Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Peregrine's Op.Cash.Burn for Q1(7-31-16) was $9.6mm (vs. Net Loss of $12.4mm) - see http://tinyurl.com/jydtkoy . We know from 9-8-16/PR-CC that Q2(10-31-16) Revs (to be rep. ~Dec8) will "be in excess of $20mm". With Avid's GP% running around 48% and depending on how much "in excess" of $20mm revs are, it'll be interesting how close we come to Positive Cash Burn for Q2. We'll find around Dec 8th or so!
Right, GF: Dec7/Dr.Gerber/IASLC’16 & Dec8/Gray-Freimark/SanAnt.BREAST-Conf., with Q2(q/e10-31-16) Financials/CC(“Avid Revs will exceed $20mm”) around Dec 8th...
Wed/12-7-16/IASLC/WCLC: UTSW’s David.Gerber to Present addl. Biomarker Data from Ph3.SUNRISE Trial. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR…
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria http://wclc2016.iaslc.org
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
IASLC = Intl. Association for the Study of Lung Cancer - http://iaslc.org
WCLC = World Conference on Lung Cancer (IASLC)
Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
David E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, H. Roder, Joe S. Shan [VP/Clin+Reg], Rachel E. Sanborn [Providence Portland Medical Ctr]
MORE: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=126689482
= = = = = = = = = =
Thur/12-8-16: Freimark/Hutchins Poster at Annual SanAntonio-Breast-Cancer-Symposium, “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors”
Dec6-10 2016: “39th Annual San Antonio Breast Cancer Symposium”
“...presented by the Cancer Therapy & Research Center at UT Health Science Center San Antonio, AACR, and Baylor College of Medicine. The driving force behind this collaboration is the shared mission of the organizations to advance progress against breast cancer. As exciting strides are made in the field of breast cancer research & treatment, our program continues to present essential up-to-the minute info. combined with engrossing discussion for basic, translational, and clinical cancer research professionals.
SABCS'16 is designed to provide state-of-the-art info. on the experimental biology, etiology, prevention, diagnosis, and therapy of breast cancer & premalignant breast disease, to an intl. audience of academic, private physicians, and researchers. We anticipate 7,500 attendees from more than 90 countries.”
http://www.sabcs.org
https://www.sabcs.org/Program/Poster-Sessions/Poster-Session-2
TRACK: Tumor Cell & Molecular Biology: Immunology & Preclin. Immunotherapy
12-8-16 7:30–9:00am Poster Session 2
#P2-04-12: “Targeting of Phosphatidylserine by Monoclonal Antibodies Enhances the Activity of Immune Checkpoint Lag3 Targeting Antibodies in Murine Breast Tumors” - Gray MJ, Gong J, Jeff Hutchins, Bruce D. Freimark (Dir.Res/Preclin.Oncology) - Peregrine Pharmaceuticals
~Dec8: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
12-7-16/IASLC/WCLC: UTSW’s David.Gerber to Present addl. Biomarker Data from Ph3.SUNRISE Trial. As VP Joe Shan said on 10-10-16, “We expect to be able to share the emerging [SUNRISE biomarker] data over the coming months at scientific & medical conferences as the more results become available”. The Lead author (presenter) is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). The Senior author is Dr. Rachel E. Sanborn, Co-Director, Thoracic Oncology Pgm, Robert W. Franz Cancer Res. Center, Earle A. Chiles Res. Inst., Providence CC, Portland, OR...
PS: "Pre-Treatment Serum Test Based" - hmmmmm. Is this new biomarker data in fact "exosome-based PS", as measured by the new assay test (7-14-16: http://tinyurl.com/zszd4fj )??
Dec4-7 2016: “WCLC’16 - IASLC’s 17th World Conf. on Lung Cancer”, Vienna, Austria
“Medical doctors, scientists, nurses, health professionals, gov’t officials, partners from the industry, health advocacy group,s and patients will come together in order to obtain and exchange information on both state-of-the-art and advances in the management of lung cancer and other thoracic malignancies, considering both global and regional aspects.”
http://wclc2016.iaslc.org
IASLC = Intl. Association for the Study of Lung Cancer - http://iaslc.org
WCLC = World Conference on Lung Cancer (IASLC)
Pgm: http://wclc2016.iaslc.org/wp-content/uploads/2016/10/WCLC-2016-Poster-Program.pdf
Poster Session with Presenters Present (ID 472) - Track: Advanced NSCLC
12/7/16 2:30-3:45pm #P3.02c-051 - ”A Pre-Treatment Serum Test Based on Complement and IL-10 Pathways Identifies Patients Benefiting from the Addition of Bavituximab to Docetaxel”
D.E. Gerber [UTSW], J. Roder, N.L. Kallinteris, L. Horn, G. Losonczy, R. Natale, M. Tang, H. Roder, J.S. Shan, Rachel E. Sanborn [Providence Portland Medical Ctr]
= = = = = = = = = =
10-10-16/PR: ESMO’16 Topline/SUNRISE; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.
10-10-16: “Peregrine Pharmaceuticals Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at European Society for Medical Oncology (ESMO) 2016 Congress”
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Additional Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development. . .
** "With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and PgmDir. of Lung Cancer Res. at the Sarah Cannon Res. Inst. and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
** Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Additional patient sample testing and analysis is ongoing and may result in other biomarkers of importance.
** Data presented at ESMO demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approximately 30% of randomized patients) achieved a statistically significant, 5.5-month improvement (13.2 months vs. 7.7 months) in median overall survival (OS) as compared to patients in the control group with the same range of B2GP1 levels [p = 0.049; hazard ratio (HR) = 0.67].
** "We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP of Clin&Reg.Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific and medical conferences as the more results become available. It is not uncommon in the cancer field for therapeutic candidates to suffer clinical trial setbacks as researchers continue to learn more about the most appropriate patient populations for those drugs. In this landscape, biomarkers play an increasingly important role in helping identify specific patient characteristics that may impact responses to a treatment. This has been seen historically with targeted cancer treatments, as well as more recently with checkpoint inhibitors including PD-1 inhibitors. We look forward to identifying the equivalent markers for bavituximab that will help guide its clinical development.”
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125687447
- - - - - - - - - - - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
10-10-16 ESMO’16 Topline/Ph3.SUNRISE Data: Biomarker B2GPI/200-240(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
EXOSOME-BASED CANCER DETECTION & MONITORING TECHNOLOGY ("Liquid Biopsy") - Excellent Exosome info: http://www.exosome-rna.com
7-14-16: Peregrine Licenses Exosome-based technology from UTSW (Inventors: Alan Schroit/Philip Thorpe) http://tinyurl.com/zszd4fj
After 3-7-14...
10-15-15: Peregrine & AstraZeneca Expand Collab. w/Ph2/2ndLine-NSCLC Trial, Bavi+durvalumab(MEDI4736), squamous or non-squamous. http://tinyurl.com/q79bkam
9-9-15 Qtly. Conf. Call (King/Shan/Worsley/Lytle) Transcript http://tinyurl.com/ph22vdn
...CEO S.King: “The Memorial Sloan Kettering & AstraZeneca collaborations are an important part of our announced plans to expand our bavituximab clinical pgm.”
8-24-15: AstraZeneca & Peregrine Collaborate on Bavi+Durvalumab Ph1/1B Trial for “multiple solid tumors” http://tinyurl.com/owlxpsf
...Durvalumab=MEDI4736(anti-PD-L1 immune checkpoint inhibitor). AZN’s Head/I-O(Robert Iannone): “Our partnership with Peregrine provides the opportunity to explore an exciting, novel combination that could deliver important clinical benefit to patients across a range of cancers."
7-14-15 Qtly. Conf. Call (King/Shan/Hutchins/Lytle) Transcript http://tinyurl.com/nw2v5u6
...CEO S.King: “We recently entered into collaboration with investigators at Memorial Sloan Kettering Cancer Ctr to continue expanding on this important work, as well as to explore other potential applications of bavituximab and other agents that target PS-signaling pathway.”
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux
12-8-16: Freimark/Hutchins Poster at Annual SanAntonio-Breast-Cancer-Symposium, “PS-Targeting Enhances Anti-LAG3 in Murine Breast Cancer Tumors”
Dec6-10 2016: “39th Annual San Antonio Breast Cancer Symposium”
“...presented by the Cancer Therapy & Research Center at UT Health Science Center San Antonio, AACR, and Baylor College of Medicine. The driving force behind this collaboration is the shared mission of the organizations to advance progress against breast cancer. As exciting strides are made in the field of breast cancer research & treatment, our program continues to present essential up-to-the minute info. combined with engrossing discussion for basic, translational, and clinical cancer research professionals.
SABCS'16 is designed to provide state-of-the-art info. on the experimental biology, etiology, prevention, diagnosis, and therapy of breast cancer & premalignant breast disease, to an intl. audience of academic, private physicians, and researchers. We anticipate 7,500 attendees from more than 90 countries.”
http://www.sabcs.org
https://www.sabcs.org/Program/Poster-Sessions/Poster-Session-2
TRACK: Tumor Cell & Molecular Biology: Immunology & Preclin. Immunotherapy
12-8-16 7:30–9:00am Poster Session 2
#P2-04-12: “Targeting of Phosphatidylserine by Monoclonal Antibodies Enhances the Activity of Immune Checkpoint Lag3 Targeting Antibodies in Murine Breast Tumors” - Gray MJ, Gong J, Jeff Hutchins, Bruce D. Freimark (Dir.Res/Preclin.Oncology) - Peregrine Pharmaceuticals
11-14-16/PR: SITC’16, PPHM+MSKCC PS-Targeting Data Presented. Dr.Jedd.Wolchok states, “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."...
11-11-16: 
11-11-16: 
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]

11-14-16/PR: Dr. Jedd Wolchok: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
11-14-16/PR: Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
- - - - - - - - - - - - - -
11-14-16/PR: New Study Demonstrates Anti-Tumor Advantages for Combination Treatment Featuring Peregrine Pharmaceuticals' PS-Targeting Antibodies in a Preclinical Melanoma Model
* Promising Results of MSK Study Evaluating Combinations of PS-Targeting Treatment, Anti-PD-1 and Radiation in Mouse B16 Melanoma Model Presented at SITC 2016
* New Data from Second Study Conducted by Peregrine Shows Triple Combination of PS-Targeting Treatment, Anti-PD-1 and Anti-LAG3 Created Long-Term Immunity in Triple Negative Breast Cancer Model; Protected Animals when Re-Challenged with Breast Cancer Cells
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=999202
TUSTIN, Nov. 14, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the presentation of positive data from multiple new preclinical studies of the company's phosphatidylserine (PS)-targeting antibodies. Study results highlight that PS-targeting antibodies similar to bavituximab synergize with checkpoint inhibitors and radiation to improve anti-tumor activity in various animal tumor models. Importantly, the improved anti-tumor activity seen in these studies was even greater when PS-targeting therapy was a part of triple combination treatment including anti-PD-1 and another therapy. Data were presented by Peregrine scientists, as well as researchers from Memorial Sloan Kettering Cancer Center (MSK), at the Society for Immunotherapy of Cancer (SITC) 2016 Annual Meeting, which was held November 9-13, 2016 in National Harbor, MD. [ http://www.sitcancer.org/2016 ]
Initial results from Peregrine's ongoing collaboration with MSK researchers were featured in a poster presented by Sadna Budhu, PhD, at SITC 2016. A team of MSK researchers led by cancer immunotherapy thought-leaders, Taha Merghoub, PhD and Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], evaluated the effects of combining PS-targeting, anti-PD-1 and radiation therapies in the mouse B16 melanoma model. Study data showed that PS-targeting antibodies synergize with both anti-PD-1 and radiation therapy to improve anti-cancer activity. PS-targeting treatment in combination with radiation, as well as triple combination of PS-targeting treatment, anti-PD-1 and radiation, led to a reduction in tumor burden. Median survival for the triple combination treatment still had not been reached at the end of the 80-day observation period with other arms in the study showing median survival that ranged from 24-70 days.
Researchers also evaluated the impact of the PS-targeting and radiation combination treatment on the level and type of immune activity. These results demonstrated that the combination led to a change in the tumor microenvironment, shifting it from immunosuppressive in which tumors are protected to immune active in which tumors are more susceptible to treatment. Analysis of local immune responses in the tumors of the treated animals showed that the combination treatment increased the number of tumor associated macrophages and shifted the macrophage polarization from the immunosuppressive M2 type to the immune active M1 type. When systemic immune responses were analyzed following triple combination of PS-targeting treatment, anti-PD-1 and radiation, researchers also saw evidence of increased immune activity. This was illustrated by key indicators of immune activity, including increases in CD8+ T-cell activation, effector cytokine production and differentiation into effector memory cells.
"Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model," stated Dr. Jedd Wolchok. "It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
"We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model," said Dr. Taha Merghoub, PhD, co-director of the Ludwig Collaborative Laboratory at MSK. "We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
[Note: FULL SITC’16 ABSTRACT #199: http://bit.ly/2dHTEVn
...Full Poster Image: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]
A second study, conducted by Peregrine, evaluated the effects of combining PS-targeting, anti-PD-1 and anti-LAG3 therapies in the E0771 triple negative breast cancer (TNBC) model. Initial findings from this study were previously reported and demonstrated that eight of the ten (80%) animals receiving the PS-targeting, anti-PD-1 and anti-LAG3 treatment combination experienced complete tumor regressions, whereas there were no animals (0/10) in the anti-PD-1 and anti-LAG3 combination treatment arm that had a complete regression. New data presented for the first time at SITC demonstrated that the triple combination established a specific and prolonged anti-tumor immune response which protected those eight animals that achieved a complete tumor regression against a re-challenge with the same E0771 TNBC model tumor cells. This sustained anti-tumor response demonstrates the ability of the triple combination treatment to trigger immune system memory and support adaptive immune responses against reemerging disease in the E0771 TNBC model.
[Note: FULL SITC’16 ABSTRACT #213: http://bit.ly/2dpUy4C ]
...Full Poster Image: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gray.pdf ]
Further highlighting the immune impact of the PS-targeting/anti-PD-1/anti-LAG3 treatment combination were initial results of a new analysis from this study using the nCounter ® PanCancer Immune Profiling Panel from NanoString Technologies ®. Data from the analysis demonstrated that the triple combination induced a greater shift in the tumor microenvironment from immunosuppressive to immune active as compared to all other treatment groups. This was evidenced by greater increases in the activity of several critical immune activating pathways, including presentation and processing of antigens and signaling and activation of T-cells, for the triple combination as compared to all other treatments.
"It is very encouraging to see the consistent increase in anti-tumor activity triggered by triple combination treatments that combine PS-targeting agents and anti-PD-1 with other cancer treatments. By demonstrating this activity across multiple studies in multiple tumor models, we are continuing to build scientific support for the therapeutic potential of adding PS-targeting therapies in combination with other cancer treatments, including checkpoint inhibitors such as anti-PD-1," said Jeff T. Hutchins, PhD, Peregrine's VP, Preclinical Research. "As cancer research continues to explore the potential of combination treatments that marry complementary mechanisms, we are pleased to see that our efforts continue to generate data supporting the role that PS-targeting agents such as bavituximab may play in this area."
Bavituximab is an investigational monoclonal antibody that targets PS. Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. Previous studies demonstrated PS-targeting antibodies shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor responses. Peregrine continues to support and guide clinical development through the evaluation of the preclinical equivalent of bavituximab, ch1N11, in animal model studies.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes the recently announced grants awarded by the National Comprehensive Cancer Network (NCCN) to support three different clinical trials of bavituximab treatment combinations [See: http://tinyurl.com/gutgwb5 ]. These trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center with the goal of evaluating combinations of bavituximab with other checkpoint inhibitors and immune stimulatory agents. The intent behind this strategy is to focus our research and development spending to further validate bavituximab's combination potential as we seek to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
Contacts:
Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = = = = = = = = =
iHub Poster FireFOX’s Commentary on the Joint MSKCC/PPHM SITC’16 News
11-15-16: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=126566507
SUBJ: IMPORTANCE OF WOLCHOK’S REMARKS IN YESTERDAY’S PR.
The key to partnerships comes from Wolchok’s confirmation that Bavi “creates a more immune active environment in which other treatments are able to have greater anti-tumor impact.”
Look how this remark echoes and reinforces comments by Dr. Perlmutter, head of IO research at Merck, in an October ESMO article on Keytruda vs. Opdivo. Dr. Perlmutter explained that higher levels of PD-L1 confer a greater benefit from PD-1 blockers and, “It turns out PD-L1 expression is just a marker for the presence of an immune response.” Here is a quote from the article:
“So what does PD-L1 expression mean — and why do higher levels confer a greater benefit from PD-1 blockers?
"It turns out PD-L1 expression is just a marker for the presence of an immune response," explained Merck's head of research, Dr. Roger Perlmutter. "Expression of PD-L1 is increased by immune response factors."
IN ENGLISH: PD-L1 is a signal that the immune system has been trying to fight the cancer. Or, as Perlmutter puts it, "Gee, there's already an immune response going on here."
http://www.cnbc.com/2016/10/07/bristol-myers-mercks-lung-cancer-treatments-square-off-this-weekend.html
This is why Wolchok’s comment yesterday is so important when he confirmed that Bavi’s MOA is to “create a more immune active environment”:
DR. JEDD WOLCHOK STATES (11-14-16), “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact.
The really important part of the MSK poster, the part that will wake up all the BP decision makers, is not the mice survival data, but rather all the detailed work MSK did to confirm that Bavi is indeed triggering an upstream immune response, e.g. increase in CD8 T-cells, shift of macrophage from M2 to M1. MSK summarizes this “creation of a more active immune environment” in the 3rd & 4th bullet points of the Poster Conclusion. This is just what Dr. Perlmutter at Merck said was missing in the 80% of patients that don’t respond to PD-1. To quote Perlmutter, Keytruda & Opdivo work better when the clinician can say "Gee, there's already an immune response going on here."
There is now strong evidence from the MSK poster and Wolchok’s summary comments that Bavi triggers an upstream immune response. This will be a huge boost to the ongoing partnership talks. *end*
= = = = = = = = = = = = = = = = = = = = = = = = = = =
MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #199: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA
ABSTRACT: http://bit.ly/2dHTEVn POSTER PDF: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf
II. 11-12-16 11:45-1pm #210: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. ABSTRACT: http://bit.ly/2dpUh1w POSTER PDF: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gong.pdf
III. 11-11-16 12:15-1:30pm #213: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. ABSTRACT: http://bit.ly/2dpUy4C POSTER PDF: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gray.pdf
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . #199/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004; Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]

