Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Ur fine...much can happen here in short term, that news and vol will lure new $$$ and some big pharma will be watching..
cashing in some subs near the HOD while using the proceeds to sit bid seems to have been a good choice.
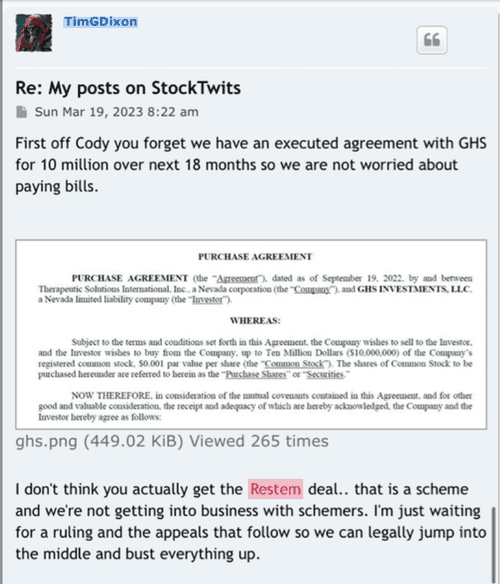
GL to us ALL as another question was finally answered, well part of it. All about the details of said agreement
Bought at 0.0190 and now down to 0.0154….smh
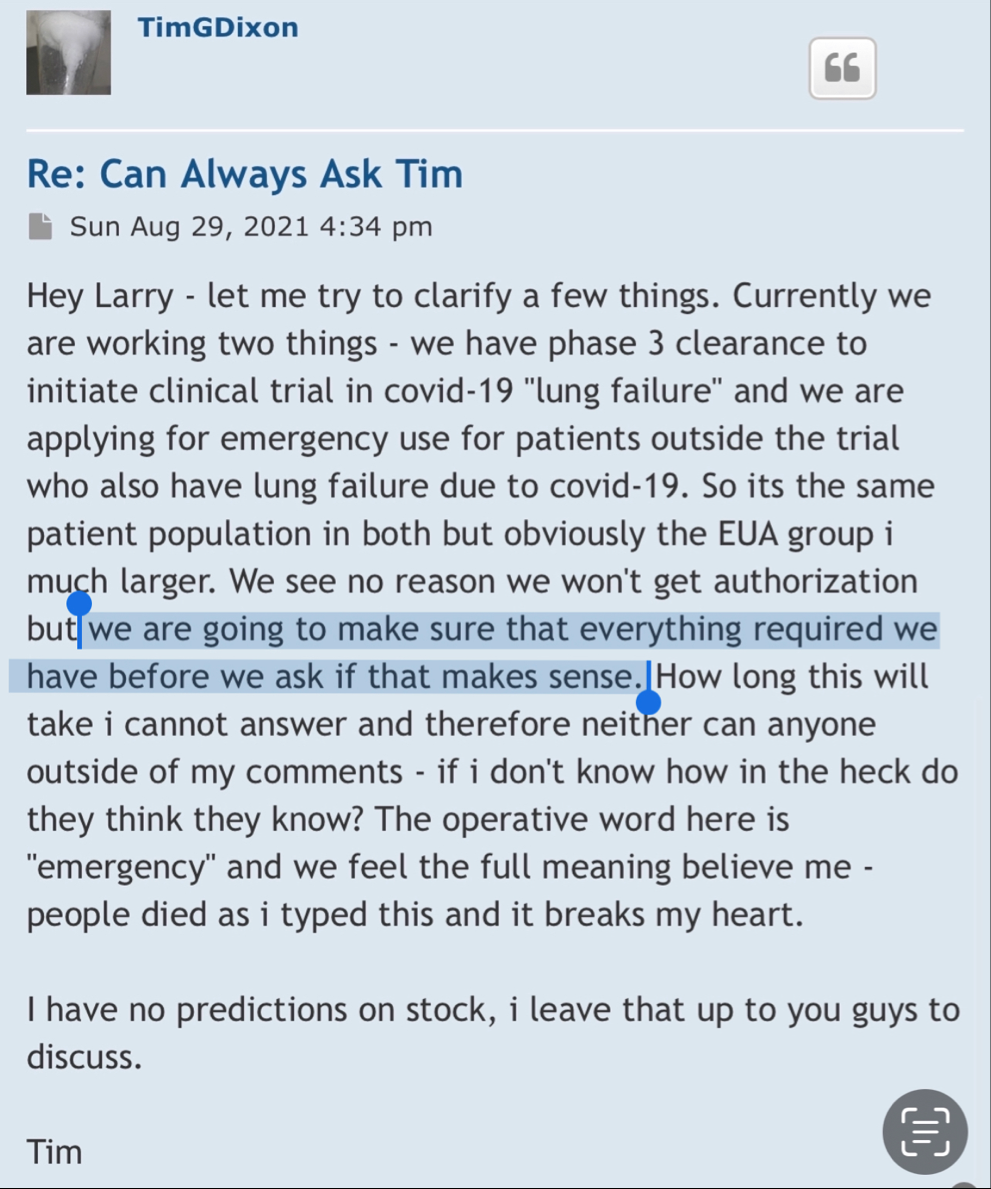
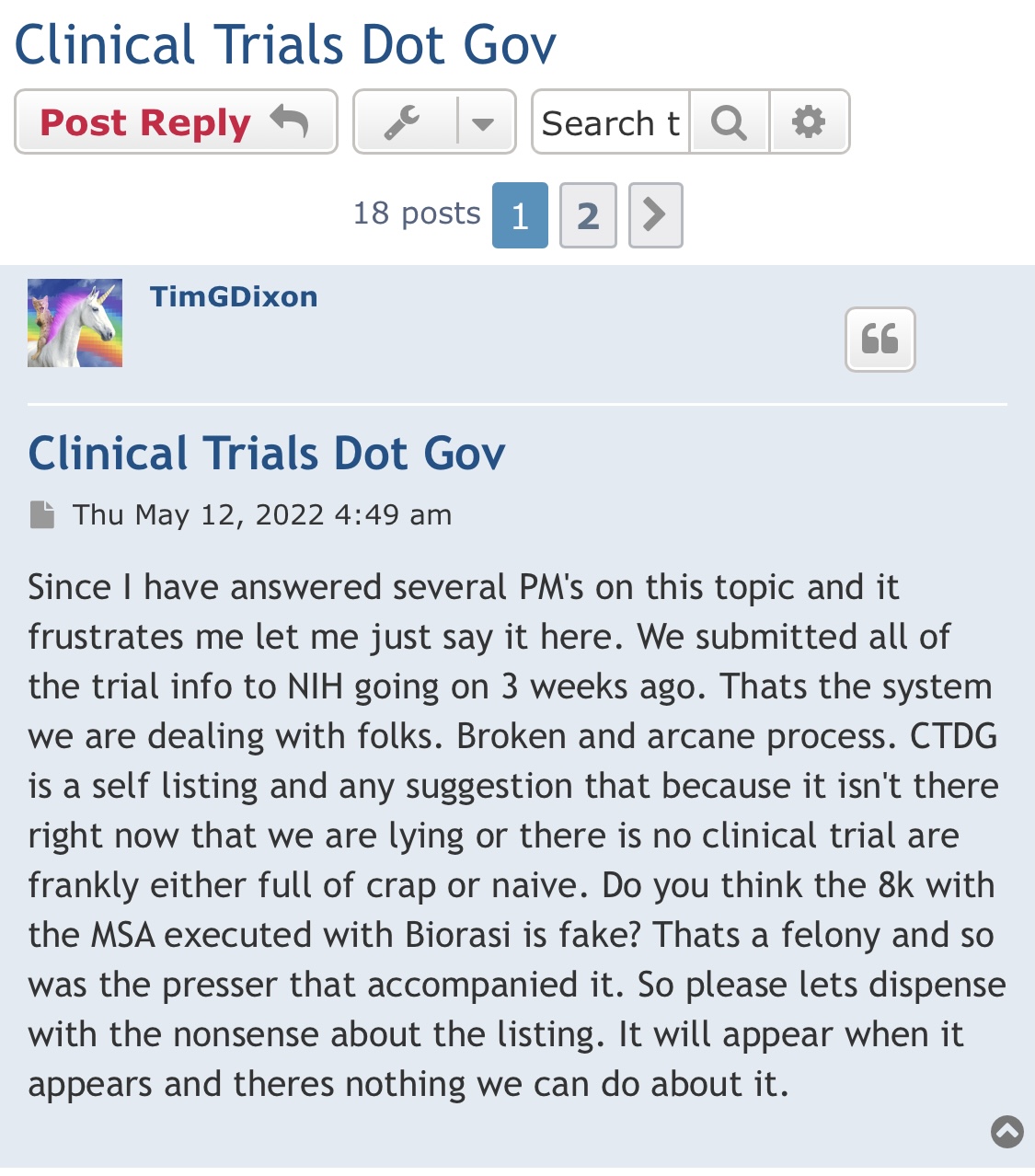
It is GREAT news that Tim has now gone public with HOW he will fund Phase 3 of the FDA clinical trials. This has always been an open issue and concern for me. Congratulations to Tim for keeping the shareholders informed !!!
