otmc.moffitt.org/startups.aspx ] and the H. Lee Moffitt Cancer Center, in collaboration with Peregrine, evaluated immune changes in 3D tumor microspheres generated from human NSCLC fresh tumors extracted at the time of surgery. These were tumor microspheres were treated with bavituximab alone, docetaxel alone, a combination of bavituximab and docetaxel, and a control. Bavituximab, alone and in combination with docetaxel, induced activation of tumor infiltrating lymphocytes as demonstrated by a statistically significant increase in key immune-stimulating cytokines such as IFN-y, TNF-a, and GM-CSF with a corresponding decrease in secretion of the immunosuppressive cytokine IL-10. Importantly, the bavituximab-affected immune response activity appeared to correlate with low PD-L1 expression on the tumor samples. "These new translational data suggest that bavituximab has the potential to activate a tumor specific immune response in patients with PD-L1 negative tumors that generally do not respond as well to immune checkpoint inhibitors. By doing so, it is believed that bavituximab may hold potential to increase the number of patients able to respond to PD-1 & PD-L1 targeting immunotherapies," said Jeff Hutchins, VP, Preclinical Dev. of Peregrine. "These findings continue to expand our collection of translational data supporting the mechanism of action for bavituximab and corroborate our wide range of previously published preclinical study results. Taken together, these data provide us with growing confidence for our bavituximab development programs, including the ongoing Phase III SUNRISE clinical trial in NSCLC which is evaluating the same treatment combination used in these studies."
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: ORAL #41 - Immune Biology, Microenvironment & Novel Targets (ID 159)
Moderators: Sukhmani K. Padda, Raphael Nemenoff
9/9/15, 6:30-8:00pm, Four Seasons Ballroom F1+F2
ORAL #41.05: “Targeting Phosphatidylserine-Mediated Immune Suppression Enhances the Efficacy of Immune Checkpoint Blockade in Pre-Clinical Tumor Models”
Rolf A. Brekken (UTSW), Bruce Freimark, Jian Gong, Carrie Baldwin, Van Nguyen, Michael Gray, Shen Yin, Jeff Hutchins, Alan Schroit, Xianming Huang (UTSW)
ABSTRACT…
BACKGROUND: Despite substantial progress, only a subset of cancer patients benefit from blockade of the PD-1 immune checkpoint. Multifocal immune suppression in the tumor microenvironment is the underlying cause for the limited efficacy of immune checkpoint blockade. Persistent immune suppression prevents the development of a robust T cell response to tumor specific antigens that is required for effective downstream immune checkpoint blockade. An underappreciated but significant contributor to immune suppression in tumors is the expression of the membrane phospholipid phosphatidylserine (PS) on the surface of tumor cells and tumor-derived microvesicles. PS is recognized by receptors on immune cells where it triggers the secretion of immune suppressive cytokines, prevents the differentiation of myeloid-derived suppressor cells (MDSCs) and inhibits dendritic cell (DC) maturation; events that prevent a productive anti-tumor T cell response. Bavituximab, a chimeric monoclonal antibody that targets PS and inhibits PS-mediated immunosuppressive signaling, drives immune activation by reducing the levels of MDSCs, by polarizing tumor-associated macrophages towards an M1 phenotype and by promoting the maturation of dendritic cells (DCs).
METHODS: The efficacy of bavituximab, anti-PD-1 and combination therapy was evaluated in multiple syngeneic, pre-clinical tumor models. Treatment efficacy was determined by inhibition of tumor growth and by immunophenotyping of spleen and tumor infiltrating leukocytes.
RESULTS: The combination of antibody-mediated PS and PD-1 blockade was significantly more effective in reducing tumor burden and promoting immune activation than single agent therapy. Combination therapy increased tumor infiltration of effector T-cells (Teff), increased the Teff:T regulatory cell ratio in the tumor and enhanced Teff function as determined by IFN-y, TNFa and granzyme B levels associated with Teff cells in the spleen and tumor. Furthermore combined blockade of PS and PD-1 signaling reduced the level of immune suppressive cells (e.g., MDSCs, M2 macrophages, and Treg) in the tumor microenvironment.
CONCLUSION: These results raise the possibility that PS blockade with bavituximab can enhance the efficacy of anti-PD-1 therapy even in patients with tumors that are unresponsive to single agent immune checkpoint therapy.
= = = = =
• RECALL: PPHM KOL/SAB Dr. David Carbone becomes President of IASLC 9-9-15 ( https://www.iaslc.org/about-us/board ). “…I also personally find it rewarding to participate in the global fight against cancer, and am honored to have been elected President of the IASLC, eff. Sept. 9, 2015." http://www.supportohiostate.org/site/TR?px=1009121&fr_id=1151&pg=personal
• IASLC Faculty & Staff Disclosures: Heather A. Wakelee, MD - Grant/Research Support: Genentech/Roche, Lilly, Pfizer, Novartis, Exelixis, Xcovery, AstraZeneca, Clovis, Regeneron; Consultant: Peregrine. http://www.gotoper.com/conferences/ilc/meetings/15th-international-lung-cancer-congress “Associate Prof. of Medicine, Div. of Oncology; Faculty Director, Cancer Clinical Trials Office; Co-leader, Lung Cancer Disease Mgt Group, Stanford Univ. School of Medicine, Stanford CA”
= = = = = = =
BAVI MOA 8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow http://tinyurl.com/qz64pzg
…“Expansion & Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
7-2015 Visiongain Report/CMO’s interviews 2 CMO’s: Avid & Fujifilm/Diosynt. “Our report predicts the world market for producing those therapeutic agents will reach $5.78B in 2019, expanding fast from 2015-2025.”
LONDON, 9-3-15: “Outsourced Biopharma Production – New Analysis Showing You Revenue Forecasts”
http://www.prnewswire.com/news-releases/biological-drug-api-manufacturing-services-world-industry-and-market-predictions-2015-2025-contract-manufacturing-organisations-cmos-serving-biopharma-300138058.html
What is the outlook for making biological active pharma ingredients (APIs) under contract? Visiongain's new report [pub. 7-2015] predicts those revenues from 2015. There you see financial data, also assessing trends, technologies, clients' needs and opportunities. Our study gives you sales forecasts to 2025 at overall world market, submarket and national level. Discover what's happening for those production services – outsourcing and off-shoring for biological drugs (biologics). That way you gain in knowledge, also helping your influence. . . Besides revenue forecasting to 2025, our new work shows you recent results, growth rates and market shares. There you find original analysis, exploring trends and developments. You gain 57 tables, 49 charts and 2 interviews with biopharmaceutical companies. . .
• First our report shows you individual revenue forecasting for 3 main submarkets to 2025, at world level, with discussions:
- Mammalian cell cultures
- Microbial fermentations (culturing)
- Other biotechnological (expression) systems for yielding medical products.
• For API revenues, our study breaks its overall world revenue forecast to 2025 into these 3 therapeutic classes:
- Monoclonal antibodies (mAbs)
- Insulin
- Vaccines
- Interferons
- Human growth hormones.
. . .
Our study explains forces affecting that industry and market from 2015, including these influences:
- Services in greatest demand for biopharma CMOs
- Development and proliferation of cell lines and expression systems, including emerging technologies
- Therapies in development (R&D) providing outsourcing opportunity for API makers
- Challenges in making vaccines and other complex protein-based and recombinant medicines
- Improving yields and efficiency in biopharmaceutical operations, including effects on downstream processing.
You also explore these aspects of the biologicals API market and industry:
- Next-generation antibody development, including antibody-drug conjugates (ADCs)
- Orphan drugs and personalized medicines – satisfying specialist production needs
- Single-use bioreactors (disposable systems) for API manufacturers
- Risks of overcapacity for biotechnological CMOs
- Regulations (GMP and other standards) affecting that biopharmaceutical contracting.
. . .
Leading companies and 2019 market value – what success is possible?
Our report predicts the world market for producing those therapeutic agents will reach $5.78bn in 2019, expanding fast from 2015-2025.
That analysis also shows you what organizations hold most potential. Companies our report profiles include these:
- Boehringer Ingelheim BioXcellence
- Celltrion
- DSM Biologics
- Lonza
- Samsung BioLogics
- FUJIFILM Diosynth Biotechnologies.
You also discover interviews with 2 companies in the industry – FUJIFILM Diosynth Biotechnologies and Avid Bioservices Inc. Hear what service providers do and think, helping you stay ahead and succeed. . .
ABOUT REPORTBUYER
Reportbuyer is a leading industry intelligence solution that provides all market research reports from top publishers. http://www.reportbuyer.com
VISIONGAIN:
7-2015: “Biological Drug API Manufacturing Services: World Industry and Market Predictions 2015-2025”
https://www.visiongain.com/blog/index.php/biological-drug-api-manufacturing-services-world-industry-and-market-predictions-2015-2025/
= = = = = = = = = = = = = = = = = = = = = = = = = = = =
• 6-17-15: Avid’s John Haney (ex-Genentech/Pfizer) speaking at BIO-INTL’5/Philly http://tinyurl.com/pnlquu3 & http://tinyurl.com/nl4vbgk
...”Designing & Implementing Avid’s New State-of-the-Art Single-Use Facility for Late Ph.3 & Commercial Prod.” - SK: "We've seen tremendous interest for production in the new facility, both from new & existing clients"
• 12-10-14: Avid to Double Mfg. Capacity (“expanding client roster; potential commercial launch of bavituximab”) http://tinyurl.com/mmc3qgy & http://tinyurl.com/kmdgq8t
• 3-24-15: Avid Receives CMO Leadership Awards for Its Commitment to Innovation & Reliability http://tinyurl.com/psep47f
• 3-12-13: Avid Q3'FY13 GP=$3.3mm; S.King 3-2012, "We have a profitable CMO, Avid Bioservices" http://tinyurl.com/l97rzm8
PPHM REVS-BY-QTR TABLE thru FY15/Q4 (fye 4-30-15), per the 4-30-15 10-K (http://tinyurl.com/ocrtkuj ) issued 7-14-15. Deferred-Revs at 4-30-15, going fwd into FY’16/Q1 (q/e 7-31-15), total $6.6mm, up from the $5.8mm of Deferred-Revs at 1-31-15 that drove into FY’15/Q4.
Total Revs since May’06: ($129.2mm/Avid + $24.1mm/Govt + $2.1mm/Lic.) = $155.5mm
Avid’s Gross-Profit over last 3 qtrs: $9.4mm on revs of $21.2mm (GM% = 44%)
=> Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GM%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 5752 0 6148
Totals: 129212 24149 2124 155485 <=since5/1/2006
.