----------------
II. #210/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gong.pdf ]
----------------
III. #213/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_gray.pdf ]
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
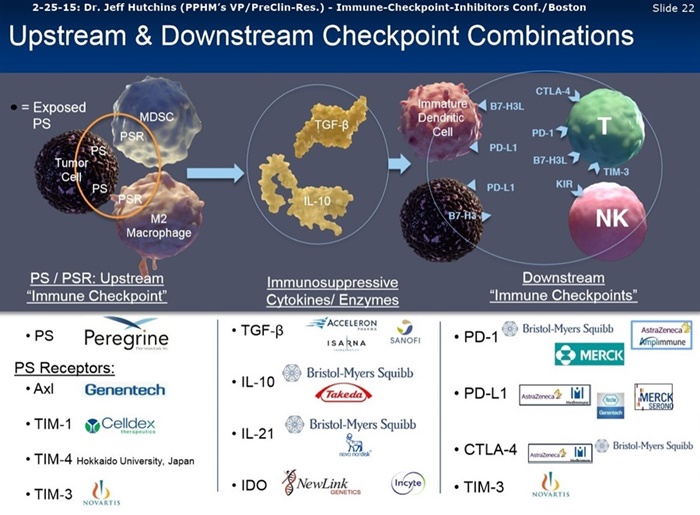
Dec7 2016: “Wistar Inst. Seminar - Combination Checkpoint Blockade”, Phila.
https://wistar.org/events/2016/12/combination-checkpoint-blockade
Lecture: Jedd D. Wolchok, MD/PhD, Chief of Melanoma & Immunotherapeutics Service at Memorial Sloan Kettering CC
= = = = = = = = = = = = = = = = = = = = = = = = = = = = = =
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray: Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, “Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
BAVI MOA 8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow http://tinyurl.com/qz64pzg
…“Expansion & Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
Conclusion Section from the Joint MSKCC/PPHM SITC’16 Poster


...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]
Dr. Jedd Wolchok: "Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."
Dr. Taha Merghoub (Co-Dir., Ludwig Collaborative Lab at MSK): "We have noted that the combination of PS-targeting treatment and radiation, as well as triple combination of PS-targeting treatment, radiation and anti-PD-1, resulted in clear advantages in anti-tumor activity in the mouse B16 melanoma model. We believe that these findings suggest the potential benefit of combining these agents to improve the outcomes of patients with cancer. With this in mind, we think this research may play an important role in designing future clinical trials of PS-targeting agents in melanoma and other cancers."
= = = = = = = = = = = = = = = = = =
SITC’16 #199/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
MORE on SITC’16: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=126542255

MOS Table from the Joint MSKCC/PPHM SITC’16 Poster

From the 11-14-16 PR: “Study data showed that PS-targeting antibodies synergize with both anti-PD-1 and radiation therapy to improve anti-cancer activity. PS-targeting treatment in combination with radiation, as well as triple combination of PS-targeting treatment, anti-PD-1 and radiation, led to a reduction in tumor burden. Median survival for the triple combination treatment still had not been reached at the end of the 80-day observation period with other arms in the study showing median survival that ranged from 24-70 days.”
...DR. JEDD WOLCHOK: “Based on these study results, we believe that the targeting of PS is having meaningful activity within the tumor microenvironment in the B16 melanoma model. It appears that this activity creates a more immune active environment in which other treatments, including radiation, are able to have a greater anti-tumor impact."

http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf
#199/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
...FULL POSTER IMAGE: http://peregrineinc.com/images/stories/pdfs/sitc_2016_budhu.pdf ]
MORE on SITC’16: http://investorshub.advfn.com/boards/read_msg.aspx?message_id=126542255
SITC’16 Today 12:15-1:30pmET: Joint MSK/WolchokLAB/PPHM AntiPS poster presentation stating that PS-Targeting “Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy” preclin.studies. Also 2 more PPHM posters at SITC’16, one today(B.Freimark, LAG3+PD1/Breast), and one tomorrow (B.Freimark, Ibrutinib+anti-PD1/TNBC)…
SITC’16 MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #P194: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #P205: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #P208: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
----------------
II. P205/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
----------------
III. P208/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."

Dec7 2016: “Wistar Inst. Seminar - Combination Checkpoint Blockade”, Phila.
https://wistar.org/events/2016/12/combination-checkpoint-blockade
Lecture: Jedd D. Wolchok, MD/PhD, Chief of Melanoma & Immunotherapeutics Service at Memorial Sloan Kettering CC
SITC’16 FRIDAY Nov.11 2016: Joint Mem.Sloan.Kett/WolchokLab + PPHM Poster – Poster Presentation Friday, Nov. 11, 2016...
SITC’16 MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #P194: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #P205: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #P208: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
----------------
II. P205/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
----------------
III. P208/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."

Dec7 2016: “Wistar Inst. Seminar - Combination Checkpoint Blockade”, Phila.
https://wistar.org/events/2016/12/combination-checkpoint-blockade
Lecture: Jedd D. Wolchok, MD/PhD, Chief of Melanoma & Immunotherapeutics Service at Memorial Sloan Kettering CC
SITC’16 MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Jedd Wolchok Lab, Memorial Sloan Kettering), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #P194: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #P205: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #P208: ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = = = = =FULL ABSTRACTS:
http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I . P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
----------------
II. P205/SITC’16/11-12-16(PPHM): “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model”
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid normally residing in the inner leaflet of the plasma membrane that becomes exposed on vascular endothelial cells and tumor cells in the tumor microenvironment, particularly in response to chemotherapy and irradiation. Binding of antibodies targeting PS induces the recruitment of immune cells and engages the immune system to destroy tumor and associated vasculature and by blocking the immunosuppressive action of PS. Recent studies have demonstrated that PS-targeting antibodies enhance the anti-tumor activity of immune checkpoint antibody blockade to CTLA-4 and PD-1 in mouse breast and melanoma tumor models. Ibrutinib [Ibrutinib=Imbruvica, dev. by Pharmacyclics & J&J, acquired by ABBVIE in 2015, proj. global sales of $1B/2016, $5B/2020] is an approved anticancer drug targeting B cell malignancies that is a selective, covalent inhibitor Bruton's tyrosine kinase (BTK) in B cell tumors. Data from recent mouse tumor studies demonstrate that ibrutinib in combination with anti-PD-1 antibody blockade inhibits growth of solid tumors, lacking BTK expression, suggesting that ibrutinib may inhibit interleukin-2 inducible T cell kinase (ITK) and promote Th1 anti-tumor responses.
METHODS:
The present study was conducted to evaluate a combination therapy including PS-targeting antibody mch1N11, ibrutinib and anti-PD-1 antibody in C57Bl/6 mice bearing triple negative E0771 breast tumors. Tumors were staged to an initial volume of ~100 mm 3 and ran- domized to treatment groups (N=10) with mch1N11 or isotype control at 10 mg/kg qw, anti-PD-1 at 2.5 mg/kg qw or ibrutinib 6 mg/kg or vehicle qd x 8. Tumor volumes were measured twice per week to determine tumor growth inhibition (TGI) relative to control treated animals. The in vitro sensitivity of E0771 tumor cells to ibrutinib was compared to the drug sensitive Jeko-1 cell line in a 72 hour growth and viability assay.
RESULTS:
The E0771 cell line is resistant in vitro to 10 mM ibrutinib. Tumor bearing mice treated with mch1N11, ibrutinib or anti-PD-1 alone had 22.2 %, 23.5 % and 32.6 % TGI respectively. The TGI for mch1N11 and ibrutinib was 30.5 %, ibrutinib and anti-PD-1 was 34.5 %, mch1N11 and anti-PD-1 was 36.1 %. The triple combination therapy had statistically greater TGI compared to control treated mice (59.9 %, p = 0.0084).
CONCLUSIONS:
Treatment of solid tumors with a combination of inhibitors that target PS, ITK and the PD-1 D-L1 axis in the tumor microenvironment provides a novel treatment for solid tumors, including triple negative breast cancer. *end*
----------------
III. P208/SITC’16/11-11-16(PPHM): ”Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors”
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm.
BACKGROUND:
Our previous work demonstrated that the addition of phosphatidylserine (PS) targeting antibodies to anti-programmed death ligand 1 (PD-1) therapy in murine triple negative breast cancers (TNBC) significantly enhanced immune system activation and tumor growth inhibition. In these studies, NanoString immune profile analysis showed that intratumoral levels of lymphocyte activation ge ne 3 (LAG3) mRNA increased in response to PS and PD-1 treatments. This suggests LAG3 may act to attenuate T cell activation in TNBC du ring I/O therapeutic regimens; however, it is unknown if PD-1 and LAG3 function cooperatively in regulating T cell anergy, and whether adding PS blocking antibodies can further enhance the effectiveness of LAG3 and/or LAG3 + PD-1 therapies.
METHODS:
Animal studies utilized C57bl/6 mice implanted with the murine TNBC model E0771. Immunoprofiling analysis was performed by flow cytometry and the NanoString nCounter PanCancer Immune Profiling Panel. Antibody treatments utilized a specific phosphatidylserine targeting antibody (ch1N11), anti-PD-1, or anti-LAG3 alone or in combination. All statistical analysis utilized the student t-test (significant with p < 0.05). RESULTS: LAG3 and PD-1 were co-expressed on T cells in E0771. Mice treated with antibodies targeting PS, PD-1, and LAG3 alone in combination with each other demonstrated that the addition of PS blocking antibodies to anti-PD-1 therapy or LAG3 had significantly greater anti- tumor activity than either single agent. Comparison of PD-1 + LAG3 combinational therapy vs. single PD-1 or LAG3 treatments showed moderately more anti-tumor activity than single treatments; however, the addition of PS blocking antibodies to either checkpoint inhibitor was as equally effective in inhibiting tumor growth as observed in the combination of LAG3 + PD-1 treatment. Further comparison of PD-1 + LAG3 vs. PS + PD-1 + LAG3 treatments demonstrated that the addition of PS blocking antibodies resulted in a significant decrease in tumor growth accompanied by complete tumor regression in a greater number of animals than observed in the PD-1 + LAG3 treatment group. FACS and NanoString immunoprofiling analysis on each treatment group showed that the addition of PS blocking antibodies to all check- point treatment groups, including the combination of PD-1 + LAG3, resulted in enhanced tumor infiltrating lymphocytes (TILs), a reduction myeloid derived suppressor cells (MDSCs), and enhanced cytokines associated with immune system activation.
CONCLUSIONS Overall, our data demonstrate that while PS, LAG3, and PD-1 therapies each have efficacy in TNBC as single agents, I/O treatments that include PS blocking antibodies offer significantly improved growth inhibition and are capable of increasing TILs compared to single and combinational treatments by T cell checkpoint targeting inhibitors alone. *end*
= = = = = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."

SITC'16 Abstracts published; here's the Joint MSKCC(Wolchok Lab)/PPHM Abstract #P194...
ABSTRACTS: http://resource-cms.springer.com/springer-cms/rest/v1/content/11027680/data/v1/Volume+4+Supplement+1+PDF+pt+2
I. P194/SITC’16/11-11-16(MSK+PPHM): “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma”
AUTHORS:
Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(Memorial Sloan Kettering SENIOR AUTHOR)
BACKGROUND:
Phosphatidylserine (PS) is a phospholipid that is exposed on the surface of apoptotic cells, some tumor cells and tumor endothelium. PS has been shown to promote anti-inflammatory and immunosuppressive signals in the tumor microenvironment. Antibodies that target PS have been shown to reactivate anti-tumor immunity by repolarizing tumor associated macrophages to a M1-like phenotype, reducing the number of MDSCs in tumors and promote the maturation of dendritic cells into functional APCs. In a B16 melanoma model, targeting PS in combination with immune checkpoint blockade has been shown to have a significantly greater anti-cancer effect than either agent alone. This combination was shown to enhance CD4+ and CD8+ T cell infiltration and activation in the tumors of treated animals. Radiation therapy is an effective focal treatment of primary solid tumors, but is less effective in treating metastatic solid tumors as a monotherapy. There is evidence that radiation induces immunogenic tumor cell death and enhances tumor-specific T cell infiltration in irradiated tumors. In addition, the abscopal effect, a phenomenon in which tumor regression occurs outside the site of radiation therapy, has been observed in both preclinical and clinical trials with the combination of radiation therapy and immunotherapy.
METHODS:
We examined the effects of combining tumor radiation therapy with an antibody that targets PS (1 N11) [Note: PGN635 (B2GPI-dep.) is Fully-Human Bavituximab=1N11=AT004 (B2GPI-depen.); Genentech studying 89Zr-PGN635 as a Tumor Imaging Agent, “indep. of cancer type”] and an immune checkpoint blockade (anti-PD-1) using the mouse B16 melanoma model. Tumor surface area and overall survival of mice were used to determine efficacy of the combinations.
RESULTS:
We examined the expression of PS on immune cells infiltrating B16 melanomas. CD11b + myeloid cells expressed the highest levels of PS on their surface whereas T cells and B16 tumor cells express little to no PS. These data suggest that targeting PS in B16 melanoma would induce a pro-inflammatory myeloid tumor microenvironment. We hypothesize that therapies that induce apoptotic cell death on tumor cells would enhance the activity of PS-targeting antibodies. We therefore examined the effects of combining a PS-targeting antibody with local tumor radiation. We found that the PS-targeting antibody synergizes with both anti-PD-1 and radiation therapy to improve anti-cancer activity and overall survival. In addition, the triple combination of the PS-targeting antibody, tumor radiation and anti-PD-1 treatment displayed even greater anti-cancer and survival benefit.
CONCLUSIONS:
This finding highlights the potential of combining these 3 agents to improve outcome in patients with advanced-stage melanoma and may inform the design of future clinical trials with PS targeting in melanoma and other cancers *end*
SITC’16 MSKCC/Wolchok-Lab & PPHM, Nov11-12, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Wolchok Lab), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #199: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering Cancer Ctr)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #210: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #213: Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."

Dec7 2016: “Wistar Inst. Seminar - Combination Checkpoint Blockade”, Phila.
https://wistar.org/events/2016/12/combination-checkpoint-blockade
Lecture: Jedd D. Wolchock, MD/PhD, Chief of Melanoma & Immunotherapeutics Service at Memorial Sloan Kettering CC
SITC’16 Abstract Titles+Times: MSKCC/Wolchok-Lab & PPHM, Nov9-13, Natl-HarborMD. 3 PS-Targeting/I-O Abstracts – note that the 1st one (#199 Nov. 11, 2016) is the joint MSKCC/PPHM one; its Senior author is Dr. Taha Merghoub (Wolchok Lab), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts
PEREGRINE/MSKCC ABSTRACTS:
I. 11-11-16 12:15-1:30pm #199: “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC (Memorial Sloan Kettering Cancer Ctr)
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. 11-12-16 11:45-1pm #210: “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-PD-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. 11-11-16 12:15-1:30pm #213: Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."

