Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Thank you for your opinion.
Here’s an old PR- second paragraph is my response to “no inventory”. No idea what will be on the quarterly but it’s something to talk about. Instead of just bashing people…
LOS ANGELES, Jan. 21, 2021 (GLOBE NEWSWIRE) -- ViaDerma, Inc., (“Company”) (OTC Pink: VDRM), announces the completion of a licensing agreement with a group of affiliated wound care medical practices. The medical practices provide advanced wound care treatment to homebound patients and are currently operating in six states. ViaDerma’s goal is to increase its presence to medical practices in sixteen states this year and to be in all fifty states by the end of 2022.
As part of their wound care treatment protocol, the medical practices will be utilizing their own private label medication based on the technology developed by ViaDerma President and CEO, Dr. Chris Otiko.
Come on. Send in the clowns. What do reasonable folks expect to see in the quarterly? Revenue from the sales of a product that no one can find or even prove exists. Maybe a list of retailers who can't sell what they don't have. Get real with the quarterly chatter.
Quarterly due next week I believe. I am expecting a PR with it, but who knows. This is a long term play for me. Not too worried or impatient. If it goes backs to 6s, we’ll I’ll get some more
WOW! Crickets. Out of excuses and nonsense it seems. Not even a hint of product availability anywhere after all this time. Come on and get real. This POS is circling the drain, or it is the best kept secret in marketing history. I don't think anyone involved with this company is that smart for that to be the case.
OTIKO =A TRUE LYING SCAMBAG.EVERYTIME.
Dr. OTIKO = STEPHEN BROWN FROM HPIL.... Same dancers!!! Same Bs.... I respect a Con-Man that a least have the integrity to let know his victims the money is gone ya fools!!! This man, leave you rot in your own fears... He hides and does not stick his head until a few months, if needed. He called another deal before of different hospitals... A year later, nothing..... I cant believe I did fall for this again... SMH
I CALLED IT.ARREST OTIKO.
ORCA
Member Level
Monday, July 11, 2022 11:48:43 PM
Re: Here Today post# 60823
Post#
60825
of 61098
Chris Otiko 4 WEEKS,AS WE KNOW,COULD BE 4 MONTHS.TOO.ALWAYS A DIFFERENT EXCUSE.IT WILL BE A MIRACLE IF HE EVER DELIVERS ON TIME.JUST LIKE THE BS $600K CONTRACT.LOL.
Dr Otiko is a failure. History tells me he hides under his desk for another 6 months
This CEO Dr. O, not sure what is mojo is but it is not in focus, cheer will or integrity to his shareholders at all. Very disrespectful his silence, sometimes he reminds me like this HPIL CEO: Stephen Brown, OMG... Always delayed on his milestone... Well, he never reached to point B. I hope this one is different
What’s new….. I don’t expect a PR until the 2nd Quarter results are released.
The DOC is out of the country won’t be back until Monday, next week. The IR will be unavailable Wednesday through the following Wednesday. Don’t expect a press release until the week of the 22nd at the earliest. We’re all frustrated waiting for some news.
I sent an email over the weekend. Awaiting reply
Thinking the OTCM snafu delayed releasing results of study. Who knows
"NO BIG DEAL" ????? Huh???
What month are we in? Year?
ViaDerma, Inc. Set to Launch Two New and Improved Versions of its Flagship Product, Vitastem and Vitastem Ultra
Press Release | 10/04/2021
LOS ANGELES, Oct. 04, 2021 (GLOBE NEWSWIRE) -- ViaDerma, Inc., (“Company”) (OTC Pink: VDRM) is pleased to announce they are launching two new and improved formulas for its top selling product, Vitastem and Vitastem Ultra.
