Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
SNGX not many sells but $3.06 on bid says they’ll pull it lower still….
GLTA
I think this company is fraud. At least I didn't put much here. Did they do a stock offering yet? if not it is coming...
Apparently they suckered you in. Hope you didn't buy this morning. This stock is dead.
And now refused so SNGX at .20 pre split
They did their RS. And then.... SLAM!!! they get a RTF from the FDA.
That's an OUCH IMO.
https://ih.advfn.com/stock-market/NASDAQ/soligenix-SNGX/stock-news/90234431/soligenix-receives-refusal-to-file-letter-from-u-s
Watching the LII's. I sold a bit ago. Like right around when they announced the RS. But I'll play this for the bounce.
I'll add... the timing of the news and the RS are suspicious. May just be my thinking.
SNGX +30% after hours…
What’s UP
GLTA
Yes. You have
Soligenix’s common stock outstanding from
43,335,174 shares to approximately 2,889,012 shares
Read my last few posts.
This spells out the way they stuck it to you even more clearly....
Proportionate adjustments will be made to the per share exercise price and the number of shares of Common Stock that
may be purchased upon exercise of outstanding stock options and warrants issued by the Company. The maximum number
of shares of Common Stock available for issuance under the Company’s 2015 Equity Incentive Plan will remain unchanged
at 6,000,000 shares. Similarly, the maximum number of shares of Common Stock for which stock options may be granted
to any person in any calendar year, the maximum benefit that may be paid to any person under performance awards in any
calendar year, and the aggregate number of shares of unrestricted stock that may be granted or sold for a purchase price that
is less than their fair market value (unless granted in lieu of cash compensation equal to such fair market value) will remain
unchanged, in each case at 200,000.
LOL....it's like watching the Comedy Channel where every show is the same...
As a result of the Reverse Stock Split, every 15 shares of issued and outstanding Common Stock will be automatically
combined into one issued and outstanding share of Common Stock, without any change in the par value per share. No
fractional shares will be issued as a result of the Reverse Stock Split. Any fractional shares that would otherwise have
resulted from the Reverse Stock Split will be rounded up to the next whole number. The Reverse Stock Split will reduce
the number of shares of Common Stock outstanding from 43,335,174 shares to approximately 2,889,012 shares, subject to
adjustment for the rounding up of fractional shares. The number of authorized shares of Common Stock under the
Certificate of Incorporation will remain unchanged at 75,000,000 shares
Found the answer myself and as suspected, another company screwing over the common shareholders....
At the effective time of the reverse stock split, every 15 shares of Soligenix's issued and outstanding common stock will be converted automatically into one issued and outstanding share of common stock without any change in the par value per share.
All authorized shares or just issued shares?
SNGX nice new OS less than 3,000,000 !
“The reverse stock split will reduce the number of shares of Soligenix’s common stock outstanding from 43,335,174 shares to approximately 2,889,012”
GLTA
Dang that may be same for me….
I can't recall if it was Kramer who first turned me on to this stock, but it has never been legit.
SNGX confirmed 1:15 gonna open tomorrw $6, and likely be back sub $2s within 2 weeks...
*Without confirmed positive movement in their pipeline*...
Skanks...
GLTA
Not sure I'd word it that way, but that's the gist. They won't convert the authorized but unissued shares.
Is it your opinion that the RS will pass because the insiders have or will have a greater percentage of the the Preferred Shares to vote?
What they promise and what they're actually doing, aren't the same.
Yeah $4.50 is likely after the 1:10 split...
SNGX assures news & progress 2023 so maybe that $4.50 post split will see $20....
GLTA
Maybe you should convince the insiders then.
1/09/2023 Watching Soligenix: Zacks Small-Cap Research Sets $4.50 Price Valuation 10:21 am ET January 9, 2023 (Benzinga)...
GLTA
...
...
.....
This has been quite the disappointment for sure.
The outstanding shares of Series D Preferred Stock will vote together with the outstanding shares of the Company’s common stock,
We'll see how this works out with the split coming. I've seen a couple RS's go ballistic lately.
SNGX must own by today 1/03/2023 to get Divi/Preffered D shares.
Buy em UP...
