Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
BP/NP business disagreement thought
There is so much synergy between Cytodyn and IncellDx. Its apparent watching the DrBeen BP interview tonight.
1. Merger. Cydy could vertically integrate and own the RANTS testing kit. They'll be doing a lot of RANTS testing for all the different diseases Leronlimab could possibly treat.
2. Partnership. Package the upcoming IncellDx Covid test kit with a full treatment course of Leronlimab in some fancy packaging for retail distribution?
Both would be a huge win for IncellDx, but a distraction for Cytodyn.
FDA wouldn't even approve a third party manufacturer (FDA Approved) labeled syringe for the latest HIV approval CYDY debacle. The IncellDx test kit isn't even FDA approved yet!
Frankly it really doesn't matter for CYDY longs at this point. Just a thought I had.
Side effects of lenzilumab
Does anyone know of any side effects or adverse reactions reported for lenzilumab?
Posts on iHub can be tough
Some posts here take a small investment of time. Its frustrating when they get lost.
I still need to read the s1 and understand this Pipe investor concept.
I think CYDY's timeline is being underestimated by most. The Tuesday release is a top line description of results. Another 3 weeks will go into the written response to FDA. FDA will then take at least 2 weeks to give an expedited EUA. FDA will probably want to see P2 M/M and P3 S/C trial results together for one approval which will delay approval to September.
There is a VERY good chance that HGEN is just as far along or further with their P3 trial but Durrant is tight lipped. I think that's probably for the best!
On the balance of it all, the HGEN P3 clinical trial sites are in COVID hot spots right now. CYDY P3 trial sites do not have as much COVID outbreak activity by comparison (even NYC).
It'll be interesting over the next few weeks for sure!
Durant doesn't want liquidity
He's running this company as a startup with a bunch of venture capitalists.
It doesn't seem he's much interested in the "public" part of this being a publicly traded company.
Absolutely everything is outsourced. Total share count is low. Stock is locked up by sophisticated investors. The company framework is setup for a buyout.
I'm still trying to figure out this company - I think I have a long way to go.
7realone7 got me good!
You're right about my misuse of terminology.
It's the traders and flippers that cause all the short term (anything less than 4 weeks) market volatility. On top of that, OTC traders are an especially impatient bunch!
Investors buy and faithfully hold stock.
HGEN is primarily owned by investors... even the float shares.
CYDY is owned by 50% investor, 40% OTC traders, and 10% CYDY groupie [unsophisticated hobbyists, for lack of a better description] based on my observations.
HGEN stock price would surge a little [I'm starting the obvious] if traders took an interest in it. It already has plenty of institutional investors IMO.
International Leronlimab Approval
How would this process happen? How would Leronlimab get approval for use in countries around the world? [At least in the countries where bribes don't work well.]
Its inconceivable CYDY would have separate localized clinical trials in each country before entering disjointed international markets to sell Leronlimab.
Can CYDY just submit all of the clinical trial data and FDA paperwork to other countries' health departments, pay an exorbitant application fee, and wait for the bureaucratic process to give approval? How much would CYDY lean on their distributor to do this for them?
Very thoughtful insight.
Share Supply for public trading from a limited float drives up price with strong demand. Absolutely spot on cowtown jay!
I think we won't hear any HGEN news until Thursday. CYDY is going to try to issue a press release Tuesday morning with top line results from their P2 M/M COVID Trial which will pull potential new $$ from HGEN. Many are fairly certain the results will be overwhelmingly positive.
BUT! Then on Wednesday CYDY is going to approve, through a special shareholders meeting, to authorize another 100M shares which will cause the stock price to drop. The CEO only plans to use 10M shares in late August after the NASDAQ uplisting; but it's a big number. I'd also expect to see a coordinated short attack that afternoon.
Thursday would be a great day for HGEN to drop some news and pick up some of those battered CYDY investors in my opinion.
Both drugs are a lot alike Part2.
The cytokine storm is bad and the human body keeps it controlled under lock & key. Lenzilumab binds to the key so it doesn't fit in the lock. Leronlimab binds to the lock so the key doesn't fit. If the biological process cannot be unlocked, then there is no cytokine storm.
1. GM-CSF is an inflammatory cytokine signaling molecule that promotes inflammation response by cells.
2. CCR5 is a type of receptor on a few types of white blood cells (including macrophage and T cells) and plays a role in inflammatory responses to infection.
3. Therapies to treat inflammatory diseases either neutralize inflammatory cytokines or their receptors.
4. Lenzilumab binds to/neutralizes the GM-CSF inflammatory cytokine.
5. Leronlimab binds to/neutralizes the CCR5 receptor.
Thanks for the nudge to do this from Fezziwig2008!
I didn't change the analysis
What I described to you about estimated net income valuation is how I originally ran the numbers.
My most recent work is in the post you were already looking at.
net income valuation
I tried to estimate net income valuation by calculating net revenue (column O) as 50% of gross revenue (column N).