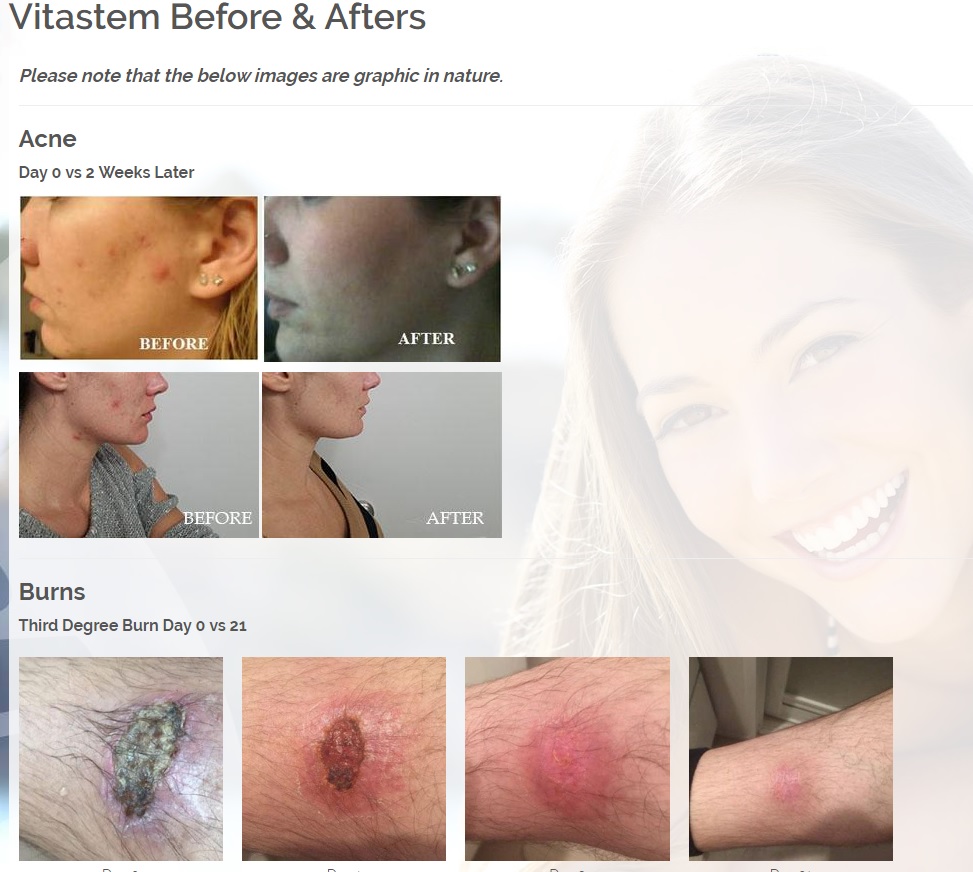
The original Vitastem is an FDA-registered drug and one of the world’s strongest topical antibiotics. Its active ingredient is tetracycline. Vitastem kills all harmful Gram-positive and Gram-negative bacteria that have been available for testing (bacteria that is associated with acne, cuts, scrapes, burns, and secondary infections associated with psoriasis and eczema). The new formula is clear and colorless compared to the brownish color of the original, so it will not stain the skin, fingernails, toenails, or anywhere else it is applied.
The Company is also unveiling its new Vitastem Ultra. The active ingredient in Vitastem Ultra is bacitracin. This provides an alternative for people who are allergic to tetracycline, as well as a way to treat tetracycline resistant bacteria. Additionally, Vitastem Ultra will open new doors overseas in many markets that prefer tetracycline alternatives. Like the new and improved Vitastem, Vitastem Ultra is colorless and non-staining. Batches of the new products are currently being produced and are expected to be available to consumers on Amazon, Vitastem.net and other ViaDerma resellers by mid-October. The old version will no longer be sold, and all new and future purchase orders will be filled with the new products.
Perhaps us investors try to reach the Dr. and implant some motivation or guilt. Perhaps both.
So we wait till next week big F******* deal
SCAMBAG OTIKO WHERE IS THE UPDATE OF THE TESTING???????YOU LYING DIRT BAG.WITHIN 4 WEEKS HUH???????????????
Tons of great posts. Here is one from July 27th:
Sure. Buying opportunity. Really? People can do what they want with their money. Can just burn it if they want. But everything is going to be all right. I know I feel better now. This company is turning into a joke with excuses more available than their product no one can find. But according to some, orders pending are huge and retailers are too numerous to count. Close your eyes. What do you see? NOTHING!
Just my opinion of course.
Here is another from July 28th:
And the hits just keep on coming. Wow! Again, I feel so much better. NOT! All, and I mean ALL evidence to the contrary. Great to talk about what used to be and how great the product was but what good is the memory of a product the public can't buy or find anywhere. Generating revenue? Really where? Except for maybe some minor runs this company seems to be circling the drain. Just excuses after excuses after more excuses. But what remains humorous is the nonsense through the prism of smoking mirrors. As always just my opinion but at least it's honest and from someone with a sizable position here.