“Princeton, NJ – December 23, 2022 – Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that its Board of Directors declared a dividend of one one-thousandth of a share of newly designated Series D Preferred Stock, par value $0.001 per share, for each outstanding share of the Company’s common stock held of record as of 5:00 p.m. Eastern Standard Time on January 3, 2023. The shares of Series D Preferred Stock will be distributed to such recipients at 5:00 p.m. Eastern Standard Time on January 4, 2023. The outstanding shares of Series D Preferred Stock will vote together with the outstanding shares of the Company’s common stock, as a single class, exclusively with respect to a reverse stock split, as well as any proposal to adjourn any meeting of stockholders called for the purpose of voting on the reverse stock split, and will not be entitled to vote on any other matter, except to the extent required under the Delaware General Corporation Law. Subject to certain limitations, each outstanding share of Series D Preferred Stock will have 1,000,000 votes per share (or 1,000 votes per one one-thousandth of a share of Series D Preferred Stock)."
GLTA
And yes, here it is as suspected it would be.....
The outstanding shares of Series D Preferred Stock will vote together with the outstanding shares of the Company's common stock, as a single class, exclusively with respect to a reverse stock split,
There hasn't been any volume here since March, to speak of.
This stock hasn't even attracted pump and dumpers.
I'm embarrassed for owning this, but actually the recommendation came from a reliable source.
Yeah yeah… SNGX volume so low today says no panic selling.
I still have ENTI fake shares from years ago same & since never authorized I can’t even have broker make them disappear
Oh, the reverse split is coming. Watch and see how they RS the preferred shares that are authorized, but not issued.
I can bet how that will go.
I saw no reverse ratio confirmed- only a vote for reverse split. My thoughts are this “distribution” of preferred could cause pps over $1 as outstanding still so low 43m can move FAST. Then no r/s needed. GLTA and Merry Christmas!
SNGX distribution of Preferred to Common stock holders as of 1/03/2023 !
“Princeton, NJ – December 23, 2022 – Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that its Board of Directors declared a dividend of one one-thousandth of a share of newly designated Series D Preferred Stock, par value $0.001 per share, for each outstanding share of the Company’s common stock held of record as of 5:00 p.m. Eastern Standard Time on January 3, 2023. The shares of Series D Preferred Stock will be distributed to such recipients at 5:00 p.m. Eastern Standard Time on January 4, 2023. The outstanding shares of Series D Preferred Stock will vote together with the outstanding shares of the Company’s common stock, as a single class, exclusively with respect to a reverse stock split, as well as any proposal to adjourn any meeting of stockholders called for the purpose of voting on the reverse stock split, and will not be entitled to vote on any other matter, except to the extent required under the Delaware General Corporation Law. Subject to certain limitations, each outstanding share of Series D Preferred Stock will have 1,000,000 votes per share (or 1,000 votes per one one-thousandth of a share of Series D Preferred Stock).
“The current market conditions have been extremely difficult for many companies both large and small, and Soligenix is no exception,” stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. “After receiving the Nasdaq deficiency letter stating that the Company no longer met the required $1.00 minimum bid price, we were hopeful that our important 2022 milestones such as the submission of the HyBryte™ new drug application (NDA), had the potential to put the Company back in compliance with Nasdaq requirements. Unfortunately, while achieving these major milestones, our stock price did not respond as we had hoped in these difficult global market conditions. Therefore, we are now faced with having to consider a reverse stock split in order to maintain our Nasdaq listing, which is very important for the Company’s future success as we move toward potential NDA approval and U.S. launch, and continue partnership discussions. It should also make our stock more attractive to larger institutional investors.”
All shares of Series D Preferred Stock that are not present in person or by proxy at the meeting of stockholders held to vote on the reverse stock split as of immediately prior to the opening of the polls at such meeting will automatically be redeemed by the Company. Any outstanding shares of Series D Preferred Stock that have not been so redeemed will be redeemed if such redemption is ordered by the Company’s Board of Directors or automatically upon the approval by the Company’s stockholders of an amendment to the Company’s certificate of incorporation effecting the reverse stock split at such meeting.