TOTAL REV’s BY YEAR (Avid+Gov’t+Lic):
FY04 4-30-04 3,314 …Avid(CMO)= 3,039 (Avid-Revs don’t incl. Govt-SVCS)
FY05 4-30-05 4,959 …Avid(CMO)= 4,684
FY06 4-30-06 3,193 …Avid(CMO)= 3,005
FY07 4-30-07 3,708 …Avid(CMO)= 3,492
FY08 4-30-08 6,093 …Avid(CMO)= 5,897
FY09 4-30-09 18,151 …Avid(CMO)= 12,963
FY10 4-30-10 27,943 …Avid(CMO)= 13,204
FY11 4-30-11 13,492 …Avid(CMO)= 8,502
FY12 4-30-12 15,233 …Avid(CMO)= 14,783
FY13 4-30-13 21,683 …Avid(CMO)= 21,333
FY14 4-30-14 22,401 …Avid(CMO)= 22,294
FY15 4-30-15 26,781 …Avid(CMO)= 26,744
...Total Gov’t Revs from 7-2008 inception thru FY11Q1(Apr’11): $24.15mm
Peregrine Exhibiting & 3-Presentations Sept6-9 at IASLC World Conf. in Denver – recall, PPHM KOL/SAB’r Dr. David Carbone becomes President of The IASLC on 9-9-15. 3 Bavi presentations (1 poster, 1 Mini-Oral, 1 Oral) by UTSW (David Gerber, Rolf Brekken, Xianming Huang) and Moffitt CC (Scott Antonia), all with Peregrine co-authors…
Sept6-9 2015: “IASLC’s 16th World Conf. on Lung Cancer”, Denver
http://wclc2015.iaslc.org/
“The World Conference on Lung Cancer (WCLC) is the world’s largest meeting dedicated to lung cancer and other thoracic malignancies. More than 7,000 delegates come from more than 100 countries to discuss the latest developments in thoracic malignancy research. Attendees include surgeons, medical oncologists, radiation oncologists, pulmonologists, radiologists, pathologists, epidemiologists, basic research scientists, nurses, allied health professionals, advocates, and patients.”
http://www.iaslc.org/events/16th-world-conference-lung-cancer
2015 Exhibitor: Peregrine Pharm. (1 of 39) - booth #1724 (adjacent to Abbvie)
• RECALL: PPHM KOL/SAB Dr. David Carbone becomes President of IASLC 9-9-15 ( https://www.iaslc.org/about-us/board ). “…I also personally find it rewarding to participate in the global fight against cancer, and am honored to have been elected President of the IASLC, eff. Sept. 9, 2015." http://www.supportohiostate.org/site/TR?px=1009121&fr_id=1151&pg=personal
• IASLC Faculty & Staff Disclosures: Heather A. Wakelee, MD - Grant/Research Support: Genentech/Roche, Lilly, Pfizer, Novartis, Exelixis, Xcovery, AstraZeneca, Clovis, Regeneron; Consultant: Peregrine. http://www.gotoper.com/conferences/ilc/meetings/15th-international-lung-cancer-congress “Associate Prof. of Medicine, Div. of Oncology; Faculty Director, Cancer Clinical Trials Office; Co-leader, Lung Cancer Disease Mgt Group, Stanford Univ. School of Medicine, Stanford CA”
PGM-Search: http://content.webges.com/library/wclc/browse/search
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: Treatment of Advanced Diseases - NSCLC
9/7/15 9:30am-5:00pm, Exhibit Hall (Hall B+C)
POSTER #P1.01-075: “Phase III, Randomized, Double-Blind Trial of Bavituximab Plus Docetaxel in Previously Treated Stage IIIb/IV Non-Squamous NSCLC (SUNRISE) (ID 1581)
David R. Spigel, Joseph Shan, Alberto Chiappori, Ulrich Keilholz, Martin Reck, Martin Edelman, Manuel Domine, Keunchil Park, Tae Won Jang, Wu-Chou Su, Rachel E. Sanborn, Leora Horn, Rebecca Heist, Paul Mainwaring, David E. Gerber (UTSW)
ABSTRACT…
BACKGROUND: Exposed phosphatidylserine (PS) in the tumor microenvironment is highly immunosuppressive. PS binding to PS receptors on myeloid derived suppressor cells (MDSC) and M2 macrophages leads to production of anti-inflammatory cytokines such as TGF-B and IL-10. Bavituximab, a first-in-class PS-targeting monoclonal antibody, counters these effects, resulting in production of pro-inflammatory cytokines such as TNF-a and IL-12, maturation of dendritic cells and induction of tumor specific cytotoxic T lymphocyte (CTL) immunity. Docetaxel has also been shown to suppress MDSCs while increasing tumor antigens and T-cell mediated cytotoxicity, thereby enhancing bavituximab’s immunomodulatory effects. In a prior double-blind Phase II trial in 2nd line non-squamous NSCLC, bavituximab 3 mg/kg + docetaxel was well-tolerated and demonstrated 60% improvement (11.7 vs 7.3 mos) in median overall survival (OS) compared to control.
METHODS: SUNRISE is a Phase III, double-blind trial where patients with previously treated Stage IIIb/IV non-squamous, non-small cell lung cancer are randomized in a 1:1 ratio to receive up to six 21-day cycles of docetaxel in combination with either weekly 3 mg/kg bavituximab or placebo, followed by maintenance with weekly bavituximab or placebo until progression or toxicity. Patients will be stratified by region (North America, Europe, or Rest of World), disease stage (IIIb or IV), and previous maintenance/targeted therapy (yes or no). This trial was initiated in Dec.2013 and accrual of 582 patients across 160+ sites in 14 countries is planned over 24 months. The primary endpoint is OS and 2 interim analyses are planned. Secondary endpoints include progression-free survival (PFS), overall response rate (ORR) and safety. Radiographic tumor response is centrally assessed every 2 cycles during combination therapy and every nine weeks during maintenance. Exploratory analysis will include the assessment of changes in circulating immune cells and cytokines to better understand the immunotherapeutic mechanism.
RESULTS: Trial in progress
CONCLUSION: Trial in progress
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: MINI ORAL #14 - Pre-Clinical Therapy (ID 119)
Moderators: Lynnette Fernandez-Cuesta Adi F. Gazdar
9/8/15, 10:45am-12:15pm, Mile High Ballroom 2c-3c
MINI-ORAL #14.07: “Bavituximab Activates CD8+ TILs in a 3D Ex Vivo System of Lung Cancer Patients Derived Tumors With Negative PD-L1 Expression”
Soner Altiok, Melanie Mediavilla Valera, Jenny Kreahling, Nikoletta L. Kallinteris, David Noyes, Tiffany N. Razabdouski, Joseph Shan, Jeff Hutchins, Kerstin Menander, Scott J. Antonia (Moffitt CC)
ABSTRACT…
BACKGROUND: Bavituximab is a chimeric monoclonal antibody that targets the membrane phospholipid phosphatidylserine (PS) exposed on endothelial cells and cancer cells in solid tumors. Bavituximab blocks PS-mediated immune suppression and activates cytotoxic T lymphocyte anti-tumor responses.
METHODS: Tissues from consented patients with adenocarcinoma of the lung were extracted at the time of surgical resection and disaggregated to characterize expression of immune checkpoint proteins such as PD-1, CTLA-4, LAG3, TIM3, BTLA and adenosine A2A receptor on both CD4+ and CD8+ tumor infiltrating cells by flow cytometry (FACS) and stained for PD-L1, CD68, and CD163 via immunohistochemistry (IHC). 3D tumor microspheres were prepared and treated ex vivo with an IgG control, F(ab)’2 version of bavituximab, bavituximab, docetaxel, anti-PD-1 or PD-L1 and combinations of bavituximab, anti-PD-1 or PD-L1 and docetaxel for 36 hours within an intact tumor microenvironment. A multiplex human cytokine assay was used to simultaneously analyze the differential secretion of cytokines. Additionally, a NanoString platform containing probes to quantitate 770 immune function genes was used to determine potential positive or negative associations between expression of immune function genes and TIL activation by treatment. In a few cases, the expanded TILs were tested in PDX models using the consented patient’s tumor.
RESULTS: Bavituximab induces activation of TILs in 3D ex vivo tumor microsphere models of lung cancer, as demonstrated by a significant increase in IFN-y, TNF-a, and GM-CSF secretion. FACS, IHC, and NanoString gene function analysis read out assays revealed that this effect was associated with low PD-1 expression on CD8+ cells, negative PD-L1 expression in the stating biopsy tissue, and a conversion of the M2 to M1 macrophage phenotype.
CONCLUSION: These data support the use of bavituximab as an immunomodulatory treatment in adenocarcinoma of the lung by enhancing the activation of CD8+ TIL derived from patients' tumors with negative PD-L1 expression; correlating with increased cytokine production by lymphoid cells and repolarization of myeloid cells from an immunosuppressive to an immune active state.
- - - - - - - - - - - - - - - - - - - - - - -
WCLC 2015 Track: ORAL #41 - Immune Biology, Microenvironment & Novel Targets (ID 159)
Moderators: Sukhmani K. Padda, Raphael Nemenoff
9/9/15, 6:30-8:00pm, Four Seasons Ballroom F1+F2
ORAL #41.05: “Targeting Phosphatidylserine-Mediated Immune Suppression Enhances the Efficacy of Immune Checkpoint Blockade in Pre-Clinical Tumor Models”
Rolf A. Brekken (UTSW), Bruce Freimark, Jian Gong, Carrie Baldwin, Van Nguyen, Michael Gray, Shen Yin, Jeff Hutchins, Alan Schroit, Xianming Huang (UTSW)
ABSTRACT…
BACKGROUND: Despite substantial progress, only a subset of cancer patients benefit from blockade of the PD-1 immune checkpoint. Multifocal immune suppression in the tumor microenvironment is the underlying cause for the limited efficacy of immune checkpoint blockade. Persistent immune suppression prevents the development of a robust T cell response to tumor specific antigens that is required for effective downstream immune checkpoint blockade. An underappreciated but significant contributor to immune suppression in tumors is the expression of the membrane phospholipid phosphatidylserine (PS) on the surface of tumor cells and tumor-derived microvesicles. PS is recognized by receptors on immune cells where it triggers the secretion of immune suppressive cytokines, prevents the differentiation of myeloid-derived suppressor cells (MDSCs) and inhibits dendritic cell (DC) maturation; events that prevent a productive anti-tumor T cell response. Bavituximab, a chimeric monoclonal antibody that targets PS and inhibits PS-mediated immunosuppressive signaling, drives immune activation by reducing the levels of MDSCs, by polarizing tumor-associated macrophages towards an M1 phenotype and by promoting the maturation of dendritic cells (DCs).
METHODS: The efficacy of bavituximab, anti-PD-1 and combination therapy was evaluated in multiple syngeneic, pre-clinical tumor models. Treatment efficacy was determined by inhibition of tumor growth and by immunophenotyping of spleen and tumor infiltrating leukocytes.
RESULTS: The combination of antibody-mediated PS and PD-1 blockade was significantly more effective in reducing tumor burden and promoting immune activation than single agent therapy. Combination therapy increased tumor infiltration of effector T-cells (Teff), increased the Teff:T regulatory cell ratio in the tumor and enhanced Teff function as determined by IFN-y, TNFa and granzyme B levels associated with Teff cells in the spleen and tumor. Furthermore combined blockade of PS and PD-1 signaling reduced the level of immune suppressive cells (e.g., MDSCs, M2 macrophages, and Treg) in the tumor microenvironment.
CONCLUSION: These results raise the possibility that PS blockade with bavituximab can enhance the efficacy of anti-PD-1 therapy even in patients with tumors that are unresponsive to single agent immune checkpoint therapy.
CEO D.Godshall presenting 9-9-15/8:50ET at Wells-Fargo
HeartWare Presentation At The 2015 Wells Fargo Healthcare Conference To Be Webcast
FRAMINGHAM, Sept. 3, 2015: Heartware Intl. Inc. (NASDAQ:HTWR), a leading innovator of less invasive, miniaturized circulatory support technologies that are revolutionizing the treatment of advanced heart failure, today announced that its President & CEO, Doug Godshall, is scheduled to present at the 2015 Wells Fargo Healthcare Conference at 8:50amET on Wednesday, Sept. 9, 2015. The conference is being held Sept. 9-10, 2015 at The Hyatt Regency, Boston.
A live webcast of the company's presentation at the conference will be available via a link provided at http://www.heartware.com . . .