Good honest posts. Certainly not everyone's opinion but I'm entitled to mine.
A lot of great posts! $VDRM share price, 0.10-0.25+ coming soon for sure!
Why choose Vitastem
Wound Care Treatment
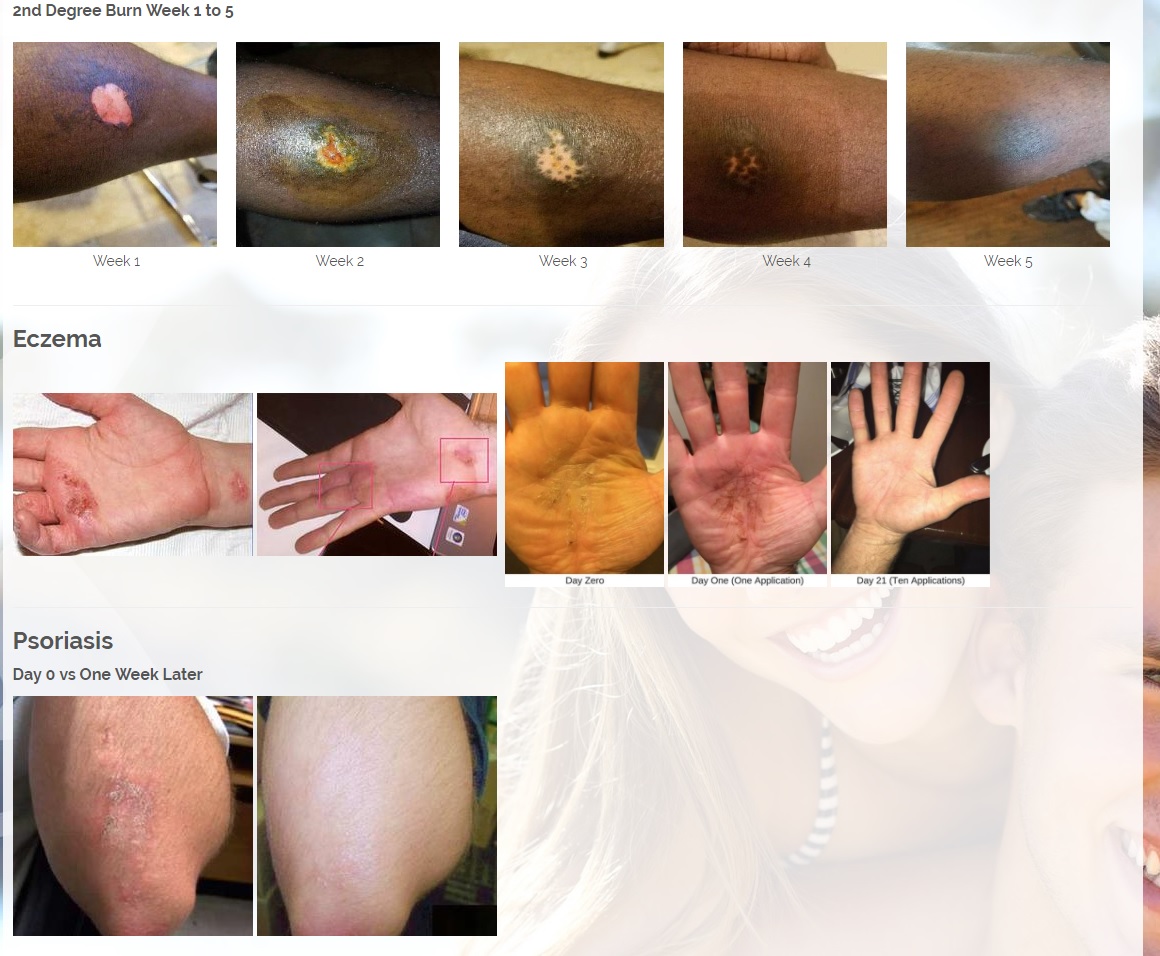
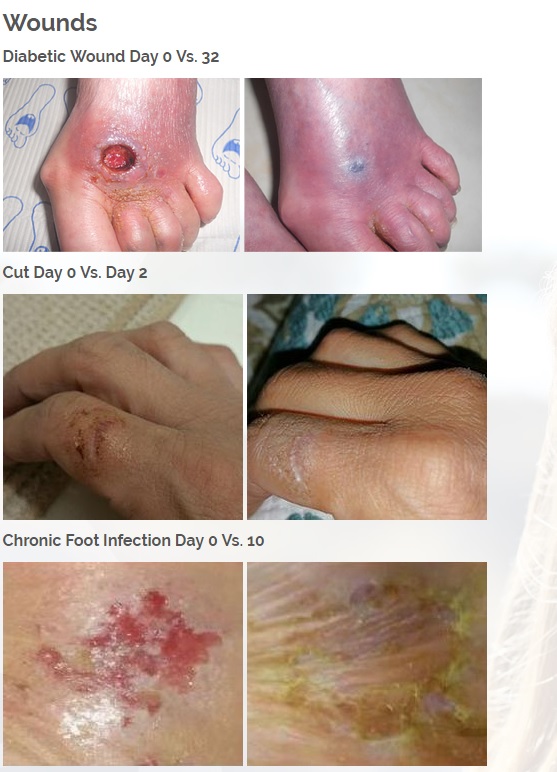
Vitastem is an FDA registered drug and one of the world’s strongest topical antibiotics. Vitastem kills all harmful Gram-positive and Gram-negative bacteria that have been available for testing (bacteria that is associated with eczema, psoriasis, acne, diabetic wounds, cuts and others).
How is this wound care treatment so powerful?
All known antibiotics (other than Vitastem) primarily use what’s called a chemical “mechanism of kill” only, whereby the antibiotic attempts to kill the bacteria slowly by applying (if topical), or absorbing (if oral) more and more of the medicine over a period of time as it is taken as prescribed. While this can be somewhat effective, it takes much longer. Unfortunately, this also allows for bacteria to do what bacteria does: evolve and develop resistance to the medicine.
That’s where Vitastem is different…
Vitastem is a wound care treatment that uses both a chemical and a physical mechanism to kill and fight pathogens. The physical mechanism of kill is a key feature of Vitastem’s strength.
Vitastem’s patent-pending, specialized combination of ingredients has hacked the delivery formula that enables medicine to be transported at 10x the strength and depth of other products in the marketplace today. This formula stimulates the cell wall such that it is up to 10x more permeable than normal, allowing for substantially more medicine to enter into the cell, overwhelming and killing the bacteria so quickly that it does not have time to adapt and develop resistance. This highly concentrated and rapid delivery of medicine to the site of treatment is what has Vitastem patients seeing results in 24 hours versus 5 to 7 days (as is the case with other products that treat the same conditions).