Dec7 2016: “Wistar Inst. Seminar - Combination Checkpoint Blockade”, Phila.
https://wistar.org/events/2016/12/combination-checkpoint-blockade
Lecture: Jedd D. Wolchock, MD/PhD, Chief of Melanoma & Immunotherapeutics Service at Memorial Sloan Kettering CC
AVID REVS: FY'17(May16-Apr17) guidance $50-55mm (committed B/L=$71mm), per 9-8-16 PR/CC (http://tinyurl.com/jydtkoy ).
Looking at Avid’s rev history, the 5qtrs ended 4-30-16 averaged ~$11mm/Qtr. <=those rev’s were ALL from AVID I (Frankin).
FY17Q1(qe Jul31) was $5.6mm (due to 3rd-party testing delays) and Q2(qe Oct31, rep. ~Dec8) “will exceed $20mm”, averaging ~$13mm/qtr, possibly more depending on how far Q2 “exceeds $20mm”.
13x4=52
15x4=60
17x4=68
In the 9-8-16 CC/Q&S Paul Lytle said this: “We recognized $44mm in revenues during FY16 (fye 4-30-16) and that all came from our original Franklin facility. In March this year, we commissioned our new Myford facility, which is built for late stage Phase III clinical & commercial production. We are currently going through the motions of multiple process validation runs and those runs right now are built into our financial projections of the $50-55mm, with our goal of turning those process validation runs, which is the final step before submitting something to the FDA under preapproval inspection in terms of producing commercial quantities for those clients. So this year is really a building year in terms of building those process validation runs for our clients, and then we’re hopeful that those will turn into commercial supply needs in future FY’s.” http://tinyurl.com/jydtkoy
Avid II (Myford) revenues were coming in as of 9-8-16, albeit via “mult. process validation runs” for clientS (<=note plural per PL). We don’t know when the FDA allows those to turn into Commercial Runs, but the trendline on Avid II revenue contributions to total Avid revenues is clearly UP – they’ve told us “up to $40mm per year” when Avid II is really humming.
Updated PPHM REVS-BY-QTR TABLE, now thru FY17'Q1(qe 7-31-16), per the 7-31-16 10-Q ( http://tinyurl.com/jmy77g3 ) issued 9-8-16.
• Total Revs since May’06: ($179.2mm/Avid + $24.1mm/Govt + $2.5mm/Lic.) = $205.8mm
• 9-8-16: FY'17 (May'16-Apr'17) Avid revs guidance $50-55mm (Committed B/L=$71mm).
• Deferred-Revs at 7-31-16 total $21.5mm, UP from $10.0mm at 4-30-16.
• Cust.Deposits at 7-31-16 total $21.7mm, DOWN from $24.2mm at 4-30-16.
• Inventories at 7-31-16 total $25.3mm, UP from $16.2mm at 4-30-16.
• Avid’s Gross-Profit over last 4 qtrs: $19.2mm on revs of $40.6mm (GP%=47%)
• Recall, Avid Rev$ from Gov’t DTRA Contract work (6/30/08 – 4/15/11, totaling $24.15mm), went into GOVT-REVS, not AVID-REVS, in the Financials.
Avid’s website: http://www.avidbio.com
AVID PROFITABILITY (GROSS*) BY QTR:
QTR Avid-Rev$ CostofMfg$ Gross-Profit$ GP%
FY13Q1 7-31-12 4,135,000 2,024,000 2,111,000 51%
FY13Q2 10-31-12 6,061,000 3,703,000 2,358,000 39%
FY13Q3 1-31-13 6,961,000 3,651,000 3,310,000 47%
FY13Q4 4-30-13 4,176,000 3,217,000 959,000 23%
FY14Q1 7-31-13 4,581,000 2,670,000 1,911,000 42%
FY14Q2 10-31-13 7,354,000 4,195,000 3,159,000 43%
FY14Q3 1-31-14 3,885,000 2,416,000 1,469,000 38%
FY14Q4 4-30-14 6,474,000 3,829,000 2,645,000 41%
FY15Q1 7-31-14 5,496,000 3,583,000 1,913,000 35%
FY15Q2 10-31-14 6,263,000 4,139,000 2,124,000 34%
FY15Q3 1-31-15 5,677,000 3,113,000 2,564,000 45%
FY15Q4 4-30-15 9,308,000 4,758,000 4,550,000 49%
FY16Q1 7-31-15 9,379,000 4,608,000 4,771,000 51%
FY16Q2 10-31-15 9,523,000 4,741,000 4,782,000 50%
FY16Q3 1-31-16 6,672,000 3,896,000 2,776,000 42%
FY16Q4 4-30-16 18,783,000 9,721,000 9,062,000 48%
FY17Q1 7-31-16 5,609,000 3,062,000 2,547,000 45%
FY17Q2 10-31-16 “we remain on track to gen. revs in excess of $20mm in Q2.”
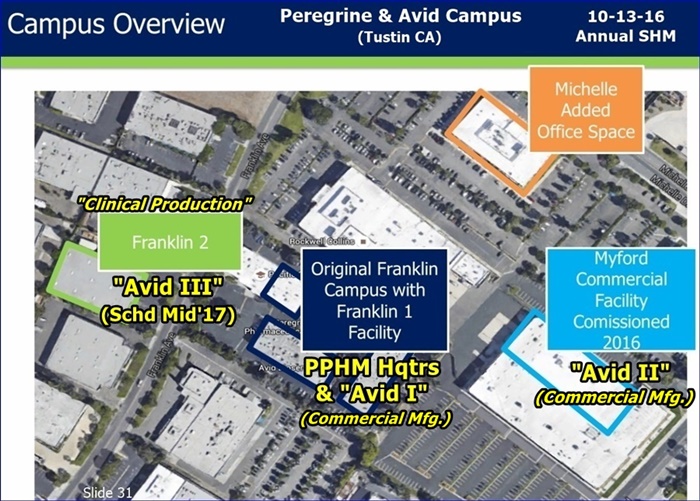
New Patent App, filed 7-12-16, pub. 10-27-16 (not yet “granted”): “Constructs Binding to Phosphatidylserine and Their Use in Disease Treatment & Imaging”. This is another “continuation” patent appl. in the PS-Targeting platform series – so complex – I have no way of telling what the “new” sections are...
U.S. Patent Application #20160311886 - Filed: 07/12/2016; Pub: 10/27/2016
“Constructs Binding to Phosphatidylserine and Their Use in Disease Treatment & Imaging”
ABSTRACT: Disclosed are new phosphatidylserine binding constructs with surprising combinations of properties, and a range of diagnostic and therapeutic conjugates thereof. The new constructs effectively bind phosphatidylserine targets in disease and enhance their destruction, and can also specifically deliver attached imaging or therapeutic agents to the disease site. Also disclosed are methods of using the new construct compositions, therapeutic conjugates and combinations thereof in tumor vasculature targeting, cancer diagnosis and treatment, and for treating viral infections and other diseases.
INVENTORS: Thorpe, Phillip E. (Dallas), Luster, Troy A. (Norfolk, MA), King, Steven W. (Tustin)
ASSIGNEES: Board of Regents, The Univ. of Texas System, Peregrine Pharmaceuticals, Inc.
http://appft1.uspto.gov/netacgi/nph-Parser?Sect1=PTO1&Sect2=HITOFF&d=PG01&p=1&u=%2Fnetahtml%2FPTO%2Fsrchnum.html&r=1&f=G&l=50&s1=%2220160311886%22.PGNR.&OS=DN/20160311886&RS=DN/20160311886
Shareholder Attendee Reports (10-13-16 ASM) – as more come in, I will delete, update, and re-post. THANKS to all attendees for sharing. This update adds FU’s by Eb0783.
WEBCAST Replay: http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
By: Eb0783 10-13-16 #275973
ASM attendees - roughly 40. 20 of us being shareholders.
FU/275979(re: Garnick?): He was a very valuable asset in the meeting.
FU/276020: From the ASM: King said (in answer to a question about B2GP1 stability, a la GJH) that B2GP1 is “relatively stable” among the patients and, although not as much is known about it compared to other proteins, there probably is no more than a +/-10% variation in a given patient.
Dr Garnick said that B2GP1 is only one (and maybe not the most important) of a number of biomarkers and they will have a number of them creating a profile. Lytle said the $$ spend on Sunrise will be finished by around end of calendar year. My take on these two statements is that they will have the analysis done and the biomarker profile will be complete. imo
Garnick shared a story about an early biomarker at Genentech with us that very few people know. Herceptin failed its first trials but one scientist identified the HER2 mutation and saw how many with HER2 lived longer. So they ran another trial which also failed. Garnick and his peers didn't believe in it but one other guy, identified a biomarker that corrolated with the ones who lived, and talked them into spending another $60mm on one more trial. Since then Herceptin has save 1000's of lives and earned $Billions of dollars. He believes in the value of biomarkers and he has been driving it for us.
As to my question of filing the BLA with what we have, [sorry CP, I did agree with your view) Dr Garnick point blank said our data is “not fileable.” We have now proof of concept and need now another trial, even a small phase II, to validate. Keep in mind however that means a smaller trial and with biomarkers involved it could be shorter than we imagine. imo
On a sidebar with just Shelley and I, Shelly Fussey said they have filed a detailed provisional patent on the Biomarkers which sets the filed date, keep it from being published/shared, and allows inexpensive additions/updates for a number of months (forget if it was 6, 9, or 12). Basically, we the public, and their competitors, will not be able to access it for 18mos. It also adds another year to the patent protection that way.
Edit: Avid III has not actually begun construction as the lease is still being negotiated. However the design, equipment models, etc is ready to begin. Some others will probably add some color because they gave 11 of us a tour of the Myford facility. I call that "shareholder friendly."
FU/276047(To HD): King told us before that ps targeting will be an astronomical value and your verbose presence here continues to confirm that for me. Thank you again for your support.
FU/276054(re: Expenses?): You are right. In fact Lytle said they have already reduced expenses by 38% and will be completely finished with Sunrise expense by end of year. Don't believe those who try to say they have not reduced.
FU/276076(Future?): LOL Loofman, You are still here for the same reason the rest of us "believers" are: The potential rewards are Astronomical (even hornet knows it as he is trying to talk us out of our PEARL every day) but it is only taking a little longer.
FU/276334: I wanted to add one more item from the Avid tour. In past Q&A during one of the latest CCs, King talked about how profitable “fill & finish” is and how he wanted to get into that part in the future. Well, part of John Haney’s presentation was a discussion on what “Fill & Finish” is and why we would want to do it (profitable due to difficulties in keeping the product pure). From his explanation, description, and excitement, my take-away is that he has the design, equipment layout, and process already to fit into the next space.
FU/276490(to ExW re: Herceptin data mining analogy): Missing the ASM made you miss this. Herceptin was NOT originally developed for HER2: data mining pointed them there. Garnick said they did a phase III after screening for HER2 and it also failed! Then one more round of "data mining" gave one scientist enough leverage to convince them to do one more. At that time they had nothing else in the pipeline. Sound familiar? With that addl. biomarker screening they ran a successful phase III and the rest is history. That is how I remember it. Some here seem to denigrate and demonize "data mining" but I see the benefit.
FU/276505(to ExW re: Herceptin analogy): I’m just telling you what I think I heard from Dr. Garnick who said, “very few people know this.” That implies to me that it is not included in any public documents. On top of that, one of the most discussed “problems” still today [there is even talk of legislation to correct it] in getting FDA approvals is that BPs often do not include “all” the information (like trial failures) in the BLAs that they file. I’m not making something up, just bringing into question rational reasons why this FDA document may not tell us about failed trials. Why would it? I may be incorrect that it was a phase III as it could have been a phase II but he said they spent $30?mm on the failed trial and this one guy convinced them (even though their committee didn't believe in it) to spend another $60mm on another trial, Phase III. ASM attendees please correct me.
FU/276539(re: Planning future trials): Yes, this is exactly what King said. The biomarker (profile) will let them filter out those who do not benefit and that will be entrance criteria for the next trial which only needs to be about 30% of the Sunrise 600 and that 30% will provide stat.sig. performance (74%?) on OS or other chosen end points. Addl. biomarkers found that can add to the profile will make it even more precise and possibly less than 200 pts could be used.
FU/276543: Brandon Cox was talking to Jeff Hutchins & Steve Worsley before the meeting started and much of that was about the aPS + aPD1 + aLAG3 data. I don’t think MSK was mentioned in that discussion (I was listening to some of it). I know he was asking about next steps for that data. I will let Brandon share that with you, if he will.
FU/277081: Jake, At the ASM Q&A King was asked is we were talking with Merck and he succinctly answered, "Yes."
FU/277180(Avid Expansion): Jake, I wish you had been there for the Avid tour so you could hear & see what is happening there. You would have gotten a great deal from it. I spent a number years doing project eng. & mgt. work myself, so I think I can see where some items can be checked off. Peregrine has been a neighbor to the tenants & landlords around them for a few decades. They are well enough known to them, have relationships with them, and have expanded a number of times in that locale over the years. I don’t think the landlords around them have any issues about their credit worthiness. They have surveyed the available sites, have leased some already for various purposes, and they know the advantages/disadvantages of each of them for the purposes they will use them for. The design for the new facility is a smaller but cookie cutter version on Avid II. John Haney also mentioned that the parking used by PPHM at Myford is considerably less than an office tenant would need and he spoke as if that was a plus for them at that location. The only hold-up (time-wise) I see right now is whether they can get the lease signed in the most desirable site or if they will relent and use one that may take a little more infrastructure build-out. It is their timeline and they will decide how much negotiation time is too much (it could be done as we type this). I might also add, imo, that if the demand for a product/service is great enough, there are a myriad of ways to get it financed, including various levels of customer input/$$. I experienced that in another industry.
By: Robert C Jonson 10-13-15 #276028
Eb, I thought I heard Rob Garrick say our trial didn’t give proof of concept, just a correlation with the Bavi + 200-240 group, and another proof of concept trial would have to be done. I asked Steve King about betabodies and he said they're still in the picture and should help out with B2GP1 problem. I also asked about earthquake provisions and he said the production labs have been designed to weather earthquakes, but the ultimate provisions are to expand our mfg. facilities to other locales, possibly to include international sites. I asked him lastly about timing to implement the RS and he said as late as possible to give other upcoming events as much time as possible to raise our pps back to the minimum required (or higher). This ASM was as good as one could be after a company has failed a phase III trial, and the NCCN preliminary data on the triple combination of Bavi, PD-L1, and LAG-3 is particularly exciting. It was particularly fun to socialize with PPHM shareholders, most of whom I knew, but some whom I met for the first time. RCJ
FU/276161: Eb, or North, or someone else who attended the ASM yesterday, please correct or expand me, but in response to a question about advancing our finding of the biomarker, Steve said he'd have to be 99% certain of the correlation first. Brandon asked if that meant a p value of .01 instead of .05 and Steve answered "yes." He then said further analysis of Sunrise data might provide addl. biomarker identification that could bring the confidence level up to 99%.
FU/276173: One person who really impressed me yesterday was John Haney, who I call Peregrine's/Avid's logistician. He conducted the first half of our tour of Avid's facilities and told us that Avid was NOT growing: it is EXPLODING! He said a major reason for this is the tremendous increase in efficiency of the manufacturing process. In the past it was only possible to obtain a few mg's of product from a batch of mix. Now, from the same amount of mix, they are obtaining 2-4 grams of product.
FU/276212: Right, JJ, another surprise for me was that we found out Avid has 10+ customers now vs the 1-2 we had heard previously!
FU/276465: Data mining may not be a preferred means of getting results, but it can be good. Rob Garnick told us at the ASM that without data mining there would be no Herceptin today, a drug that's sold $8-10bb.
FU/276485(to IFU): Rob Garnick brought up Herceptin as an analogy to how/why PPHM is analyzing the Sunrise data.
By North40000 10-14-16 #276066
The attendees at the ASM did not care how timely the meeting started---why should you? Attendees, including us, were quite satisfied talking with management and BOD members pre ASM. The meeting started at 10 or shortly thereafter.
FU/276071: Reverting to an earlier exwannabe post prior to start of ASM, ex is correct no BLA will or would be filed today, per Dr. Garnick's opinion expressed post-close of original ASM presentations...it would be "laughed out of Washington" he said. I rejoined that a lot of funny things happen in Washington these days. Dr. Garnick responded with reliance on his Herceptin experience with Genentech[DNA], and the need--in his opinion--for completion of analysis of Sunrise trial to determine existence of further biomarker data or other data that would provide concrete evidence of validity of the proof of concept data now extant today.
More on this issue when I return to East Coast next week, with access to transcripts or audio record, if any, on what was said.
FU/276075: SK: the RS split will not be immediate, answering your speculation about today or tomorrow. Rather, he said, any RS will be delayed as long as possible. Others who attended likely have already commented to same effect.
FU/276077(Worsley?): Worsely was there. I had no discussions with him, and have no idea what others may have discussed with him before or after ASM.
FU/276086(To Eb0783): We enjoyed visiting with you again, as well as the Avid facilities tour.
FU/276088: As further proxies were collected from attendees at ASM, and were yet to be counted, the actual vote count on proposal 1 was not announced during the meeting itself, only the result. Results themselves were obviously apparent from data and votes on hand during the ASM.
FU/276204: I noted that PPHM had obtained a new stock transfer agent some time ago, and asked whether PPHM had monitored who, or what entity, was buying, selling or accumulating shares of PPHM; I further noted that share price had been cabined in a lid of some sort day to day for some time. PL, CFO, responded to my question and comment. As CP has noted previously on other occasions, and consistent with PL's response, the stock transfer agent is not necessarily aware of certain trades itself known by a certain acronym. As to trades known by the agent, PPHM is aware of them as well as any accumulation > 5%. He reminded us of shareholder protection plan in place that enables certain activities by the BOD. I will not detail those here - they can be posted by others as necessary or desirable.
FU/276323(re: AZN?): The analysis of the Sunrise data in regards to biomarkers is critical to the design of all future trials. Peregrine knows it and so does AZN. AZN is not gone. The future of biomarkers is the ability to zero in on target patients to greatly improve the odds of success while helping patients survive. Any person who attended the ASM would have heard that message from King, Garnick, Shan and others. They have valuable data that could lead to a lighted pathway. According to King, that analysis should be completed by yr-end. The Herceptin story was used as an example by Garnick to illustrate a drug developed from data after failing 2 clinical trials. Garnick also commented that the MOA of Herceptin is still unknown, but as science advances we gain the ability to identify and target specific biomarkers that improve patient outcomes.
FU/276472(re: Avid II & III in Calif): Training purposes for later duplicate facilities to built elsewhere: Dr Garnick's words, not mine.
FU/276598: Dr. Garnick spoke with at least some PPHM ASM attendees, including my wife and me, after the meeting was adjourned: 1) Herceptin experience as he[Dr. Garnick] related it...DNA had only prelim. data that targeting Her2 worked or gave good results in some patients... [DNA] needed further addl. data from another, prospective trial---that trial needed to include the pre-identified Her2 patients. Only a very few, including the original project leader and perhaps Garnick himself, thought THAT addl. trial would produce info that would be useful or that Her2 was in fact useful as a target; top mgt did not. But DNA had nothing more in its pipeline at the time. Finally, or ultimately, top mgt of DNA was convinced to invest what Garnick said was $60mm for that addl. trial. 2) As a further side-note, CJ told me directly that, following the ASM, he and the BOD would convene an extended meeting until what he expected to be early evening hours to discuss various matters/issues....what those might be he did not say, nor did I ask.
FU/276514: "BJ", one of our guides on the tour of the Avid facilities on the afternoon of 10/13/2016, was a neuro-scientist himself with PPHM before switching over to Avid, for how long I did not ask. He originally was from the bay area of SanFran. John Haney, who began our tour, is a mechanical engineer with a great deal of experience in property location & acquisition for purposes intended. Like SK, John had a great many slides shown to us that he employs to demonstrate Avid facilities to customers & visitors. I have not looked on Avid web-site to see what slide decks may be there.
By Djohn 10-13-16 #276058
Expanding head count? Of course. Avid is expanding like wildfire. FDA, biosimilars. 12 customers, $20mm in revenue turned down in 2017 because of capacity constraints.
FU/276225: I would say The GM of biotech in 10 years. From what I saw yesterday PPHM has perfected the manufacturing assemble line of biotech. It will be replicated all over the world IMO. Nice meeting all of you at the ASM and Avid tour!!
By JJ1223 10-14-16 #276197
John Haney was well versed and impressive with his presentation on future Avid growth. I was surprised that Peregrine had to turn down $20MM of business in the past year due to capacity. He made it easy to see Avid's vision of growth and why SK see's total corporate profitability within the next 21mos. Also impressed by his comment of 1-2 new customer visits per month, and their enthusiasm to "get" started. No one I spoke with was happy with the Sunrise results, but the Biomarker data could surprise. We will know by year-end. I am not happy with delay, but I walked away with a clear picture and understanding that my long term investment is probably more secure than at any time in the past. For those who believe Steve King was not positive, I would have to disagree.
FU/276237(to HD): Not true at all. Fact is they tried to lease addl. in the Myford building, but the landlord had made improvements and wanted an unreasonable price. The addl. space in that building remains vacant today. Avid leased across the street at a much better price/sf and a more accommodating landlord. BTW, 6 new clients have already booked for the new facility expected to be operational mid 2017. The search for addl. space is underway at present, with a vision of major expansion planned. "Explosion of business", and "perfect storm" in terms of the business environment were the themes we heard during the Avid tour. Avid now employs 220 people (growing 6 just this week) and the facilities are massive and very impressive. If anyone gets the opportunity to hear John Harney speak about Avid, or tour the facility, you should do so. Sounds like Biosimiliars will play a big role in the future growth of the business at Avid. IMO, Pete Gagnon plays a very important role here.
FU/276487(to IFU re: Herceptin data mining analogy) Perhaps you missed Garnick's point since you did not hear it directly from him. He stressed the importance of analyzing the data by saying that Herceptin failed a PI and PII trial dramatically. No one at Genetech wanted to proceed any further. It was dead until one scientist noticed something in the data and convinced others to take a look. The comparison to Herceptin is not the point. In a Biomarker world, it is critical to understanding.
By Hawkfan1 10-15-16 #276290
I also think that John Haney (Avid Sr.Proj.Mgr, prev: Genentech, Pfizer) is a very impressive gentleman, and his enthusiasm is contagious. It's too bad that he couldn't finish the rest of the Avid tour with us. The tour that Paul and I got with him last year was much better than this year's. I haven't seen anyone else post about this, so I thought I'd add a few details about Avid's current state and future plans. Anyone who was at the ASM, please feel free to make additions or correct any errors. Avid has leased 2 buildings since last year. The one that they are calling the Michelle building (it's a half a block northeast on Michelle Drive) is all office space. They have also leased another building on the other side of Franklin Ave. (I think they called it Franklin II, if I'm not mistaken). They leased this one to be the home of Avid III, or a water purification plant when they couldn't work out a deal with landlord of the Myford building. He had decided to improve his building in the hopes of enticing a Google type business at three times the rate that we are paying to lease Avid II. He added all kinds of amenities (including an outdoor seating area with a fire pit!) to attract creative programmer types, but a warehouse district is not the most desirable location for this type of business, and it has been vacant for the past year. John has been negotiating with the owner, and while he stopped short of saying that he is becoming more reasonable, he noted that he has investors too, and John is hopeful that they can come to some kind of suitable arrangement. He would like to lease the other half of the Myford building to build a water purification plant to support Avid. Currently, Avid buys the water that they use in the cell growing process in barrels. I've forgotten how many gallons John said that they use, but each run costs about $150,000.00 just for the water that they buy. If he is successful in negotiating a lease of the Myford building, then the Franklin II building would be used for warehouse space. The Myford building has a limited amount, but not enough. John also talked a little about some pf the challenges of building the water purification plant. Interestingly, in it's finished state, the water is so pure that it can leach the ions out of the stainless steel pipes that carry it, and they must keep the water circulating to prevent this. Before John had to leave, he also made the comment that he is already looking for his next building, which I assume would be the home of Avid IV, since he has already made other plans for the other 2 buildings.
FU/276506(to ExW re: Herceptin data-mining analogy): My purely uneducated guess as to why we didn't data mine the phII is that there were insufficient numbers in that study to achieve statsig. in any subgroups that they might have found, so it wouldn't have meant much. Just a guess… Regarding the Herceptin trial, I have in my ASM notes that the PhI showed safety, but the phII didn't show much, if any, benefit at all. Most of the powers that be at Genentech (including Garnick) were for dropping the drug, but as eb points out, they had nothing else in the pipeline. Then Dr. Cowen re-analyzed the data, and found that a subgroup of patients that over-expressed Her2 had benefited. He developed a test for Her2 expression, and convinced Genentech to run a $60mm PhIII that pre-selected for the over-expression of Her2, and the rest is history. Hope that clarifies some of the confusion.
By Copper888 10-18-16 #276580
Thanks to all attendees of the ASM for your great recaps and it was a great pleasure to meet you and spend some time together! Everyone did a great job of recapping the discussion portion of the meeting, so I don't have much to add except for a few observations and verifications.
1. B2GP1 discussion - I wrote in my notes several times the phrase "layering of biomarker data" and "layering selection criteria" They have the B2GP1 data but are looking for all the data to come in to further "explain the effects" of the stat sig population. New Data is coming in "every day" and should be complete in the next 3 months. They stated that they want to be 99% sure before moving forward with any confirmatory trials, whether that is a registrational PHII or Phase 3 trial.
2. Avid - John Haney indeed described the business as exploding and attributed the growth to what he called the "perfect storm" of market factors aiding growth including:
i) FDA now has a clear path for approval of biosimilars
ii) Production Tech has now improved to the point that, in his words, "little Avid" can produce a worldwide supply of a given drug and compete with the largest producers of biologics.
He also mentioned that the speed to enter into production agreements used to be a year long courtship. Now companies are begging to get on the schedule and using early test runs just to get a relationship with Avid and hopefully try to jump the line.
They expressed their interest in building new facilities - mentioning places like Europe, Singapore, and other parts of the US taking advantage of local tax incentives to build and to decentralize for risk mitigation (earthquakes and such).
Now comes my personal gut feeling about what I heard...let the chastising begin :)
I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there. Well, that is it...I tried to furiously write direct quotes as much as I could. For those in attendance, Please feel free to correct or add to anything written here… Hope that this is of some value!
WEBCAST Replay (10-13-16 ASM): http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
Slideshow PDF (45 Slides): http://files.shareholder.com/downloads/PPHM/2879585795x0x911689/C8068896-AF8B-4CAA-A08C-FB483775B2AD/20161013-_PPHM_Corporate_Overview_-_ASM_FINAL.pdf
EXCERPTS:

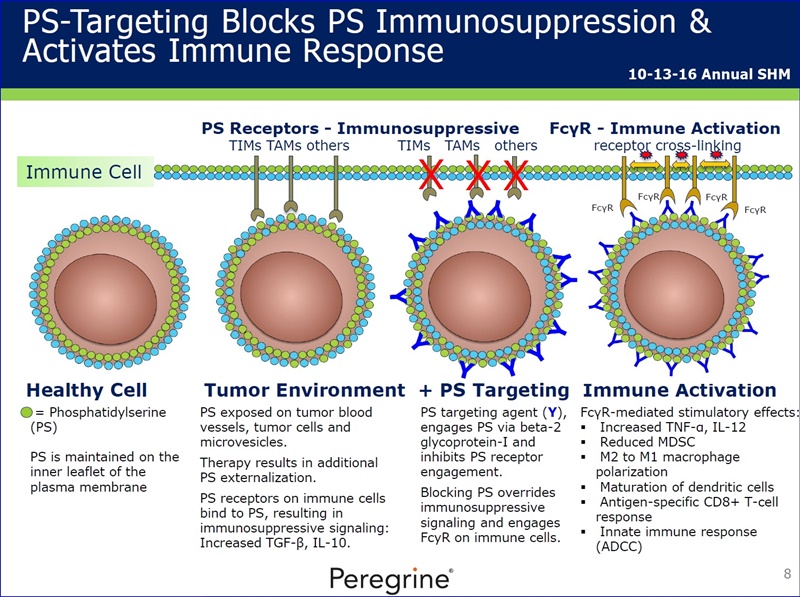


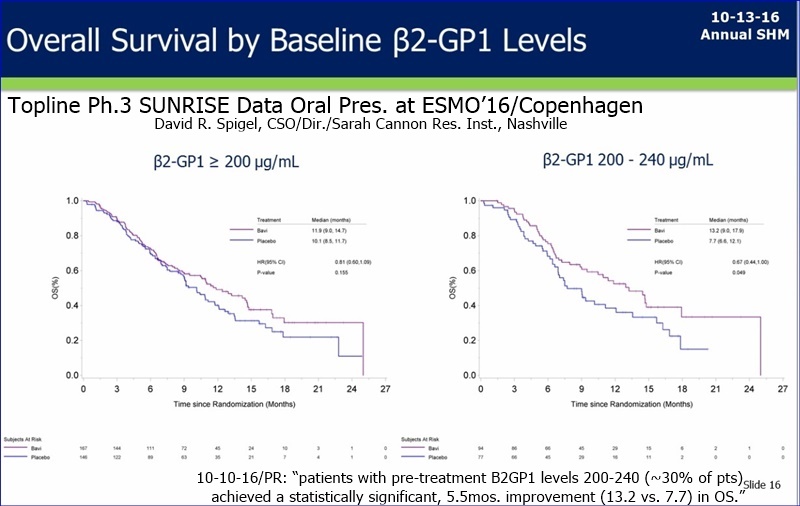
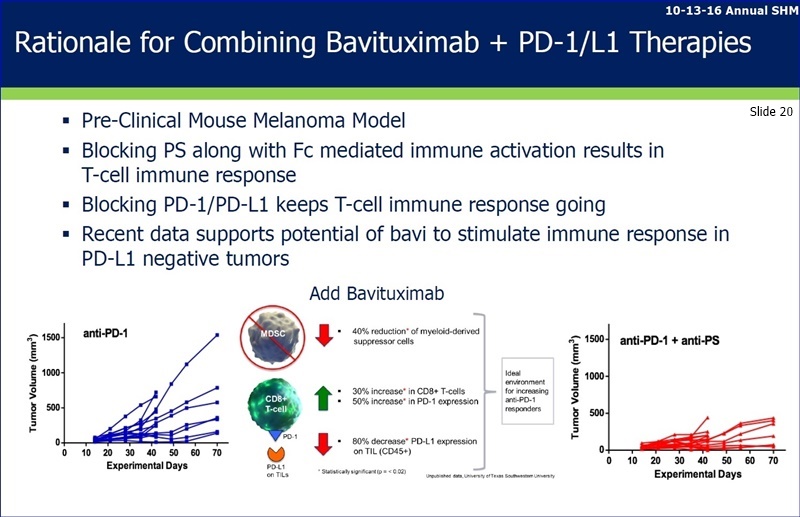

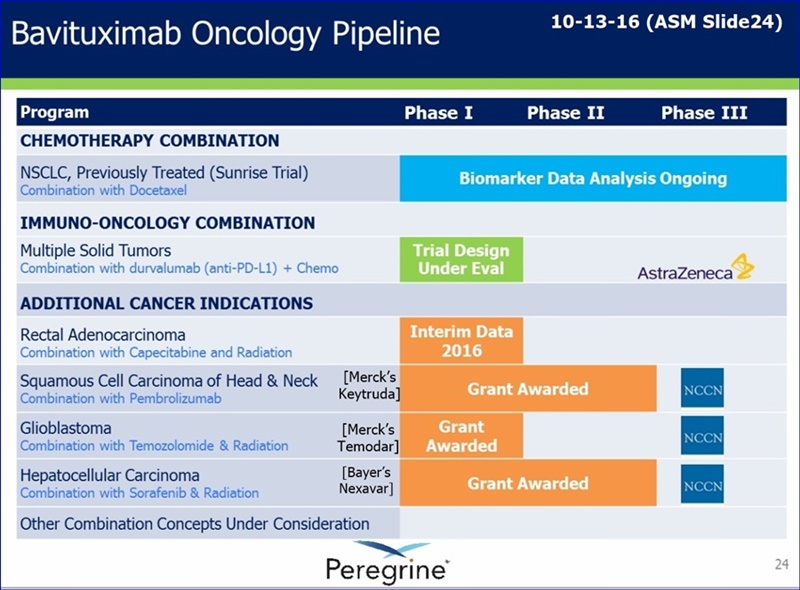


----------
BAVI MOA 10-22-16: Duke’s Herbert K. Lyerly (w/PPHM) poster on AntiPS/TNBC data at AACR’s Tumor Immunotherapy Conf./Boston http://tinyurl.com/zzryfok
...”Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
Nov9-13 2016: (SITC’16) Society for Immunotherapy of Cancer 31st Annual Meeting, Natl-Harbor MD http://tinyurl.com/j4tw5p9
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
...I. “PS Targeting Antibody in Combo w/Checkpoint Blockade & Tumor Rad. Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC
...II. “Antibody Targeting of PS Enhances the Anti-Tumor Responses of Ibrutinib & anti-Pd-1 Therapy in a Mouse Triple Neg. Breast Tumor Model” - Jian Gong, PhD, PPHM
...III. Monoclonal Antibodies Targeting PS Enhance Combinational Activity of the the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD, PPHM
10-24-16: Duke’s Herbert.Lyerly presents AntiPS/TNBC data at AACR’s “Tumor Immunology and Immunotherapy Conference” in Boston. We first learned of the DUKE/PPHM Cancer Collab. in Bavi/TNBC back in Nov. 2015 at SITC'15 (See 11-9-15: Duke’s Dr. Herbert K. Lyerly http://tinyurl.com/pbof95w )...
10-24-16: Preclinical Research Demonstrates Peregrine Pharmaceuticals' PS-Targeting Antibodies Enhance the Anti-Tumor Activity of PD-L1 Checkpoint Inhibitors in Model of Triple Negative Breast Cancer (TNBC)
* Combination of PS-Targeting Antibody and Anti-PD-L1 Therapy, With or Without Chemotherapy, Led to Greater Anti-Tumor Activity Than Single Agent Treatment or Dual Combinations With Chemotherapy
* Additional Experiments Demonstrate that PS Expression is Upregulated on Cancer Cells Following Chemotherapy, Radiation or Photodynamic Therapy
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=994994
TUSTIN, Oct. 24, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced the presentation of preclinical study data demonstrating that phosphatidylserine (PS)-targeting antibodies similar to bavituximab are able to enhance the anti-tumor activity of anti-PD-L1 therapy in a model of triple negative breast cancer (TNBC). Data showed that a combination of anti-PS and anti-PD-L1 therapies, with or without paclitaxel, led to greater anti-tumor responses than any of the treatments administered as single agents or dual treatment combinations with paclitaxel, in the well-characterized E0771 murine model of TNBC. Study results were presented by researchers from Duke University Medical Center at the American Association for Cancer Research's [AACR’s] Tumor Immunology and Immunotherapy Conference held October 20-23, 2016 in Boston, MA.
[ http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=92
10-22-16 POSTER SESSION B:
#B36 Modulating The Tumor Microenvironment to Enhance Cancer Immunotherapy by Inducing Phosphatidylserine Expression on the Tumor Surface”
Kensuke Kaneko 1, Takuya Osada 1, Bruce D. Freimark 2, Herbert Kim Lyerly ** (Duke Univ.) 1
1=Duke University, Durham, NC
2=Peregrine Pharmaceuticals, Inc.
**Dr. Herbert Kim Lyerly: https://immunology.duke.edu/people/herbert-kim-lyerly-md (George Barth Geller Professor, Duke Univ. MC)
In addition to evaluating the anti-tumor activity of the various treatment combinations, researchers also examined the impact of various traditional cancer therapies on PS expression in cancer cells. Study results confirmed that levels of PS expression were upregulated in E0771 and 4T1 TNBC cells following treatment with chemotherapy, radiation or photodynamic therapy. Photodynamic therapy also was shown to increase PS expression on tumor cells.
"These study results provide the latest support for the belief that PS-targeting therapies can enhance the anti-tumor activity of checkpoint inhibitors such as anti-PD-L1 therapy in the treatment of TNBC. Just last month, we announced results from another preclinical study [9-27-16/AACR-CRI: http://tinyurl.com/zy9yv78 ] in TNBC demonstrating that 80% of animals receiving the triple combination of anti-PS, anti-PD-1 and anti-LAG3 therapies experienced complete tumor regressions, whereas there were no animals in the anti-PD-1 and anti-LAG3 combination treatment arm that had a complete regression," said Jeff T. Hutchins, Ph.D., Peregrine's VP, Preclinical Research. "Additionally, these latest study findings related to increased PS expression on the surface of tumor cells following traditional cancer treatments demonstrate important activity within the tumor microenvironment that offers rationale for the potential of anti-PS agents in combatting cancer. We plan to continue to work with our collaborators at Duke University Medical Center to further study the therapeutic potential of PS-targeting agents in combination with checkpoint inhibitors like anti-PD-L1 and conventional therapies that augment immunotherapy mechanisms."
Bavituximab is an investigational monoclonal antibody that targets PS. Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. Previous studies demonstrated PS-targeting antibodies shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor responses. Peregrine evaluates the preclinical equivalent of bavituximab, ch1N11, in animal model studies to guide clinical development.
Peregrine's clinical development strategy for bavituximab currently focuses on small, early-stage, proof-of-concept trials evaluating the drug in combination with other cancer treatments. This approach includes the recently announced grants by the National Comprehensive Cancer Network (NCCN) to support three different clinical trials of bavituximab treatment combinations [See: http://tinyurl.com/gutgwb5 ]. Those trials will evaluate novel bavituximab combinations in glioblastoma, head and neck cancer, and hepatocellular carcinoma including an immunotherapy combination. Additionally, Peregrine continues to advance its pre-clinical collaboration with Memorial Sloan Kettering Cancer Center with the goal of evaluating combinations of bavituximab with other checkpoint inhibitors and immune stimulatory agents [see MSKCC Wolchok Lab SITC'16 Nov9-13 2016: http://tinyurl.com/j4tw5p9 ]. The intent behind this strategy is to focus our research and development spending to further validate bavituximab's combination potential as we seek to advance the program though a pharmaceutical or biotechnology partner.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. ( http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = = =
BAVI MOA 9-27-16 AACR-CRI/Dr. Michael Gray: Preclin. Triple-Combo Bavi+PD1+LAG3 TNBC data in TNBC (80% Compl. Regression, Stat-Sig. Incr. in Key Tumor Fighting Immune Cells) http://tinyurl.com/zy9yv78
BAVI MOA 5-11-16 Breast Cancer Res. article, B.Freimark/CW.Hughes-et-al, “PS-Targeting/Bavi Combo w/Anti-PD1/PDL1 in Triple.Neg-MBC” http://tinyurl.com/zxu882y
...”our observations demonstrate that including PS-targeting antibodies such as bavituximab can enhance the anti-tumor activity of anti-PD-1/PD-L1 treatments, not only by increasing TIL responses but also by inhibiting cytokines stimulated by single-agent anti-PD-1 therapy that serve to suppress the immune response & promote tumor progression.”
BAVI MOA 4-20-16/AACR'16 & 4-4-16/Cancer-Immunology-Res.(AACR) article - preclin. data: Bavi combo w/anti-PD-1/anti-CTLA-4 “induces a shift in tumor microenvironment from immunosuppressive to immune active” http://tinyurl.com/jyox458
BAVI MOA 1-18-16: CEO Steve King explains PPHM's direct PS-Targeting advantage vs. the “individual-receptors” PS-binding approach of others like: Axl Mer TIM-3 RAGE Tyro3 GAS6 CD300a BAI1 MFG-E8 etc. http://tinyurl.com/h2h87mc
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, ”Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
–----
11-9-15: BREAST CANCER - Researchers from Duke University and Peregrine evaluated the combination of ch1N11 (preclinical bavituximab equivalent) and anti-PD-1 therapy versus anti-PD-1 stand-alone therapy in well-characterized murine breast cancers, including the triple negative breast cancer (TNBC) model E0771. Study data showed that the combination therapy significantly enhanced overall survival (p=0.0016) and was capable of mediating complete tumor regressions in a greater number of subjects compared to single agent treatments (60% vs. 20%). Data also demonstrated that animals receiving combination treatment had significant increases in tumor associated indicators of immune system activation, including CD45+, CD8+ and CD3+ T-cells. Importantly, the combination treatment led to a prolonged anti-tumor immune response which protected the animals against a re-challenge with the same tumor. This sustained anti-tumor response suggests the potential of the combination therapy to trigger immune system memory and support adaptive immune responses against reemerging disease in breast cancers. All study animals experienced no signs of adverse effects following repeated doses of all therapeutic agents.
BAVI MOA 8-26-15: Jeff Hutchins(VP/PreClin-Res) ImVacS’15 Talk – 29pg. Slideshow http://tinyurl.com/qz64pzg
…“Expansion & Activation of T Cells via the Targeting of the Immunosuppressive Ligand PS: Combination Strategy with Conventional, Targeted, and Checkpoint Inhibitor Therapy”
5-31-15: ASCO’15 Roundtable (webcast), “Raising the Immuno-Oncology Bar” - 7 panel members, incl. 3 Sloan Kettering researchers http://tinyurl.com/qxu4w2x
• PGN632 (B2GPI-indep.) is the Duke-PPHM-HIV candidate=11.31=AT005; also being studied by PPHM+LSU vs. Ocular Herpes (Acute HSV-1 Keratitis), see http://tinyurl.com/cax9a4p
A zoom-in of the PS-receptor AXL section in Slide9 from the 10-13-16 ASM.

Raymond Birge(Rutgers) and Rolf Brekken(UTSW, PPHM SAB) and others co-author 2-2016 article, “Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” - many references to the Axl PS receptor, like, “Studies with the related TAM receptor Axl provides further mechanistic insight into how PS receptors transmit immune inhibitory signals. Unlike Mertk that is constitutively expressed on macrophages and DCs, under basal conditions the expression of Axl in DCs is low, but is significantly upregulated as a consequence of TLR engagement to resolve and break inflammation in anticipation of the end of an inflammatory cycle… Mertk & Axl are expressed on infiltrating macrophages and DCs, but also frequently expressed on the tumor cells themselves... This combined effect of PS & PS receptors may provide a ‘perfect storm’ that accentuates immune escape.”
http://www.nature.com/cdd/journal/v23/n6/full/cdd201611a.html
= = = = = = = = = = =

.
.