The Series D Preferred Stock will be uncertificated, and no shares of Series D Preferred Stock will be transferable by any holder thereof except in connection with a transfer by such holder of any shares of the Company’s common stock held by such holder. In that case, a number of one one-thousandths of a share of Series D Preferred Stock equal to the number of shares of the Company’s common stock to be transferred by such holder would be transferred to the transferee of such shares of common stock.
Further details regarding the Series D Preferred Stock will be contained in a report on Form 8-K to be filed by the Company with the Securities and Exchange Commission.”
GLTA
Soligenix's pipeline and assets are way undervalued by the market IMO. With a solid Balance Sheet and by carefully managing expenses, the company is now in an advantageous position to dramatically enhance its value. It is incredible that the stock is selling for $0.66+/- a share and has not been acquired or strategically aligned with a major Pharma company.
SNGX p/r 12/19/2022: Soligenix Initiates Phase 2 Clinical Trial of SGX302 (synthetic hypericin) for the Treatment of Mild-to-Moderate Psoriasis. 7:30 am ET December 19, 2022
Study HPN-PSR-01 opens patient enrollment
Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that patient enrollment has been opened for its Phase 2a study (protocol number HPN-PSR-01) evaluating SGX302 (synthetic hypericin) in the treatment of mild-to-moderate psoriasis. Psoriasis is an ongoing unmet medical need, with as many as 7.5 million people in the U.S. and 60-125 million people worldwide affected by this incurable disease.
- "We are excited to expand synthetic hypericin's development into different cutaneous T-cell diseases such as psoriasis, as a component of our long-term strategy to enhance the value of this unique compound," stated Christopher J. Schaber, PhD, President and Chief Executive Officer of Soligenix. "Given our promising published results with hypericin to date, including a small Phase 1/2 proof of concept clinical trial in mild-to-moderate psoriasis, and the Phase 3 FLASH study in cutaneous T-cell lymphoma, where we filed a New Drug Application (NDA) this month, we are hopeful synthetic hypericin will have a role to play in helping patients suffering from this difficult to treat and chronic disease."
- The Phase 2a clinical trial (protocol number HPN-PSR-01) will target enrollment of up to 42 patients ages 18 years or older with mild to moderate, stable psoriasis covering 2 to 30% of their body. In both Parts A and B, all patients will apply the study drug twice per week and will activate the drug with visible light 24 ± 6 hours later using the supplied visible light devices and according to the manufacturer's instructions. Patients will undergo treatments for a total of 18 weeks and, on completion, will be followed for a 4-week follow-up period in which patients will not receive other psoriasis treatments. In Part A, 5-10 patients will be assigned open-label SGX302 (0.25% hypericin) at the time of enrollment. Once the tolerability and response to SGX302 has been established, Part B of the protocol will commence. In Part B, patients will be randomized to double-blind treatment groups at a ratio 1:1 of active drug to placebo ointment.
- Active dermatologic assessment of treated lesions for adverse events will be performed immediately before and during light treatments. Patients will be assessed for overall disease status through 4 weeks of follow-up. Efficacy endpoints will include the extent of lesion clearance and patient reported quality of life indices. Routine safety laboratories also will be collected.
About Synthetic Hypericin:
- Visible light-activated synthetic hypericin is a novel, first-in-class, photodynamic therapy (PDT) that is expected to avoid much of the long-term risks associated with other PDT treatments. Synthetic hypericin is a potent photosensitizer that is topically applied to skin lesions and taken up by cutaneous T-cells. With subsequent activation by safe, visible light, T-cell apoptosis is induced, addressing the root cause of psoriasis lesions. Other PDTs have shown efficacy in psoriasis with a similar apoptotic mechanism, albeit using ultraviolet (UV) light associated with more severe potential long-term safety concerns. The use of visible light in the red-yellow spectrum has the advantage of deeper penetration into the skin (much more than UV light) potentially treating deeper skin disease and thicker plaques and lesions, similar to what was observed in the positive Phase 3 FLASH (Fluorescent Light Activated Synthetic Hypericin) study in CTCL. Synthetic hypericin or HyBryte™ (the tradename used in CTCL) was demonstrated in this study to be equally effective in treating both plaque (42% treatment response rate after 12 weeks treatment, p<0.0001 relative to placebo treatment) and patch (37%, p=0.0009) lesions in this orphan disease caused by malignant T-cells. In a published Phase 1/2 proof of concept clinical study using synthetic hypericin, efficacy was demonstrated in patients with CTCL (58.3% response, p=0.04) as well as psoriasis (80% response, p<0.02).