SUNRISE at UTSW, “with lung cancer medical oncologist Dr. David Gerber leading the charge.” Goggle says updated 2 days ago, but my guess is the Bavi/Gerber ref. (the only example given) was already there…
UT SOUTHWESTERN MEDICAL CENTER – PATIENT CARE
http://www.utswmedicine.org/cancer/programs/lung/
LUNG CANCER
Lung cancer, the leading cause of cancer deaths in men & women in the United States, often grows silently without causing symptoms. This can be scary for those at high risk for the disease, such as smokers, but there’s reason for hope. Treatments are improving, and some lung cancer patients are living longer with the development of new drugs, early detection methods, and innovative techniques in surgery and radiation – all happening right here at UTSW Harold C. Simmons Comprehensive Cancer Center. UTSW-MC – the only National Cancer Institute (NCI)-designated cancer center in North Texas – is the place to go in North Texas for the diagnosis and treatment of lung cancer.
PIONEERING DRUG TREATMENTS
UT Southwestern is a leader in new drug development for lung cancer, and through clinical trials our patients can gain access to treatments not available elsewhere.
For example, bavituximab, an immunotherapeutic antibody developed by UT Southwestern researchers, is now being evaluated in a Phase III clinical trial [ http://www.utsouthwestern.edu/research/fact/detail.html?studyid=STU%20102013-077 ] for the treatment of NSCLC, with lung cancer medical oncologist David Gerber, M.D., leading the charge. [ http://profiles.utsouthwestern.edu/profile/53487/david-gerber.html ]
In addition, our patients get special access to nationwide group trials for lung cancer because of an NCI grant given to only 30 research institutions in the U.S. …
8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow
…See Images of Dr. Hutchins’ 29-screen Presentation Slideshow below…
8-26-15: Peregrine Pharmaceuticals Presents Data at Annual Immunotherapy and Vaccine Summit (ImVacS) Supporting Ability of Bavituximab to Mediate Anti-Tumor T Cell Responses Across Multiple Tumor Types
• Increasing Activated T Cells in Tumors Demonstrates Potential Complement to anti-PD-1 and anti-PD-L1 Checkpoint Inhibitors
• Clinical and Preclinical Studies Demonstrate Estimated Survival Curves that Plateau
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=928906
TUSTIN, Aug. 26, 2015: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM, PPHMP), a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer, today announced the presentation of a range of clinical, translational and pre-clinical study results highlighting the ability of bavituximab, Peregrine's investigational phosphatidylserine (PS)-signaling pathway inhibitor, to promote anti-tumor T cell mediated activity in several tumor types. The data were presented today by Jeff T. Hutchins, Ph.D., VP, Preclinical Research at Peregrine Pharmaceuticals and chairperson of the Combination Immunotherapy Strategies session at the 10th Annual Immunotherapy and Vaccine Summit (ImVacS), being held August 24-28, 2015 in Boston, Massachusetts.