Shareholder Attendee Reports (10-13-16 ASM): http://tinyurl.com/zrv38fl
Shareholder Attendee Reports (10-13-16 ASM) – as more come in, I will delete, update, and re-post. THANKS to all attendees for sharing. This update adds Copper888 and FU’s by Eb0783 and North40000.
WEBCAST Replay: http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
By: Eb0783 10-13-16 #275973
ASM attendees - roughly 40. 20 of us being shareholders.
FU/275979(re: Garnick?): He was a very valuable asset in the meeting.
FU/276020: From the ASM: King said (in answer to a question about B2GP1 stability, a la GJH) that B2GP1 is “relatively stable” among the patients and, although not as much is known about it compared to other proteins, there probably is no more than a +/-10% variation in a given patient.
Dr Garnick said that B2GP1 is only one (and maybe not the most important) of a number of biomarkers and they will have a number of them creating a profile. Lytle said the $$ spend on Sunrise will be finished by around end of calendar year. My take on these two statements is that they will have the analysis done and the biomarker profile will be complete. imo
Garnick shared a story about an early biomarker at Genentech with us that very few people know. Herceptin failed its first trials but one scientist identified the HER2 mutation and saw how many with HER2 lived longer. So they ran another trial which also failed. Garnick and his peers didn't believe in it but one other guy, identified a biomarker that corrolated with the ones who lived, and talked them into spending another $60mm on one more trial. Since then Herceptin has save 1000's of lives and earned $Billions of dollars. He believes in the value of biomarkers and he has been driving it for us.
As to my question of filing the BLA with what we have, [sorry CP, I did agree with your view) Dr Garnick point blank said our data is “not fileable.” We have now proof of concept and need now another trial, even a small phase II, to validate. Keep in mind however that means a smaller trial and with biomarkers involved it could be shorter than we imagine. imo
On a sidebar with just Shelley and I, Shelly Fussey said they have filed a detailed provisional patent on the Biomarkers which sets the filed date, keep it from being published/shared, and allows inexpensive additions/updates for a number of months (forget if it was 6, 9, or 12). Basically, we the public, and their competitors, will not be able to access it for 18mos. It also adds another year to the patent protection that way.
Edit: Avid III has not actually begun construction as the lease is still being negotiated. However the design, equipment models, etc is ready to begin. Some others will probably add some color because they gave 11 of us a tour of the Myford facility. I call that "shareholder friendly."
FU/276047(To HD): King told us before that ps targeting will be an astronomical value and your verbose presence here continues to confirm that for me. Thank you again for your support.
FU/276054(re: Expenses?): You are right. In fact Lytle said they have already reduced expenses by 38% and will be completely finished with Sunrise expense by end of year. Don't believe those who try to say they have not reduced.
FU/276076(Future?): LOL Loofman, You are still here for the same reason the rest of us "believers" are: The potential rewards are Astronomical (even hornet knows it as he is trying to talk us out of our PEARL every day) but it is only taking a little longer.
FU/276334: I wanted to add one more item from the Avid tour. In past Q&A during one of the latest CCs, King talked about how profitable “fill & finish” is and how he wanted to get into that part in the future. Well, part of John Haney’s presentation was a discussion on what “Fill & Finish” is and why we would want to do it (profitable due to difficulties in keeping the product pure). From his explanation, description, and excitement, my take-away is that he has the design, equipment layout, and process already to fit into the next space.
FU/276490(to ExW re: Herceptin data mining analogy): Missing the ASM made you miss this. Herceptin was NOT originally developed for HER2: data mining pointed them there. Garnick said they did a phase III after screening for HER2 and it also failed! Then one more round of "data mining" gave one scientist enough leverage to convince them to do one more. At that time they had nothing else in the pipeline. Sound familiar? With that addl. biomarker screening they ran a successful phase III and the rest is history. That is how I remember it. Some here seem to denigrate and demonize "data mining" but I see the benefit.
FU/276505(to ExW re: Herceptin analogy): I’m just telling you what I think I heard from Dr. Garnick who said, “very few people know this.” That implies to me that it is not included in any public documents. On top of that, one of the most discussed “problems” still today [there is even talk of legislation to correct it] in getting FDA approvals is that BPs often do not include “all” the information (like trial failures) in the BLAs that they file. I’m not making something up, just bringing into question rational reasons why this FDA document may not tell us about failed trials. Why would it? I may be incorrect that it was a phase III as it could have been a phase II but he said they spent $30?mm on the failed trial and this one guy convinced them (even though their committee didn't believe in it) to spend another $60mm on another trial, Phase III. ASM attendees please correct me.
FU/276539(re: Planning future trials): Yes, this is exactly what King said. The biomarker (profile) will let them filter out those who do not benefit and that will be entrance criteria for the next trial which only needs to be about 30% of the Sunrise 600 and that 30% will provide stat.sig. performance (74%?) on OS or other chosen end points. Addl. biomarkers found that can add to the profile will make it even more precise and possibly less than 200 pts could be used.
FU/276543: Brandon Cox was talking to Jeff Hutchins & Steve Worsley before the meeting started and much of that was about the aPS + aPD1 + aLAG3 data. I don’t think MSK was mentioned in that discussion (I was listening to some of it). I know he was asking about next steps for that data. I will let Brandon share that with you, if he will.
By: Robert C Jonson 10-13-15 #276028
Eb, I thought I heard Rob Garrick say our trial didn’t give proof of concept, just a correlation with the Bavi + 200-240 group, and another proof of concept trial would have to be done. I asked Steve King about betabodies and he said they're still in the picture and should help out with B2GP1 problem. I also asked about earthquake provisions and he said the production labs have been designed to weather earthquakes, but the ultimate provisions are to expand our mfg. facilities to other locales, possibly to include international sites. I asked him lastly about timing to implement the RS and he said as late as possible to give other upcoming events as much time as possible to raise our pps back to the minimum required (or higher). This ASM was as good as one could be after a company has failed a phase III trial, and the NCCN preliminary data on the triple combination of Bavi, PD-L1, and LAG-3 is particularly exciting. It was particularly fun to socialize with PPHM shareholders, most of whom I knew, but some whom I met for the first time. RCJ
FU/276161: Eb, or North, or someone else who attended the ASM yesterday, please correct or expand me, but in response to a question about advancing our finding of the biomarker, Steve said he'd have to be 99% certain of the correlation first. Brandon asked if that meant a p value of .01 instead of .05 and Steve answered "yes." He then said further analysis of Sunrise data might provide addl. biomarker identification that could bring the confidence level up to 99%.
FU/276173: One person who really impressed me yesterday was John Haney, who I call Peregrine's/Avid's logistician. He conducted the first half of our tour of Avid's facilities and told us that Avid was NOT growing: it is EXPLODING! He said a major reason for this is the tremendous increase in efficiency of the manufacturing process. In the past it was only possible to obtain a few mg's of product from a batch of mix. Now, from the same amount of mix, they are obtaining 2-4 grams of product.
FU/276212: Right, JJ, another surprise for me was that we found out Avid has 10+ customers now vs the 1-2 we had heard previously!
FU/276465: Data mining may not be a preferred means of getting results, but it can be good. Rob Garnick told us at the ASM that without data mining there would be no Herceptin today, a drug that's sold $8-10bb.
FU/276485(to IFU): Rob Garnick brought up Herceptin as an analogy to how/why PPHM is analyzing the Sunrise data.
By North40000 10-14-16 #276066
The attendees at the ASM did not care how timely the meeting started---why should you? Attendees, including us, were quite satisfied talking with management and BOD members pre ASM. The meeting started at 10 or shortly thereafter.
FU/276071: Reverting to an earlier exwannabe post prior to start of ASM, ex is correct no BLA will or would be filed today, per Dr. Garnick's opinion expressed post-close of original ASM presentations...it would be "laughed out of Washington" he said. I rejoined that a lot of funny things happen in Washington these days. Dr. Garnick responded with reliance on his Herceptin experience with Genentech[DNA], and the need--in his opinion--for completion of analysis of Sunrise trial to determine existence of further biomarker data or other data that would provide concrete evidence of validity of the proof of concept data now extant today.
More on this issue when I return to East Coast next week, with access to transcripts or audio record, if any, on what was said.
FU/276075: SK: the RS split will not be immediate, answering your speculation about today or tomorrow. Rather, he said, any RS will be delayed as long as possible. Others who attended likely have already commented to same effect.
FU/276077(Worsley?): Worsely was there. I had no discussions with him, and have no idea what others may have discussed with him before or after ASM.
FU/276086(To Eb0783): We enjoyed visiting with you again, as well as the Avid facilities tour.
FU/276088: As further proxies were collected from attendees at ASM, and were yet to be counted, the actual vote count on proposal 1 was not announced during the meeting itself, only the result. Results themselves were obviously apparent from data and votes on hand during the ASM.
FU/276204: I noted that PPHM had obtained a new stock transfer agent some time ago, and asked whether PPHM had monitored who, or what entity, was buying, selling or accumulating shares of PPHM; I further noted that share price had been cabined in a lid of some sort day to day for some time. PL, CFO, responded to my question and comment. As CP has noted previously on other occasions, and consistent with PL's response, the stock transfer agent is not necessarily aware of certain trades itself known by a certain acronym. As to trades known by the agent, PPHM is aware of them as well as any accumulation > 5%. He reminded us of shareholder protection plan in place that enables certain activities by the BOD. I will not detail those here - they can be posted by others as necessary or desirable.
FU/276323(re: AZN?): The analysis of the Sunrise data in regards to biomarkers is critical to the design of all future trials. Peregrine knows it and so does AZN. AZN is not gone. The future of biomarkers is the ability to zero in on target patients to greatly improve the odds of success while helping patients survive. Any person who attended the ASM would have heard that message from King, Garnick, Shan and others. They have valuable data that could lead to a lighted pathway. According to King, that analysis should be completed by yr-end. The Herceptin story was used as an example by Garnick to illustrate a drug developed from data after failing 2 clinical trials. Garnick also commented that the MOA of Herceptin is still unknown, but as science advances we gain the ability to identify and target specific biomarkers that improve patient outcomes.
FU/276472(re: Avid II & III in Calif): Training purposes for later duplicate facilities to built elsewhere: Dr Garnick's words, not mine.
FU/276598: Dr. Garnick spoke with at least some PPHM ASM attendees, including my wife and me, after the meeting was adjourned: 1) Herceptin experience as he[Dr. Garnick] related it...DNA had only prelim. data that targeting Her2 worked or gave good results in some patients... [DNA] needed further addl. data from another, prospective trial---that trial needed to include the pre-identified Her2 patients. Only a very few, including the original project leader and perhaps Garnick himself, thought THAT addl. trial would produce info that would be useful or that Her2 was in fact useful as a target; top mgt did not. But DNA had nothing more in its pipeline at the time. Finally, or ultimately, top mgt of DNA was convinced to invest what Garnick said was $60mm for that addl. trial. 2) As a further side-note, CJ told me directly that, following the ASM, he and the BOD would convene an extended meeting until what he expected to be early evening hours to discuss various matters/issues....what those might be he did not say, nor did I ask.
FU/276514: "BJ", one of our guides on the tour of the Avid facilities on the afternoon of 10/13/2016, was a neuro-scientist himself with PPHM before switching over to Avid, for how long I did not ask. He originally was from the bay area of SanFran. John Haney, who began our tour, is a mechanical engineer with a great deal of experience in property location & acquisition for purposes intended. Like SK, John had a great many slides shown to us that he employs to demonstrate Avid facilities to customers & visitors. I have not looked on Avid web-site to see what slide decks may be there.
By Djohn 10-13-16 #276058
Expanding head count? Of course. Avid is expanding like wildfire. FDA, biosimilars. 12 customers, $20mm in revenue turned down in 2017 because of capacity constraints.
FU/276225: I would say The GM of biotech in 10 years. From what I saw yesterday PPHM has perfected the manufacturing assemble line of biotech. It will be replicated all over the world IMO. Nice meeting all of you at the ASM and Avid tour!!
By JJ1223 10-14-16 #276197
John Haney was well versed and impressive with his presentation on future Avid growth. I was surprised that Peregrine had to turn down $20MM of business in the past year due to capacity. He made it easy to see Avid's vision of growth and why SK see's total corporate profitability within the next 21mos. Also impressed by his comment of 1-2 new customer visits per month, and their enthusiasm to "get" started. No one I spoke with was happy with the Sunrise results, but the Biomarker data could surprise. We will know by year-end. I am not happy with delay, but I walked away with a clear picture and understanding that my long term investment is probably more secure than at any time in the past. For those who believe Steve King was not positive, I would have to disagree.
FU/276237(to HD): Not true at all. Fact is they tried to lease addl. in the Myford building, but the landlord had made improvements and wanted an unreasonable price. The addl. space in that building remains vacant today. Avid leased across the street at a much better price/sf and a more accommodating landlord. BTW, 6 new clients have already booked for the new facility expected to be operational mid 2017. The search for addl. space is underway at present, with a vision of major expansion planned. "Explosion of business", and "perfect storm" in terms of the business environment were the themes we heard during the Avid tour. Avid now employs 220 people (growing 6 just this week) and the facilities are massive and very impressive. If anyone gets the opportunity to hear John Harney speak about Avid, or tour the facility, you should do so. Sounds like Biosimiliars will play a big role in the future growth of the business at Avid. IMO, Pete Gagnon plays a very important role here.
FU/276487(to IFU re: Herceptin data mining analogy) Perhaps you missed Garnick's point since you did not hear it directly from him. He stressed the importance of analyzing the data by saying that Herceptin failed a PI and PII trial dramatically. No one at Genetech wanted to proceed any further. It was dead until one scientist noticed something in the data and convinced others to take a look. The comparison to Herceptin is not the point. In a Biomarker world, it is critical to understanding.
By Hawkfan1 10-15-16 #276290
I also think that John Haney (Avid Sr.Proj.Mgr, prev: Genentech, Pfizer) is a very impressive gentleman, and his enthusiasm is contagious. It's too bad that he couldn't finish the rest of the Avid tour with us. The tour that Paul and I got with him last year was much better than this year's. I haven't seen anyone else post about this, so I thought I'd add a few details about Avid's current state and future plans. Anyone who was at the ASM, please feel free to make additions or correct any errors. Avid has leased 2 buildings since last year. The one that they are calling the Michelle building (it's a half a block northeast on Michelle Drive) is all office space. They have also leased another building on the other side of Franklin Ave. (I think they called it Franklin II, if I'm not mistaken). They leased this one to be the home of Avid III, or a water purification plant when they couldn't work out a deal with landlord of the Myford building. He had decided to improve his building in the hopes of enticing a Google type business at three times the rate that we are paying to lease Avid II. He added all kinds of amenities (including an outdoor seating area with a fire pit!) to attract creative programmer types, but a warehouse district is not the most desirable location for this type of business, and it has been vacant for the past year. John has been negotiating with the owner, and while he stopped short of saying that he is becoming more reasonable, he noted that he has investors too, and John is hopeful that they can come to some kind of suitable arrangement. He would like to lease the other half of the Myford building to build a water purification plant to support Avid. Currently, Avid buys the water that they use in the cell growing process in barrels. I've forgotten how many gallons John said that they use, but each run costs about $150,000.00 just for the water that they buy. If he is successful in negotiating a lease of the Myford building, then the Franklin II building would be used for warehouse space. The Myford building has a limited amount, but not enough. John also talked a little about some pf the challenges of building the water purification plant. Interestingly, in it's finished state, the water is so pure that it can leach the ions out of the stainless steel pipes that carry it, and they must keep the water circulating to prevent this. Before John had to leave, he also made the comment that he is already looking for his next building, which I assume would be the home of Avid IV, since he has already made other plans for the other 2 buildings.

FU/276506(to ExW re: Herceptin data-mining analogy): My purely uneducated guess as to why we didn't data mine the phII is that there were insufficient numbers in that study to achieve statsig. in any subgroups that they might have found, so it wouldn't have meant much. Just a guess… Regarding the Herceptin trial, I have in my ASM notes that the PhI showed safety, but the phII didn't show much, if any, benefit at all. Most of the powers that be at Genentech (including Garnick) were for dropping the drug, but as eb points out, they had nothing else in the pipeline. Then Dr. Cowen re-analyzed the data, and found that a subgroup of patients that over-expressed Her2 had benefited. He developed a test for Her2 expression, and convinced Genentech to run a $60mm PhIII that pre-selected for the over-expression of Her2, and the rest is history. Hope that clarifies some of the confusion.
By Copper888 10-18-16 #276580
Thanks to all attendees of the ASM for your great recaps and it was a great pleasure to meet you and spend some time together! Everyone did a great job of recapping the discussion portion of the meeting, so I don't have much to add except for a few observations and verifications.
1. B2GP1 discussion - I wrote in my notes several times the phrase "layering of biomarker data" and "layering selection criteria" They have the B2GP1 data but are looking for all the data to come in to further "explain the effects" of the stat sig population. New Data is coming in "every day" and should be complete in the next 3 months. They stated that they want to be 99% sure before moving forward with any confirmatory trials, whether that is a registrational PHII or Phase 3 trial.
2. Avid - John Haney indeed described the business as exploding and attributed the growth to what he called the "perfect storm" of market factors aiding growth including:
i) FDA now has a clear path for approval of biosimilars
ii) Production Tech has now improved to the point that, in his words, "little Avid" can produce a worldwide supply of a given drug and compete with the largest producers of biologics.
He also mentioned that the speed to enter into production agreements used to be a year long courtship. Now companies are begging to get on the schedule and using early test runs just to get a relationship with Avid and hopefully try to jump the line.
They expressed their interest in building new facilities - mentioning places like Europe, Singapore, and other parts of the US taking advantage of local tax incentives to build and to decentralize for risk mitigation (earthquakes and such).
Now comes my personal gut feeling about what I heard...let the chastising begin :)
I think there is a shift on how the company execs and BOD view the business. As mentioned by multiple posters, SK said that they are still looking to hit a "homerun" with Bavi. But I think that they are now doing that in a framework of risk avoidance, and profitability as their primary goals. With every initiative mentioned, SK would talk about partnering in the next sentence. Exosome testing with a partner; potential of exploring the utility of Beta Bodies - would "advance aggressively with a partner"; If Sunrise data warrants a small study to confirm, they would "partner the next step", etc. I think that for good or bad...the new company directive is the march toward profitability. He also said that the company is worth multiples of its current market cap and that they want to delay the RS as much as they can. "I am focused on getting the Share price over a dollar" He mentioned that there may be many events between now and April that may get us there. Well, that is it...I tried to furiously write direct quotes as much as I could. For those in attendance, Please feel free to correct or add to anything written here… Hope that this is of some value!
WEBCAST Replay (10-13-16 ASM): http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
Slideshow PDF (45 Slides): http://files.shareholder.com/downloads/PPHM/2879585795x0x911689/C8068896-AF8B-4CAA-A08C-FB483775B2AD/20161013-_PPHM_Corporate_Overview_-_ASM_FINAL.pdf
EXCERPTS:










Cash Burn Rate for Q1(q/e 7-31-16) rep. 9-8-16: $9,607,000
OPER. CASH BURNS* BY QTR (FROM THE 10-Q/K’S):
FY16Q1 7-31-15 12,306,000 (from 10Q pg.25)
FY16Q2 10-31-15 11,701,000 (Q1+Q2: 24,007,000 10Q pg.26)
FY16Q3 1-31-16 15,086,000 (Q1+Q2+Q3: 39,093,000 10Q pg.27)
FY16Q4 4-30-16 10,112,000 (FY'16: 49,205,000 10K pg.39)
FY17Q1 7-31-16 9,607,000 (from 7-31-16 10Q pg.22 http://tinyurl.com/jmy77g3 )
9-8-16/PR: “Avid's current mfg. revenue backlog is $71 million… The company remains on track to generate revenue in excess of $20mm in Q2FY17 (q/e 10-31-16 rep. ~12-8-16)” http://tinyurl.com/jydtkoy (Cash 7-31-16=$44.2mm)
• Avid’s Gross-Profit over last 4 qtrs(thru 7-31-16): $19.2mm on revs of $40.6mm (GP%=47%)
- - - - - - - - - - - - - - - - -
Latest 10K 4-30-16 iss. 7-14-16 http://tinyurl.com/zgognwz PR: http://tinyurl.com/h8eqtg5 (Cash 4-30-16=$61.4mm)
Latest 10Q 7-31-16 iss. 9-8-16 http://tinyurl.com/jmy77g3 PR: http://tinyurl.com/jydtkoy (Cash 7-31-16=$44.2mm)
ALL SEC filings for PPHM: http://tinyurl.com/6d4jw8
*The 10-Q’s define OPER.BURN as, ”Net cash used in operating activities before chgs. in operating assets & liabilities”.
The 7-21-2001 10Q explains OP.BURN:
“RESULTS OF OPERATIONS. Before we discuss the Company's total expenses (cash & non-cash expenses), we would like to discuss the Company's operational burn rate (cash expenses used in operations, net of interest and other income) for q/e July 31, 2001 compared to the same period in the prior year. The operational burn rate is calculated by taking the net income (loss) from operations and subtracting all non-cash items, such as the recognition of deferred license revenue, depreciation and amortization and stock-based compensation expense.”
Shareholder Attendee Reports (10-13-16 ASM) – as more come in, I will delete, update, and re-post. THANKS to all attendees for sharing. This update adds Hawkfan1, and also FU’s by Eb0784, North40000, and JJ1223.
WEBCAST Replay: http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
By: Eb0783 10-13-16 #275973
ASM attendees - roughly 40. 20 of us being shareholders.
FU/275979(re: Garnick?): He was a very valuable asset in the meeting.
FU/276020: From the ASM: King said (in answer to a question about B2GP1 stability, a la GJH) that B2GP1 is “relatively stable” among the patients and, although not as much is known about it compared to other proteins, there probably is no more than a +/-10% variation in a given patient.
Dr Garnick said that B2GP1 is only one (and maybe not the most important) of a number of biomarkers and they will have a number of them creating a profile. Lytle said the $$ spend on Sunrise will be finished by around end of calendar year. My take on these two statements is that they will have the analysis done and the biomarker profile will be complete. imo
Garnick shared a story about an early biomarker at Genentech with us that very few people know. Herceptin failed its first trials but one scientist identified the HER2 mutation and saw how many with HER2 lived longer. So they ran another trial which also failed. Garnick and his peers didn't believe in it but one other guy, identified a biomarker that corrolated with the ones who lived, and talked them into spending another $60 million on one more trial. Since then Herceptin has save 1000's of lives and earned $Billions of dollars. He believes in the value of biomarkers and he has been driving it for us.
As to my question of filing the BLA with what we have, [sorry CP, I did agree with your view) Dr Garnick point blank said our data is “not fileable.” We have now proof of concept and need now another trial, even a small phase II, to validate. Keep in mind however that means a smaller trial and with biomarkers involved it could be shorter than we imagine. imo
On a sidebar with just Shelley and I, Shelly Fussey said they have filed a detailed provisional patent on the Biomarkers which sets the filed date, keep it from being published/shared, and allows inexpensive additions/updates for a number of months (forget if it was 6, 9, or 12). Basically, we the public, and their competitors, will not be able to access it for 18 months. It also adds another year to the patent protection that way.
Edit: Avid III has not actually begun construction as the lease is still being negotiated. However the design, equipment models, etc is ready to begin. Some others will probably add some color because they gave 11 of us a tour of the Myford facility. I call that "shareholder friendly."
FU/276047(To HD): King told us before that ps targeting will be an astronomical value and your verbose presence here continues to confirm that for me. Thank you again for your support.
FU/276054(re: Expenses?): You are right. In fact Lytle said they have already reduced expenses by 38% and will be completely finished with Sunrise expense by end of year. Don't believe those who try to say they have not reduced.
FU/276076(Future?): LOL Loofman, You are still here for the same reason the rest of us "believers" are: The potential rewards are Astronomical (even hornet knows it as he is trying to talk us out of our PEARL every day) but it is only taking a little longer.
FU/276334: I wanted to add one more item from the Avid tour. In past Q&A during one of the latest CCs, King talked about how profitable “fill & finish” is and how he wanted to get into that part in the future. Well, part of John Haney’s presentation was a discussion on what “Fill & Finish” is and why we would want to do it (profitable due to difficulties in keeping the product pure). From his explanation, description, and excitement, my take-away is that he has the design, equipment layout, and process already to fit into the next space.
By: Robert C Jonson 10-13-15 #276028
Eb, I thought I heard Rob Garrick say our trial didn’t give proof of concept, just a correlation with the Bavi + 200-240 group, and another proof of concept trial would have to be done. I asked Steve King about betabodies and he said they're still in the picture and should help out with B2GP1 problem. I also asked about earthquake provisions and he said the production labs have been designed to weather earthquakes, but the ultimate provisions are to expand our manufacturing facilities to other locales, possibly to include international sites. I asked him lastly about timing to implement the RS and he said as late as possible to give other upcoming events as much time as possible to raise our pps back to the minimum required ( or higher). This ASM was as good as one could be after a company has failed a phase III trial, and the NCCN preliminary data on the triple combination of Bavi, PD-L1, and LAG-3 is particularly exciting. It was particularly fun to socialize with PPHM shareholders, most of whom I knew, but some whom I met for the first time. RCJ
FU/276161: Eb, or North, or someone else who attended the ASM yesterday, please correct or expand me, but in response to a question about advancing our finding of the biomarker, Steve said he'd have to be 99% certain of the correlation first. Brandon asked if that meant a p value of .01 instead of .05 and Steve answered "yes." He then said further analysis of Sunrise data might provide addl. biomarker identification that could bring the confidence level up to 99%.
FU/276173: One person who really impressed me yesterday was John Haney, who I call Peregrine's/Avid's logistician. He conducted the first half of our tour of Avid's facilities and told us that Avid was NOT growing: it is EXPLODING! He said a major reason for this is the tremendous increase in efficiency of the manufacturing process. In the past it was only possible to obtain a few mg's of product from a batch of mix. Now, from the same amount of mix, they are obtaining 2-4 grams of product.
FU/276212: Right, JJ, another surprise for me was that we found out Avid has 10+ customers now vs the 1-2 we had heard previously!
By North40000 10-14-16 #276066
The attendees at the ASM did not care how timely the meeting started---why should you? Attendees, including us, were quite satisfied talking with management and BOD members pre ASM. The meeting started at 10 or shortly thereafter.
FU/276071: Reverting to an earlier exwannabe post prior to start of ASM, ex is correct no BLA will or would be filed today, per Dr. Garnick's opinion expressed post-close of original ASM presentations...it would be "laughed out of Washington" he said. I rejoined that a lot of funny things happen in Washington these days. Dr. Garnick responded with reliance on his Herceptin experience with Genentech[DNA], and the need--in his opinion--for completion of analysis of Sunrise trial to determine existence of further biomarker data or other data that would provide concrete evidence of validity of the proof of concept data now extant today.
More on this issue when I return to East Coast next week, with access to transcripts or audio record, if any, on what was said.
FU/276075: SK: the RS split will not be immediate, answering your speculation about today or tomorrow. Rather, he said, any RS will be delayed as long as possible. Others who attended likely have already commented to same effect.
FU/276077(Worsley?): Worsely was there. I had no discussions with him, and have no idea what others may have discussed with him before or after ASM.
FU/276086(To Eb0783): We enjoyed visiting with you again, as well as the Avid facilities tour.
FU/276088: As further proxies were collected from attendees at ASM, and were yet to be counted, the actual vote count on proposal 1 was not announced during the meeting itself, only the result. Results themselves were obviously apparent from data and votes on hand during the ASM.
FU/276204: I noted that PPHM had obtained a new stock transfer agent some time ago, and asked whether PPHM had monitored who, or what entity, was buying, selling or accumulating shares of PPHM; I further noted that share price had been cabined in a lid of some sort day to day for some time. PL, CFO, responded to my question and comment. As CP has noted previously on other occasions, and consistent with PL's response, the stock transfer agent is not necessarily aware of certain trades itself known by a certain acronym. As to trades known by the agent, PPHM is aware of them as well as any accumulation > 5%. He reminded us of shareholder protection plan in place that enables certain activities by the BOD. I will not detail those here - they can be posted by others as necessary or desirable.
FU/276323(re: AZN?): The analysis of the Sunrise data in regards to biomarkers is critical to the design of all future trials. Peregrine knows it and so does AZN. AZN is not gone. The future of biomarkers is the ability to zero in on target patients to greatly improve the odds of success while helping patients survive. Any person who attended the ASM would have heard that message from King, Garnick, Shan and others. They have valuable data that could lead to a lighted pathway. According to King, that analysis should be completed by year end. The Herceptin story was used as an example by Garnick to illustrate a drug developed from data after failing two clinical trials. Garnick also commented that the MOA of Herceptin is still unknown, but as science advances we gain the ability to identify and target specific biomarkers that improve patient outcomes.
By Djohn 10-13-16 #276058
Expanding head count? Of course. Avid is expanding like wildfire. FDA, biosimilars. 12 customers, $20mm in revenue turned down in 2017 because of capacity constraints.
FU/276225: I would say The GM of biotech in 10 years. From what I saw yesterday PPHM has perfected the manufacturing assemble line of biotech. It will be replicated all over the world IMO. Nice meeting all of you at the ASM and Avid tour!!
By JJ1223 10-14-16 #276197
John Haney was well versed and impressive with his presentation on future Avid growth. I was surprised that Peregrine had to turn down $20MM of business in the past year due to capacity. He made it easy to see Avid's vision of growth and why SK see's total corporate profitability within the next 21mos. Also impressed by his comment of 1-2 new customer visits per month, and their enthusiasm to "get" started. No one I spoke with was happy with the Sunrise results, but the Biomarker data could surprise. We will know by year-end. I am not happy with delay, but I walked away with a clear picture and understanding that my long term investment is probably more secure than at any time in the past. For those who believe Steve King was not positive, I would have to disagree.
FU/276237(to HD): Not true at all. Fact is they tried to lease addl. in the Myford building, but the landlord had made improvements and wanted an unreasonable price. The addl. space in that building remains vacant today. Avid leased across the street at a much better price/sf and a more accommodating landlord. BTW, 6 new clients have already booked for the new facility expected to be operational mid 2017. The search for addl. space is underway at present, with a vision of major expansion planned. "Explosion of business", and "perfect storm" in terms of the business environment were the themes we heard during the Avid tour. Avid now employs 220 people (growing 6 just this week) and the facilities are massive and very impressive. If anyone gets the opportunity to hear John Harney speak about Avid, or tour the facility, you should do so. Sounds like Biosimiliars will play a big role in the future growth of the business at Avid. IMO, Pete Gagnon plays a very important role here.
By Hawkfan1 10-15-16 #276290
I also think that John Haney (Avid Sr.Proj.Mgr, prev: Genentech, Pfizer) is a very impressive gentleman, and his enthusiasm is contagious. It's too bad that he couldn't finish the rest of the Avid tour with us. The tour that Paul and I got with him last year was much better than this year's. I haven't seen anyone else post about this, so I thought I'd add a few details about Avid's current state and future plans. Anyone who was at the ASM, please feel free to make additions or correct any errors. Avid has leased 2 buildings since last year. The one that they are calling the Michelle building (it's a half a block northeast on Michelle Drive) is all office space. They have also leased another building on the other side of Franklin Ave. (I think they called it Franklin II, if I'm not mistaken). They leased this one to be the home of Avid III, or a water purification plant when they couldn't work out a deal with landlord of the Myford building. He had decided to improve his building in the hopes of enticing a Google type business at three times the rate that we are paying to lease Avid II. He added all kinds of amenities (including an outdoor seating area with a fire pit!) to attract creative programmer types, but a warehouse district is not the most desirable location for this type of business, and it has been vacant for the past year. John has been negotiating with the owner, and while he stopped short of saying that he is becoming more reasonable, he noted that he has investors too, and John is hopeful that they can come to some kind of suitable arrangement. He would like to lease the other half of the Myford building to build a water purification plant to support Avid. Currently, Avid buys the water that they use in the cell growing process in barrels. I've forgotten how many gallons John said that they use, but each run costs about $150,000.00 just for the water that they buy. If he is successful in negotiating a lease of the Myford building, then the Franklin II building would be used for warehouse space. The Myford building has a limited amount, but not enough. John also talked a little about some pf the challenges of building the water purification plant. Interestingly, in it's finished state, the water is so pure that it can leach the ions out of the stainless steel pipes that carry it, and they must keep the water circulating to prevent this. Before John had to leave, he also made the comment that he is already looking for his next building, which I assume would be the home of Avid IV, since he has already made other plans for the other 2 buildings.

WEBCAST Replay (10-13-16 ASM): http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
Slideshow PDF (45 Slides): http://files.shareholder.com/downloads/PPHM/2879585795x0x911689/C8068896-AF8B-4CAA-A08C-FB483775B2AD/20161013-_PPHM_Corporate_Overview_-_ASM_FINAL.pdf
EXCERPTS:








ASM Webcast Link (10amPT) – will start in a few minutes after everybody settles in and meeting called to order...
WEBCAST: http://edge.media-server.com/m/p/afyt8u2w (45 Slides)
From:
http://ir.peregrineinc.com/events.cfm
Chg. the 6000 to 30000 - that was a hypothetical number to show the progression to the general population.
Not what I was saying, but that's OK, KU.
30% of pts in the SUNRISE Trial (~200pts) who were positive for B2GPI(200-240) showed Stat.Sig. Improvement in Overall Survival, from 7.7mos to 13.2mos, ~70% longer lives.
~600 pts treated in Ph.3 SUNRISE 2ndLine NSCLC trial: Doce v. Bavi+Doce.
~600 pts' "pre-specified" biomarker samples were tested.
~200 (~30%) tested positive for B2GPI(200-240).
Those 200 received Stat.Sig. Improvement in Overall Survival (7.7=>13.2mos, ie, lived +70% longer)
Let’s say somebody decided to run SUNRISE II, and screen 6,000pts instead of 600.
This time, include ONLY those who test positive for B2GPI(200-240).
~1800 (30%) pts would be selected for SUNRISE II (if follows orig. SUNRISE I actual data)
900 would get Doce ONLY, 900 would get Bavi+Doce.
At the end of SUNRISE II (and if survival follows orig. SUNRISE I actual data), the 900 that rec’d BAVI+DOC would benefit from an Stat.Sig. Improvement in OS – their Median Survival would be 13.2mos vs. 7.7mos. for the 900 in the Doce ONLY arm.
And on and on: 60,000 Pt. Trial, and ultimately the entire 2ndLine NSCLC patient universe. All extrapolated from actual Stat.Sig. data from Peregrine's SUNRISE I Ph3 Trial.
= = = = = = = = = = = = =
10-10-16/PR: ESMO’16 Topline/SUNRISE Data; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
“The study protocol pre-specified the collection of thousands of patient samples for exploratory analyses over a wide range of possible biomarkers, including pre-treatment levels of beta-2 glycoprotein-1 (B2GP1). Data presented at ESMO demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approximately 30% of randomized patients) achieved a statistically significant, 5.5-month improvement (13.2 months vs. 7.7 months) in median overall survival (OS) as compared to patients in the control group with the same range of B2GP1 levels [p = 0.049; hazard ratio (HR) = 0.67].”
From FTM's 6-2012 post, the MEAN OS of a bunch of Ph3's for DOCE-Only in 2ndLine NSCLC was 7.4mos. vs. SUNRISE of 10.8mos.
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=76905176
= = = = = =
10-10-16/PR: ESMO’16 Topline/SUNRISE Data; B2GPI/200-240 Biomarker(30% of pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
10-10-16/PR: ESMO’16 Topline/SUNRISE; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos.
10-10-16: Peregrine Pharmaceuticals Reports Top-Line and Initial Biomarker Data from Phase III SUNRISE Trial of Bavituximab in Oral Presentation at European Society for Medical Oncology (ESMO) 2016 Congress
-- Company Has Identified Beta-2 Glycoprotein-1 (B2GP1) as a Biomarker that Correlates with Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination Compared to Chemotherapy Alone
-- Ongoing SUNRISE Trial Biomarker Analysis Expected to Identify Additional Biomarkers Associated with Patients Benefiting from Bavituximab Treatment that Will Help Guide Program's Future Clinical Development
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=992804
TUSTIN, Oct. 10, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today reported that top-line data from the Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC) were presented in an oral presentation at the European Society for Medical Oncology (ESMO) 2016 Congress. The presentation included interim efficacy and safety outcomes, as well as initial findings from the company's ongoing biomarker analysis of samples collected during the study. The SUNRISE Phase III trial was discontinued earlier this year based on a pre-specified interim analysis although patient treatment and follow-up in the study were allowed to continue. The pre-planned biomarker analysis has been taking place as patient follow-up has continued and available results were evaluated as part of the recent top-line data analysis.
The study protocol pre-specified the collection of thousands of patient samples for exploratory analyses over a wide range of possible biomarkers, including pre-treatment levels of beta-2 glycoprotein-1 (B2GP1). Data presented at ESMO demonstrated that patients with pre-treatment B2GP1 levels between 200 and 240 (representing approximately 30% of randomized patients) achieved a statistically significant, 5.5-month improvement (13.2 months vs. 7.7 months) in median overall survival (OS) as compared to patients in the control group with the same range of B2GP1 levels [p = 0.049; hazard ratio (HR) = 0.67]. A similar trend was observed with pre-treatment B2GP1 levels ≥ 200 µg/mL (representing approximately 50% of randomized patients) with 11.9 months vs. 10.1 months median OS in favor of the bavituximab-containing group (p = 0.155; HR = 0.81). Taken together, this strongly suggests B2GP1 levels may be useful for identifying patients who are more likely to benefit from a bavituximab containing therapeutic regimen. Numerous additional biomarkers are currently being analyzed with the goal of developing a multi-marker signature that can potentially identify patients that are likely to receive significant clinical benefit from a bavituximab-containing therapeutic regimen.
Top-line results reported at ESMO today were based on a data cut-off after 70% (330/473) of the targeted OS events had been reached and demonstrated the addition of bavituximab to docetaxel did not result in improvement of the study's primary endpoint of OS in the intent-to-treat population. Median OS for the bavituximab plus docetaxel group was 10.7 months as compared to 10.8 months for the placebo plus docetaxel control group (HR = 1.110; p = 0.382). Median progression free survival (PFS) for the bavituximab-containing group was 4.1 months compared to 3.9 months for the control group (HR = 0.97; p = 0.803). Objective response rates based on independent central review are currently 13% and 11% (p = 0.53) for the bavituximab-containing and control groups, respectively. Additionally, the safety profile of the combination of bavituximab with docetaxel was similar to placebo plus docetaxel.
"With every clinical trial we conduct, we are constantly reminded of the difficulty involved in treating patients with NSCLC. This continues to prove to be a very challenging cancer to combat and the need for effective treatments remains high," David R. Spigel, MD, CSO and Program Director of Lung Cancer Research at the Sarah Cannon Research Institute and one of the lead investigators in the SUNRISE trial. "The findings with regard to B2GP1 that have been collected as part of the ongoing SUNRISE trial data analysis are interesting and support further investigation."
Peregrine intends to further evaluate the role of B2GP1 levels in response to bavituximab therapy in future clinical trials. The company has filed a new patent application directed to the use of this initial biomarker discovery. Additional patient sample testing and analysis is ongoing and may result in other biomarkers of importance.
"We would once again like to thank all of the patients, clinical investigators and scientists who participated in the SUNRISE trial and have made it possible for us to continue to collect and analyze a range of key data from the study. While we were disappointed with the trial being discontinued earlier in the year, we are excited by the fact that we are beginning to learn important information from the trial through the ongoing biomarker analysis program that will be critical in helping guide the future clinical development of bavituximab," said Joseph Shan, VP of Clinical & Regulatory Affairs at Peregrine. "It is encouraging that the initial biomarker analysis has identified an important biomarker early in the process and we are optimistic that additional biomarkers associated with improved outcomes for bavituximab-containing treatments will be identified as the analysis continues. We expect to be able to share the emerging data over the coming months at scientific and medical conferences as the more results become available."
Mr. Shan continued, "It is not uncommon in the cancer field for therapeutic candidates to suffer clinical trial setbacks as researchers continue to learn more about the most appropriate patient populations for those drugs. In this landscape, biomarkers play an increasingly important role in helping identify specific patient characteristics that may impact responses to a treatment. This has been seen historically with targeted cancer treatments, as well as more recently with checkpoint inhibitors including PD-1 inhibitors. We look forward to identifying the equivalent markers for bavituximab that will help guide its clinical development."
Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses.
Peregrine's clinical development strategy for bavituximab is currently focused on small, early-stage proof-of-concept trials evaluating the drug in combination with other cancer treatments. The intent behind this strategy is to control research and development costs, while continuing to generate clinical data to further validate bavituximab's combination potential that will be critical to bringing onboard a partner to help advance the program.
ABOUT PEREGRINE PHARMACEUTICALS, INC.
Peregrine Pharmaceuticals, Inc. is a biopharmaceutical company committed to improving the lives of patients by delivering high quality pharmaceutical products through its contract development and manufacturing organization (CDMO) services and through advancing and licensing its investigational immunotherapy and related products. Peregrine's in-house CDMO services, including cGMP manufacturing and development capabilities, are provided through its wholly-owned subsidiary Avid Bioservices, Inc. (http://www.avidbio.com ), which provides development and biomanufacturing services for both Peregrine and third-party customers. The company is also working to evaluate its lead immunotherapy candidate, bavituximab, in combination with immune stimulating therapies for the treatment of various cancers, and developing its proprietary exosome technology for the detection and monitoring of cancer. For more information, please visit http://www.peregrineinc.com .
Safe Harbor *snip*
CONTACTS:
Stephanie Diaz (Investors), Vida Strategic Partners, 415-675-7401, sdiaz@vidasp.com
Tim Brons (Media), Vida Strategic Partners, 415-675-7402, tbrons@vidasp.com
= = = = = = = = = = = = = = = =
Oct7-11 2016: “41st ESMO European Cancer Congress”, Copenhagen, Denmark
http://www.esmo.org/Conferences/ESMO-2016-Congress
ESMO = European Society for Medical Oncology, ECCO = European CanCer Organization
PPHM EXHIBITING: Booth #418 (Floorplan: http://tinyurl.com/zqhv88v )
Pgm: http://www.esmo.org/Conferences/ESMO-2016-Congress/Programme
I. 10-9-16 1:00-2:00pm: POSTERS - Topic: Immunotherapy of Cancer
#1074P: “Antibody Mediated Blockade of Phosphatidylserine Improves Immune Checkpoint Blockade by Repolarizing Immune Suppressive Mechanisms of the Tumor Microenvironment” - Jeff Hutchins(VP/PreClin.Res), Peregrine Pharmaceuticals (pg.165)
9-8-16/CC/JeffH: “Exciting new internal work on PPHM’s I-O preclin. studies (Jeff Hutchins) will be presented.”
- - - - - - - - -
II. 10-10-16 Type: Proffered Paper Session* - “NSCLC/Metastatic2”
*Proffered Paper Session = Oral presentations by authors presenting original data of superior quality, followed by expert discussion and perspectives.
Chairs: Fiona Blackhall(GB); Tony S.K. Mok(Hong Kong)
10-10-16 9:15-9:30am (3:15-3:30amET) #LBA45: “Top-line Results from SUNRISE: A Phase III, Randomized, Double-Blind, Placebo-Ctl’d Multicenter Trial of Bavituximab + Docetaxel in Patients with Previously Treated Stage IIIb/IV Non-Squamous NSCLC”
David R. Spigel###(LEAD AUTHOR: CSO/Dir. Lung Cancer Res. Pgm. at Sarah Cannon Res. Inst., Nashville) 1, I. Bondarenko 2, G. Losonczy 3, J. Mezger 4, H. Kalofonos 5, M. Reck 6, R. Palmero 7, T. Jang 8, R. Natale 9, R. Sanborn 10, J. Lai 11, N. Kallinteris 12, M. Tang 11, J. Shan 13, David E. Gerber***(SENIOR AUTHOR: UTSW/Dallas) 14
1=Nashville TN; 2=Dnepropetrovsk, UA, 3=Budapest, HU, 4=Karlsruhe, DE, 5=Patras, GR, 6=Grosshansdorf, DE, 7=Barcelona, ES, 8=Busan, KR, 9=Los Angeles CA, 10=Portland OR, 11/12/13=Tustin CA; 14=UTSW/Dallas
--------
###6-1-16: “Sarah Cannon Appoints David Spigel, MD to CSO… For more than 10yrs, Dr. Spigel, has been instrumental in bringing the latest targeted therapies to patients through our lung cancer research program...” - see: http://tinyurl.com/jgxhjdn . Many Dr. David Spigel interviews on lung cancer here: https://www.youtube.com/results?search_query=David+Spigel – esp. interesting, the 4th one down: “Dr. David Spigel on Fox News Discussing Novel Therapies at ASCO 2015” https://www.youtube.com/watch?v=eb2L5xRedhQ (at 1:45 he discusses the emergence of I-O therapies: “It’s a very exciting time in oncology.”)
***UTSW’s Dr. David Gerber presented Prelim. SUNRISE Data 5-31-14 at ASCO’14 – see http://tinyurl.com/nv4jloo
= = = = = = = = = = = = =
BAVITUXIMAB "SUNRISE" PHASE III TRIAL: http://www.SunriseTrial.com
A. Phase III Bavi+Doce vs. 2nd-Line NSCLC "SUNRISE" (randomized, double-blind, placebo-ctl'd, n=582)
USA Protocol: http://www.clinicaltrials.gov/ct2/show/NCT01999673
...2 ARMS: A=BAVI/3mg+DOCE(Weekly), B=Doce+Placebo(Weekly)
...161 sites a/o 7-9-15 (USA/39 Aus/9 Bel/7 Fr/9 Ger/15 Greece/10 Hungary/7 Italy/10 Korea/9 Rom/6 Rus/8 Spain/16 Taiwan/10 Ukraine/6) - Growth: http://tinyurl.com/qbemrr2
=>10-10-16: ESMO’16 Topline/SUNRISE Data; B2GPI Biomarker(30%pts) StatSig OS 7.7=>13.2mos. http://tinyurl.com/hp73njt
2-25-16: IDMC Halts SUNRISE at 1st Look-in. Bavi+Doce arm “OS performing as expected”; Doce arm “dramatically outperforming OS expectations” http://tinyurl.com/jbg48vs
- - - - - - -
5-31-14 ASCO’14: David Gerber/Joe Shan Poster on Ph3/SUNRISE Trial (#TPS8129) http://tinyurl.com/nv4jloo
9-21-16/PR About ESMO’16/Copenhagen:
Peregrine Pharmaceuticals Provides Update on Oral Presentation of Top-Line Data from Phase III SUNRISE Trial of Bavituximab at European Society for Medical Oncology (ESMO) 2016 Congress
“Ongoing Biomarker Analysis Has Identified a Biomarker that is Associated with a Statistically Significant Improvement in Overall Survival for Patients Receiving the Bavituximab Combination”
http://ir.peregrineinc.com/releasedetail.cfm?ReleaseID=990296
TUSTIN, Sept. 21, 2016: Peregrine Pharmaceuticals, Inc. (NASDAQ:PPHM/PPHMP), a biopharmaceutical company committed to improving patient lives by manufacturing high quality products for biotechnology and pharmaceutical companies and advancing its proprietary R&D pipeline, today announced that top-line data from the Phase III SUNRISE trial of bavituximab in patients with previously treated locally advanced or metastatic non-squamous non-small cell lung cancer (NSCLC) will be presented as a late-breaking oral presentation at the upcoming European Society for Medical Oncology (ESMO) 2016 Congress. Presented data will include a biomarker in the SUNRISE trial that correlated with a statistically significant improvement in overall survival for patients treated with bavituximab in combination with docetaxel compared to patients treated with docetaxel alone. Peregrine will file a new patent application directed to the use of the biomarker prior to the presentation of results at the ESMO 2016 Congress, which is being held October 7-11, 2016 in Copenhagen, Denmark.
Details of the Phase III SUNRISE trial data presentation are as follows:
Presentation #LBA45
Presentation Title: “Top-line results from SUNRISE: A Phase III Randomized Double-Blind, Placebo-Controlled Multicenter Trial of Bavituximab Plus Docetaxel in Patients with Previously Treated Stage IIIb/IV Non-Squamous NSCLC”
Date: Monday, October 10, 2016 Time: 9:15am (local time in Copenhagen)
"I want to start by thanking the patients, investigators and scientists involved in the SUNRISE trial that have made possible the continuing collection and analysis of important data from the study. While the current interim analysis is still ongoing, it is exciting to already see that a biomarker associated with positive outcomes for patients receiving the combination of bavituximab with docetaxel has been identified. In the evolving cancer therapeutic space, biomarker identification is playing an increasingly critical role in guiding clinical development strategies and trial designs," said Steven W. King, President and CEO of Peregrine. "It is important to note that we are undertaking a broad biomarker analysis effort as part of the SUNRISE trial and this initial set of data is just the first of what we hope will be several findings that will help guide the bavituximab clinical program going forward. We look forward to presenting results from this ongoing analysis effort at the ESMO 2016 Congress, as well as other medical conferences as the additional data becomes available."
The primary goal of the biomarker analysis is to identify a biomarker profile for patients that receive the most benefit from a bavituximab-containing therapeutic regimen. As specified in the study protocol, thousands of patient samples were collected to potentially identify biomarkers associated with improved outcome for patients receiving bavituximab. Peregrine is in the process of filing a new patent application directed to the use of the initial biomarker discovery which will be presented at the ESMO 2016 Congress. Additional patient sample testing and analysis is ongoing and may result in other biomarkers of importance.
Bavituximab is an investigational chimeric monoclonal antibody that targets phosphatidylserine (PS). Signals from PS inhibit the ability of immune cells to recognize and fight tumors. Bavituximab is believed to override PS mediated immunosuppressive signaling by blocking the engagement of PS with its receptors as well as by sending an alternate immune activating signal. PS targeting antibodies have been shown to shift the functions of immune cells in tumors, resulting in multiple signs of immune activation and anti-tumor immune responses.
Peregrine's clinical development strategy for bavituximab is currently focused on small, early-stage proof-of-concept trials evaluating the drug in combination with other cancer treatments. The intent behind this strategy is to control research and development costs, while continuing to generate clinical data to further validate bavituximab's combination potential that will be critical to bringing onboard a partner to help advance the program.
ABOUT PEREGRINE PHARMACEUTICALS, INC. *snip*
Safe Harbor *snip*
= = = = = = = = = =
9-8-16/CC CEO Steve King: “Topline data from our Phase III SUNRISE trial have been accepted for a late-breaking oral presentation [10-10-16, Dr. David Gerber, UTSW, Lead Author – see below] at the upcoming European Society of Medical Oncology (ESMO) Congress to be held in Copenhagen in early October [Oct7-11]. ESMO is the premier European oncology meeting, attended by thousands of oncologists. The presentation at ESMO will be a great opportunity to share clinical data from the study in conjunction with initial results from ongoing biomarker analysis, which are already highly encouraging. Biomarker analysis was built into the SUNRISE trial from the beginning, including the collection of thousands of patient samples that could be analyzed once of the trial was unblinded. The primary goal the biomarker analysis is to identify a biomarker pattern, present in patients that receive the most benefit from a bavituximab containing therapeutic regimen, and we look forward to sharing the results of the ongoing analysis, with more data expected later in the year. The impact of the effective biomarker identification is already quite evident in oncology drug development, with the latest evidence being PD1/PD-L1 in the development of Keytruda. These types of biomarkers can only be identified through analysis of larger patient populations, such as in the SUNRISE trial. The results of identifying effective biomarkers can potentially have a huge impact on clinical development, potentially reducing the size of future trials, including making possible biomarker-driven clinical designs which could provide even more rapid readouts. Taken together, these developments are setting the stage for new data throughout the rest of 2016 and into 2017.” http://tinyurl.com/jmy77g3
9-8-16/CC: ROBERT GARNICK (Head of Reg. Affairs):
“I’d like to emphasize the strategic importance of the biomarker study, which, as Joe said, was built into the SUNRISE trial. The identification of a positive biomarker or relevant biomarker pattern with clinical efficacy is an important facet of clinical trial design that can be used to inform addl. new studies that in turn might be used to select patients who would best benefit from bavituximab therapy. For example, the identification of the HER2 biomarker was critical in the development of Herceptin. Its importance was only critically established based on the results of a Phase II clinical trial. As Steve previously described, the critical role of the PD-L1 biomarker is emerging as a major factor in the selection of patients for the clinical use of Opdivo & Keytruda. In the case of bavituximab, once a promising biomarker or biomarker pattern is identified, we strategically plan to include these results into potential new clinical trials. I will now turn the call over to Jeff Hutchins, VP of Preclin. Research.” http://tinyurl.com/jydtkoy