- This treatment approach avoids the risk of secondary malignancies (including melanoma) inherent with both the frequently used DNA-damaging drugs and other phototherapies that are dependent on UV A or B exposure. The use of synthetic hypericin coupled with safe, visible light also avoids the risk of serious infections and cancer associated with the systemic immunosuppressive treatments used in psoriasis.
- The Phase 3 FLASH trial enrolled a total of 169 patients (166 evaluable) with Stage IA, IB or IIA CTCL. The trial consisted of three treatment cycles. Treatments were administered twice weekly in 6-week cycles. In the first double-blind treatment cycle, 116 patients received HyBryte™ treatment and 50 received placebo treatment of their index lesions. A total of 16% of the patients receiving HyBryte™ achieved at least a 50% reduction in their lesions (using the standard Composite Assessment of Index Lesions Severity [CAILS] score) compared to only 4% of patients in the placebo group after just 6 weeks of treatment (p=0.04). Further treatment with HyBryte™ increased the number of treatment successes to 40% and 49% after 12 and 18 weeks, respectively (p<0.0001 for both). Additional analyses also indicated that HyBryte™ is equally effective in treating both plaque (42% treatment response rate after 12 weeks treatment, p<0.0001 relative to placebo treatment in Cycle 1) and patch (37%, p=0.0009) lesions of CTCL, a particularly relevant finding given the historical difficulty in treating plaque lesions. This is also relevant to psoriasis where the lesions can be thicker than the patches observed in CTCL.
- In a subset of patients evaluated during their third treatment cycle, it was demonstrated that HyBryte™ is not systemically available, consistent with the general safety of this topical product observed to date. At the end of Cycle 3, HyBryte™ continued to be well tolerated despite extended and increased use of the product to treat multiple lesions.
About Psoriasis :
Psoriasis is a chronic, non-communicable, itchy and often painful inflammatory skin condition for which there is no cure. Psoriasis has a significantly detrimental impact on patients' quality of life, and is associated with cardiovascular, arthritic, and metabolic diseases, as well as psychological conditions such as anxiety, depression and suicide. Many factors contribute to development of psoriasis including both genetic and environmental factors (e.g., skin trauma, infections, and medications). The lesions develop because of rapidly proliferating skin cells, driven by autoimmune T-cell mediated inflammation. Of the various types of psoriasis, plaque psoriasis is the most common and is characterized by dry, red raised plaques that are covered by silvery-white scales occurring most commonly on the elbows, knees, scalp, and lower back. Approximately 80% of patients have mild-to-moderate disease. Mild psoriasis is generally characterized by the involvement of less than 3% of the body surface area (BSA), while moderate psoriasis will typically involve 3-10% BSA and severe psoriasis greater than 10% BSA. Between 20% and 30% of individuals with psoriasis will go on to develop chronic, inflammatory arthritis (psoriatic arthritis) that can lead to joint deformations and disability. Studies have also associated psoriasis, and particularly severe psoriasis, with an increased relative risk of lymphoma, particularly CTCL. Although psoriasis can occur at any age, most patients present with the condition before age 35.
- Treatment of psoriasis is based on its severity at the time of presentation with the goal of controlling symptoms. It varies from topical options including PDT to reduce pain and itching, and potentially reduce the inflammation driving plaque formation, to systemic treatments for more severe disease. Most common systemic treatments and even current topical photo/photodynamic therapy such as UV A and B, carry a risk of increased skin cancer.
- Psoriasis is the most common immune-mediated inflammatory skin disease. According to the World Health Organization (WHO) Global Report on Psoriasis 2016, the prevalence of psoriasis is between 1.5% and 5% in most developed countries, with some suggestions of incidence increasing with time. It is estimated, based upon review of historic published studies and reports and an interpolation of data that psoriasis affects 3% of the U.S. population or more than 7.5 million people. Current estimates have as many as 60-125 million people worldwide living with the condition. The global psoriasis treatment market was valued at approximately $15 billion in 2020 and is projected to reach as much as $40 billion by 2027."