If I misunderstood, let me know. I'd like to get it right.
The drug treatment cost of column K doesn't include markups from distribution channels. That's why I assumed net revenue would be 50% gross revenue... and not something like 20% of gross revenue.
Both drugs are a lot alike.
For COVID at least, Lenzilumab and Leronlimab function VERY similarly.
Leronlimab has a small advantage over Lenzilumab. 1) It blocks one of the key pathways that COVID virus uses to infect cells. 2) It calms the cytokine storm at the top level source receptor and returns the immune system to normal conditions. 3) It reduces viral load (due to 1 & 2). Link to summary of preprint manuscript.
Lenzilumab does #2 by interrupting a downstream biological process and helps the body immune system naturally do #3.
They are both amazing drugs.
Both companies are a lot alike.
Combined they have less than 25 employees.
CYDY has manufacturing agreements in place with Samsung Biologics (practically unlimited capacity if given enough $$$). Inventory will be ready as soon as FDA approval is given, just like HGEN.
They are using American Regent for distribution in USA but it is a subsidiary of a larger worldwide pharma distribution company.
CYDY has a shareholder vote July 22nd to increase the total number of authorized shares of common stock from 700,000,000 to 800,000,000 shares. They would use about 30% to self finance for the NASDAQ uplisting cash reserve requirements. I included the 800M number in my dilution analysis. The other 70% would not be released unless there was critical need.
I agree with you're points Ohm20.
LOL. I've seen "FDA doesn't approve on anecdotal evidence" about both HGEN and CYDY. Which is why we have the bureaucratic FDA who only caters to big pharma trying to slow down both CYDY and HGEN by requiring these lengthy but necessary clinical trials. Gilead didn't have to wait months to push their COVID drug!
Pourhassan said on a recent conference call that he expects CYDY stock to be in the three figure range once he get's the FDA approvals. Now that's a buyout deal I'd like to be part of!
Durrant really don't say much at all about anything - very frustrating.
My Analysis of Target Prices
Cowtown Jay, spot on! You said in a couple of sentences what I'm going to repeat in 'chapter book' form.
You can invest $20,000 in company A or company B. They both have similar stock prices (for now), are both going to enter the same market with comparable products at the same time. Your return on investment comes down to the total number of shares outstanding in each company if both companies are going to have equivalent market caps.
When both HGEN and CYDY get their FDA approvals for COVID treatment, essentially both companies should have the same valuation. Ignore all the 'side' projects such as HIV, cancer, etc for this thought exercise for simplicity. That stuff matters - just not in the next 3 months.
I saw some numbers thrown around on CYDY's iHub forum for price per share after the COVID FDA approval ($30 to $100/share). If CYDY stock price is $40/share based on current outstanding shares, it would have a total market cap of $20.8B.
If HGEN also has the same total (hypothetical) market cap of $20.8B but with a far fewer number of outstanding shares, the stock price would be $147/share.
This analysis shows the effect of share dilution on stock price between the two companies - regardless of the amount of share float.
I think the market cap of $20.8B is VERY low for either company based on the global need for a COVID therapeutic. [I have a separate post on that.] Therefore, the actual stock price should be much higher for both companies... but proportionally higher. Again, this ignores the likelihood that one company will be better positioned in particular local or regional markets based on their distribution deals, the amount of drive & expertise of the company's management team, and a fair bit of luck.
The John Hopkins COVID Dashboard reports 13.7M total confirmed COVID19 cases right now. Let's assume for every 1 confirmed case, there are 9 that are unconfirmed. Let's say 137M worldwide COVID (confirmed + unconfirmed) cases today and assume no one will get re-infected. The global adult population is 5.6B. The world had a very long way to go until herd immunity is achieved.
I don't know where Long Tkt got his Low and High end target prices. But, given that both companies are currently trading at about the same price, depending on the day, it's a slam dunk in favor of HGEN. Far fewer shares to claim a piece of that market cap.
Avoid debate of Leronlimab vs. Lenzilumab
I don't want to get into the medical debate about Leronlimab vs. Lenzilumab for COVID. I've seen misunderstandings about the drugs' EIND results from both sides of the fence.
For both treatments, patients get significantly better in 5 days or less; compared to 12 days for current standard of care.
Everyone agrees there is a COVID market big enough for both products and both will likely get FDA approvals very soon based on anecdotal evidence. Neither CYDY nor HGEN will be able to fully/solely supply the COVID treatment global demand to calm the cytokine storm.
When the COVID crisis is over, however; I think the two companies will have very different futures. And that warrants a separate big picture with a different financial analysis.
Let's just hope neither company is bought out at current bargain basement prices given the potential revenue from the COVID crisis.