Bavituximab is an investigational immunotherapy designed to assist the body's immune system by targeting and modulating the activity of phosphatidylserine (PS), a highly immune-suppressive signaling molecule expressed broadly on the surface of cells in the tumor microenvironment. Peregrine's PS signaling pathway inhibitor candidates, including bavituximab, reverse the immunosuppressive environment that many tumors establish in order to proliferate and fight cancer by activating macrophages and cytotoxic T cells in tumors. Preclinical data demonstrate that combining the enhanced T cell anti-tumor activity of bavituximab-like antibodies with checkpoint inhibitors, such as anti-PD-1 antibodies, results in significantly improved tumor control in multiple models of cancer.
Dr. Hutchins' presentation highlighted key findings from several recent bavituximab-focused studies including:
• The potential of bavituximab to shift the tumor microenvironment from immuno-suppressive in which tumors evade immune detection to a state of immune activation in which the immune system recognizes and fights the tumor. Presented findings demonstrate that bavituximab-like antibodies significantly increase the prevalence of tumor infiltrating CD8+ T-cells and immune-activating cytokines, while decreasing macrophages and myeloid cells that allow the tumor to evade immune detection. This elucidation and confirmation of bavituximab's mechanism of action highlights the potential of bavituximab to enhance the anti-tumor effects of both chemotherapy and immune checkpoint inhibitors.
• Bavituximab increases the number of activated CD8+ cells in the tumor, which stimulates PD-1 expression, thereby upregulating the target for checkpoint inhibitors such as anti-PD-1 and anti-PD-L1. Importantly, translational study data across multiple cancers indicated that tumors with low PD-L1 or PD-1 expression on tumor infiltrating T cells showed promising signs of immune activation after treatment with bavituximab. This suggests the potential for bavituximab to activate a tumor specific immune response in patients with PD-L1 negative tumors that generally do not respond as well to PD-1 and PD-L1 inhibitors. By doing so, it is believed that bavituximab may hold potential to increase the number of patients able to respond to PD-1 and PD-L1 targeting immunotherapies. Furthermore, the combination of bavituximab-like antibodies and anti-PD-1 antibodies resulted in enhanced, synergistic anti-tumor activity in animal models of multiple tumor types, as compared to either agent alone. In some cases, complete tumor regressions were achieved, highlighting the anti-tumor potential of bavituximab in combination with checkpoint inhibitors such as anti-PD-1 antibodies.
• Results from several clinical and preclinical studies in a range of tumor types show that bavituximab and bavituximab-like antibodies, in combination with conventional therapy, have consistently demonstrated estimated survival curves that plateau.
"We continue to generate a broad collection of pre-clinical, translational and clinical data highlighting bavituximab's novel mechanism of action and synergistic activity for a range of combination treatments. These study results, particularly as they relate to the potential synergies between bavituximab and checkpoint inhibitors, create great excitement for us as we begin work with our new collaborators at Memorial Sloan Kettering Cancer Center and AstraZeneca, while continuing our long-standing relationship with the University of Texas Southwestern Medical Center where this technology was originally developed," said Dr. Hutchins. "By aligning with these world leaders in cancer immunotherapy to study novel immuno-oncology combination therapies, we are best positioning ourselves to maximize the potential role that bavituximab can play in this new era of innovative cancer treatments."