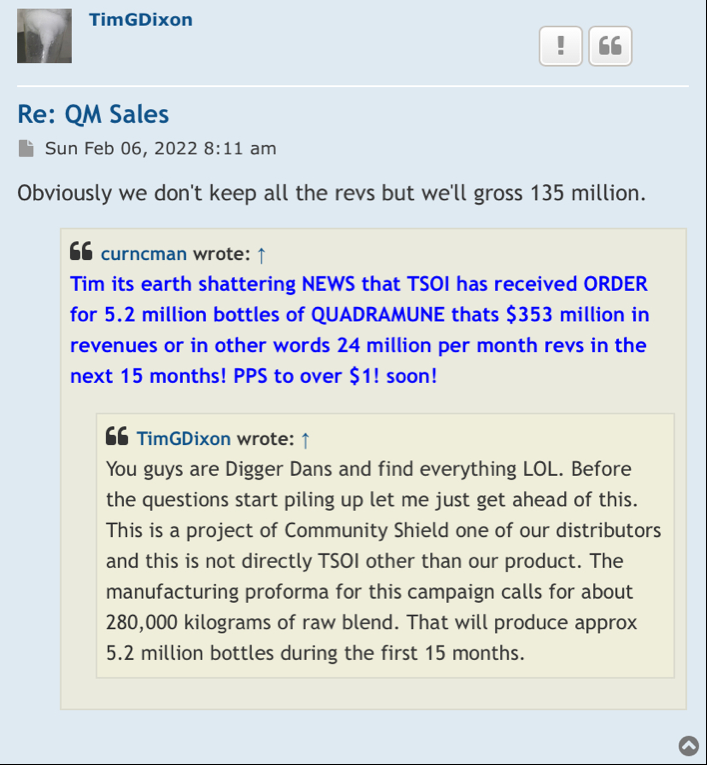
TSOI what should the market cap be for a company that is in Phase III Trial and funded?
![]()
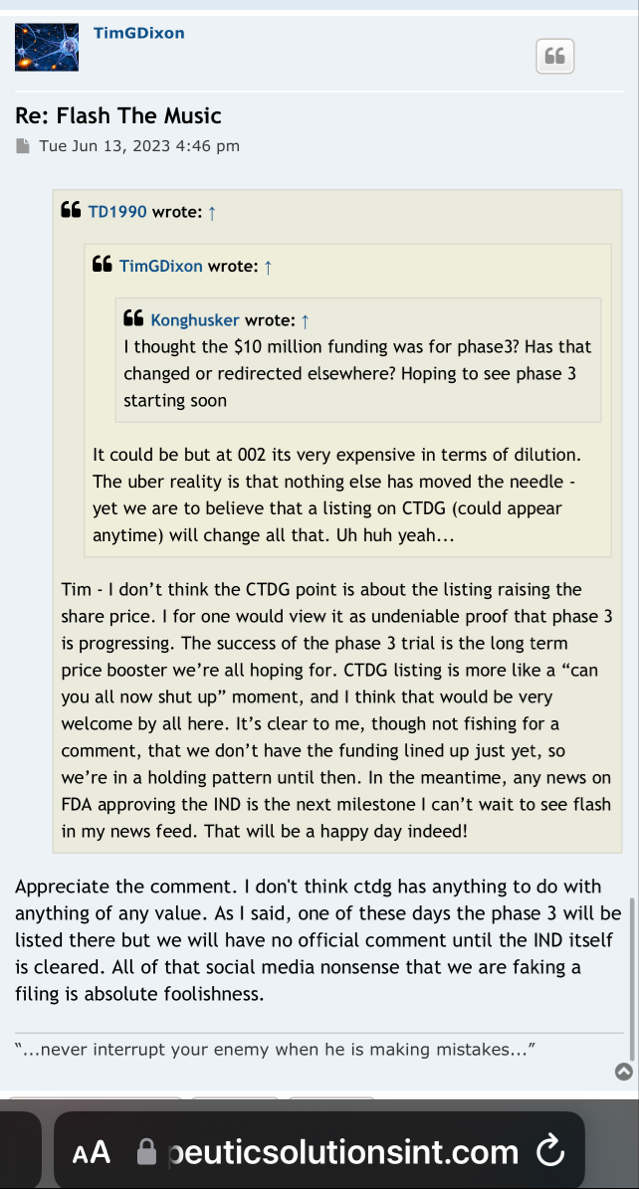
Tripping here may cross 02 huge got funding for phase III clinicals
TSOI Therapeutic Solutions International Inc () 0.017625
0.007525 (74.50%)
Volume: 13,643,191
Day Range: 0.0101 - 0.0192
TSOI MM's shaking it hard, lets see how thin to .02+ now
TSOI Lets see if .02 gets annihilated ![]()
TSOI He He, getting some Whales attention yet?
BoooooooYAAAAAAAAAAA!!!
TSOI might see a Turbo Boost after .016, imo $$$$$
TSOI Bid support coming in, lets break .015 HOD
Good TSOI morning subs, great news out this morning ![]()
https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2328196
Been nice accumulating shares all along the way...Those who sold, thanks!!! I got a nice stash!
Funding should attract much larger investors as News disseminates over the next few days, $$$$, imo
TSOI
Yessir morgan & so much for all the uneducated noise foolishly claiming that Biorasi was supposedly not doing the trials, as there's no mistaking what the headline itself states for those who actually understand all the inherent Phase III trial costs involved in Biorasi's completion over the coming months!
Hopefully we get 6 month trial results prior to EOY woot woot>>>
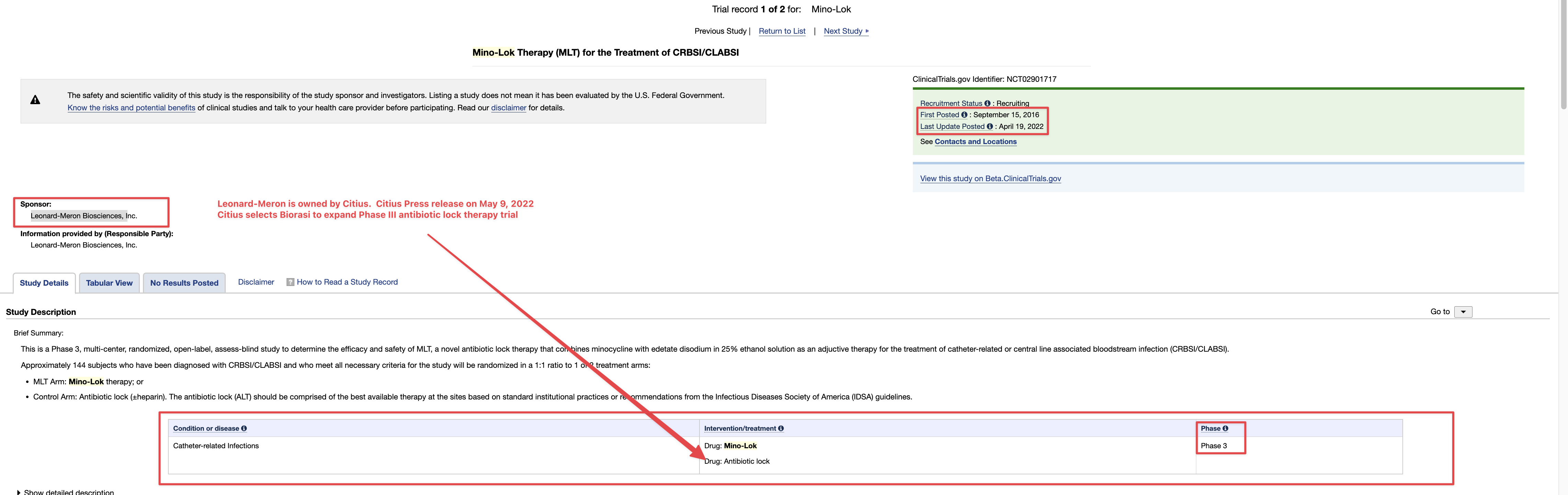
Therapeutic Solutions International Secures Financing to Complete Current Phase III Stem Cell Clinical Trial
That right Doog! Non-Toxic Funding..This isn't a here, I'll give you this money...Its here, I'm gonna INVEST in your company by PURCHASING $10mm in stock...Sounds to me like their is some profound factual strength in this company. Love it...
Great News, congrats to all the TSOI team. Acceleration time, $$$$, imo
TSOI lookin green doog, this Good Tuesday Morning, up over 40% already ![]() with good news out - https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2328196
with good news out - https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2328196
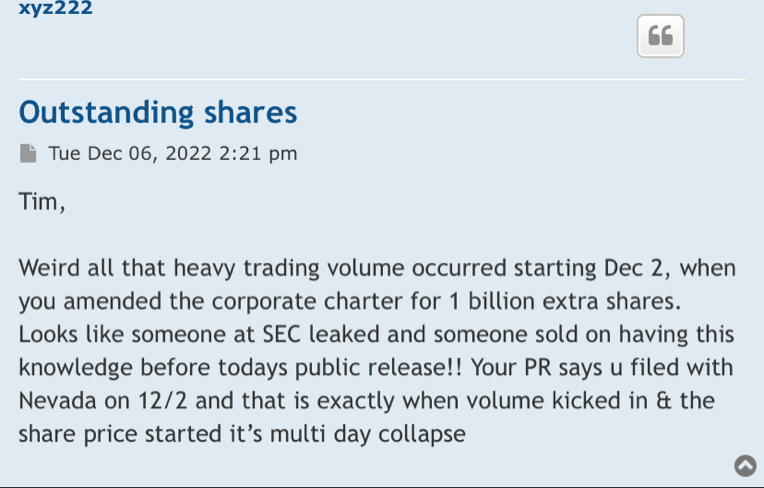
absolutely Held at DTC 1,350,524,672 09/07/2022
while awaiting next T/A update
yea
1,350,524,672 is what matters its what is out in the float
ELK CITY, Idaho--(BUSINESS WIRE)-- Therapeutic Solutions International (TSOI), a clinical stage regenerative medicine and immunotherapy company, announced today it has entered into an agreement with GHS Investments, LLC ("GHS") to purchase up to $10,000,000 of Registered Common Stock.