In addition to treating the condition, Vitastem also infuses skin cells damaged by the infection with vitamin D3 and vitamin C (ascorbic acid). Skin is left treated and rejuvenated!
Because Vitastem is a topical antibiotic, it is safer for your liver than oral OTC or prescription treatments.
KEY FEATURES OF VITASTEM:
Vitastem was originally designed over 10 years ago to heal diabetic ulcers and wounds in which it boasts 96% of patients seeing improvement within just 2 weeks, dwarfing the largest name brand Rx competitor at 50% of patients seeing improvements at 20 weeks. Vitastem has saved countless patients from amputations thus reducing their hospital stays and increasing their life expectancy.
Vitastem kills all known bacteria it has ever been tested against, including MRSA (flesh-eating bacteria) and Staph Infection.
Vitastem is the only antibiotic in the world that kills with both a physical and chemical mechanism, giving it a unique ability to overcome drug resistant bacteria.
Vitastem has a unique ability to heal and regenerate tissue unlike any other medication on the market (see before and after pictures in case studies section).
Since Vitastem is a topical antibiotic it does not have the same ill-effects on the internal organs of the body as oral antibiotics. For example, unlike oral antibiotics, Vitastem will not harm the kidneys or the good bacteria in the stomach.
In the era of Corona virus, Ebola and Swine Flu, and when 80% of the world’s antibiotics are made in China, our product is proudly Made in the USA where pharmaceutical standards and quality are the finest the world has to offer.
Vitastem’s patent-pending, specialized combination of ingredients has hacked the delivery formula that enables medicine to be transported at 10x the strength and depth of other products in the marketplace
What is Vitastem?