ABOUT BAVITUXIMAB: A TARGETED INVESTIGATIONAL IMMUNOTHERAPY
Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab, the lead compound in Peregrine's immuno-oncology development program, blocks PS to alter this immunosuppressive signal and sends an immune activating signal. Targeting PS with bavituximab has been shown to shift the functions of immune cells in tumors, resulting in anti-tumor immune responses.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials focused on the treatment of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
Contacts:
• Jay Carlson, Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
• Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
• Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = =8-26-15 Dr. Hutchins Presentation PDF posted to http://www.peregrineinc.com/images/stories/pdfs/imvacs_hutchins-2015.pdf
...Here are images of his 8-26-15 29-screen talk:


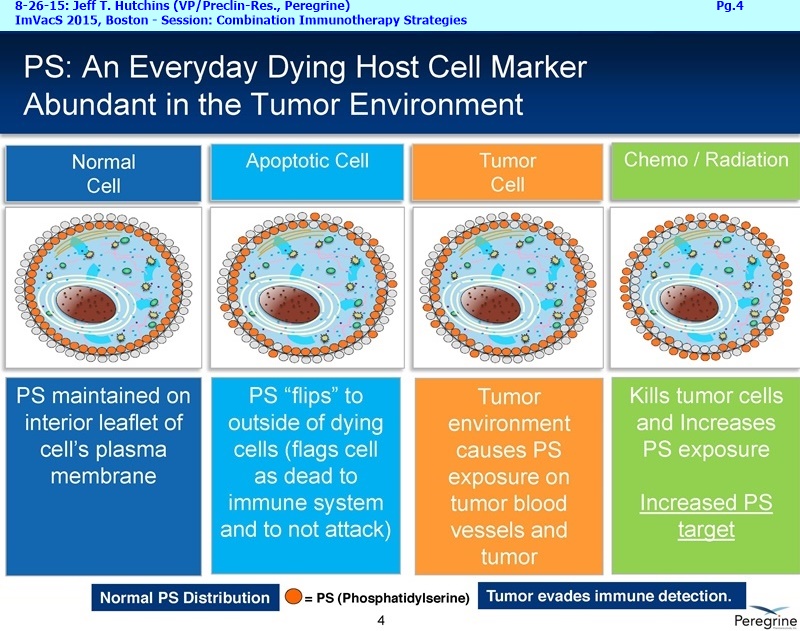
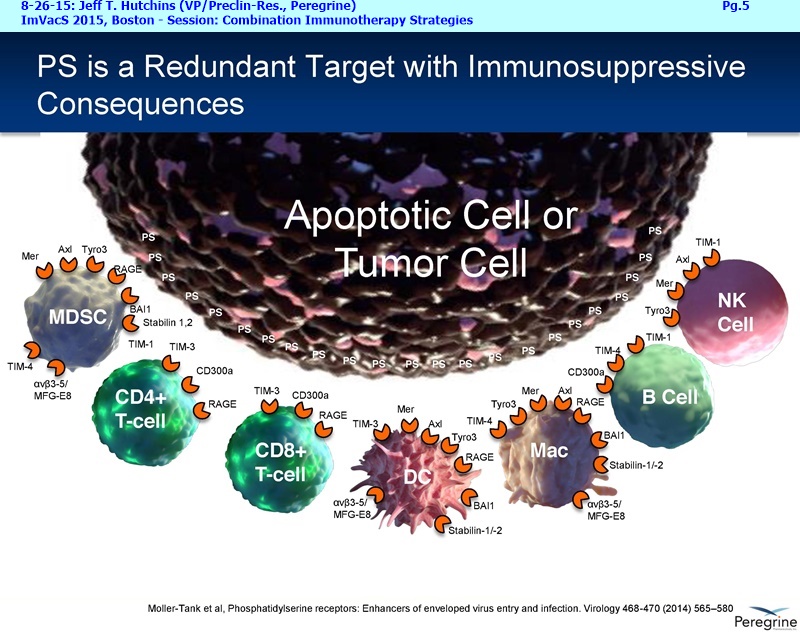
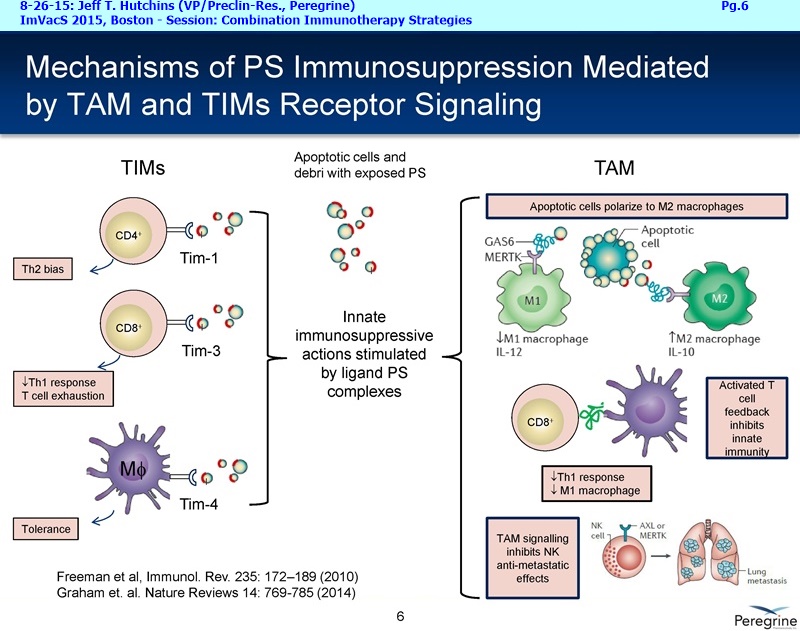

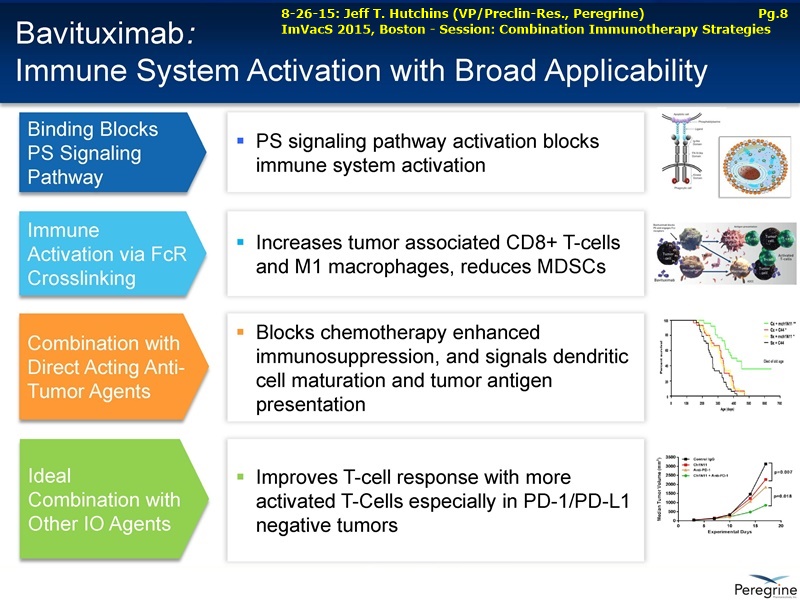
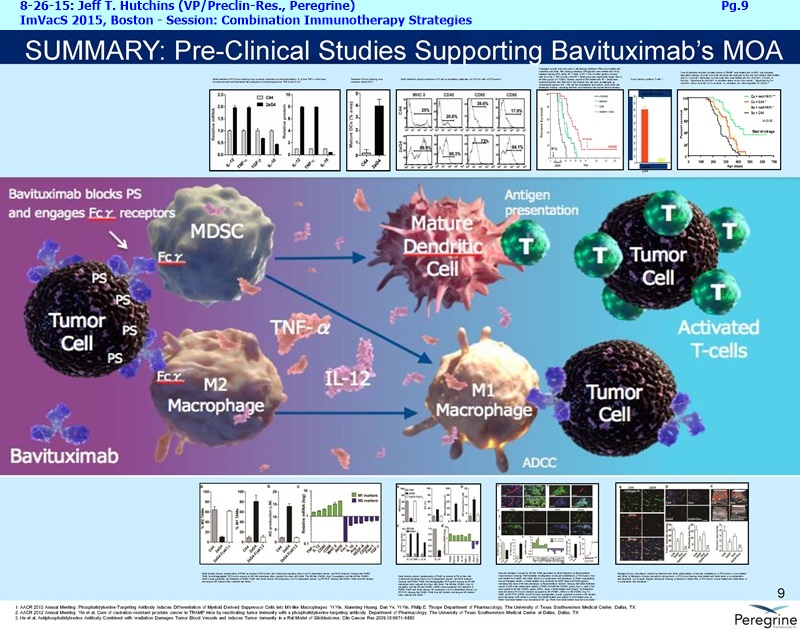
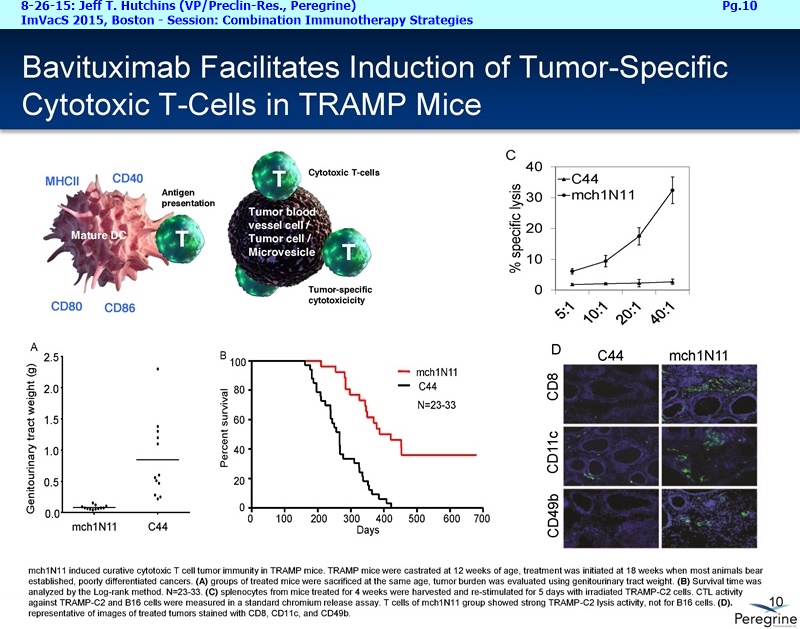
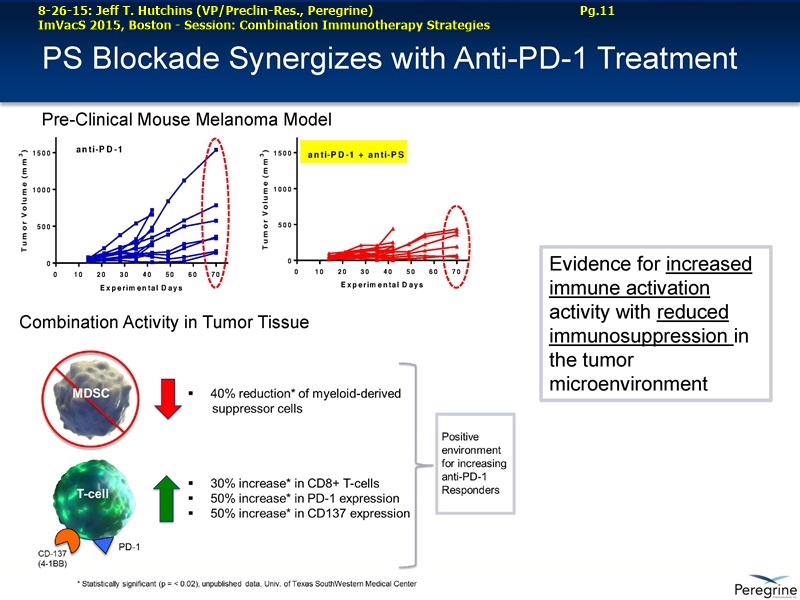
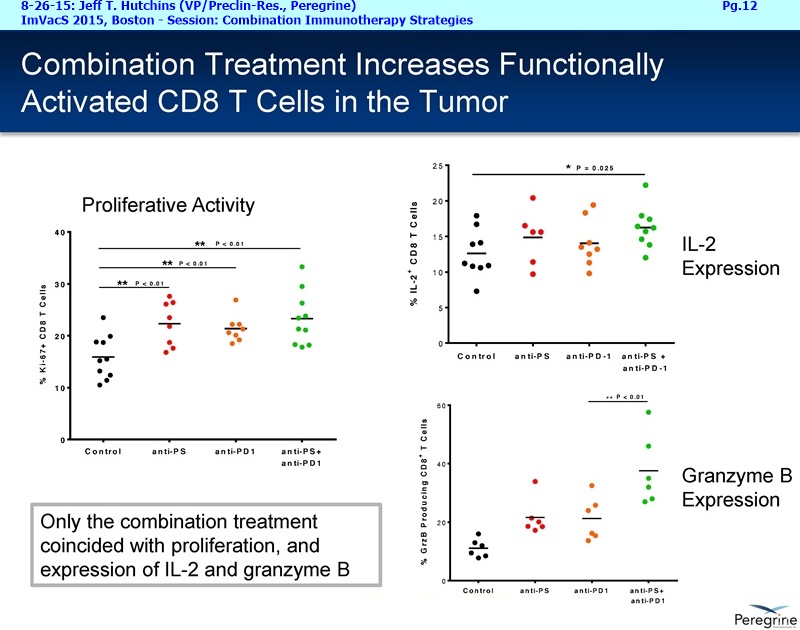
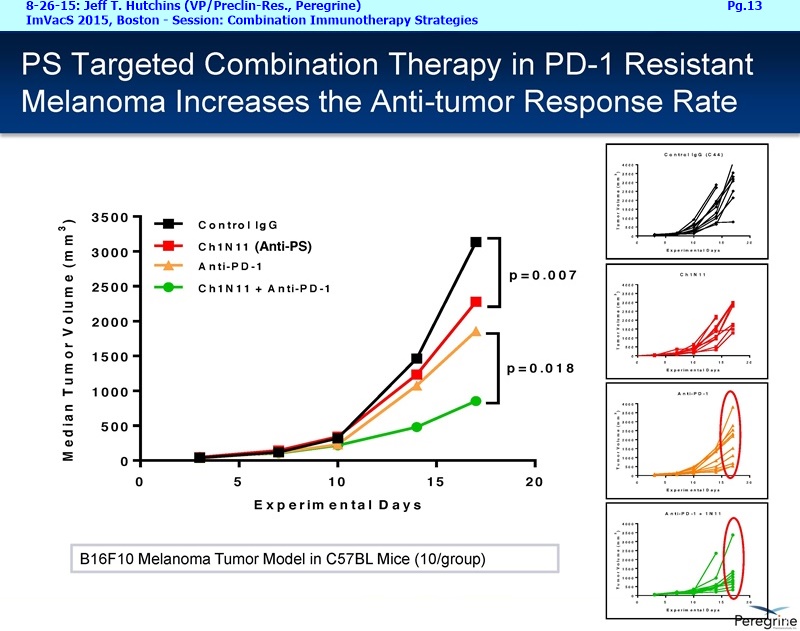
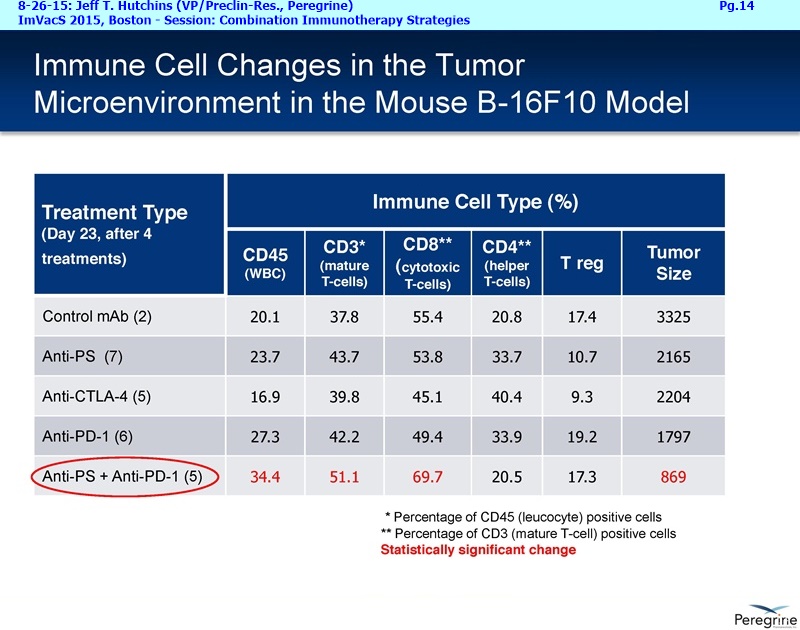
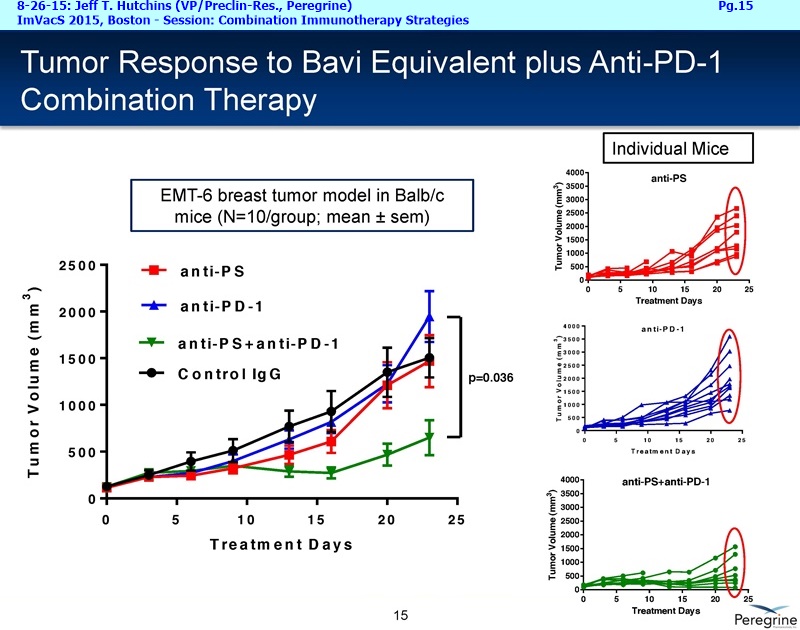

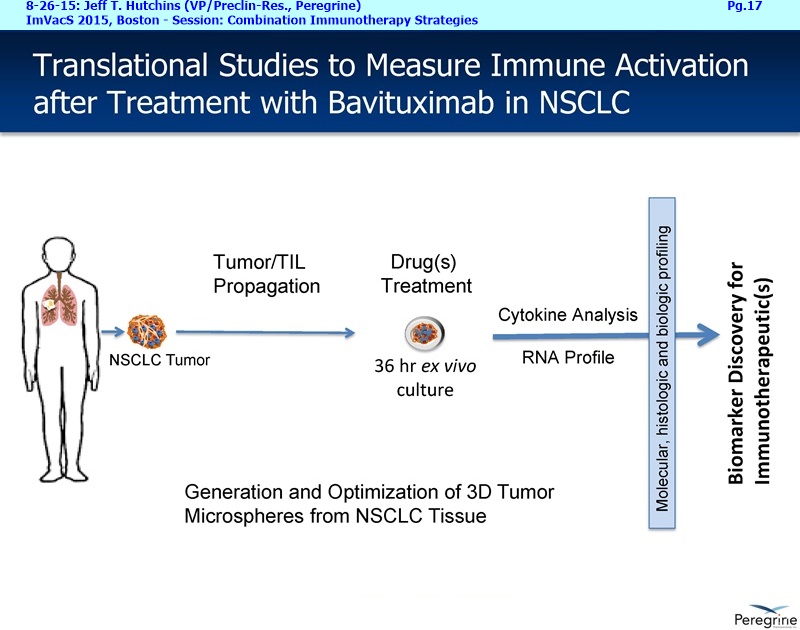
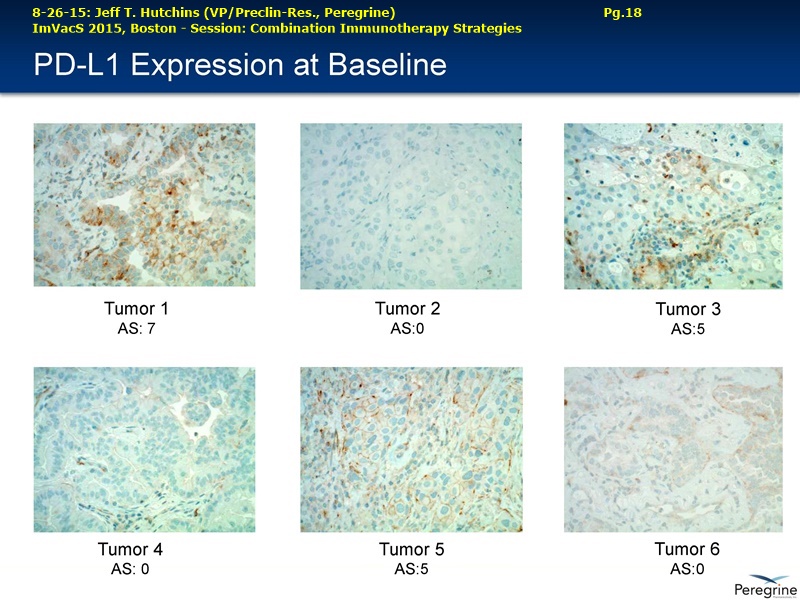
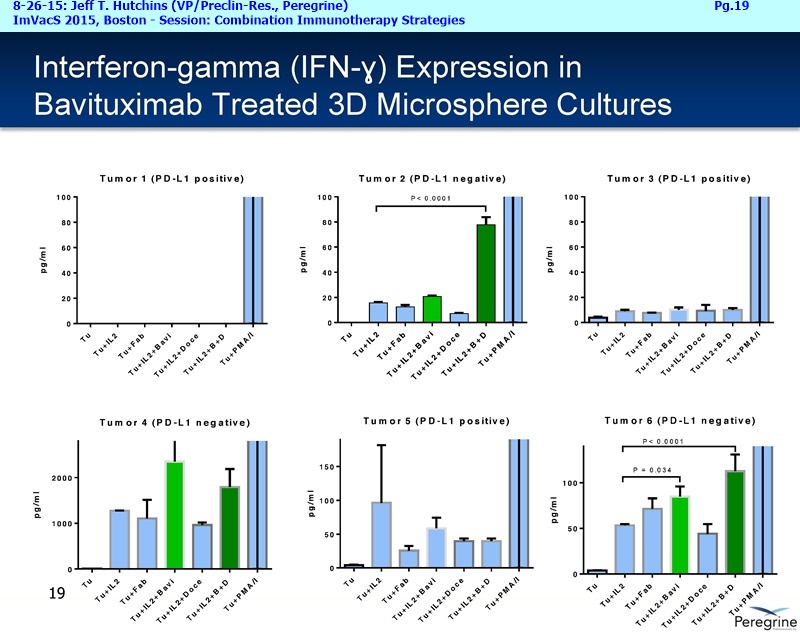
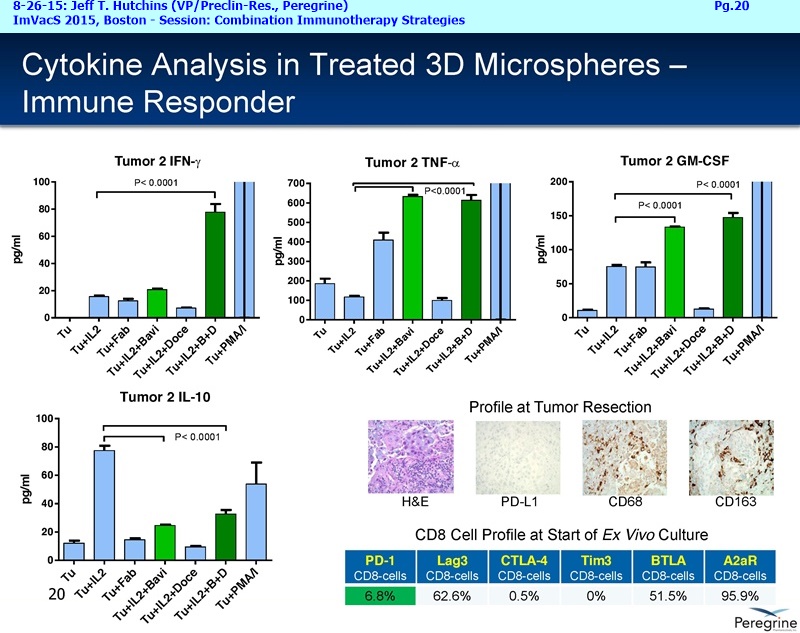
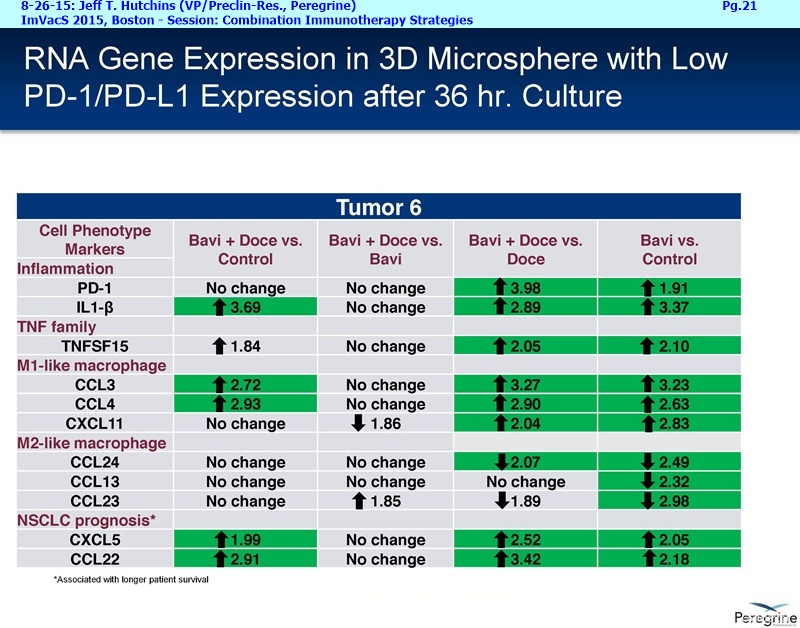
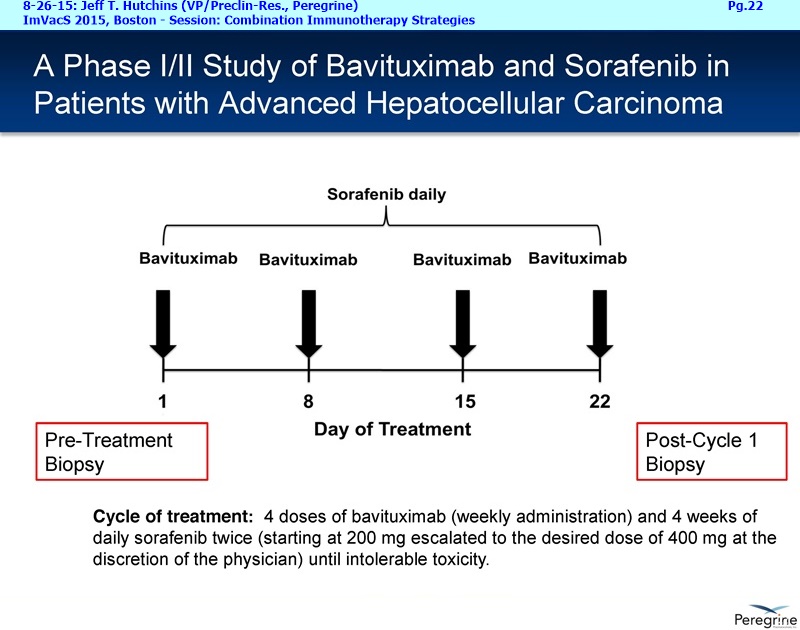
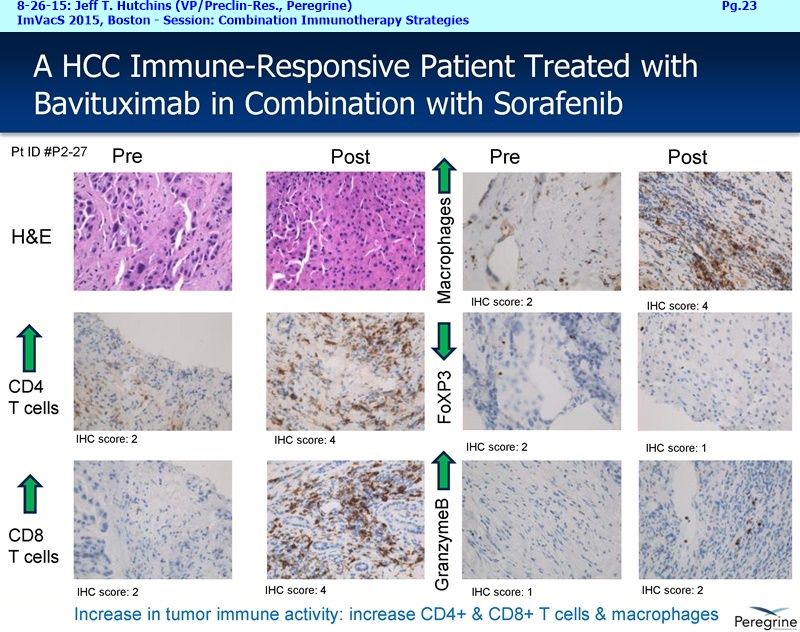
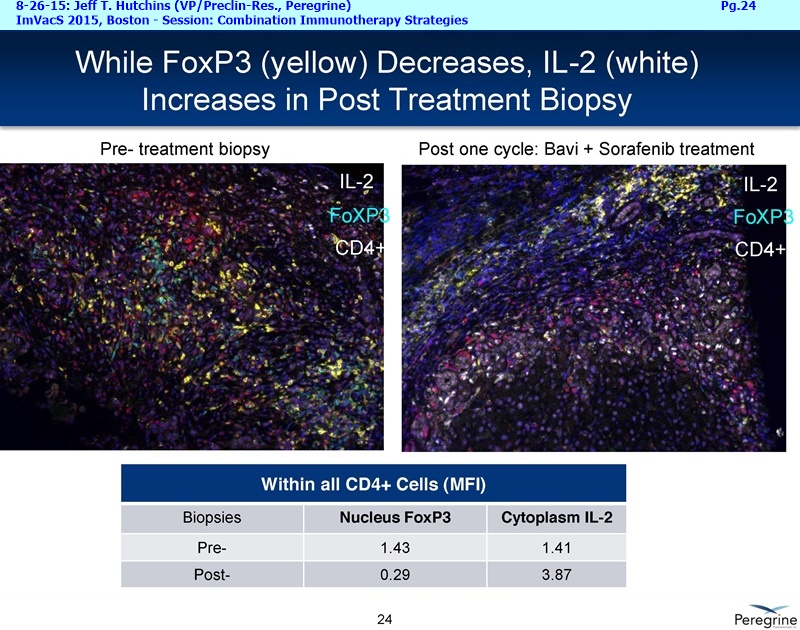
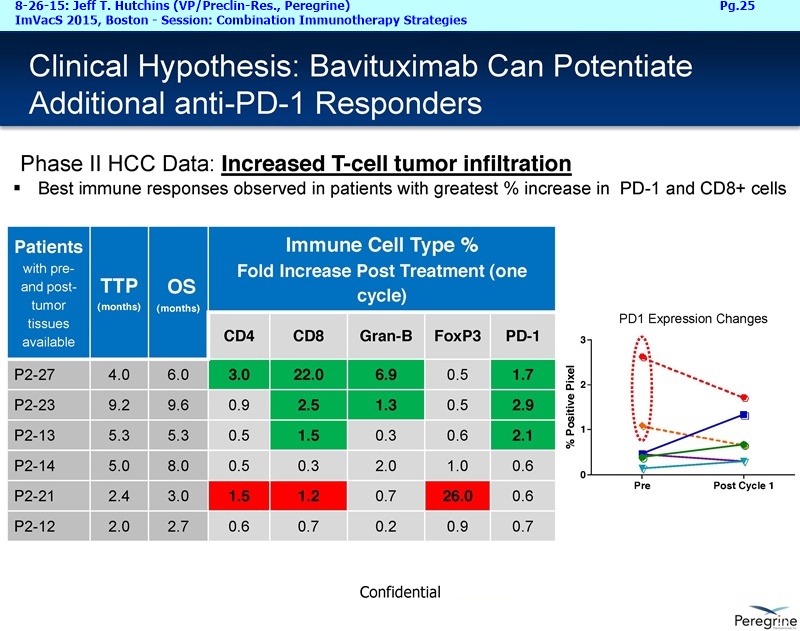
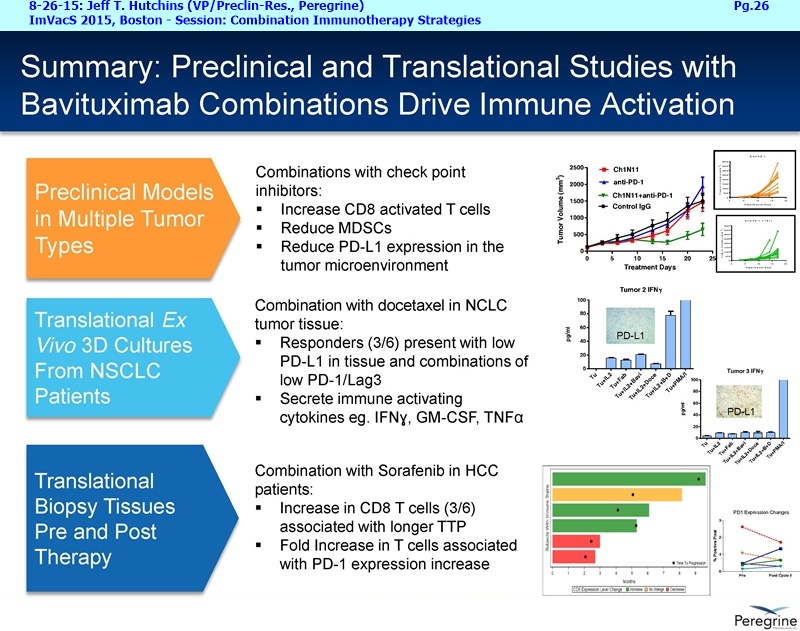

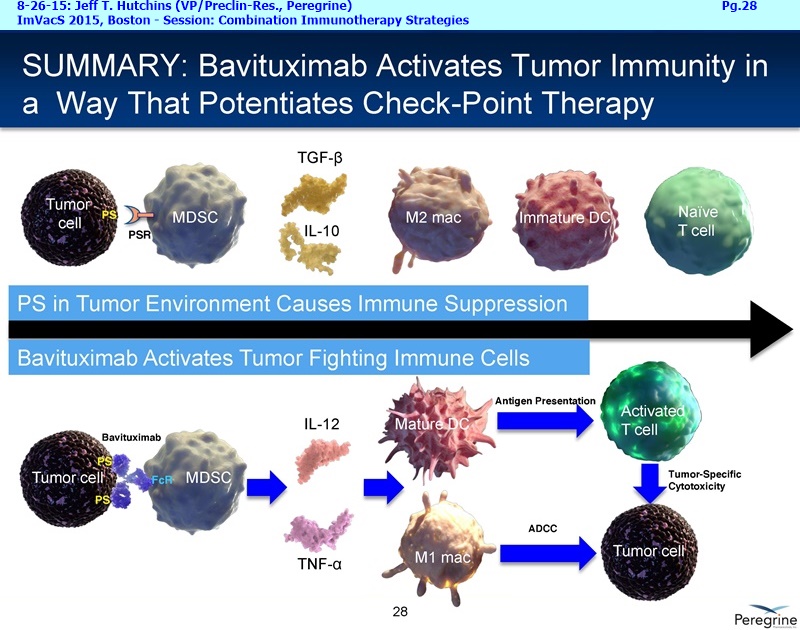

= = = = = = = = = = = = = = = = = = =
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
Durvalumab=MEDI4736, an anti-PD-L1 immune checkpoint inhibitor.
…AZN’s Head/I-O(Robert Iannone): “Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
Durvalumab (MEDI4736, anti-PD-L1) has a Net Present Value of $6.5B…

7-14-15 Qtly. Conf. Call (King/Shan/Hutchins/Lytle) Transcript http://tinyurl.com/nw2v5u6
...CEO S.King: “We recently entered into collaboration with investigators at Memorial Sloan Kettering Cancer Ctr to continue expanding on this important work, as well as to explore other potential applications of bavituximab and other agents that target PS-signaling pathway.”
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/qxu4w2x
= = = = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
BAVITUXIMAB MOA & CLINICAL DATA: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s

5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
= = = = = = = = = = = = = = = = =
8-26-15: Jeff T. Hutchins (VP/Preclin-Res., Peregrine) - ImVacS 2015, Boston (Session: Combination Immunotherapy Strategies)