GLTA
..
.....
Tomorrow 12/16 is Quadruple Witch Friday so should make SNGX current short interest very interesting…
“The four types of derivatives expiring on quadruple witching are:
- Stock index futures contracts
- Single-stock options
- Options on stock-index futures
- Stock index options”
GLTA
I've held on to my small position here, because frankly it's not worth selling, however, there is no indication whatsoever that this stock is getting ready to surge.
There is a resistance at .71 and again at $1.13 currently. Until it breaks above that second resistance, this stock is bearish.
Submission of the NDA is a true milestone and accomplishment for the Soligenix Team.
Bravo Zulu!
Level II is showing a little action. This puppy WANTS the $$!!!
SNGX p/r 12/15/2022: "Soligenix Announces Submission of New Drug Application to the FDA for HyBryte™ in the Treatment of Cutaneous T-cell Lymphoma
7:30 am ET December 15, 2022 (PR Newswire) Print
Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that it submitted a new drug application (NDA) to the United States (U.S.) Food and Drug Administration (FDA) for HyBryte™ (synthetic hypericin) in the treatment of early stage cutaneous T-cell lymphoma (CTCL), a rare cancer and area of unmet medical need affecting over 25,000 patients in the U.S. The Company estimates the potential worldwide market for HyBryte™ to be in excess of $250 million for the treatment of CTCL.
https://mma.prnewswire.com/media/1514137/HyBryte_High_Resolution_Logo.jpg
"The submission of our NDA is a significant milestone forSoligenix, positioning us well to fulfill our promise to offer an innovative first-in-class therapy to the CTCL community of patients, families and healthcare professionals," statedChristopher J. Schaber, PhD, President and Chief Executive Officer ofSoligenix. "CTCL is an incredibly difficult to treat orphan disease and remains an area of unmet medical need with a very limited number of safe and effective treatment options. The clinical results we've generated throughout the development program support the potential for HyBryte™ to serve as an important front-line treatment in the management of this chronic cancer. We look forward to working with the FDA through the review process. As the NDA has been granted both FDA orphan drug and fast track designations, we anticipate potential approval in the second half of 2023 as we continue advancing our preparations for U.S. launch, which is currently targeted for first quarter 2024."
About HyBryte™
HyBryte™ (research name SGX301) is a novel, first-in-class, photodynamic therapy utilizing safe, visible light for activation. The active ingredient in HyBryte™ is synthetic hypericin, a potent photosensitizer that is topically applied to skin lesions that is taken up by the malignant T-cells, and then activated by visible light approximately 24 hours later. The use of visible light in the red-yellow spectrum has the advantage of penetrating more deeply into the skin (much more so than ultraviolet light) and therefore potentially treating deeper skin disease and thicker plaques and lesions. This treatment approach avoids the risk of secondary malignancies (including melanoma) inherent with the frequently employed DNA-damaging drugs and other phototherapy that are dependent on ultraviolet exposure. Combined with photoactivation, hypericin has demonstrated significant anti-proliferative effects on activated normal human lymphoid cells and inhibited growth of malignant T-cells isolated from CTCL patients. In a published Phase 2 clinical study in CTCL, patients experienced a statistically significant (p=0.04) improvement with topical hypericin treatment whereas the placebo was ineffective. HyBryte™ has received orphan drug and fast track designations from the FDA, as well as orphan designation from the European Medicines Agency (EMA).
The recently published Phase 3 FLASH trialenrolled a total of 169 patients (166 evaluable) with Stage IA, IB or IIA CTCL. The trial consisted of three treatment cycles. Treatments were administered twice weekly for the first 6 weeks and treatment response was determined at the end of the 8th week of each cycle. In the first double-blind treatment cycle, 116 patients received HyBryte™ treatment (0.25% synthetic hypericin) and 50 received placebo treatment of their index lesions. A total of 16% of the patients receiving HyBryte™ achieved at least a 50% reduction in their lesions (graded using a standard measurement of dermatologic lesions, the CAILS score) compared to only 4% of patients in the placebo group at 8 weeks (p=0.04) during the first treatment cycle (primary endpoint). HyBryte™ treatment in the first cycle was safe and well tolerated.