Outstanding Shares 2,565,802,627 09/07/2022
Authorized Shares 3,500,000,000 09/07/2022
Transfer Agent Verified
YET MORE TSOI BULLSHIT: No legitimate Medical "Scientist" employs terminology like this in a press release!!! NOR would a REAL Medical Scientist even launch a stupid PR to announce that they now have the mere FUNDING to perform their so-call "FAR-REACHING" research --- with MICE, or in a petri dishes again????
"The implications of this trial are far-reaching ..." (In FACT, they are NO SUCH thing, Scamming "Company"!!)
/////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////
agreed sad the OS isnt lower ugh
A lookback provision bases the purchase price not on the stock price at the time of purchase but, rather, on the price either at the beginning of the offering period or at the end of the purchase period, whichever is lower.
just keeping it real & imho positive considering
current pps low 01s & hopefully rising prior to the beginning of the offering period, yet for now we do not seem to have that info
The implications of this trial are far-reaching in that success will open the door for entry into other pulmonary indications including ‘long COVID’ which remains a significant health problem.”
Awesome news PennyWorld, now we'll see how things will go.
Therapeutic Solutions International Secures Financing to Complete Current Phase III Stem Cell Clinical Trial
Company Announces $10,000,000 Common Stock Purchase Agreement with GHS Investments, LLC.
ELK CITY, Idaho--(BUSINESS WIRE)-- Therapeutic Solutions International (TSOI), a clinical stage regenerative medicine and immunotherapy company, announced today it has entered into an agreement with GHS Investments, LLC ("GHS") to purchase up to $10,000,000 of Registered Common Stock.
The agreement is on a look-back basis and there are no make-up provisions, a true non-toxic equity investment. GHS is a leading private investment and management group providing financial solutions for high potential small cap enterprises. GHS has made investments into multiple successful portfolio companies in the nano-market cap space that are seeking capital to measurably grow and scale their existing businesses to take advantage of emerging market sectors.
“This financing will allow the acceleration of our current Phase III clinical trial assessing efficacy of JadiCells, a novel adult stem cell, for treatment of COVID-19 associated lung injury,” said Timothy Dixon, President, and CEO of Therapeutic Solutions International. “The implications of this trial are far-reaching in that success will open the door for entry into other pulmonary indications including ‘long COVID’ which remains a significant health problem.”
Therapeutic Solutions International has 66 filed and issued patents, multiple IND’s covering Chronic Traumatic Encephalopathy (CTE), Acute Respiratory Distress Syndrome (ARDS), Chronic Obstructive Pulmonary Disease (COPD), and solid tumor Cancer’s as well as numerous preclinical and pilot clinical data around immunotherapy, oncology, and stem cell therapeutics.
About Therapeutic Solutions International, Inc.
Therapeutic Solutions International is focused on immune modulation for the treatment of several specific diseases. The Company's corporate website is www.therapeuticsolutionsint.com.
https://cts.businesswire.com/ct/CT?id=bwnews&sty=20220920005776r1&sid=acqr8&distro=nx&lang=en
View source version on businesswire.com: https://www.businesswire.com/news/home/20220920005776/en/
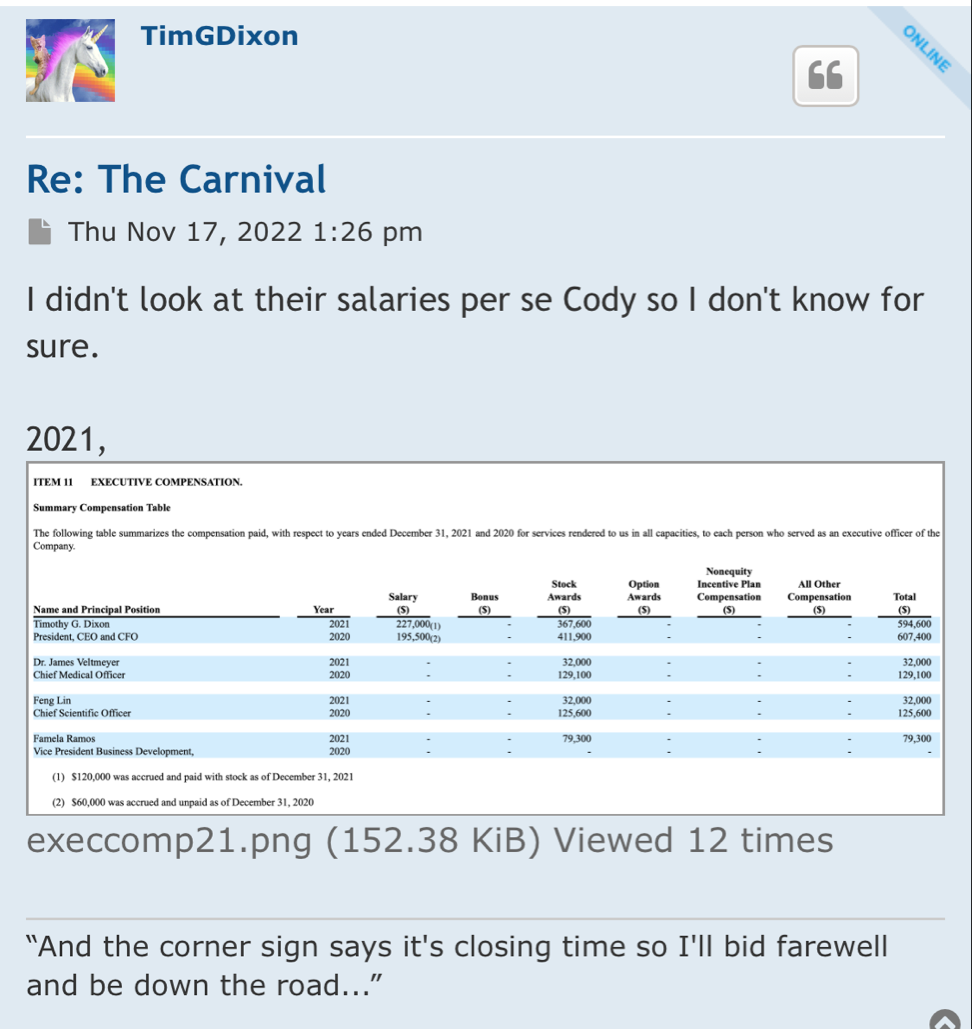
Timothy G. Dixon
ir@tsoimail.com
Source: Therapeutic Solutions International, Inc.
https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2328196
that's some great news !
How many OTC Pubco's ever remotely position themselves to get this kind of quality friendly financial backing right bb:)
The agreement is on a look-back basis and there are no make-up provisions, a true non-toxic equity investment.
nice funding for phase III https://www.otcmarkets.com/stock/TSOI/news/story?e&id=2328196
BusinessWire, GlobeNewswire and PR Newswire News
Therapeutic Solutions International Inc TSOI:OTC Pink - Current Information
Therapeutic Solutions International Secures Financing to Complete Current Phase III Stem Cell Clinical Trial
BusinessWire
9:00 AM ET
Therapeutic Solutions International (TSOI), a clinical stage regenerative medicine and immunotherapy company, announced today it has entered into an agreement with GHS Investments, LLC ("GHS") to purchase up to $10,000,000 of Registered Common Stock.
The agreement is on a look-back basis and there are no make-up provisions, a true non-toxic equity investment. GHS is a leading private investment and management group providing financial solutions for high potential small cap enterprises. GHS has made investments into multiple successful portfolio companies in the nano-market cap space that are seeking capital to measurably grow and scale their existing businesses to take advantage of emerging market sectors.
"This financing will allow the acceleration of our current Phase III clinical trial assessing efficacy of JadiCells, a novel adult stem cell, for treatment of COVID-19 associated lung injury," said Timothy Dixon, President, and CEO of Therapeutic Solutions International. "The implications of this trial are far-reaching in that success will open the door for entry into other pulmonary indications including 'long COVID' which remains a significant health problem."
Therapeutic Solutions International has 66 filed and issued patents, multiple IND's covering Chronic Traumatic Encephalopathy (CTE), Acute Respiratory Distress Syndrome (ARDS), Chronic Obstructive Pulmonary Disease (C OPD), and solid tumor Cancer's as well as numerous preclinical and pilot clinical data around immunotherapy, oncology, and stem cell therapeutics.
About Therapeutic Solutions International, Inc.
Therapeutic Solutions International is focused on immune modulation for the treatment of several specific diseases. The Company's corporate website is www.therapeuticsolutionsint.com.
View source version on businesswire.com: http s://www.businesswire.com/news/home/20220920005776/en/
SOURCE: Therapeutic Solutions International, Inc."><Property FormalName="PrimaryTwitterHandle" Value="@TSOISupplement
<img alt="" src="https://cts.businesswire.com/ct/CT?id=bwnews&sty=20220920005776r1&sid=cmtx6&distro=nx&lang=en"; style="width:0;height:0" />
Timothy G. Dixon ir@tsoimail.com COMTEX_414870123/1006/2022-09-20T09:00:09
EDUCATE YOURSELF
Let me explain how an experiment work.