Previous Look at the Global COVID Market
The COVID vaccine won't be ready for mass distribution until June 2021. It will be similar to the flu vaccine so 50% of the population choose to get the vaccine and the vaccine is 50% effective for those that receive it. That means only 25% of the world will be successfully vaccinated from COVID by October 2021.
There is also growing evidence that the general population can be re-infected by COVID only a few months after recovering from a first infection; but this analysis doesn't take that into account. Research also suggests that people with COVID who are asymptomatic will likely be re-infected with a more severe immune response. Yes, currently 20% of COVID infections result in a hospital stay but I'm ignoring who is hospitalized vs. outpatient.
Someone on the CYDY forum mentioned that the company should offer a dividend. If HGEN is NOT bought out, then I think there is potential for HGEN to do so as well.
CYDY Global COVID Market iHub post
Here's a version for HGEN:


I'm pretty comfortable with $1.3 trillion dollar global market prediction for COVID-19 treatment. Here are my reasons why.
I think HGEN has nearly limitless COVID revenue potential; even if they only capture 10% of the global market.
Comparison of HGEN and CYDY
Both companies have a great treatment for COVID. They will both be welcomed by the global market. Ignoring future product development potential, let's assume both companies will have the same "New Market Cap" when their product receives FDA approval for COVID treatment.
I'm trying to wrap my head around the differences for COVID only investing.
What other comparisons did I leave out?

Follow-up to Millstone's HGEN interim analysis timeline.
So... It's crunch time between two OTC companies!
HGEN will soon (likely) receive their Phase 3 S/C trial interim analysis (to halt the trial because of its clear success exceeding current standard of care). Very little public info available but one tiny possible hint - The recent Twitter posts with FDA tagged.
CYDY will unblind it's completed Phase 2 M/M trials late this week, per conference call. Announcement of positive results during the week of July 27th or August 3rd.
Based on gut feeling, who do you think we'll hear clinical trial results from first: HGEN or CYDY? And, why.
Great job by the way, Millstone!
Interim Results Strategy. Great Post Errol315.
I'm fascinated by the idea of building Interim Analysis into clinical trials as a circuit breaker to end the trial early. Here me out here.
FDA's independently acting Data Safety Monitoring Board (and only them) gets both the data and 'the keys' to unblind that data. When they look at the data, the can 'call an audible' for the following:
1) Modifications of the study protocol based upon the review of the safety data;
2) Suspension or early termination of the study or of one or more study arms because of serious concerns about subjects’ safety, inadequate performance, or rate of enrollment;
3) Suspension or early termination of the study or of one or more study arms because study objectives have been obtained according to pre-established statistical guidelines;
Source: NIS-DSMB
If Lerlonlimab (or any drug for that matter) is such a great product and clearly demonstrates it exceeds the objectives of the clinical study by (say) being 50% better than the current Standard of Care, I'd sure want the FDA DSMB to terminate the study early so the drug approval can occur quicker.
Considering that a clinical trial costs a company $40,000 per patient (on average), I think it's a great idea to manage some of the risk by having the FDA do a sanity check on the trial progress.
Engineers see the world differently
Everything is analyzed, ranked, and compared. Nothing is ever black and white. The simplest and most elegant solution is always the most difficult to find. Processes should always be tweaked and perfected. It's just the engineer's way.
I'm not a big fan of smooth talking romeos either. I am who I am, and I'm certainly not always right. I'm always putting my foot in my mouth.
But, I don't mind sharing my thoughts or asking difficult questions despite the risk of harsh criticism.
I try to see both sides of the coin. How can anyone see the full picture unless you pick something apart, look at it from many different perspectives, and try to understand both the good and the bad?
I live on the East Coast. I'm just another guy who's taken an interest in CYDY. I've never even heard of Nader/CYDY until about 2 months ago. There's no way to convince anyone here if I'm a long or a short. Just as there's no way to know if you secretly work for CytoDyn or Citron Research.
I'll do my best.
I don't remember which Glorified CYDY Podcast I heard this story from. Someone with better memory than me - please feel free to correct this.
CYDY was originally setup to find a new HIV treatment drug. They had a bunch of research scientists and everything. Nader became CEO of Cytodyn just as it was about to go bankrupt with $2 in its bank account [DrBeen Interview]. Needless to say, CYDY was not having much success.
Progenics was researching different HIV drugs and PRO-140 was one of them. Paul Madden invented PRO-140 in 1996 and saw the potential of the drug but was upset that Progenics was going to move forward and commercialize a different research drug.
PRO-140 development was originally funded by the National Institute of Allergy and Infectious Diseases (National Institutes of Health). The projected cost to complete the research and bring the product to market was hundreds of millions of dollars based on the funding agreement with NIH. That's why Progenics wanted to give up on PRO-140 and develop a different drug that had lower development costs.
Nader was introduced to Paul Madden through a mutual contact. Nader was struggling to find a drug for CYDY to commercialize. Madden had a drug no one wanted to develop. It was fate. It was a chance encounter. Call it whatever you want.