In the second open-label treatment cycle (Cycle 2), all patients received HyBryte™ treatment of their index lesions. Evaluation of 155 patients in this cycle (110 receiving 12 weeks of HyBryte™ treatment and 45 receiving 6 weeks of placebo treatment followed by 6 weeks of HyBryte™ treatment), demonstrated that the response rate among the 12-week treatment group was 40% (p<0.0001 vs the placebo treatment rate in Cycle 1). Comparison of the 12-week and 6-week treatment groups also revealed a statistically significant improvement (p<0.0001) between the two groups, indicating that continued treatment results in better outcomes. HyBryte™ continued to be safe and well tolerated. Additional analyses also indicated that HyBryte™ is equally effective in treating both plaque (response 42%, p<0.0001 relative to placebo treatment in Cycle 1) and patch (response 37%, p=0.0009 relative to placebo treatment in Cycle 1) lesions of CTCL, a particularly relevant finding given the historical difficulty in treating plaque lesions in particular.
The third (optional) treatment cycle (Cycle 3) was focused on safety and all patients could elect to receive HyBryte™ treatment of all their lesions. Of note, 66% of patients elected to continue with this optional compassionate use / safety cycle of the study. Of the subset of patients that received HyBryte™ throughout all 3 cycles of treatment, 49% of them demonstrated a positive treatment response (p<0.0001 vs patients receiving placebo in Cycle 1). Moreover, in a subset of patients evaluated in this cycle, it was demonstrated that HyBryte™ is not systemically available, consistent with the general safety of this topical product observed to date. At the end of Cycle 3, HyBryte™ continued to be well tolerated despite extended and increased use of the product to treat multiple lesions.
Overall safety of HyBryte™ is a critical attribute of this treatment and was monitored throughout the three treatment cycles (Cycles 1, 2 and 3) and the 6-month follow-up period. HyBryte's™ mechanism of action is not associated with DNA damage, making it a safer alternative than currently available therapies, all of which are associated with significant and sometimes fatal, side effects. Predominantly these include the risk of melanoma and other malignancies, as well as the risk of significant skin damage and premature skin aging. Currently available treatments are only approved in the context of previous treatment failure with other modalities and there is no approved front-line therapy available. Within this landscape, treatment of CTCL is strongly motivated by the safety risk of each product. HyBryte™ potentially represents the safest available efficacious treatment for CTCL. With very limited systemic absorption, a compound that is not mutagenic and a light source that is not carcinogenic, there is no evidence to date of any potential safety issues.
The Phase 3 CTCL clinical study was partially funded by the National Cancer Institute via a Phase II SBIR grant (#1R44CA210848-01A1) awarded to Soligenix, Inc.
About Cutaneous T-Cell Lymphoma (CTCL)
CTCLis a class of non-Hodgkin's lymphoma (NHL), a type of cancer of the white blood cells that are an integral part of the immune system. Unlike most NHLs which generally involve B-cell lymphocytes (involved in producing antibodies), CTCL is caused by an expansion of malignant T-cell lymphocytes (involved in cell-mediated immunity) normally programmed to migrate to the skin. These malignant cells migrate to the skin where they form various lesions, typically beginning as patches and may progress to raised plaques and tumors. Mortality is related to the stage of CTCL, with median survival generally ranging from about 12 years in the early stages to only 2.5 years when the disease has advanced. There is currently no cure for CTCL. Typically, CTCL lesions are treated and regress but usually return either in the same part of the body or in new areas.
CTCL constitutes a rare group of NHLs, occurring in about 4% of the approximate 700,000 individuals living with the disease. It is estimated, based upon review of historic published studies and reports and an interpolation of data on the incidence of CTCL that it affects over 25,000 individuals in the U.S., with approximately 3,000 new cases seen annually."
GLTA
..
....
"We expect to submit our new drug application (NDA) application to the FDA for HyBryte(TM) during this quarter. Additionally, we are continuing our expansion into new disease indications with synthetic hypericin having received FDA investigational new drug (IND) clearance for a Phase 2a clinical trial in mild-to-moderate psoriasis, also expected to begin before year end.
https://finance.yahoo.com/news/soligenix-inc-showcase-2022-milestones-120000272.html
What are you doing? I thought we covered this already?
This should be well over the $ imo.