Vitastem is an FDA registered drug and one of the world’s strongest topical antibiotics. Vitastem kills all harmful bacteria that have been available for testing. It is the only antibiotic in the world that kills bacteria with both a physical and a chemical mechanism.
Vitastem is registered with the FDA as a first aid antibiotic to help prevent infection in minor cuts, scrapes and burns. In addition, our research and customer feedback have shown success in treating the symptoms common with secondary infections associated with eczema, psoriasis, acne, chronic wounds, diabetic wounds, and a host of bacterial infections
Has Vitastem been clinically tested?
Yes. Vitastem has over 10 years of clinical testing behind it. It was developed by a team of scientists and physicians with a combined expereince of 60 years in Biochemistry, Molecular Biology, and Wound Care. In the lab, Vitastem has demonstrated a 99% efficacy rate in killing all harmful bacteria on which it has been tested. This includes MRSA (flesh-eating bacteria), VRE, Pseudomonas to name just a few. Vitastem is recommended by both physicians and patients worldwide and has been found effective in treating a variety of skin conditions
Good stuff - thanks for sharing
I have used it with great results, thanks
And the fabulous testing results should be announced.
I expect news this week, big orders, new state contracts. Should be all good from here. Been a long time coming. We do need some international things to happen after we get up and rolling.
That’s a very conservative approach, based on the slow way this CEO works and comunícate his updates with the company milestones. However, I don’t think it will take him 9 months time frame to retrieve the test results he’s already past due (30 days+) to reveal , in order to give green light to the pending orders in place. He said massive orders pending!!! I am aiming 0.45c before Christmas
Should be .40+!
Within a 9 month window I believe we can get close to.25.
Been a good week. Glad when so many were bad mouthing I was buying all I could at 55+6. Happy now.
Thanks for reposting some of my factual posts
This is not a stock for the weak minded investor
Just a matter of days PLENTY TO SELL
What a foolish thing to post JUST WATCH
Are you saying they will never have product again?
What would a salesperson sell? Have you seen anything that resembles a product from this company that is marketable lately? All there is is noise. And unsubstantiated noise at that. Tough to sell noise.
*ANY* revenue is a good thing**
Agree. And revenue is good IMO. Plenty of stinkies out there with zero revs and covered in debt
I would love to see the Dr use some of this reoccurring revenue to hire a couple of sales people. What’s your opinion on this?
The most logical outcome for the 2nd Qtr Report would seem to be maintenance of the nine (9) state licenses and thus, $135K revenue.
Quarterly due soon. Maybe they’ll pair that with an update RE study
Well we should be free now low volume.
you will be bleeding $ Green $ : )
Expect anything less???
But remember, FURTHER testing is now needed? ODD, as Brewer sells the new version and has for a while :)
ViaDerma, Inc. Set to Launch Two New and Improved Versions of its Flagship Product, Vitastem and Vitastem Ultra
Press Release | 10/04/2021
LOS ANGELES, Oct. 04, 2021 (GLOBE NEWSWIRE) -- ViaDerma, Inc., (“Company”) (OTC Pink: VDRM) is pleased to announce they are launching two new and improved formulas for its top selling product, Vitastem and Vitastem Ultra.
The original Vitastem is an FDA-registered drug and one of the world’s strongest topical antibiotics. Its active ingredient is tetracycline. Vitastem kills all harmful Gram-positive and Gram-negative bacteria that have been available for testing (bacteria that is associated with acne, cuts, scrapes, burns, and secondary infections associated with psoriasis and eczema). The new formula is clear and colorless compared to the brownish color of the original, so it will not stain the skin, fingernails, toenails, or anywhere else it is applied.
The Company is also unveiling its new Vitastem Ultra. The active ingredient in Vitastem Ultra is bacitracin. This provides an alternative for people who are allergic to tetracycline, as well as a way to treat tetracycline resistant bacteria. Additionally, Vitastem Ultra will open new doors overseas in many markets that prefer tetracycline alternatives. Like the new and improved Vitastem, Vitastem Ultra is colorless and non-staining. Batches of the new products are currently being produced and are expected to be available to consumers on Amazon, Vitastem.net and other ViaDerma resellers by mid-October. The old version will no longer be sold, and all new and future purchase orders will be filled with the new products.