Nader learned the upside potential of PRO-140 from Paul Madden and decided it was the drug to save CYDY. Nader worked a deal to purchase the patent from Progenics for $3.5M, two future milestone payments of $1.5M and $5M plus 5% royalties from any future sales for the length of the original PRO-140 patent. This was October 2012.
One of the first things Nader did, was end the NIH research funding agreement for PRO-140 because he realized it wasn't attainable for a small company like CYDY. Then he fired all the research scientists and hired a bunch of staff with expertise bringing Leronlimab to market.
If Nader didn't step in, Progenics would have ended research of PRO-140. All that knowledge would be in a 3-ring binder somewhere on a bookshelf [or in the trash heap, lol].
How did Nader 'know' PRO-140 would be great? The same way you or I found out about it. I'm a civil engineer and know nothing about HIV drug development. I did my own due diligence, made sure the information I read passed the BS meter, and kept reading up on it until I felt I understood enough about Leronlimab to make an informed investment decision.
Do I think it's great? Yeah, I think Leronlimab is to RANTS what penicillin was to bacteria. When they first discovered penicillin, they didn't really have a deep understanding about the bacteria it was used against. I think the current state of medical science doesn't really understand the roll of RANTS in many seemingly unrelated diseases. Who would have thought that Alzheimer's could possibly be a RANTS auto-immune disease?
It's really all an incredibly unbelievable story.
My Struggle Understanding the Clinical Trial Process
I've been researching this for about 30 minutes [making me a subject matter expert]. Actually it just makes me realize how much I don't know. I seek answers - always and about everything. I'm a curious person.
I never considered all the steps involved in a clincal trial. When we hear progress reports about it and discuss it here, I never realized how much the process had been oversimplified!
I now understand what NP meant when he said that executing the Phase 2 M/M COVID clinical trial from study design to closing enrollment in ~90 days is "actually very fast and very hard to do" when DrBeen asked why it was taking CYDY so long. [I'm paraphrasing a little].


Source: AstraZeneca
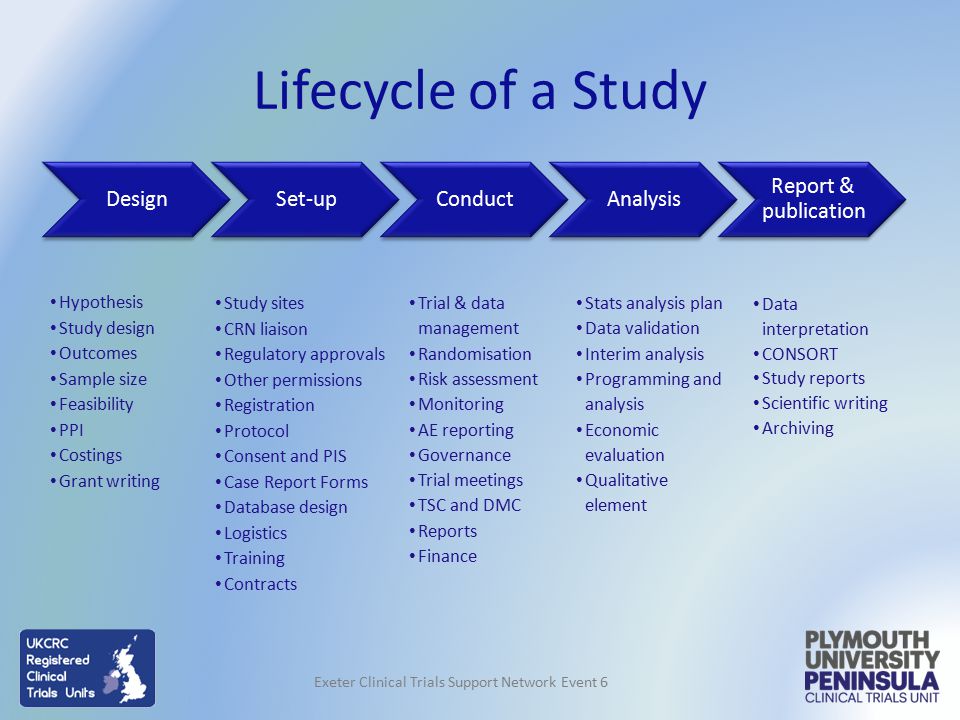
Plymouth University Peninsula Clinical Trials Unit

Announce World Changing News on Glorified Podcast
You need to take a minute to understand the people of CYDY. Both their strengths and weaknesses. Especially their leader Nader Pourhassan who charts the course for the company.
The back story is everything!
[short version of his life] NP was born in Iran. He moved to the United States at 14 with only his brother and mother. They were homeless and had barely enough money to pay for one year's tuition at the school he was going to attend. [Which is likely where he learned his thriftiness]. Nader started is the USA with absolutely nothing but willpower and perseverance. He was able to get a PH.D. in engineering and then become CEO of a company in a field he knew nothing about.