Aug 24-28 2015: “CHI’s 10th Annual Immunotherapy & Vaccine Summit (ImVacS ’15)”, Boston
CHI = Cambridge Healthtech Institute, http://www.healthtech.com
“…This year's event has been expanded to 5 days with coverage on adjuvants, vaccine & immunotherapy technologies, immunomodulatory therapeutic antibodies for cancer, combination cancer immunotherapy, and T cell target discovery. ImVacS 2015 promises to be a must-attend event for commercial and academic entities to continue advancing immunotherapies and vaccines through technology and innovation.”
Event: http://www.imvacs.com/
PGM: Rational Combination Cancer Immunotherapy (1 of 6)
SESSION: COMBINATION IMMUNOTHERAPY STRATEGIES
8-26-15 9-9:30 Jeff T. Hutchins, PhD, VP/Preclin-Res., Peregrine Pharmaceuticals
“Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand Phosphatidylserine (PS): Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
SUMMARY:
The underlying cause for the failure of current therapies is the persistent and multifocal immune suppression in the tumor microenvironment that drives the absence of pre-existing antitumor T cells. Bavituximab blocks PS-mediated immunosuppression (decreasing MDSCs) by reprograming immune cells in the tumor microenvironment to enhance anticancer activity. Pre-clinical, translational, and clinical results using bavituximab with conventional and immunotherapy combinations promotes a robust, anti-tumor T cell mediated response to enhance cancer therapy.
http://ir.peregrineinc.com/events.cfm
Dr.Hutchins(VP/Preclin-Res.) speaks today at 9amET at the 10th Annual Immunotherapy & Vaccine Summit (ImVacS 2015) in Boston – in session, “Combination Immunotherapy Strategies”…
Aug 24-28 2015: “CHI’s 10th Annual Immunotherapy & Vaccine Summit”, Boston
CHI = Cambridge Healthtech Institute, http://www.healthtech.com
“…This year's event has been expanded to 5 days with coverage on adjuvants, vaccine & immunotherapy technologies, immunomodulatory therapeutic antibodies for cancer, combination cancer immunotherapy, and T cell target discovery. ImVacS 2015 promises to be a must-attend event for commercial and academic entities to continue advancing immunotherapies and vaccines through technology and innovation.”
Event: http://www.imvacs.com/
PGM: Rational Combination Cancer Immunotherapy (1 of 6)
SESSION: COMBINATION IMMUNOTHERAPY STRATEGIES
8-26-15 9-9:30 Jeff T. Hutchins, PhD, VP/Preclin-Res., Peregrine Pharmaceuticals
“Expansion and Activation of T Cells via the Targeting of the Immunosuppressive Ligand Phosphatidylserine (PS): Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
SUMMARY:
The underlying cause for the failure of current therapies is the persistent and multifocal immune suppression in the tumor microenvironment that drives the absence of pre-existing antitumor T cells. Bavituximab blocks PS-mediated immunosuppression (decreasing MDSCs) by reprograming immune cells in the tumor microenvironment to enhance anticancer activity. Pre-clinical, translational, and clinical results using bavituximab with conventional and immunotherapy combinations promotes a robust, anti-tumor T cell mediated response to enhance cancer therapy.
http://ir.peregrineinc.com/events.cfm
Yesterday’s statement by AstraZeneca’s Head of Immuno-Oncology about what he termed “Our Partnership with Peregrine” - every time I read it, it gets better...