COMMON SENSE
Every Biopharma since very long long time ago, after they medicated a human with certain medicine and that human died, many years ago, the Entity in charge of the pharmaceutical approvals decided to use rats for experiments? Why? Because the life of a human being is consider more valuable.
LETS START!!!
So how i know a medicine works in a rat???
Thru induction ( this is just one of several methods). When you induce some kind of chemicals into an animal which will cause the condition you are targeting, then the game is on.
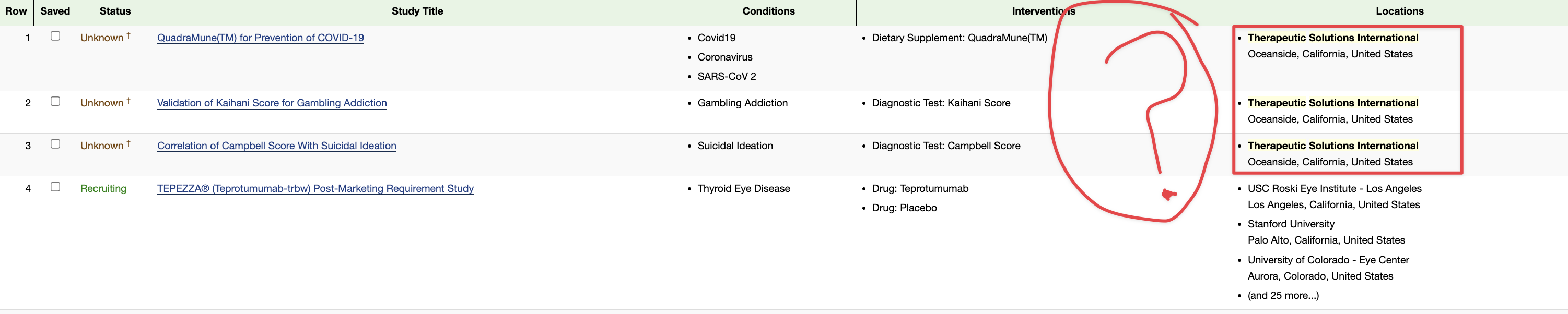
Once you prove that the rat is infected, then you can try your method to see if it does works or not. For example, let see some extract from the TSOI US Patent for Nutraceuticals for suppressing indolamine 2,3 deoxygenase Patent (Patent # 11,229,674)
Type: Grant
Filed: Oct 23, 2020
Date of Patent: Jan 25, 2022
Assignee: Therapeutic Solutions International, Inc. (Oceanside, CA)
Inventors: Thomas E. Ichim (Oceanside, CA), Timothy G. Dixon (Oceanside, CA), James Veltmeyer (Oceanside, CA)
Primary Examiner: Aaron J Kosar
Application Number: 16/901,028
EXAMPLES ( this is how the experiment was conducted)
Example 1: QUADRAMUNE™ Preserves Memory in Inflammation Associated Memory Decline Model
Female BALB/c mice were treated with control, daily LPS treatment, and some with LPS and QUADRAMUNE™.QUADRAMUNE™ was administered daily by gavage at a concentration 1 (100 ug of broccoli sprout extract, Nigella sativa, and green tea extract, and 50 ug of pterostilbene), and concentration 2 (200 ug of broccoli sprout extract, Nigella sativa, and green tea extract, and 100 ug of pterostilbene).
To assess memory function, water filled basin which was 120 cm in diameter was broken into 4 quadrants. 10 cm diameter platform placed 1 cm below water. Mice were forced to swim to find the hidden platform, starting from all four different quadrants, each day for 7 days. The time was recorded if they could find the hidden platform in 60 s . . . . If not, mice are guided toward the platform and allowed to stand on it for 10 s. Results are shown in FIG. 1.
Example 2: QUADRAMUNE™ Reduces Inflammation Associated Kynurenine Elevation