“I am very excited to get these two new products on the market,” said Dr. Chris Otiko, ViaDerma President and CEO. “Vitastem has always received great reviews, but the one suggestion that came up from time to time was if we can make it non-staining. I am happy to announce we have addressed that issue.”
Dr. Otiko also said, “Our new Ultra product is a great alternative for people who cannot use tetracycline. Ultra will provide the same great results as the original.”
About ViaDerma, Inc.
ViaDerma, Inc. (OTC: VDRM) is a publicly traded specialty pharmaceutical company committed to bringing new products to market and licensing its innovative technology to current leaders in the pharmaceutical industry in a wide variety of therapeutic areas. For more information, visit: www.viadermalicensing.com
Any forecast of future performance is a "forward looking statement" under securities laws. Such statements are included to allow potential investors the opportunity to understand management’s beliefs and opinions with respect to the future so that they may use such beliefs and opinions as one factor among many in evaluating an investment.
Can't wait to puncher myself when the new clear stuff comes out.
Agreed to all
The way we are getting interest in the stock I would say we have smoke let's see if we get fire.
ON ANY ticker....that is.
**Any surprise here?**
ViaDerma, Inc. Set to Launch Two New and Improved Versions of its Flagship Product, Vitastem and Vitastem Ultra
Press Release | 10/04/2021
LOS ANGELES, Oct. 04, 2021 (GLOBE NEWSWIRE) -- ViaDerma, Inc., (“Company”) (OTC Pink: VDRM) is pleased to announce they are launching two new and improved formulas for its top selling product, Vitastem and Vitastem Ultra.
The original Vitastem is an FDA-registered drug and one of the world’s strongest topical antibiotics. Its active ingredient is tetracycline. Vitastem kills all harmful Gram-positive and Gram-negative bacteria that have been available for testing (bacteria that is associated with acne, cuts, scrapes, burns, and secondary infections associated with psoriasis and eczema). The new formula is clear and colorless compared to the brownish color of the original, so it will not stain the skin, fingernails, toenails, or anywhere else it is applied.
The Company is also unveiling its new Vitastem Ultra. The active ingredient in Vitastem Ultra is bacitracin. This provides an alternative for people who are allergic to tetracycline, as well as a way to treat tetracycline resistant bacteria. Additionally, Vitastem Ultra will open new doors overseas in many markets that prefer tetracycline alternatives. Like the new and improved Vitastem, Vitastem Ultra is colorless and non-staining. Batches of the new products are currently being produced and are expected to be available to consumers on Amazon, Vitastem.net and other ViaDerma resellers by mid-October. The old version will no longer be sold, and all new and future purchase orders will be filled with the new products.
“I am very excited to get these two new products on the market,” said Dr. Chris Otiko, ViaDerma President and CEO. “Vitastem has always received great reviews, but the one suggestion that came up from time to time was if we can make it non-staining. I am happy to announce we have addressed that issue.”
Dr. Otiko also said, “Our new Ultra product is a great alternative for people who cannot use tetracycline. Ultra will provide the same great results as the original.”
Still no products available for purchase on Amazon or Viaderma website … surprise
|
Followers
|
619
|
Posters
|
|
|
Posts (Today)
|
2
|
Posts (Total)
|
74586
|
|
Created
|
10/27/08
|
Type
|
Free
|
| Moderators | |||

ViaDerma's proprietary transdermal delivery system allows for rapid mass transfer of the pharmaceutical active ingredient across the skin and into the body to provide immediate localized therapy.
The technology allows transfer of chemicals through the stratum corneum (outer layer of skin) with a diffusion constant which is 10,000 times higher than the diffusion constant which characterizes water movement through the stratum corneum.
This enables ViaDerma to pair almost any active ingredient with the technology and provide rapid transport of the medicine right to the site of action.
The first product is a broad spectrum tetracycline-based topical antibiotic is the only antibiotic in the world that kills bacteria both a physical and a chemical mechanism. All known antibiotics (other than ours) primarily use only a chemical mechanism of kill. The physical mechanism of kill is a key feature of what we call Rapid Active Ingredient Delivery System (RAIDS). One result of RAIDS is that tetracycline is carried in higher concentrations, more quickly, to and through the cell walls, where the tetracycline can become more effective than if conventional antibiotics were used. Conventional antibiotics require more time (usually prescribed for 5 to 7 days for best results), whereas our tetracycline-based products usually produce desirable results in 24 hours (or less) because of the RAIDS effect.