8-24-15: Robert Iannone, Head of Immuno-Oncology, Global Medicines Development, at AstraZeneca said, "We believe that combination therapy in immuno-oncology has the potential to be a novel and highly effective approach to treating cancer. Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
= = = = = = = = =
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
Durvalumab=MEDI4736, an anti-PD-L1 immune checkpoint inhibitor.
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors”…
Durvalumab=MEDI4736, an anti-PD-L1 immune checkpoint inhibitor.
AZN’s Head/I-O(Robert Iannone): “…Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
= = = = = = = = = = = = = =
8-24-15: AstraZeneca and Peregrine Pharmaceuticals to Collaborate on Immuno-Oncology Combination Clinical Trial
• Collaboration to Focus on Cancer Immunotherapy Combination of Peregrine's PS-Targeting Bavituximab and AstraZeneca's PD-L1 Inhibitor MEDI4736
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=928488
TUSTIN, Aug. 24, 2015: AstraZeneca (NYSE:AZN) and Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer, today announced that they have entered into a cancer immunotherapy clinical trial collaboration. The collaboration will evaluate Peregrine's investigational phosphatidylserine (PS)-signaling pathway inhibitor, bavituximab, in combination with AstraZeneca's investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab (MEDI4736). The planned Phase I/Ib trial will evaluate the safety and efficacy of bavituximab in combination with durvalumab in multiple solid tumors.
Peregrine and AstraZeneca will collaborate on a non-exclusive basis, to evaluate the combination of bavituximab and durvalumab with chemotherapy as a potential treatment in various solid tumors. The Phase I part of the trial is expected to establish a recommended dose regimen for the combination and the Phase Ib part of the trial will assess the safety and efficacy of the investigational combination. Under the terms of the agreement, the initial trial will be conducted by Peregrine.
Robert Iannone, Head of Immuno-Oncology, Global Medicines Development, at AstraZeneca said, "We believe that combination therapy in immuno-oncology has the potential to be a novel and highly effective approach to treating cancer. Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
Bavituximab and durvalumab are investigational immunotherapies with different mechanisms that assist the body's immune system in fighting cancer. Bavituximab targets and modulates the activity of phosphatidylserine, a highly immune-suppressive molecule expressed broadly on the surface of cells in the tumor microenvironment. The treatment increases activated T-cells in tumors and fights cancer by reversing the immunosuppressive environment that many tumors establish in order to proliferate. MEDI4736 is a monoclonal antibody directed against programmed cell death ligand 1 (PD-L1). Signals from PD-L1 help tumors avoid detection by the immune system. Preclinical data have demonstrated that combining the enhanced T-cell mediated anti-tumor activity of bavituximab with checkpoint inhibitors, like PD-L1 antibodies, prolong the ability of tumor-specific T-cells to continue attacking the tumor.
"Data generated to date have shown significant potential for combining bavituximab with agents targeting the PD-1/PDL-1 pathway and we're excited to further explore this approach in studies with AstraZeneca's durvalumab," said Steven W. King, President and CEO of Peregrine. "AstraZeneca is a recognized leader in the immuno-oncology field and this collaboration will play a key role as we continue to fully explore the potential of bavituximab in combination immunotherapies for a variety of clinical applications."
ABOUT BAVITUXIMAB: A Targeted Investigational Immunotherapy
Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab, the lead compound in Peregrine's immuno-oncology development program, blocks PS to remove this immunosuppressive signal and sends an alternate immune activating signal. Targeting PS with bavituximab has been shown to shift the functions of immune cells in tumors, resulting in robust anti-tumor immune responses.
ABOUT DURVALUMAB (MEDI4736)
MEDI4736 is an investigational human monoclonal antibody directed against programmed cell death ligand 1 (PD-L1). Signals from PD-L1 help tumors avoid detection by the immune system. MEDI4736 blocks these signals, countering the tumor's immune-evading tactics. MEDI4736 is being developed, alongside other immunotherapies, to empower the patient's immune system and attack the cancer.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company with a pipeline of novel drug candidates in clinical trials focused on the treatment of cancer. The company's lead immunotherapy candidate, bavituximab, is in Phase III development for the treatment of second-line non-small lung cancer (the "SUNRISE trial") along with several investigator-sponsored trials evaluating other treatment combinations and additional oncology indications. Peregrine also has in-house cGMP manufacturing capabilities through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. For more information, please visit http://www.peregrineinc.com .
ABOUT ASTRAZENECA IN ONCOLOGY?
Oncology is a therapeutic area in which AstraZeneca has a deep-rooted heritage. It will be potentially transformational for the company's future, becoming the sixth growth platform. Our vision is to help patients by redefining the cancer treatment paradigm and one day eliminate cancer as a cause of death. By 2020, we are aiming to bring six new cancer medicines to patients.
Our broad pipeline of next-generation medicines is focused on four main disease areas - ovarian, lung, breast and haematological cancers. These are being targeted through four key platforms - immuno-oncology, the genetic drivers of cancer and resistance, DNA damage repair and antibody drug conjugates.
ABOUT ASTRAZENECA
AstraZeneca is a global, innovation-driven biopharmaceutical business that focuses on the discovery, development and commercialization of prescription medicines, primarily for the treatment of cardiovascular, metabolic, respiratory, inflammation, autoimmune, oncology, infection and neuroscience diseases. AstraZeneca operates in over 100 countries and its innovative medicines are used by millions of patients worldwide. For more information please visit: http://www.astrazeneca.com
Safe Harbor *snip*
Contacts:
• Jay Carlson, Peregrine Pharmaceuticals, Inc., (800) 987-8256, info@peregrineinc.com
• Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
• Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = = = = = = = = = = =
Durvalumab (MEDI4736, anti-PD-L1) has a Net Present Value of $6.5B.
AZN’s Head/I-O(Robert Iannone): “…Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."

= = = = = = = = = = = = = = =
SCIENCE: "Evolution has favored pathogenesis that resembles apoptosis."
Dr. Judah Folkman: ”This [Thorpe’s VTA research] is very promising and very elegant work... The whole goal is really 2-part, reducing the harsh side effects of cancer treatment, and reducing the chance that some cancer cells will evade treatment. That would be a big step in the next decade, and anti-vascular therapy will play a major role." (’97 & ’02 http://tinyurl.com/k5qe96g & http://tinyurl.com/n6vh9hp )
Peregrine's Bavituximab Clinical Trials website: http://PeregrineTrials.com
BAVITUXIMAB MOA & CLINICAL DATA: http://www.peregrineinc.com/pipeline/bavituximab-oncology.html
...Bavi Publications: http://www.peregrineinc.com/publications/publications.html
...Bavi Posters & Presentations: http://www.peregrineinc.com/publications/posters-and-presentations.html
Bavi MOA: Video (3:34) added ~3-2014 http://vimeo.com/87116642 "Bavituximab: A Novel Immunotherapy Candidate Targeting an Upstream Immune Checkpoint to Fight Cancer"
Bavi MOA: Video (1:33) on Bavi’s Immunotherapeutic MOA added to Youtube on 3-27-14 https://www.youtube.com/watch?v=Esewl35JD8s

5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
BAVI MOA 3-25-15: PPHM/VP Dr. Jeff Hutchins’ presentation at "Immune Checkpoint Inhibitors Conf.", Boston - PDF(34 Slides): http://tinyurl.com/ooxkhq7
BAVI MOA 2-9-15: PPHM/VP Dr. Bruce Freimark’s presentation at KEYSTONE "Tumor Immunology Meeting", Banff/CN – PR & Slides: http://tinyurl.com/q6cx4w6
BAVI MOA 12-12-14 San Antomio Breast Cancer Symposium, Dr. Bruce Freimark, Bavi+anti-PD-1 vs. Breast Cancer http://tinyurl.com/p5ng6vs
BAVI MOA 11-2014 SITC’14: 3 posters on preclin. Bavi+aCTLA4/aPD1 combo data (Hutchins Freimark Brekken Huang etal) http://tinyurl.com/pchzr6h
BAVI MOA 8-28-14: Italy’s Dr. Federico Cappuzzo, Bavi/NSCLC profile article (DovePress OpenAccess, 7pgs) - http://tinyurl.com/n3oa7bc
BAVI MOA 8-11-14: PPHM/VP Dr. Jeff Hutchins’ presentation at ImVacS "Immunotherapies & Vaccine Summit", Boston - PR: http://tinyurl.com/lpjy3u7 ; PDF(31 Slides): http://tinyurl.com/oueldme
...MLV’s Dr. George Zavoico’s 8-27-14 report on Dr. Jeff Hutchins’ 8-11-14 ImVacS/Boston Bavi MOA presentation: http://tinyurl.com/l3tw63c
BAVI MOA 5-28-14: Dr. Rolf Brekken’s 47min CRI “Cancer Immunotherapy” webinar about Bavituximab as an Upstream/Global Immune Checkpoint Inhibitor – REPLAY: http://tinyurl.com/lxgftyx
. . .CRI=Cancer Research Institute (NYC – Supported by BMS): http://www.cancerresearch.org - Facebook: http://tinyurl.com/pbmhb2z , https://twitter.com/CancerResearch
. . .CRI launches “Answer to Cancer” (cancer immunotherapy) website http://www.theanswertocancer.org
. . .8-12-14: CRI adds Youtube links to the 5-28-14 CRI Immunotherapy webinar, incl. Dr. Brekken's talk "about Bavi and how it works against lung, liver, and other kinds of cancers" http://tinyurl.com/ps5h6h8
BAVI MOA 3-25-14: Dr. Rolf Brekken’s 40min talk at NYAS Lung Cancer Symposium http://tinyurl.com/lq9stnk (45 Slides)
. . .Dr.Brekken’s talk: “Antibody-mediated Inhibition of PS - A Novel Strategy for Immune Checkpoint Blockade” (the 5 speakers: Jessica Donington, Roy Herbst, Balazs Halmos, Suresh Ramalingam, Rolf Brekken)
12-10-13: With recent scientific insights highlighting bavi’s immunostimulatory moa, these additions to PPHM’s SAB: Dimitry Gabrilovich, Scott Antonia, David Carbone**, Hakan Mellstedt http://tinyurl.com/mw776mk
......**A/o 9-2014, Dr. David Carbone (PPHM SAB/KOL) is President-Elect of IASLC https://www.iaslc.org/about-us/board
BAVI MOA: 12-2013 Bavi’s Immunotherapeutic MOA overviewed by UTSW’s Brekken/Huang in Pan European Networks Jrnl. http://tinyurl.com/lnb46pq
BAVI MOA 11-9-13: Annual SITC (WashDC) – 2 posters about Bavi’s Immunostimulatory MOA http://tinyurl.com/mjaweu5
...“We are actively working towards initiating a clinical trial in the coming months to further investigate the potential synergistic effects of bavituximab and an approved [anti-CTLA-4] immunotherapy in patients with Melanoma."