Blood samples were collected from mice in Example 1 and assessed for Kynurenine content. Augmented levels of kynurenine were observed in LPS treated mice as compared to controls. Results are shown in FIG. 2.
QUADRAMUNE HAD BEEN SCIENTIFICALLY PROVEN TO HAVE GREAT RESULTS. THIS ARE THE CLAIMS
:
Claims
1. A method of inhibiting indolamine 2,3 deoxygenase (IDO) expression or activity, comprising: administration to a patient in need a therapeutic combination comprising: a) Green Tea and/or extract thereof; b) Blueberry and/or extract thereof; c) Nigella sativa and/or extract thereof; and d) broccoli and/or extract thereof.
2. The method of claim 1, wherein said green tea extract is epigallocatechin-3-gallate or an analogue thereof.
3. The method of claim 1, wherein said blueberry extract is pterostilbene or an analogue thereof.
4. The method of claim 1, wherein said Nigella sativa extract is thymoquinone or an analogue thereof.
5. The method of claim 1, wherein said broccoli extract is sulforaphane or an analogue thereof.
6. The method of claim 1, wherein inhibition of IDO expression and/or activity in the host is associated with enhancement of natural killer cell activity.
7. The method of claim 6, wherein said natural killer cell activity is quantified by ability to lyse a virally infected cell.
8. The method of claim 6, wherein said natural killer cell activity is quantified by ability to lyse K562 cells.
9. The method of claim 6, wherein said natural killer cell activity is quantified by ability to lyse YAC-1 cells.
10. The method of claim 1, wherein inhibition of IDO expression and/or activity in the host is associated with enhancement of interferon production.
11. The method of claim 1, wherein inhibition of IDO expression and/or activity in the host is accomplished by enhancement of T cell activation.
12. The method of claim 11, wherein said T cell activation is induction of T helper cell 1 activity.
13. The method of claim 12, wherein said T helper cell 1 activity comprises production of interferon gamma.
14. The method of claim 11, wherein said T cell activation is induction of T cytotoxic cell activity.
15. The method of claim 1, wherein said patient is suffering from a tumor, and said therapeutic combination is administered at a dosage and frequency sufficient to suppress growth of a tumor.