10-10-16@3:15amET: Topline SUNRISE Data Oral Pres. at ESMO’16/Copenhagen – A ‘Proffered Paper Session’, def. “Oral presentations by authors presenting original data of superior quality, followed by expert discussion and perspectives.” LEAD author is David R. Spigel (CSO/Dir./Sarah Cannon Res. Inst., Nashville) and SENIOR author is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14).
From the 9-21-16 PR: “Presented data will include a biomarker in the SUNRISE trial that correlated with a statistically significant improvement in OS for patients treated with bavituximab+Doce vs. Doce alone.”
...Also, Peregrine is Exhibiting: Booth #418.
MORE DETAILS HERE: http://tinyurl.com/zcsa4md
.
.
II. 10-10-16 Type: Proffered Paper Session* - “NSCLC/Metastatic2”
*Proffered Paper Session = Oral presentations by authors presenting original data of superior quality, followed by expert discussion and perspectives.
Chairs: Fiona Blackhall(GB); Tony S.K. Mok(Hong Kong)
10-10-16 9:15-9:30am (3:15am-3:30amET) #LBA45: “Top-line Results from SUNRISE: A Phase III, Randomized, Double-Blind, Placebo-Ctl’d Multicenter Trial of Bavituximab + Docetaxel in Patients with Previously Treated Stage IIIb/IV Non-Squamous NSCLC”
David R. Spigel###(LEAD AUTHOR: CSO/Dir. Lung Cancer Res. Pgm. at Sarah Cannon Res. Inst., Nashville) 1, I. Bondarenko 2, G. Losonczy 3, J. Mezger 4, H. Kalofonos 5, M. Reck 6, R. Palmero 7, T. Jang 8, R. Natale 9, R. Sanborn 10, J. Lai 11, N. Kallinteris 12, M. Tang 11, J. Shan 13, David E. Gerber***(SENIOR AUTHOR: UTSW/Dallas) 14
1=Nashville TN; 2=Dnepropetrovsk, UA, 3=Budapest, HU, 4=Karlsruhe, DE, 5=Patras, GR, 6=Grosshansdorf, DE, 7=Barcelona, ES, 8=Busan, KR, 9=Los Angeles CA, 10=Portland OR, 11/12/13=Tustin CA; 14=UTSW/Dallas
--------
###6-1-16: “Sarah Cannon Appoints David Spigel, MD to CSO… For more than 10yrs, Dr. Spigel, has been instrumental in bringing the latest targeted therapies to patients through our lung cancer research program...” - see: http://tinyurl.com/jgxhjdn . Many Dr. David Spigel interviews on lung cancer here:
Endo, from the 9-8-16/CC re: SUNRISE - “some pts are continuing to receive bavituximab, over a year now. At ESMO, we will be presenting...”
JOE SHAN (VP/Clin.&Reg. Affairs) – CLINICAL TRIALS:
“As Steve has stated, we are very excited to have the opportunity for a late-breaking oral presentation on the Phase III SUNRISE trial at the ESMO Congress about a month from now [Oct7-11 http://www.esmo.org/Conferences/ESMO-2016-Congress SEE: http://tinyurl.com/jxdppyo ]. Even as the trial continues to wind down, treatment, follow-up, and data collection are still ongoing for a number of patients, with some continuing to receive bavituximab over a year. At ESMO, we will be presenting both topline clinical data from the trial as well as initial results from our ongoing biomarker analysis, the results of which have been promising to-date. Because these data have been accepted for presentation at ESMO, they are under embargo until the actual presentation, but we will provide addl. details on the ESMO presentation and our findings as soon as we can. As a reminder, the biomarker program was prospectively built into the SUNRISE protocol, and is designed to help us better understand how bavituximab works and which patients may benefit most. This is a massive effort, and though there are still much ongoing sample testing and data analysis, our goal is to share these results as they become available throughout the rest of this year and into 2017.” http://tinyurl.com/jydtkoy
Q&A:
STEVE KING: “As we get more and more information from the SUNRISE study, particularly the biomarker analysis, and other bits of information that will help guide the program… We’re trying to do is learn as much as we can from SUNRISE, because it's a great opportunity and it’s not often you get a set of data this big in which we had already prospectively built in a lot of biomarker and other types of analysis into the study. And we actually think that the data, even though it’s a docetaxel study, those results will be actually critical in advancing this in I-O combinations in the future as well. And so, it’s just gathering data, discussing the data that's available, but more data is literally coming to light on almost daily basis, it seems. And that's going to continue for the next few months.” http://tinyurl.com/jydtkoy
Known Upcoming Events – Avid VP Pete Gagnon spoke today at an IBC Conf. in Boston…
Oct4-7/Avid/Booth#716: IBC's BioProcess Intl. Conf., Boston http://www.ibclifesciences.com/BPI/overview.xml & http://tinyurl.com/zyblds7
...10-5-16/8-8:30am: Peter Gagnon(Avid's VP/Process-Svcs), ”Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”
Oct7-11: 41st ESMO European Cancer Congress, Copenhagen, Denmark http://tinyurl.com/jxdppyo (Oral Pres. of Topline Data from Ph3 SUNRISE trial, incl. initial Biomarker analysis)
...10-10-16 9:15-9:30am: David R. Spigel (CSO/Dir./Sarah Cannon Res. Inst., Nashville) Proffered Oral Presentation, “Top-line Results from Ph3/SUNRISE..." (Senior-Author: David Gerber/UTSW) - 9-21-16 PR said, "Presented data will include a biomarker in the SUNRISE trial that correlated with a statistically significant improvement in OS for patients treated with Bavi+Doce vs. Doce alone.” http://tinyurl.com/jxdppyo
...10-9-16 1:00-2:00pm: Jeff Hutchins(VP/PreClin.Res), “Antibody Mediated Blockade of PS Improves Immune Checkpoint Blockade...”
Oct13/10am: Annual SHM, Avenue of the Arts Hotel, Costa Mesa – Final Proxy: http://tinyurl.com/gsrmgs2
Nov9-13: (SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD http://www.sitcancer.org/2016
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
…...9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.): “The goal of our work [with The MSKCC] is to evaluate combinations of PS targeting with other checkpoint inhibitors & immune stimulatory agents for the purpose of developing new & increasingly effective anticancer treatments. These pgms are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists, and radiation….” http://tinyurl.com/jmy77g3
~Dec8: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & I-O”, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Bruce Freimark(Dir.Res/Preclin.Oncology), ”Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
10-10-16: Topline SUNRISE Data Oral Pres. at ESMO’16/Copenhagen – A ‘Proffered Paper Session’, def. “Oral presentations by authors presenting original data of superior quality, followed by expert discussion and perspectives.” LEAD author is David R. Spigel (CSO/Dir./Sarah Cannon Res. Inst., Nashville) and SENIOR author is UTSW’s Dr. David Gerber (previously presented Ph2/NSCLC data and Prelim. SUNRISE data at AACR’14). As JD found on ESMO.org, the 1st author listed is the presenting author, so it’s gonna be DR. DAVID R. SPIGEL!
From the 9-21-16 PR: “Presented data will include a biomarker in the SUNRISE trial that correlated with a statistically significant improvement in OS for patients treated with bavituximab+Doce vs. Doce alone.”
...Also, Peregrine is Exhibiting: Booth #418.
ESMO’16 DETAILS HERE, Endo:
http://investorshub.advfn.com/boards/read_msg.aspx?message_id=125381225
Avid exhibiting @IBC/Boston/Oct4-7, Pete Gagnon(VP) speaks tomorrow…
Oct4-7 2016: “IBC's BioProcess Intl. Conference & Exhibition”, Boston
”The largest bioprocessing event bringing you new ideas, demystifying technology and fostering partnerships in highly engaging formats to move drug candidates closer to approval.”
http://www.ibclifesciences.com/BPI/overview.xml
10-5-16/8-8:30am Stream: Recovery & Purification
“Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”, Peter Gagnon, VP-Process-Svcs/Peregrine Pharmaceuticals
https://lifesciences.knect365.com/bioprocessinternational/agenda/2
*AVID: Visit us at Booth #716
= = = = = = = = = = =
5-23-16: Pete Gagnon (100pubs/presentations, 12+patents, 30 pending patents) Appointed VP/Process-Services of Avid http://tinyurl.com/hg6abdq – See Oct'15:
SITC’16 Abstract Titles: MSKCC/Wolchok-Lab & PPHM, Nov9-13, Natl-HarborMD – Reg. Abstract Titles released today! Take a look at #1, the joint MSKCC/PPHM one! Its Senior author is Dr. Taha Merghoub (Wolchok Lab), who said this in the 5-29-15 PPHM/MSK Collab. Announcement PR (see below): "A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize & destroy cancer. This collaboration will allow us to focus on the role & contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical & translational work will potentially guide the design of the next generation of clinical studies with bavituximab."
Nov9-13 2016: “(SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD
”The premier destination for scientific exchange, education, and networking in the Cancer Immunotherapy Field”
SITC = The Society for Immunotherapy of Cancer http://www.sitcancer.org
SITC 2016 Meeting: http://www.sitcancer.org/2016
Abstracts: http://www.sitcancer.org/2016/abstracts “Reg. Abstract Titles Avail. 9-29-16, Late-Breakers Early Oct2016”
----------
PEREGRINE ABSTRACTS:
I. “Phosphatidylserine Targeting Antibody in Combination with Checkpoint Blockade & Tumor Radiation Therapy Promotes Anti-Cancer Activity in Mouse Melanoma” - Sadna Budhu, PhD - Ludwig Collaborative Lab, MSKCC
AUTHORS: Sadna Budhu(MSKCC PRESENTING AUTHOR) 1, Olivier De Henau 2, Roberta Zappasodi 1, Kyle Schlunegger 3, Bruce Freimark 4, Jeff Hutchins 5, Christopher A. Barker 6, Jedd D. Wolchok 7, Taha Merghoub(MSKCC SENIOR AUTHOR) 1 [<=See 5-29-15 PPHM/MSKCC PR below]
1/2 Ludwig Collaborative Lab, Memorial Sloan Kettering CC, NYC
6 Memorial Sloan Kettering CC, NYC
7 Dept of Medicine, Memorial Sloan Kettering CC, NYC
3/4/5 Peregrine Pharmaceuticals, Tustin, CA http://bit.ly/2dHTEVn
II. “Antibody Targeting of Phosphatidylserine Enhances the Anti-Tumor Responses of Ibrutinib & anti-Pd-1 Therapy in a Mouse Triple Negative Breast Tumor Model” - Jian Gong, PhD - Peregrine Pharmaceuticals
AUTHORS: Jian Gong, Michael Gray, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUh1w
III. Monoclonal Antibodies Targeting Phosphatidylserine Enhance Combinational Activity of the the Immune Checkpoint Targeting Agents LAG3 & PD-1 in Murine Breast Tumors” - Michael Gray, PhD - Peregrine Pharmaceuticals
AUTHORS: Michael Gray, Jian Gong, Jeff Hutchins, Bruce Freimark - Peregrine Pharm. http://bit.ly/2dpUy4C
= = = = = = = = =
9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.) http://tinyurl.com/jmy77g3
“...I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering Cancer Center. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the AACR/CRI Immunotherapy Meeting in New York later this month [Sept25-28] and at ESMO in early October [Oct7-11: http://tinyurl.com/jxdppyo ]. We expect the first results from our collaboration with MSK’s Jedd Wolchok Lab investigators [See MSK Collab: http://tinyurl.com/zkvebh6 ] to be presented at SITC in November [Nov9-13: http://www.sitcancer.org/2016 Natl.Harbor MD] and we will provide more detailed information as that presentation becomes available.”
= = = = = = = = = = = = = = = =
5-29-15: Peregrine & Sloan Kettering Enter Collab. to “Investigate Novel PS-Targeting Immunotherapy Combos” http://tinyurl.com/o3k9ux8
...The studies at MSK will be performed under the direction of Taha Merghoub, PhD, [ http://www.mskcc.org/research-areas/labs/members/taha-merghoub-01 ] Associate Attending Biologist, Melanoma and Immunotherapeutics Service, Ludwig Collaborative and the Swim Across America Laboratory, a part of the laboratory of Jedd D. Wolchok, MD, PhD [ http://www.mskcc.org/research-areas/labs/jedd-wolchok ], a leader in the field of cancer immunotherapy. Dr. Wolchok serves as the Chief, Melanoma and Immunotherapeutics Service, Lloyd J. Old Chair for Clinical Investigation as well as an Associate Director of the Ludwig Center for Cancer Immunotherapy at MSK.
----
”The phosphatidylserine (PS) signaling pathway is a very interesting target for modulating the immune system's response to cancer. We look forward to exploring the potential of PS-targeting agents alone and with other immune modulators that may lead to novel advances in cancer therapy," said Dr. Jedd Wolchok.
–--
As part of the collaboration, researchers at MSK will conduct research to further explore the combination of PS-targeting agents, including bavituximab, that block a primary immunosuppressive pathway thereby allowing anti-tumor immune responses with other immuno-stimulatory agents that enhance immune responses. Specifically, MSK researchers will examine the combination of bavituximab alongside models of checkpoint blockade that are unresponsive to inhibition or co-stimulation given the ability of bavituximab to reprogram myeloid derived suppressor cells (MDSC) and increase tumoricidal T-cells in tumors, a mechanism of action that is complementary to checkpoint blockade and T-cell activation.
–--
"A key focus of the Wolchok Lab's research is studying novel immunotherapy combinations that work together to enable the immune system to recognize and destroy cancer. This collaboration will allow us to focus on the role and contribution of PS blockade therapy in determining which combination of the current and next generation of immune modulators is likely to increase the extent and amplitude of anti-tumor response. This important pre-clinical and translational work will potentially guide the design of the next generation of clinical studies with bavituximab," said Dr. Taha Merghoub.
–--
"We are delighted to be working with a world-renowned pioneer and leader in the immuno-oncology space, recognizing that there remains significant research in order for more cancer patients to realize the benefits of combination immune therapy," said Jeff T. Hutchins, PhD, VP of Preclinical Research at Peregrine. ”Our internal and collaborative research presented over the last year has established a robust foundation of PS-targeting activity on which to initiate this next chapter in PS research and development."
–--
"This collaboration is an important extension of our established research efforts to further explore and understand the potential of our PS-targeting platform including bavituximab our lead clinical candidate. This research will focus on better understanding how treatment with PS-targeting agents can assist other anti-tumor immunotherapies in order to work better," said Steven King, CEO of Peregrine. ”Our goal is to change the way cancer patients are treated by allowing their immune system to recognize and fight their disease. This collaboration will undoubtedly assist us in identifying potential new opportunities to better treat patients with cancer."
.
.

Known Upcoming Events...
Oct4-7/Avid/Booth#716: IBC's BioProcess Intl. Conf., Boston http://www.ibclifesciences.com/BPI/overview.xml & http://tinyurl.com/zyblds7
...10-5-16/8-8:30am: Peter Gagnon(Avid's VP/Process-Svcs), ”Across the Great Divide: The Upstream Origins & Downstream Ramifications of a Newly Discovered Contaminant Class”
Oct7-11: 41st ESMO European Cancer Congress, Copenhagen, Denmark http://tinyurl.com/jxdppyo (Oral Pres. of Topline Data from Ph3 SUNRISE trial, incl. initial Biomarker analysis)
...10-10-16 9:15-9:30am: David R. Spigel (CSO/Dir./Sarah Cannon Res. Inst., Nashville) Proffered Oral Presentation, “Top-line Results from Ph3/SUNRISE..." (Senior-Author: David Gerber/UTSW) - 9-21-16 PR said, "Presented data will include a biomarker in the SUNRISE trial that correlated with a statistically significant improvement in OS for patients treated with Bavi+Doce vs. Doce alone.” http://tinyurl.com/jxdppyo
...10-9-16 1:00-2:00pm: Jeff Hutchins(VP/PreClin.Res), “Antibody Mediated Blockade of PS Improves Immune Checkpoint Blockade...”
Oct13/10am: Annual SHM, Avenue of the Arts Hotel, Costa Mesa – Final Proxy: http://tinyurl.com/gsrmgs2
Nov9-13: (SITC) Society for Immunotherapy of Cancer 31st Annual Meeting”, Natl-Harbor MD http://www.sitcancer.org/2016
...”First Results” from our collaboration with Jedd Wolchok Lab investigators (MSK) to be presented” (per 9-8-16/Ccall/Hutchins)
…...9-8-16/CC - JEFF HUTCHINS (VP/PreClinical Res.): “The goal of our work [with The MSKCC] is to evaluate combinations of PS targeting with other checkpoint inhibitors & immune stimulatory agents for the purpose of developing new & increasingly effective anticancer treatments. These pgms are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists, and radiation….” http://tinyurl.com/jmy77g3
~Dec8: FY'17Q2 (qe 10-31-16) Financials & Conf. Call - http://ir.peregrineinc.com/events.cfm
Dec11-15/Avid/Booth#209: IBC's Antibody Eng. & Therapeutics 2016”, SanDiego http://www.ibclifesciences.com/AntibodyEng/overview.xml
Feb20-22 2017: “CHI’s 5th Translational Models in Oncology & I-O”, SanFran http://www.triconference.com/Pre-Clinical-Oncology-Models
...Bruce Freimark(Dir.Res/Preclin.Oncology), ”Blockade of PS-Mediated Tumor Immune Suppression to Enhance Immune Checkpoint Therapies”
I'm sure today's B119 (Birge+PPHM) AACR-CRI Poster Image will be released soon - often times they put them up on PeregrineInc.com. I'll keep checking. Take care.
Track: MECHANISTIC MERGING OF TREATMENT MODALITIES
II. #B119 “Characterization of a Phosphatidylserine, TAM Receptor (Tyro3, Axl, Mertk), PDL1 Axis in Breast Cancer”
Canan Kasikara 1, Sushil Kumar 1, Ke Geng 1, Viral Davre 1, Cyril Empig 2, Bruce Freimark 2, Michael Gray 2, Kyle Schlunegger 2, Jeff Hutchins 2, Sergei V. Kotenko 1, Raymond B. Birge [SENIOR AUTHOR] 1
1=Rutgers, New Jersey Medical School, Newark, NJ
2=Peregrine Pharmaceuticals, Tustin
N40k, re: Birge+PPHM today, go to this link http://www.cancerimmunotherapyconference.org/abstracts
...Click on POSTER B. It's a PDF I think. It lists All the AACR-CRI Posters (Set B) - pg.14 is B119 (Birge+PPHM)... It only lists the Title and the Authors, not the full abstract or the poster image. Hope it works for you - it does for me!
N40K, re: today's Birge+PPHM AACR-CRI/NYC, click on the ABSTRACTS link in my post. Then, POSTERS B - it's on pg.14 (B119)
TODAY: Rutgers’ Raymond Birge w/Peregrine co-authors, AACR-CRI/Sept26/NYC Poster. This involves Peregrine’s “preclinical I-O focused internal efforts”, which Jeff Hutchins (VP/PreClin.Res) described in the 9-8-16 CC as, “advancing well, and we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation.” - see Dr. Hutchin’s 9-8-16 comments below. Note that Dr. Birge is the Senior Author of B119.
Sept25-28 2016: “2nd CRI-AACR-CIMT-EATI Intl. Cancer Immunotherapy Conf.”, NYC
“The pgm will focus on “Translating Science into Survival” and feature talks from more than 60 leaders in the field covering all areas of inquiry in cancer immunology and immunotherapy.”
http://www.aacr.org/Meetings/Pages/MeetingDetail.aspx?EventItemID=101
ABSTRACTS: http://www.cancerimmunotherapyconference.org/abstracts
9-26-16 5:15-7:45pm: Poster Session B
-----
Track: NEW AGENTS AND THEIR MODE OF ACTION IN ANIMALS AND HUMANS
I. #B019 “LAG3 is an Immunotherapeutic Target in Murine Triple- Breast Cancers, Whose Activity is Significantly Enhanced in Combination with Phosphatidylserine Targeting Antibodies”
Michael J. Gray, Jian Gong, Jeff Hutchins, Bruce Freimark (Peregrine Pharmaceuticals, Dir.Res/Preclin.Oncology)
-----
Track: MECHANISTIC MERGING OF TREATMENT MODALITIES
II. #B119 “Characterization of a Phosphatidylserine, TAM Receptor (Tyro3, Axl, Mertk), PDL1 Axis in Breast Cancer”
Canan Kasikara 1, Sushil Kumar 1, Ke Geng 1, Viral Davre 1, Cyril Empig 2, Bruce Freimark 2, Michael Gray 2, Kyle Schlunegger 2, Jeff Hutchins 2, Sergei V. Kotenko 1, Raymond B. Birge 1
1=Rutgers, New Jersey Medical School, Newark, NJ
2=Peregrine Pharmaceuticals, Tustin
- - - - -
9-8-16 CC/Jeff Hutchins: “I would now like to provide an update on Peregrine's preclinical I-O focused internal efforts and our collaboration with Memorial Sloan Kettering CC. The goal of this work is to evaluate combinations of PS targeting with other checkpoint inhibitors and immune stimulatory agents for the purpose of developing new and increasingly effective anticancer treatments. These programs are advancing well and to-date, we have seen impressive signs of activity with new combinations of PS targeting and other treatment modalities such as checkpoint blockers, T-cell agonists and radiation. These new combinations are improving overall survival accompanied with increases in cyto-reactive T cells into the tumor tissue. This exciting new internal work will be presented at the CRI/AACR Immunotherapy Meeting in NY later this month and at ESMO in early October. We expect the first results from our collaboration with the Wolchok Lab investigators to be presented at SITC in November and we will provide more detailed information as that presentation becomes available.” http://tinyurl.com/jydtkoy
= = = = = = =
2-2016: Rutgers' Dr. Raymond Birge's relationship with Peregrine & UTSW's Dr. Rolf Brekken and his 2-26-16 article, “Phosphatidylserine is a Global Immunosuppressive Signal in Efferocytosis, Infectious Disease, and Cancer” http://tinyurl.com/z5d9qt9 poster B019, “LAG3 is an Immunotherapeutic Target in Murine Triple- Breast Cancers, Whose Activity is Significantly Enhanced in Combo with PS-Targeting Antibodies”
11-9-15 SITC'15: New Bavi+Checkpoint Inhibitors preclin. data (UTSW/DUKE's Herbert K. Lyerly) http://tinyurl.com/pbof95w
...Also, collab. with Dr. Bernard Fox (Immunotherapist/Earle A. Chiles Res.Inst.) on new Immuno-Profiling Clinical Test (Opal 6-plex quantitative IF Assay), PPHM roundtable with Raymond Birge (Rutgers), Douglas Graham (Emory), Dmitry Gabrilovich (Wistar), Rolf Brekken (UTSW), Maria Karasarides (AstraZeneca) - ”Combining Bavi w/anti-PD-1 significantly enhanced O/S… significant incr. CD45+, CD8+ and CD3+ T-cells… led a prolonged anti-tumor immune response which protected the animals against a re-challenge w/same tumor.”
= = = = = = = =DR. BIRGE:
...Dr. Raymond Birge has authored ~85 scientific publications in molecular & cancer biology. http://www.ncbi.nlm.nih.gov/pubmed/?term=birge+rb
“The Birge laboratory conducts basic science focused on the eradication of cancer.”
http://birgelab.org => http://birgelab.org/biography.html
Robert Garnick’s 9-8-16 comments re: upcoming ESMO’16/SUNRISE/Biomarker data, to be presented 10-10-16 in a proffered late-breaking oral presentation by lead author Dr. David R. Spigel (CSO/Dir. Lung Cancer Res. Pgm. at Sarah Cannon Res. Inst., Nashville)...
9-8-16/CC: ROBERT GARNICK (Head of Reg. Affairs):
“I’d like to emphasize the strategic importance of the biomarker study, which, as Joe said, was built into the SUNRISE trial. The identification of a positive biomarker or relevant biomarker pattern with clinical efficacy is an important facet of clinical trial design that can be used to inform addl. new studies that in turn might be used to select patients who would best benefit from bavituximab therapy. For example, the identification of the HER2 biomarker was critical in the development of Herceptin. Its importance was only critically established based on the results of a Phase II clinical trial. As Steve previously described, the critical role of the PD-L1 biomarker is emerging as a major factor in the selection of patients for the clinical use of Opdivo & Keytruda. In the case of bavituximab, once a promising biomarker or biomarker pattern is identified, we strategically plan to include these results into potential new clinical trials...” http://tinyurl.com/jydtkoy