A real underdog!! I would invest in anything this guy does.
NP has nurtured CYDY from the very beginning. He and his family have taken personal risks. He choose to be CEO of a near bankrupt company with no product and took no salary his first year. He found Leronlimab just as it was about to be sent to a trash heap - and bought it. He has saved CYDY from failure multiple times. He had been ousted by an internal coup who didn't understand the simplified, focused, and thrifty execution plan NP had.
The company personality changed under new leadership who proceeded to exponentially increase share dilution (running boutique CYDY like a Big Pharma) and accomplishing next to nothing.
NP regained the helm to become CEO again! He has invested personal funds to keep Samsung Biologics manufacturing on schedule and also kept debtors from seeking CYDY bankruptcy. NP seeks out those who can fill his knowledge gaps (like any good leader would do). He respects and listens to Scott Kelley - not as chairman of the board but as a mentor.
No one has more blood, sweat, and tears invested in CYDY than Nader. Isn't it a good idea to find out who you've trusted your investment with?
So with that said:
Do you really think that when NP makes the BIG announcement that HIS team from HIS company will save the world from COVID using Leronlimab... do you really think he's going to do it using Wall Street Reporter or Proactive as his communications platform?
I'll tell you what would get me really excited. An investor news release saying CNBC Squakbox pre-market or Fox Business Cavuto mid-day is going to have Nader and Kelly on for a company announcement.
This is the comment of a daytrader:
Pure short BS. Good try but really lame and weak. CYDY is solid like no other OTC stock.
I pointed out this tension following the July 4th call. It's team dynamic gossip stuff that's interesting to watch for as an obsessed CYDY groupie.
That's the point of this forum -> talk about all things CYDY! Good and Bad.
If anything, BP should be frustrated with NP. BP had his paper ready for publication ~6 weeks ago which was pulled at the last moment because NP spilled the beans and named the Medical Journal.
It's important to try and understand the personality traits of the team members to understand how the company runs.
Some investors set NP, BP, and Scott Kelly on too high a pedestal. That makes it hard for some to believe there may be day-to-day friction between the cast of characters. The same way some celebrity super-fans think their idols can never do wrong or ever be criticized. [Any 2010-2012 era Justin Bieber fans here? No... Well, moving on]
It shouldn't have much affect on the company's long term outcome. It is helpful when trying to anticipate how CYDY will zig or zag with day-to-day decisions based on the leaderships' personalities. This is the type of stuff that big shot stock analysts miss because they don't take the time to learn the inner workings of the company. It's emotional intelligence stuff.
Black-Ops,
How did you ever end up in the OTC market with CYDY?
Putting aside the COVID pandemonium for a moment, how has your thinking about CYDY changed over all these years being a long time shareholder?
I'm not even going to attempt to look back through your almost 800 past iHub posts! Sometimes, it takes me a good portion of night just to keep up with the days discussions.
Diversify and hedge your bets??
BlackOps had a post that ever so briefly mentioned portfolio diversity as a fleeting thought after spending time among the trees. So I chewed on his wisdom quite a bit like I always do, took a deep breath, and tried to take a critical look at the forest. [See what I did there, clever huh?]
So, on the theme of controversial posts following my BP/NP post, I have an even more uncomfortable question for those already sporting their CYDY tattoos for the upcoming shareholder meeting.
First, I'm a huge fan of CYDY. I have my big picture 5-year $1000/share conservative forecast.
And I'm trying to understand NP's strengths and weaknesses - which becomes CYDY's in a sense. NP is a super busy guy but he seems finds time to email responses to investor questions and be the company's spokesman. He has a big heart, is careful with shareholders' money, and has kept CYDY moving forward all these years.
One part of the DrBeen YouTube interview I keep thinking about is CYDY's slow or lack of progress with clinical trials.
1) It seems that NP's team is really good at 'passing the hot potato' back to the foreign health bureaucrats so no one is waiting on CYDY for paperwork. Then CYDY waits days/weeks for a response.
2) When the FDA COVID 19 trial enrollments began, it was kind of a wait and see all the new patients get enrolled. But instead during the first few weeks, enrollment was surprisingly slow.
3) And if Brazil, UK, or India wants to do a clinical trial then NP will wait for them to contact him.
Why wasn't CYDY leadership all over that like a ... well you pick your own analogy.
It seems that NP's management style is the opposite of a nagging manger who stops by your desk twice a day to see how work is progressing and how soon it can be finished. NP instead seems to innately trusts that once a task is passed along, the next step will be completed, and everything will move forward orderly. [Am I projecting? I myself fall into the later category]
I kind of want to see someone specifically be assigned as the 'nagging manager role' if its not going to be NP. Could that role have been filled be the chief medical officer Scott Kelly about a month into the clinical trials? It seems to be a possibility, but I don't think anyone really knows that answer.