16. The method of claim 15, wherein said tumor growth in the host is associated with suppression of cancer angiogenesis.
17. The method of claim 1, wherein said patient is suffering from COVID 19.
18. The method of claim 1, wherein the patient is suffering from memory loss.
19. The method of claim 1, wherein the patient has elevated levels of Kynurenine
AND I INVITE YOU TO THE JUSTIA SITE TO FIND HOW THE EXPERIMENTS WERE CONDUCTED SUCCESSFULLY FOR EVERY PATENT.
DO YOU SEE HOW TSOI IS WORKING TO IMPROVE THE LIFE QUALITY OF MILLIONS OF PEOPLE THAT COULD BE OUR RELATIVES?
Please do your own DD.
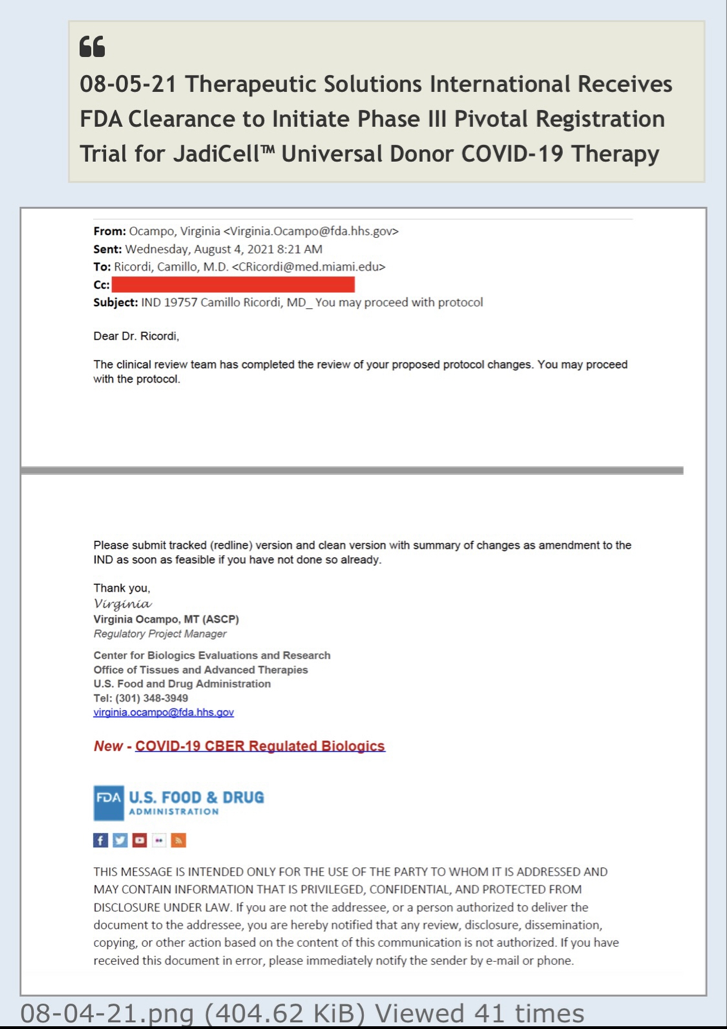
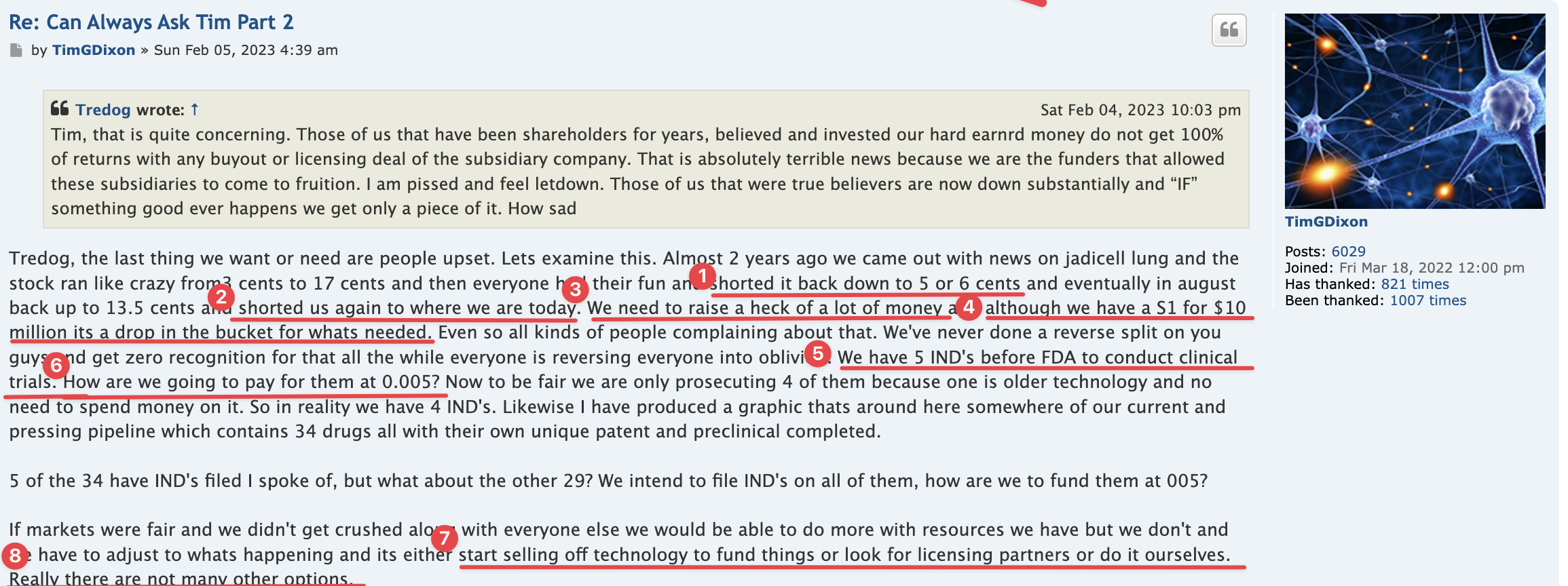
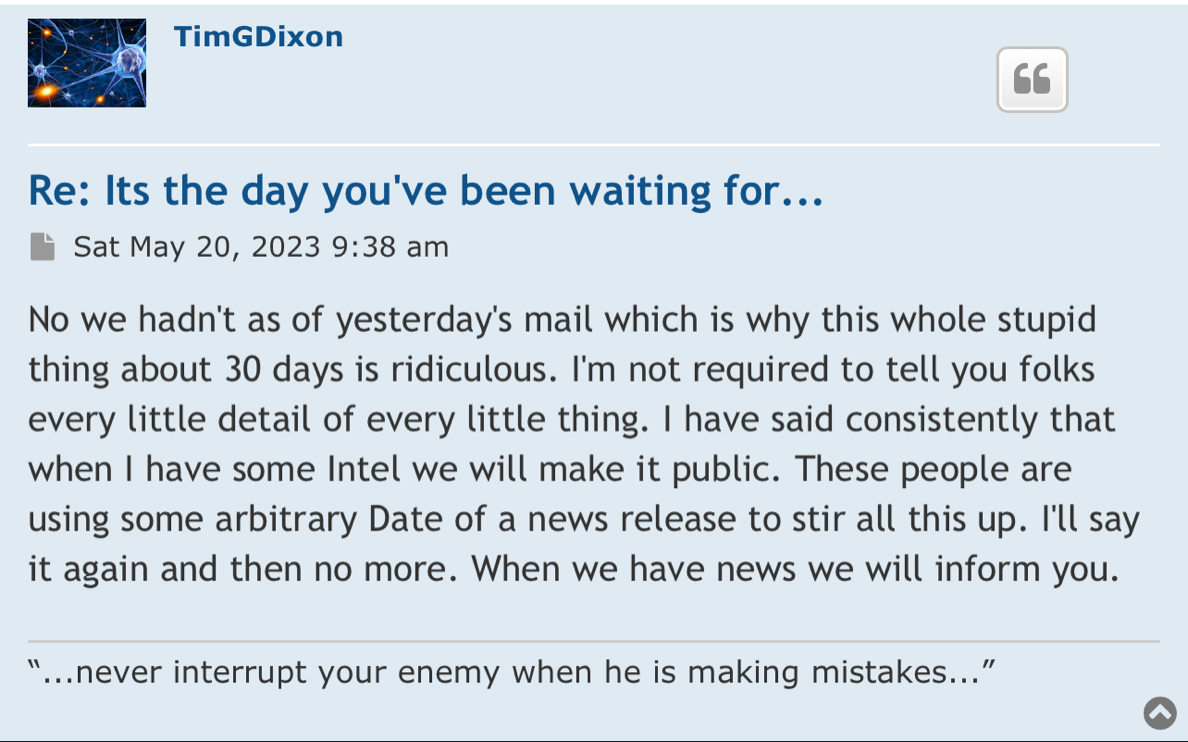
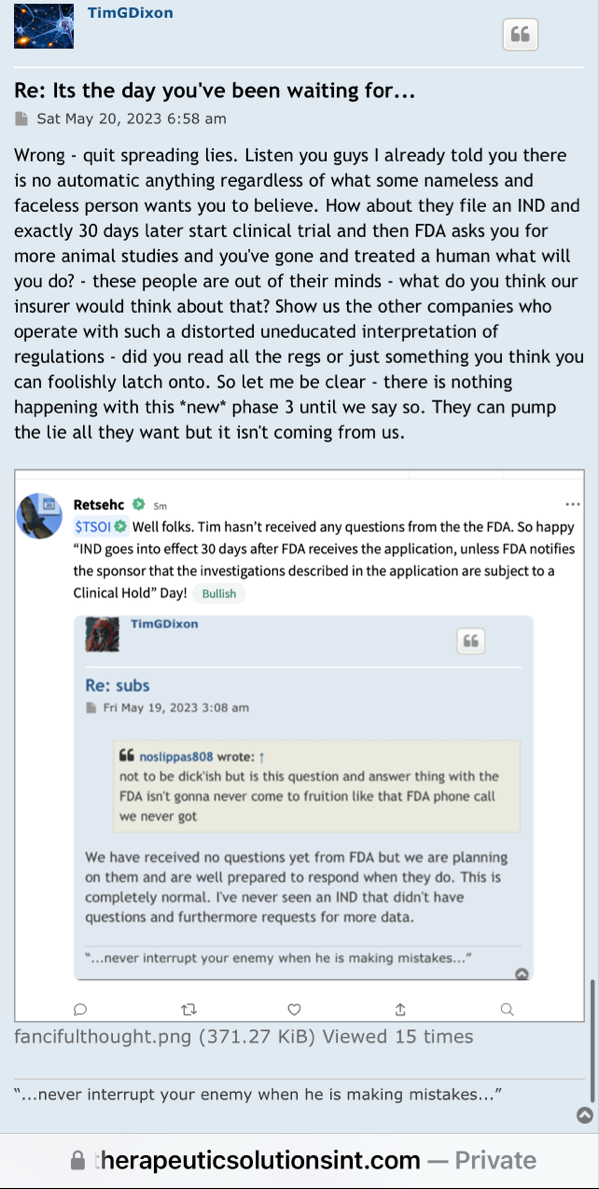
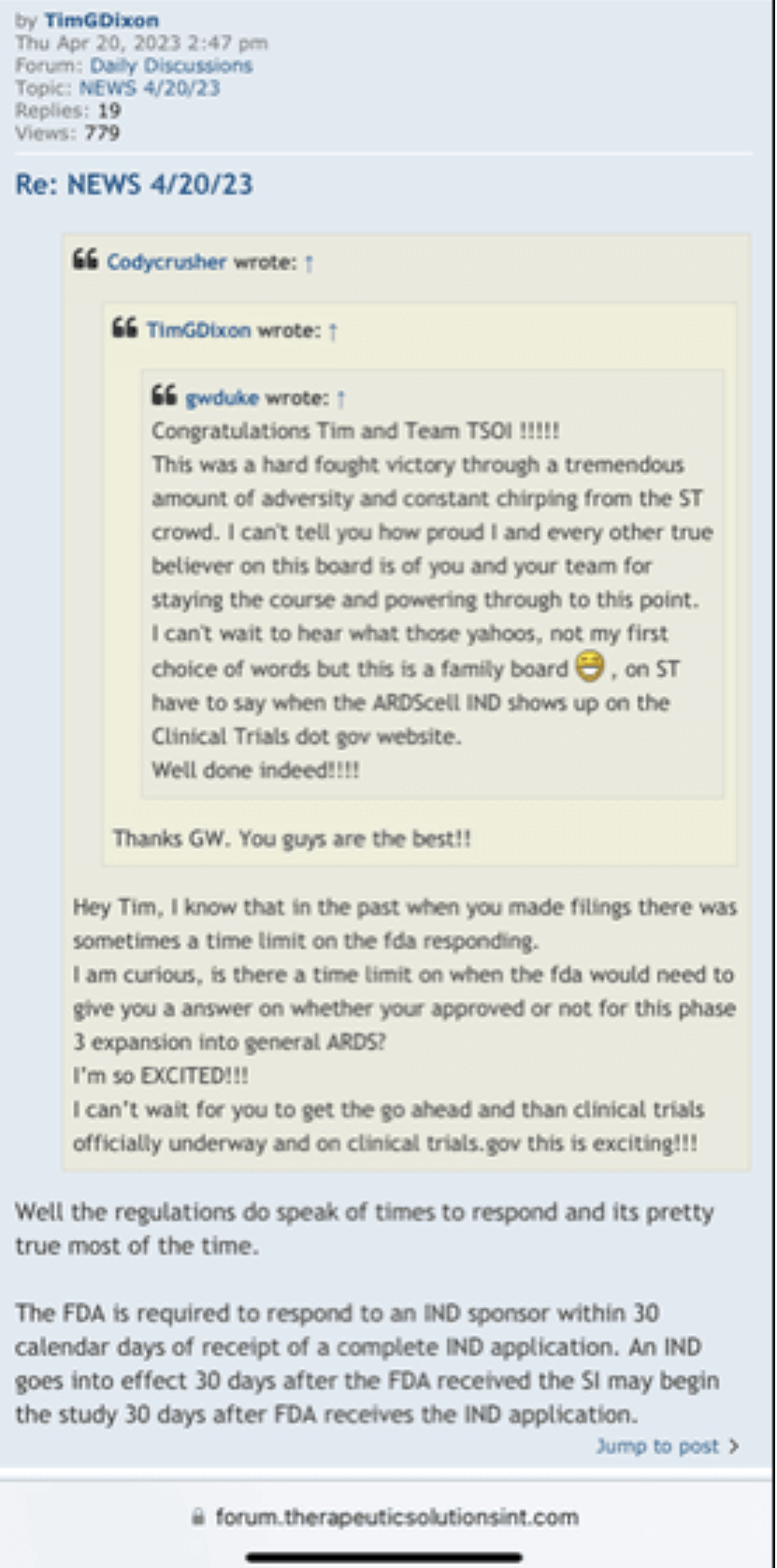
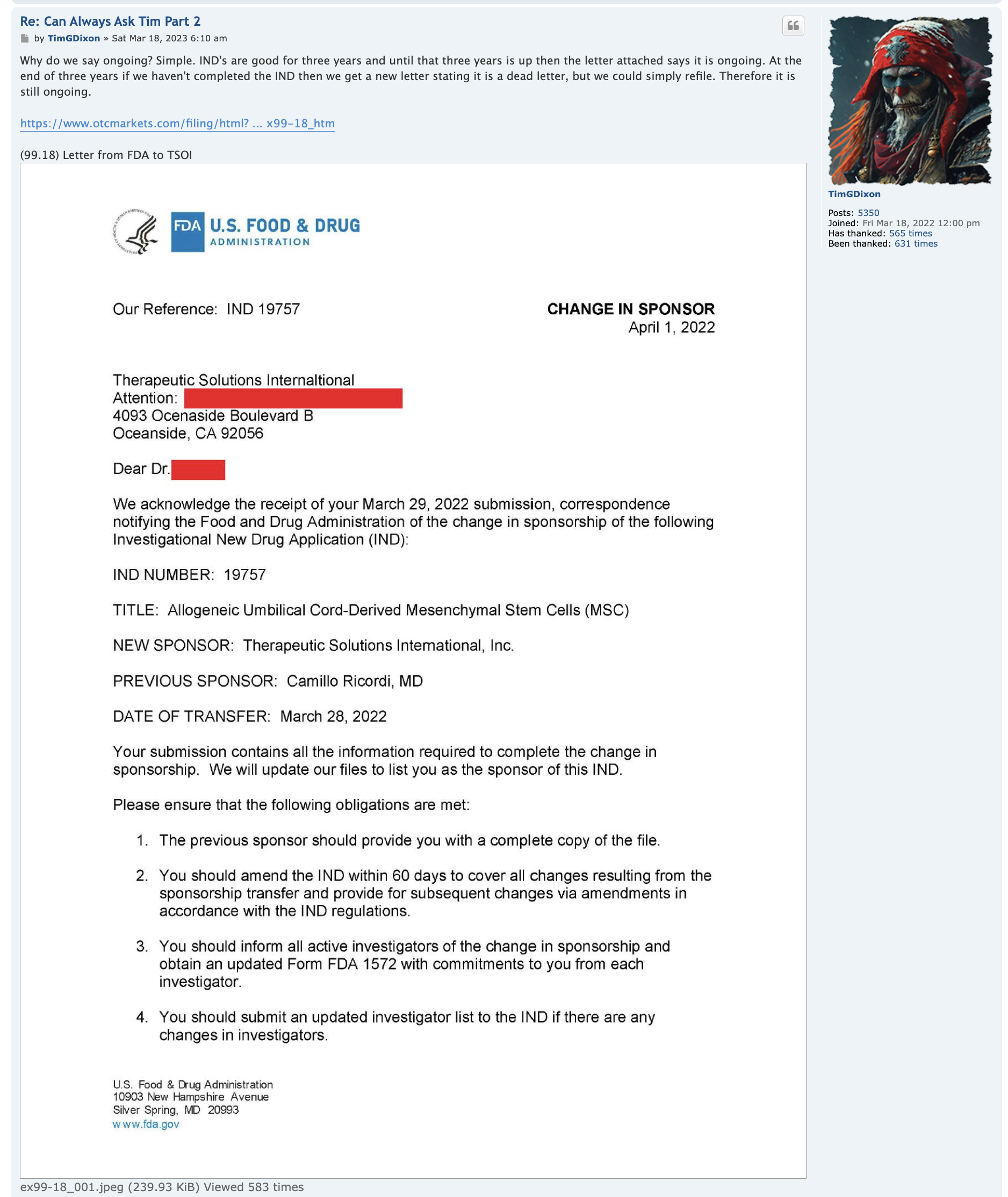
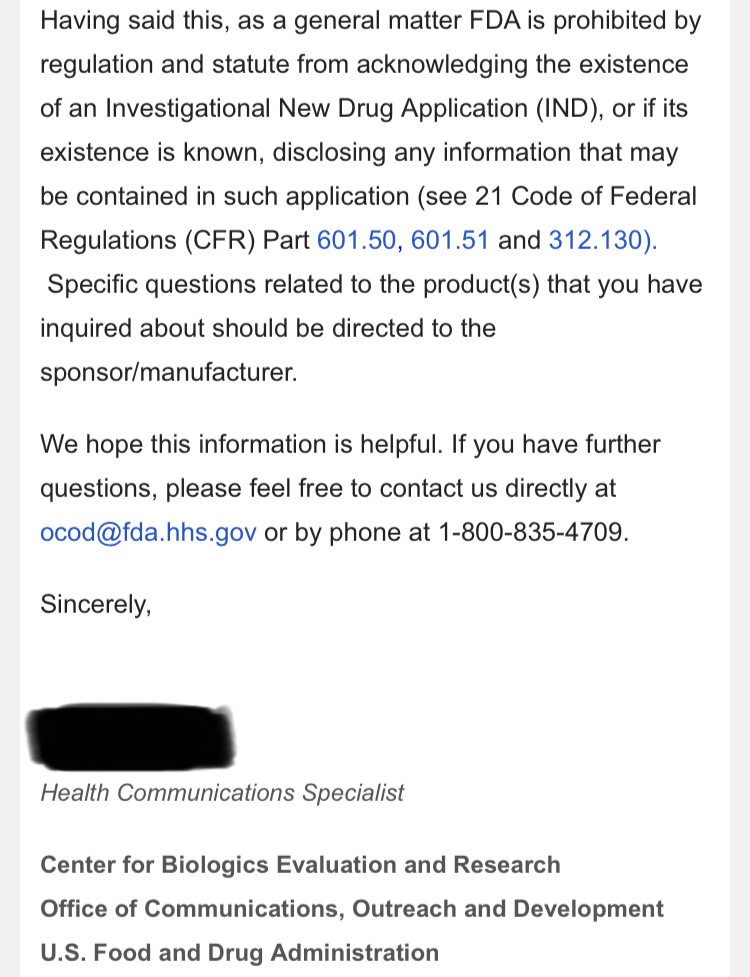
Regarding FDA Response Time for Re: NEWS 8/25/22
https://www.fda.gov/drugs/investigational-new-drug-ind-application/ind-application-reporting-protocol-amendments
https://www.businesswire.com/news/home/20220825005259/en/
NEWS 8/25/22 Therapeutic Solutions International Files with FDA to Expand ongoing Phase III Clinical Trial to Include All Causes of Acute Respiratory Distress Syndrome
https://www.businesswire.com/news/home/20220825005259/en/
ELK CITY, Idaho--(BUSINESS WIRE)--Therapeutic Solutions International announced today filing with the Food and Drug Administration (FDA) an amendment to its currently open Phase III clinical trial requesting expansion of eligible patients with all patients suffering from Acute Respiratory Distress Syndrome (ARDS) regardless of cause.
“We are honored to be guided by Key Opinion Leaders such as our SAB member Dr. Donald Banerji, former Head of Novartis Respiratory Medicine, during this process of taking JadiCell from an experimental research project into what appears to be a lifesaving medicine”
Tweet this
The Company is currently only recruiting patients with advanced ARDS caused by COVID-19. The newly proposed criteria will allow all patients suffering from this condition to be eligible for entry into the clinical trial.