A second important result of RAIDS is that the topical antibiotic kills all harmful Gram positive and Gram negative bacteria that have been available for testing. We believe this is the world’s strongest broad-spectrum topical antibiotic available.
The potential commercial impact is immense. Drug developers believe it takes much longer for bacteria to develop drug resistance to a physical kill mechanism. This is because it is relatively easy for bacteria to change their response to a chemical threat, but it takes numerous generations for bacteria to grow a new kind of cell wall structure to respond to a physical threat.
Our novel approach to overcome drug resistance of antibiotics is designed to sustain the effectiveness of antibiotics and other topical drugs for many years. This gives new topical drug products a longer useful lifetime and therefore more commercial value. This technology, when licensed to larger pharmaceutical companies, may provide stronger incentive for the discovery and development of new antimicrobial drugs. In recent years, the dearth of new antibiotics has been largely due to the uncertain new-drug commercial lifetime which is diminished when bacteria develop immunity to that drug.
In addition the technology can be licensed to other companies to convert existing oral drugs to transdermal medications extending the profitably of the existing drug.


We are developing products in the following fields of use;
Treats all types of bacteria including MRSA, VRE and other flesh eating bacteria. Fights drug resistant bacteria. $6B/year global market.

Onychomycosis, a fungal infection of the toenails, is a major health problem. It is estimated that there are in excess of 40 million sufferers with this condition in the USA. It is a problem throughout the world. A recent European study showed that the prevalence of Onychomycosis may be as high as 26.9%. Fungal resistance can occur when the oral antifungal agents are used on a long-term basis. Topically applied antifungal drugs may work somewhat better adjunctive to surgical removal or chemical dissolution of the nail plate. Yet, this often ineffective and traumatic procedure leaves the subject without a nail for months at risk for re-infection. Given the limitations of current treatment options in this $3B market, there is a great need for a simple, nontoxic and effective alternative treatment. Estimated Global prevalence rate: 140 million.
Treats symptoms of Influenza which is a$4B/ year global market: Common
Diabetic foot wounds.

Per 3/8/18 PR: ViaDerma’s technology is currently being used in Elixr Cannabis products; Topical Balm, Topical Serum and Topical Spray. Sales have begun in Canada.
Per 12/7/17 PR: The Company has signed an MOU and will start production of its Patent Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent #62466209, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent # 62433964 for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinary uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments.
Per 1/23/18 PR: The Company has also a licensing and distribution agreement in place with its Canadian partners to produce a CBD Topical Serum, which has a 92% absorption rate powered by ViaDerma's Patent Pending Dual Carrier Technology. The CBD and Terpene profile (enriched with specific essential oils and vitamins C, and D) is crafted to alleviate Chronic Dry Skin, Psoriasis, Eczema, Rosacea, and many other skin conditions. The serum has anti-inflammatory, anti-bacterial, and chronic pain relief properties that are absorbed by the skin and provide overall healing benefits. Sales are expected to begin in the first quarter of 2018 and are projected to generate approximately $2 million annual revenues.
The Patented Pending CBD or Cannabinoids product line. The Company would combine its two Patent pending technologies, the MMJ Patent, a patent for delivering medical marijuana / cannabis to the body by applying the medication onto the skin in an ointment base topical solution and the second Provisional patent for enhanced antibiotic and drug delivery for "Aqueous Topical Applications" for human and veterinarian uses. The Company would combine its proprietary solutions with CBD's and other natural products that treat psoriasis, fibromyalgia for pain and other ailments. The Company plans to continue to expand its (IP) "Intellectual Property Portfolio" in 2018.

CBD is a compound found in the Cannabis family of plants such as hemp. CBD is combined into our topical body care products like Elixr's Topical Balm, Serum, and Spray.
CBD is an antioxidant. Antioxidants protect our skin from damaging exposure to smoke, UV rays, and environmental pollutants. Cannabinoid receptors are located all throughout the skin, Topical CBD products interact with those receptors resulting homeostasis and healing. Studies show that CBD can help treat an array of skin conditions such as eczema, psoriasis, rosacea, and acne.
| "What is Intellectual Property?" - A Publication by the World Intellectual Property Organization -
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