[Now I'm going to put this all together.]
If one spends all their time becoming an obsessive subject matter expert on CYDY, then when does one step back to see where BCRX and HGEN are in the race?
For example, by a show of hands... [err um never-mind] did you know HGEN has about 4 weeks left for the NASDAQ uplisting process? Where's CYDY in this process? [ugh, I sound like a short] Is it also conceivable that CYDY is just a little ever so slightly behind COVID treatment competitors in a few other areas?
We all know that the first new COVID treatment to market has the real advantage. It doesn't have to be the best treatment - being just better than remdesivir is good enough. [OK, so take a deep breath.] It feels like both BCRX and HGEN are maybe just a few weeks ahead of CYDY.
So that begs the question, does it make sense in the next 6 weeks to diversify one's portfolio among the three companies who share very similar short term outcomes and upside potential?
When all three companies get FDA approval for COVID treatment, their stock prices will all shoot to $30/share or higher based on the general consensus in each of the respective investor hub stock forums.
BTW, I think I'll get a green CYDY tattoo in 3D bubble letters with little COVID mushroom numbs attached to it.
I wanted to hear others points of view, so I pointed out the NP/BP thing.
In the big scheme of things, if there is something to that observation, then it should probably stay just a very minor thing. Even superheros like Iron Man (BP) and Captain America (NP) get into arguments sometimes.
Grip It and Sip It had some good insight into NP.
"Nader has a history of power struggles with people.... he doesn’t like to be challenged."
You need to know the CEO's strengths and weaknesses. Right?
So did Upside08's post:
"Dr. Patterson is in a position that does not allow him to speak in a public forum until results are completed."
and "The FDA requires complete silence at this point." which appears to be a very hard thing for NP to do.
NP Public Speaking Skills
I was in the Toastmasters Public Speaking group for a couple of years. It teaches people how to think on their feet in front of a crowd, refine content presentations, and polish their speaking skills. Why is this background important?
I've noticed that NP is getting much much better with his video chats, interviews, investor updates, & paid promotions. It's great to see but also a little disappointing.
As an investor, I feel its getting a lot harder to read between the lines of how the updates are being delivered. NP is not sharing nearly as much emotion or anecdotal thought about every topic he talks about. He's probably burning the candle at both ends and is plain tired.
A few weeks ago when he talked about CYDY's initial reactions on the progress of the FDA COVID 19 trials, we could basically infer by reading between the lines that CYDY could definitively see the lab data difference between patients with Leronlimab vs. placebo. Some also thought they heard NP slip in a live version of the recent Proactive News Update that unfortunately patients have died during the FDA S/C trial in the placebo group; which might have been edited out later. Remember, the data is still blinded.
In the DrBeen Youtube interview, NP says nothing of the sort. Instead he says that all the data is blinded and CYDY has no way to tell what's going on with the patients in the clinical trial until it is unblinded and the analysis is complete. A definitive company party line type of answer with a complete lack of any candor or personal insight.
It seems that someone finally impressed upon NP that "Loose Lips Sink Ships". Great for CYDY but bad for the curious cat.
BP + NP Relationship
But, do you agree that there seems to be some type of awkwardness in that relationship? Something is going on and it's anyone's guess what it is.
You seem to be offering some thoughts why that relationship could have changed, just as I tried to do. I just couldn't quite generalize my observation about the BP/NP dynamic.
BP presents himself as medical lab rock star and CYDY followers love it! People with that type of 'promoter' personality always seem to invite themselves to the party. I'd expect to hear more from BP in the context of COVID 19 + CYDY. Feel free to disagree or defend BP (I think BP is a blast to listen too) - it's just my opinion.
Friction between NP and BP?
Am I the only one who sensed some behind the scene friction between NP and BP on the DrBeens live stream?
NP went out of his way to explain that BP is only doing lab testing, its CYDY's data at the end of the day, and that CYDY has many other lab testing partners. It was an odd defense/attack on a simple little question about BP.
I think NP is trying to phase out BP's 15 minutes of fame.
CYDY Patent Protection
I've looked into this a little.
New patent protection generally encompasses narrow improvements or new uses for the pharmaceutical not disclosed or suggested in the original patent.
Merck originally developed, patented, and marketed Finasteride for treatment for benign prostate enlargement under the brand name Proscar. Additional patent protection and FDA approval were sought when a new use for Finasteride – treating male pattern baldness – was identified. Finasteride for the treatment of hair loss is marketed under the brand name Propecia.
CYDY discussions noticeably similar to TSLA.
I bought TSLA at $195/share and still follow it today the same way everyone follows CYDY here - lots of DD. You can literally swap TSLA for CYDY with the headlines of the past to capture CYDY naysayers current sentiment.
TSLA wont be able to scale up production.
Motortrend: TSLAs product is just vaporware.
JPMorgan: TSLA doesnt have a shot once big auto catches up.