“In my opinion as a physician, as well as our Scientific Advisory Board, and our various collaborators, we strongly feel that the existing safety and efficacy profile of the JadiCell warrants expansion to other patients suffering from ARDS besides the COVID-19 associated population,” said Dr. James Veltmeyer, Chief Medical Officer of the Company. “We are confident that this decision will not only accelerate completion of the clinical trial but will also allow for treatment of a significantly larger patient population.”
Before the COVID-19 outbreak ARDS was believed to be approximately 200,000 cases per year in the United States and represents a multi-billion-dollar market. Currently there are no curative interventions available for advanced ARDS.
“We are honored to be guided by Key Opinion Leaders such as our SAB member Dr. Donald Banerji, former Head of Novartis Respiratory Medicine, during this process of taking JadiCell from an experimental research project into what appears to be a lifesaving medicine,” said Timothy Dixon, President, and CEO of the Company. “Based on the positive progress being made in understanding the JadiCell at both a clinical and molecular level, I am optimistic that we will be given the opportunity to address the larger problem of ARDS.”
About Therapeutic Solutions International, Inc.
Therapeutic Solutions International is focused on immune modulation for the treatment of several specific diseases. The Company's corporate website is www.therapeuticsolutionsint.com.
Contacts
Timothy G. Dixon
ir@tsoimail.com
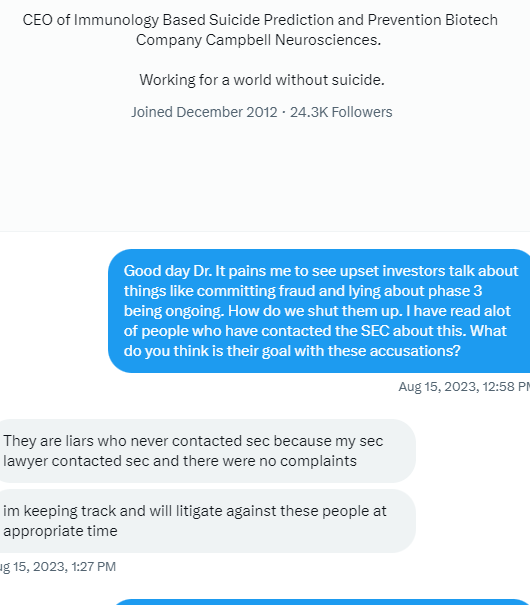
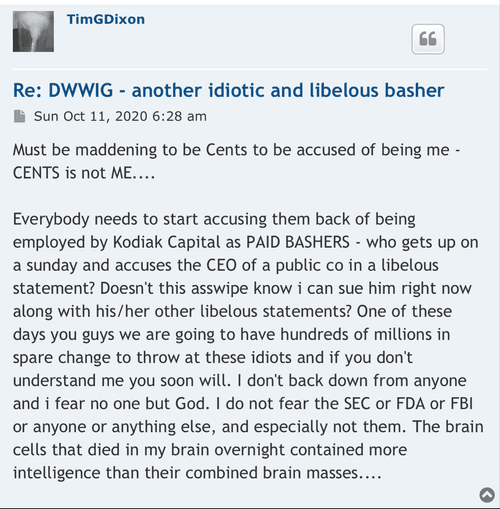
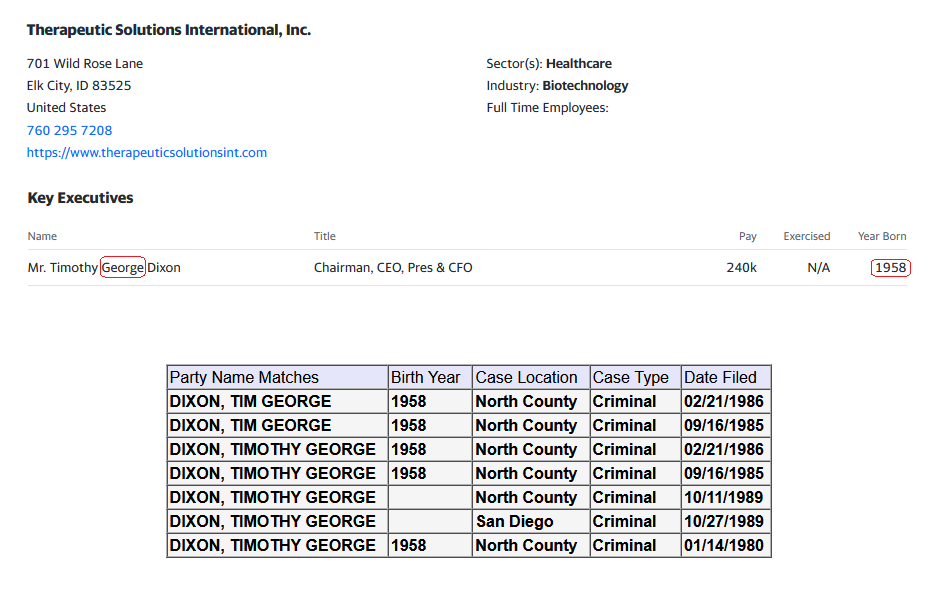
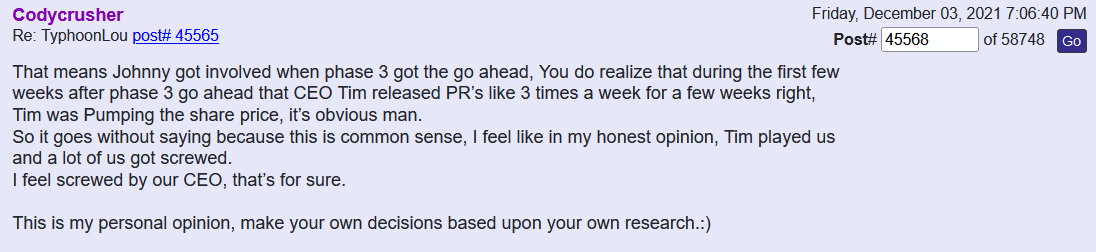
I am starting to see more new handles show up every day to post weather pro or con. But one handle that was showing up regularly I don’t see any more and that’s December to remember where is he gone better yet December of what year after all we were told we got the All-Star team we got this we got that this is being down that being then and hell what are we have now five dozen intellectual properties and patents that can barely hold a penny.
Many of us are asking the question now what exactly is wrong with this company. In my opinion I totally believe in the science of it so to me it Hass to be money and management. Just because a new PR comes out once in a while doesn’t mean jack shit to me cause nothings going to happen.
Is it this week the deadline that the FDA hast to announce to us at where we’re trying to incorporate COPD into the Covid phase 3 for some reason I thought they had to respond within 30 days and I thought this was the fourth week I might be wrong on the timing. But it’s obvious these PR‘s that’s put out even the spin off one that they were talking about today and blah blah blah may nothing right now down the line they might right now absolutely nothing.
In my opinion it’s quite a shame the shape that this company is in I do believe if we had a partnership this thing would be blossoming right now or a hell of a lot farther along than where we’re at deep down in the cellar.
Just like Mark Cuban said the other day 50% of a watermelon is a hell of a lot better than 100% of a grape and I totally believe that
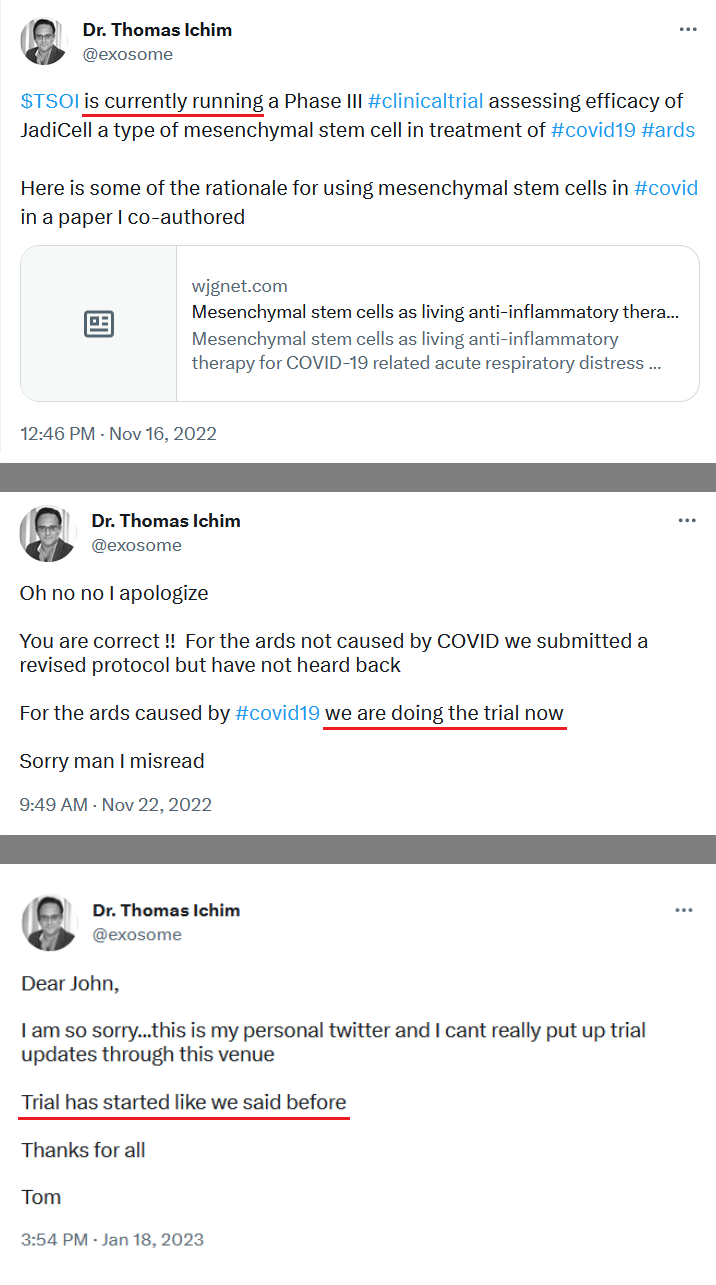
“Having used various stem cell therapies in my practice, including JadiCell, for which we are currently running a Phase III clinical trial, I have seen the potent effects this regenerative approach can have on many branches of medicine,” said Dr. James Veltmeyer, Chief Medical Officer of the Company, and co-inventor of the patent. “I am proud to be working with Ms. O’Connor and her team at Campbell Neurosciences in creating, what I believe to be, the first science-based approach to diagnosing and treating suicide as a disease and not a choice.”