10-28-13 IASLC/Sydney: “Immune Checkpoints in the Tumor Environment: Novel Targets & the Clinical Promise of Combined Immunotherapies” http://tinyurl.com/mjaweu5
…Symposium speakers: Scott J. Antonia/MD-PhD(H.Lee Moffitt CC), Dmitry I. Gabrilovich/MD-PhD(Wistar Inst), Rolf A. Brekken/PhD(UTSW), David E. Gerber/MD(UTSW)
BAVI MOA: 8-19-13 Data Supporting Bavituximab’s Immunotherapy MOA Published in “Cancer Immunology Research” (AACR) - http://tinyurl.com/mhjftka (PDF)
…“PS-Targeting Antibody Induces M1 Macrophage Polarization & Promotes Myeloid-Derived Suppressor Cell Differentiation” (Thorpe etal)
BAVI MOA: 8-13-13 PPHM/VP Dr. Jeff Hutchins’ Presentation on the Downstream Immunostimulatory Effects/Moa of PS-targeting antibodies (like Bavi) at CHI’s “Immunotherapies Congress”/Boston http://tinyurl.com/m6h2tvt
BAVI MOA: 10-12-12 NMB article on how Bavi "Induces Innate & Specific Anti-tumor Responses" http://tinyurl.com/cw9odb8
BAVI MOA: 5-1-12 Dr. Phil Thorpe's 46min talk at NYAS PS-Targeting Symposium http://tinyurl.com/9792gl5
. . .Symposium title: "Phosphatidylserine (PS) Asymmetry - Therapeutic Apps. in Cancer & Infectious Disease Symposium"
. . .Replays of 5 speakers: Alan Schroit, Chris Reutlingsperger, David Ucker, Ari Helenius, Philip Thorpe
Durvalumab (MEDI4736, anti-PD-L1) has a NPV of $6.5B.
AZN’s Head/I-O(Robert Iannone): “…Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."

8-24-15: AstraZeneca and Peregrine Pharmaceuticals to Collaborate on Immuno-Oncology Combination Clinical Trial
• Collaboration to Focus on Cancer Immunotherapy Combination of Peregrine's PS-Targeting Bavituximab and AstraZeneca's PD-L1 Inhibitor MEDI4736
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=928488
TUSTIN, Aug. 24, 2015: AstraZeneca (NYSE:AZN) and Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company focused on developing therapeutics to stimulate the body's immune system to fight cancer, today announced that they have entered into a cancer immunotherapy clinical trial collaboration. The collaboration will evaluate Peregrine's investigational phosphatidylserine (PS)-signaling pathway inhibitor, bavituximab, in combination with AstraZeneca's investigational anti-PD-L1 immune checkpoint inhibitor, durvalumab (MEDI4736). The planned Phase I/Ib trial will evaluate the safety and efficacy of bavituximab in combination with durvalumab in multiple solid tumors…
. . .
Robert Iannone, Head of Immuno-Oncology, Global Medicines Development, at AstraZeneca said, "We believe that combination therapy in immuno-oncology has the potential to be a novel and highly effective approach to treating cancer. Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
. . .
"Data generated to date have shown significant potential for combining bavituximab with agents targeting the PD-1/PDL-1 pathway and we're excited to further explore this approach in studies with AstraZeneca's durvalumab," said Steven W. King, President and CEO of Peregrine. "AstraZeneca is a recognized leader in the immuno-oncology field and this collaboration will play a key role as we continue to fully explore the potential of bavituximab in combination immunotherapies for a variety of clinical applications."
I wonder what spurred Dana-Farber/Harvard’s Dr. Kathleen Mahoney (or co-authors Dr. Gordon Freeman/Harvard / Paul Rennert/Consultant) to add this to her new Nature-Reviews article on Combination Cancer Immunotherapy:
Pg19: “Bavituximab might also be beneficial when paired with a targeted therapy such as the VEGFR2-specific antibody ramucirumab (Cyramzain, developed by Eli Lilly & Co.), which has reported positive Phase III trial data as 2nd-Line therapy in combination with docetaxel for NSCLC.
I’d love to know if it just a simple observation she came up with on her own, or whether someone else provided input.
= = = = = = = = = = = =
NATURE REVIEWS Drug Discovery (series: Cancer Immunotherapy)
Aug. 2015 (Pub. online: 7-31-15)
“Combination Cancer Immunotherapy & New Immunomodulatory Targets”
KATHLEEN M. MAHONEY, Paul D. Rennert, Gordon J. Freeman
Dana–Farber Cancer Institute, Harvard Medical School, Dept of Medical Oncology, Boston
http://www.nature.com/nrd/journal/v14/n8/abs/nrd4591.html
PDF: http://www.sugarconebiotech.com/wp/wp-content/uploads/2015/07/nrd4591-1.pdf
- - - - - - -
Note: On 12-12-14, the FDA approved Eli Lilly’s ramucirumab (trade name Cyramzain) combination with docetaxel, for treatment of metastatic NSCLC with disease progression on or after platinum-containing chemotherapy.
http://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm444496.htm
• K.Mahoney: http://www.conquercancerfoundation.org/kathleen-m-mahoney-md-phd
• G.Freeman: http://www.dfhcc.harvard.edu/insider/member-detail/member/gordon-j-freeman-phd
• Paul Rennert: http://www.sugarconebiotech.com/?author=2 (SugarCone Biotech “A consortium managed by Paul D Rennert, a well-known and highly respected biotechnology scientist. Consortium members are highly skilled and experienced professionals from the biotechnology sector…Our core strength is Strategic Analysis and Consulting to support successful corporate development. We engage with our clients in academic technology transfer, start-up creation and financing, corporate in-licensing, due diligence, competitive intelligence, program development and execution, outsourcing and vendor selection, out-licensing, defining critical transition stages and milestones, and identifying academic and corporate partnerships.”)

Aug2015 NatureReviews article by Dana-Farber/Harvard Oncologists profiles Bavi in 20pg article, “Combination Cancer Immunotherapy & New Immunomodulatory Targets”
Nature Reviews Drug Discovery (series: Cancer Immunotherapy)
Aug. 2015 (Pub. online: 7-31-15)
“Combination Cancer Immunotherapy & New Immunomodulatory Targets”
KATHLEEN M. MAHONEY, Paul D. Rennert, Gordon J. Freeman
Dana–Farber Cancer Institute, Harvard Medical School, Dept of Medical Oncology, Boston
http://www.nature.com/nrd/journal/v14/n8/abs/nrd4591.html
PDF: http://www.sugarconebiotech.com/wp/wp-content/uploads/2015/07/nrd4591-1.pdf
= = = = = = = = =
Interesting last sentence by Dr. Mahoney:
Pg19: “Bavituximab might also be beneficial when paired with a targeted therapy such as the VEGFR2-specific antibody ramucirumab (Cyramzain, developed by Eli Lilly & Co.), which has reported positive Phase III trial data as 2nd-Line therapy in combination with docetaxel for NSCLC.
- - - - - - -
Note: On 12-12-14, the FDA approved Eli Lilly’s ramucirumab (trade name Cyramzain) combination with docetaxel, for treatment of metastatic NSCLC with disease progression on or after platinum-containing chemotherapy.
http://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm444496.htm
TEXT OF ARTICLE (pg.18-19):
Phosphatidylserine: Phosphatidylserine is a membrane marker exposed during the process of programmed cell death or apoptosis and serves as an ‘eat me’ signal for phagocytes. Phosphatidylserine is also expressed on rapidly dividing cells, notably proliferating lymphocytes, tumor cells and the tumor vasculature. Profligate tumor cell proliferation leads to the death of large numbers of cells. Because apoptosis is a controlled mechanism of cell death it does not normally trigger immune activation, and therefore phosphatidylserine expression on tumor cells does not induce an antitumor immune response. Indeed, tumor cells may further evade infiltrating immune cells via binding of phosphatidylserine to TIM3 expressed on T cells.
The phosphatidylserine-specific antibody bavituximab (developed by Peregrine Pharmaceuticals) acts by dimerizing soluble B2-glycoprotein 1, converting it from a low to a high avidity binder of phosphatidylserine 182. This allows an antitumor immune response to develop, possibly by coating the tumor cells with antibody or by converting apoptotic cell death to a more immunostimulatory type of cell death. A Phase II trial of bavituximab in advanced NSCLC yielded positive results on progression-free survival and, more importantly, improved overall survival from less than 6 mos. to 12 mos. 183, leading the FDA to grant a Fast Track Designation for bavituximab as a 2nd-Line therapy. A Phase III trial of bavituximab as a second-line therapy for advanced NSCLC is underway [ http://www.clinicaltrials.gov/ct2/show/NCT01999673 ]. Recent presentations showed synergy of phosphatidylserine-specific antibody treatment with CTLA4-specific and PD1-specificantibodies in mouse models, through effects on MDSC and lymphocyte activity 184,185.
Bavituximab might also be beneficial when paired with a targeted therapy such as the VEGFR2-specific antibody ramucirumab (Cyramzain, developed by Eli Lilly & Co.), which has reported positive Phase III trial data as 2nd-Line therapy in combination with docetaxel for NSCLC 186.

Thanks very much for catching that, BioBS. Just got it corrected B4 my 15min. edit window ran out!
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GM%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY13 TOTAL: 21,333,000 12,595,000 8,738,000 41%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY14 TOTAL: 22,294,000 13,110,000 9,184,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
*Avid Net-Profit (ie, incl. Selling, G&A) not split out from PPHM-Corp. in the financials.
.
PPHM REVENUES (in thousands) DEFERRED
-------REVENUES------- REVENUES INVEN-
Quarter Avid Govt Lic. TOTAL Avid Govt TORIES
FY07Q1 7-31-06 398 0 23 421 317 0 971
FY07Q2 10-31-06 636 0 48 684 1388 0 1899
FY07Q3 1-31-07 347 0 16 363 2202 0 1325
FY07Q4 4-30-07 2111 0 129 2240 1060 0 1916
FY08Q1 7-31-07 1621 0 4 1625 1820 0 2363
FY08Q2 10-31-07 1863 0 29 1892 1338 0 3500
FY08Q3 1-31-08 1662 0 13 1675 1434 0 2394
FY08Q4 4-30-08 751 0 150 901 2196 0 2900
FY09Q1 7-31-08 1193 324 0 1517 4021 980 4628
FY09Q2 10-31-08 983 958 0 1941 6472 1701 6700
FY09Q3 1-31-09 5778 1048 0 6826 4805 3262 5547
FY09Q4 4-30-09 5009 2683 175 7867 3776 3871 4707
FY10Q1 7-31-09 2070 4671 9 6750 5755 2332 6177
FY10Q2 10-31-09 5308 1510 78 6896 4260 3989 5850
FY10Q3 1-31-10 2945 6854 78 9877 3052 76 3861
FY10Q4 4-30-10 2881 1461 78 4420 2406 78 3123
FY11Q1 7-31-10 983 2111 115 3209 3719 47 4692
FY11Q2 10-31-10 3627 966 78 4671 2447 35 3555
FY11Q3 1-31-11 1922 882 79 2883 4300 40 3915
FY11Q4 4-30-11 1970 681 78 2729 5617 0 5284
FY12Q1 7-31-11 5439 0 216 5655 4145 0 4481
FY12Q2 10-31-11 4154 0 78 4232 2012 0 3178
FY12Q3 1-31-12 3203 0 78 3281 2552 0 2722
FY12Q4 4-30-12 1987 0 78 2065 3651 0 3611
FY13Q1 7-31-12 4135 0 116 4251 6056 0 5744
FY13Q2 10-31-12 6061 0 78 6139 6221 0 5426
FY13Q3 1-31-13 6961 0 78 7039 5061 0 4635
FY13Q4 4-30-13 4176 0 78 4254 4171 0 4339
FY14Q1 7-31-13 4581 0 107 4688 4164 0 5679
FY14Q2 10-31-13 7354 0 0 7354 3468 0 4033
FY14Q3 1-31-14 3885 0 0 3885 4329 0 5224
FY14Q4 4-30-14 6474 0 0 6474 5241 0 5530
FY15Q1 7-31-14 5496 0 0 5496 4670 0 5998
FY15Q2 10-31-14 6263 0 37 6300 3612 0 5379
FY15Q3 1-31-15 5677 0 0 5677 5752 0 6148
FY15Q4 4-30-15 9308 0 0 9308 5752 0 6148
Totals: 129212 24149 2124 155485 <=since5/1/2006