SEC is coming after TSLA CEO for misleading statements. (Had to throw this one in for the shorts!!)
I could go on, but I proved my point.
Just wait until CYDY is uplifted. Paid Stock Analysts will start issuing press releases making really bad CYDY predictions that are so off the mark because they only do about 15min of research.
Please click the links!! Its comical to read those old articles with TSLA at $1200/share.
Global Market Size vs. Leronlimab Treatment Disease List
I put this bit of research together for consideration.
Easy to Quantify Global Market Size
$78.3B Diabetes (type 1 and 2)
$40.4B Allergies (anaphylactic shock)
$33.0B Asthma
$30.8B Multiple Sclerosis
$25.8B HIV
$19.8B COPD
$19.8B Emphysema
$17.7B Alzheimer's
$17.6B Sjogren’s Syndrome
$13.4B Inflammatory Bowel Disease/Colitis/Crohn's Disease
$10.5B Atherosclerosis
$10.0B Atopic Dermatitis
$8.4B Parkinson's Disease
$8.3B NASH
$7.0B Pneumonia
$3.2B Neurofibromatosis
$2.9B Duchenne Muscular Dystrophy/Polymyositis (possibly other muscular dystrophies also)
$1.1B Lupus
$0.9B Dermatomyositis
$0.9B Grave’s Disease/Hashimoto’s Thyroiditis (US Only)
$0.9B Scleroderma
$0.9B Tuberculosis
$0.7B Coeliac Disease
$0.7B Psoriasis
$0.6B Guillain-Barre Syndrome
$0.6B GVHD
$0.5B Myasthenia Gravis
$0.4B Idiopathic Pulmonary Fibrosis
$0.4B Rheumatoid Arthritis
$0.4B Vasculitis
$0.2B Sarcoidosis
$0.2B Chronic Fatigue Syndrome
$0.1B Bullous diseases (Pemphigus)
$0.1B Lambert-Eaton Syndrome
===========
$356B Sub-Total
$1,316.9B COVID-19
Difficult to Quantify Market Sizes
$?.?B Chronic Kidney Disease (underlying pathologies)
$?.?B Fibromyalgia
$?.?B Cancer (20+ types)
$?.?B Hepatitis (autoimmune)
$?.?B Influenza (severe)
$?.?B Kawasaki Syndrome
$?.?B Marshall’s syndrome (PFAPA)
$?.?B Myocarditis
$?.?B Post-Myocardial Infarction Syndrome
$?.?B Rheumatic Fever
$?.?B Sickle Cell Disease (anti-inflammatory effect)
$?.?B Stroke
Today's short attack made someone big money.
Definitely CYDY raising capital for uplisting & profit taking - LOL. SEC will uncover this latest crime as the investigation continues moving forward to uncover the CEO lying about the FDA clinical trials that don't exist and the incomplete FDA submissions. /s
Is it fair to say the person/group who orchestrated today's attack will try again sometime soon once the stock price stabilizes?
Or do you think this would be a one time ninja attack? Most of the stop losses that were triggered would not be in play for a second round and some of the less experienced investors might have just become a little bit wiser.
I don't think these are questions anyone can answer.
Cydy 15% gain comparison
I love math.
$3.0/share + 15% day gain = $3.45/share
$8.7/share + 15% day gain = $10/share
$8.7/share × 375% loss = does not compute
2 active leronlimab to 1 placebo ratio
Yup, your right. I had the numbers inversed. Thanks!
Preliminary results from this patient population led to the FDA’s recent clearance for CytoDyn’s Phase 2b/3 clinical trial for 390 patients, which is randomized, placebo-controlled with 2:1 ratio (active drug to placebo ratio).
CYDY Strategy for Mexican Phase 3 Trial
When the trial goes well in Mexico, Leronlimab will likely be green-lighted for use in Mexico for COVID. But also...
It's a possibility that the Mexican Phase 3 could also be a way to hedge CYDY's risky bet and gather more data as a safety net in the unforeseen event that the FDA determines the statistical significance of patient data was not high enough in the Leronlimab arm of the phase 2b/3 severe & critically ill.
The Mexican study would be an additional pool of severe and critically ill patients that CYDY has thus far struggled to find in the USA clinical trial.
Memorandum of Understanding (MOU) to conduct a COVID-19 clinical trial with leronlimab for severe and critically ill patients, with the potential to collaborate on additional COVID-19 trials.
. . . and we believe with a small Phase 3 trial of only 25 patients, leronlimab could receive approval in Mexico very quickly. This Phase 3 trial is similar to our Phase 3 trial protocol in the U.S., with the exception of the number of patients.”
New CNN Article vs What We Know
CNN: After saving his own life with a repurposed drug, a professor reviews every drug being tried against Covid-19. Here's what he's found
So the thing is: This guy doesn't have leronlimab in his database of 'every drug being tried against Covid 19'.