Don’t you disrespect the mune! Supplements rule. Haha. I mean medical breakthroughs and we’re trading at a penny. SMH.
TSOI: Homo sapiens are NOT as dumb as TSOI Company thinks!! Planet Earth knows FULL WELL that even the SUGGESTION that (Bipolar) Mice/Rat studies would have any correllation whatsoever to Humans is patently ABSURD on its face!!! (This TSOI Company --- & @HALB to boot --- are the two most CONSPICUOUSLY SCAMMING firms in the entire history of this Planet!! If you own TSOI, well, then you surely own some @HALB too, right!!)
If you are so dumb as to believe that COMPLEX PSYCHIATRIC disorders --- e.g., Bipolar --- can be fixed with a PILL, then you would likely believe that CANCER, HEART DISEASE, THE AGING PROCESS itself, etc., can be cured easily with psychological COUNSELING!!!
But you still don't believe ME??? Well, then simply watch the RED PRICE of this TSOI crap!!! No matter WHAT kind of patently-stupid PR's they publish!!!
$TSOI Twitter chatter! https://twitter.com/search?q=%24TSOI&src=typed_query&f=live
“Having used various stem cell therapies in my practice, including JadiCell, for which we are currently running a Phase III clinical trial, I have seen the potent effects this regenerative approach can have on many branches of medicine,” said Dr. James Veltmeyer, Chief Medical Officer of the Company, and co-inventor of the patent. “I am proud to be working with Ms. O’Connor and her team at Campbell Neurosciences in creating, what I believe to be, the first science-based approach to diagnosing and treating suicide as a disease and not a choice.”
Therapeutic Solutions International Spin-Off Company Campbell Neurosciences Announces Successful Preclinical Studies of CampbellCell for Treatment of Bipolar Disorder
Validation of Efficacy for Second Suicide-Associated Psychiatric Indication Supports Push for Clinical Entry of Novel Brain-Healing Stem Cell Type
ELK CITY, Idaho--(BUSINESS WIRE)-- Therapeutic Solutions International announced positive data and filing of a patent covering the use of CampbellCell for treatment of Bipolar Disorder by the Company’s suicide prevention based spin-off Campbell Neurosciences.
In a series of animal experiments using an established animal model of bipolar disorder, superior inhibition of behavioral abnormality as measured in the “Open Field Test”, as well as reduction in brain inflammation was observed in animals treated with CampbellCell as compared to control stem cells. Furthermore, it was found that protection from behavioral abnormalities could be transferred to naïve mice by T cell infusions, indicating that an immunological effect was present.
“We initially demonstrated a biological basis to suicidality by our clinical trial demonstrating validity of the Campbell Score1,2. We subsequently demonstrated that the CampbellCell, which possesses potent regenerative and anti-inflammatory activities is capable of treating Schizophrenia in animal models3. Today we disclosed that the CampbellCell is also capable of treating bipolar disorder models,” said Kalina O’Connor, President, and CEO of Campbell Neurosciences. “We are advancing toward clinical trials in both Schizophrenia and Bipolar Disorder, which we believe will position the Company as the first to merge psychiatry with regenerative medicine.”
“Having used various stem cell therapies in my practice, including JadiCell, for which we are currently running a Phase III clinical trial, I have seen the potent effects this regenerative approach can have on many branches of medicine,” said Dr. James Veltmeyer, Chief Medical Officer of the Company, and co-inventor of the patent. “I am proud to be working with Ms. O’Connor and her team at Campbell Neurosciences in creating, what I believe to be, the first science-based approach to diagnosing and treating suicide as a disease and not a choice.”
Bipolar Disorder and Schizophrenia are recognized as major contributors to suicide and have been associated with various immunological abnormalities. For a review of the inflammatory nature of suicide-associated mental illness, the reader is referred to a paper published by Ms. O’Conner4.
“Campbell Neurosciences was capable of successfully raising capital, running two clinical trials for its diagnostic platform, and now has late preclinical data on two significant suicide-associated indications. This speaks volumes for our model of incubating technologies and spinning them off,” said Timothy Dixon, President, and CEO of Therapeutic Solutions International and co-inventor. “Through cross-fertilizing and sharing resources and talent between our companies, I am excited to see acceleration in clinical development and hopefully new treatments for this immensely important and underserved area of mental health.”
Rumor has it traders trader over on ST trying to panel a bunch of hotdogs and buns he’s afraid no picnic for 2023
Someone needs to tell him he could use them as catfish bait neck creek of his in Idaho running down the middle of the property for catfish
You to Pier, Anony, Transgender and Antisnakeoilsalesman! Let’s get back to work on Monday…. I can’t wait to hear more amusing stories about it being time to take action!
I remember all the dreamers with big dreams. Lol The dream turned into a nightmare lol
|
Followers
|
528
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
64546
|
|
Created
|
10/04/08
|
Type
|
Free
|
| Moderators BigBadWolf johnnytrader33 JMC$ Yooperman Hogwarts | |||













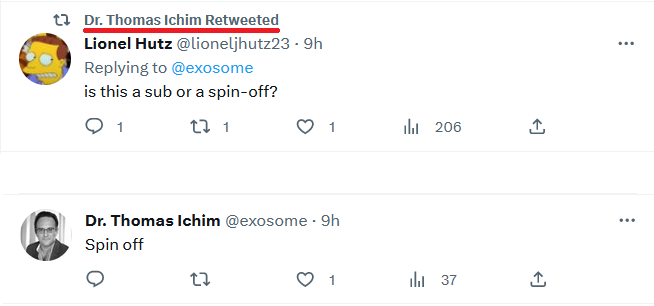










































Preclinical Data Suggests QuadraMune™ Prevents Stress-Induced Suppression of Neurogenesis More Effectively than Prozac
OCEANSIDE, Calif., Dec. 9, 2020 /PRNewswire/ -- Therapeutics Solution International, Inc., (OTC Markets: TSOI), announced today new data suggesting the possibility that QuadraMune™ may mediate neuroprotective activity through preserving the ability of regenerative brain cells to proliferate subsequent to psychological stress.
The experiments, which involved exposing mice to established stressors, demonstrated that specific areas of the brain associated with production of new brain cells are damaged by stress. In agreement with previously published research, administration of fluoxetine (Prozac™) protected the brain from stress-induced damage. Surprisingly, QuadraMune™ administration appeared superior to Prozac™ at stimulating proliferation of new brain cells.
"QuadraMune™ which is currently in a clinical trial for prevention of COVD-191, has also been demonstrated to possess anti-inflammatory activity in other clinical trials, suppressing cytokines such as IL-62, which are known to be involved in depression3 and suicide4" said Kalina O'Connor, Director of Campbell Neurosciences and co-inventor on the patent. "Given major depressive disorder causes a significant risk for suicide, we are highly interested in exploring the use of QuadraMune™ for preventing suicide."
"Although much enthusiasm has been generated over the planned distribution of the COVID vaccine, at present little is being done to address mental health issues that are being exacerbated by the current pandemic" said Dr. James Veltmeyer, co-inventor of the patent, and Chief Medical Officer of the Company. "If current results are reproducible, the possibility that a nutraceutical would concurrently boost immunity while preserving mental health is highly enticing."
"It has not escaped us that COVID-19 is associated with increased inflammatory cytokines in the blood of patients, cytokines that also predispose to depression" said Famela Ramos, Vice President of Business Development for the Company. "It may be that the recent increase in suicides and suicide attempts is related biologically to activities of the coronavirus. It will be interesting to examine whether QuadraMune™ may modify putative negative mental effects of the virus."
"An estimated 17.3 million adults in the United States had at least one major depressive episode. This number represented 7.1% of all U.S. adults" stated Timothy Dixon, President and CEO of the Company. "We believe the Mission of our Company is not just providing a return on investment to our shareholders, but also increasing the quality of life for Americans. We are extremely pleased to report this unexpected finding with significant potential implications to advancing non-toxic means of helping patients with this terrible condition."
1 QuadraMune(TM) for Prevention of COVID-19 - Full Text View - ClinicalTrials.gov
2 Therapeutic Solutions International Announces Positive Preclinical and Clinical Evaluation of Nutritional Supplement QuadraMune™, Designed to Protect Against COVID-19 | BioSpace
3 Ting et al. Role of Interleukin-6 in Depressive Disorder. Int J Mol Sci. 2020 Mar 22;21(6):2194.
4 O'Donovan et al. Suicidal ideation is associated with elevated inflammation in patients with major depressive disorder. Depress Anxiety. 2013 Apr;30(4):307-14.
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