How does it feel to be part of a very small group of people across the globe that know about leronlimab? We have subject matter experts right here in this forum that know more about it then people who's job it is to search for a COVID treatment.
I truly think we've developed a mastermind in this forum with everyone bring their own unique expertise.
I have a secret big picture plan too.
I choose conservative numbers to model the future stock price so it's easily defensible. Hey, this can be a tough crowd full of diverse thinkers! And that's what I value most about ALL of you.
So, now what I really believe:
The PE will be more like 50. CYDY doesn't have anywhere near the bureaucratic overhead or research expenses of Big Pharma. They are going to run a bunch of clinical trials, sure. But they are not going to R&D new experimental drug candidates with unknown outcomes. Heck, even their corporate office looks very spartan on Google StreetView! I like the focus of CYDY on a single product with very modest window dressing. The only argument for a low PE would be that the COVID pandemic is currently viewed as a one time event.
The CYDY global market share should be more like 40%. There are a few similar products that may be used as less expensive alternatives but there will be a tradeoff of more potential side effects and longer/more expensive patient COVID recovery. I'm not at all worried about market share erosion with the model set at 10% global market share for CYDY. I just think market share will be higher.
I don't want to troll you, but I also don't want to recalculate the absolutely ridiculous share price I come up with based on my beliefs. Plus, it wouldn't be a secret anymore! LOL
I'm pretty comfortable with $1.3 trillion dollar global market prediction for COVID-19 treatment.
Wow, the message thread on this forum is moving faster than the stock price!
Someone said that very few people (less than 2%) will need the drug. I think that in third world countries - you'd be close (and I modeled that). The more money someone has, the more likely they will be to seek treatment though. Also, I'm not (1) including the risk of COVID reinfection after recovery, (2) a person's immune protection lasts 12 months or less, (3) the virus significantly mutates, (4) mistreatment using Leronlimab for people with flu/cold symptoms, (5) abuse of Leronlimab by ultra-wealthy for COVID immunity (1 shot a week until the pandemic is over).
Other Data:
50% with COVID show no symptoms (and likely get reinfected)
25% with COVID have mild symptoms
25% with COVID require Hospitalization
6% with require ICU admission (in USA)
2% to 5% of cases result in death WHO
Ultimately in September 2021 and beyond, I think if someone gets COVID - they'll get Leronlimab at a CVS MinuteClinic (like Tamiflu for the flu) to minimize recovery time, reduce risk of transmission, and (politically) keep the economy going as Standard of Care in the United States.
I assumed a drug target cost of $2,000 for everyone in the income bracket >$10k. For the very poor <$10k/year, I went with a $200 drug target because our CEO has demonstrated compassionate tendencies with his use of eInds.
CYDY 2018 market pricing per patient for HIV = $24,000 during the clinical study
That's $2,000 per patient per month. Way under priced, IMO.
CytoDyn is expected to price leronlimab at a similar price to its main rival . . . which commands an exceptionally high annual list price of approximately $118,000 per patient.
That's $9,800 per patient per month! Over priced, maybe.
Leronlimab would be a Tier 6 Specialty Drug and command a premium price tag between $2,000 and $9,800 per month. A full treatment for COVID is equivalent to 1 month of HIV treatment with Leronlimab.
Think about the cost of medicine:
$25/dose of aspirin at hospital
$187/vial for generic insulin
$390/dose for generic Epipen
$700/dose Name brand Epipen
$4500 for Remdesivir
$5747 for infliximab
$6421 for Udenyca
$7238 for certolizumab
$8590 for Adalimumab
Costs and markups for Distribution, Pharmacy overhead/profit, and international licensing should be considered as ADD-ONS to the $2,000 drug treatment cost. The total patient cost should probably be in the $5k to $7k range. Insurance company costs are recovered from insurance premiums and doctor/hospital administration are separate charges from drug cost.
I don't see the supply limitation of 1.2M vials as much of a hurdle. Yea, Yea... you can't sell what you didn't make. I think the supply issue will not be hard to solve. Pay Samsung Bio to setup a couple more manufacturing lines to run simultaneously 24/7 with a healthy federal/private health grant - problem solved.
Investors of all sorts flock towards COVID related companies which seems to be the recent trend to hedge against the uncertain market conditions right now. Heck, that's how I found CYDY after doubling my modest investment in Moderna. That means a PE of 40 might not be too unreasonable. But, to be ULTRA conservative, let's use a PE of 20.
As a reference:
GlaxoSmithKline has a PE of 19.5
Merck has a PE of 19.4
AbbVie has a PE of 39.8
Sanofi has a PE of 41.96
If Cydy gets a 10% global market share (ULTRA conservative) with $110 of net revenue per share (over 2 years) with a PE of 20 then:
The stock price should be valued at $1,100/share
Again, that's assuming $2000 drug cost and ONLY 10% global market share with 50% net revenue.