Thinking the fuse has been lit!
10/27/2022: Soligenix Invited to Submit BARDA Contract Proposal for Development of Thermostabilized Subunit Vaccines for Sudan Ebola and Marburg Viruses.
" - Emerging infectious diseases where no protective vaccines are currently available.
- SuVax™ targets Sudan ebolavirus, origin of a current disease outbreak in Uganda.
PRINCETON, N.J. , Oct. 27, 2022 /PRNewswire/ -- Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the Company has been invited by the Biomedical Advanced Research and Development Authority (BARDA) Division of Chemical, Biological, Radiological and Nuclear (CBRN) Medical Countermeasures to submit a full contract proposal for the development of single-vial, adjuvanted, heat stable subunit vaccines to prevent filovirus infection. This submission supports a potential multi-year, multi-million dollar contract to develop SuVax™ and MarVax™ vaccine candidates as medical countermeasures (MCM) for use in the event of a Sudan ebolavirus or Marburg marburgvirus outbreak.
Soligenix was invited to submit this proposal following a review of its white paper entitled, "SuVax™: A Safe and Thermostable Single-Vial Adjuvanted Subunit Vaccine with Rapid Onset Immunity to Sudan ebolavirus", which proposed development of SuVax™ through Phase 1 clinical studies in a $25 million program of work over 5 years. BARDA also requested that additional options be built into the full proposal including addressing a separate potential Marburg marburgvirus vaccine candidate, MarVax™, which has the potential to increase the value of the program. BARDA's mission is to develop and procure needed MCMs, including vaccines, therapeutics, diagnostics, and non-pharmaceutical countermeasures, against a broad array of public health threats, whether natural or intentional in origin.
In a non-human primate model of Sudan viral disease, SuVax™ demonstrated 100% protection four weeks after vaccination with a 3-dose series of SuVax™. Similar potency has been demonstrated for MarVax™. The development of these vaccines is part of an ongoing long-term collaboration with Soligenix's academic partner, the University of Hawai?i at Manoa. This work was partially supported by a $1.5 million Small Business Innovation Grant (#1R44AI157593-01) awarded to Soligenix and a grant to the University of Hawaii at Manoa (R01-AI132323) from the National Institute of Allergy and Infectious Diseases (NIAID).
"We are excited about the favorable review of our white paper by BARDA and by their invitation to submit a full contract proposal," stated Christopher J. Schaber, PhD, President & Chief Executive Officer of Soligenix. "Although a contract award is not guaranteed, we believe that we are well-positioned to receive BARDA development support for this indication allowing us to further demonstrate the growing body of compelling scientific evidence supporting our heat stable filovirus vaccine platform, including vaccine candidates directed towards Sudan ebolavirus and Marburg marburgvirus. This is particularly relevant given the outbreak of Sudan viral disease in Uganda for which there is no current protective vaccine available. We look forward to continued productive interactions with BARDA, NIAID and FDA as we move these programs forward."
The submission of a contract proposal is non-binding and does not guarantee the award of a BARDA contract. The contract award will require a favorable technical and scientific review by BARDA followed by negotiation of fair and reasonable contract terms.
About Filovirus Infection
Ebola Virus Disease is caused by one of six species of Ebolavirus, four of which are known to cause disease in humans, including its best-known member, Zaire ebolavirus (Ebola virus), with Sudan ebolavirus (Sudan virus) being the second-most common cause of human infection in this family. All species of ebolavirus belong to the Filoviridae family, a family that further contains the equally human pathogenic Marburg marburgvirus (Marburg virus). Filoviruses are believed to be harbored in various animal species in Africa, particularly bats, although the specific reservoir host for many of these viruses is still unknown. There have been several known Ebola and Marburg Virus Disease outbreaks since 1967, with the largest outbreak starting in 2014 in Western Africa, and involved over 26,000 confirmed/probable/suspected cases with an estimated death toll of over 11,000 people according to the Centers for Disease Control and Prevention (CDC). These numbers also include some cases of virus introduction and limited spread in Europe and the United States. Morbidity after viral infection is also significant, with surviving patients reporting symptoms including fatigue, memory problems, shortness of breath, sleep problems and joint pain for years after infection. Most recently, an outbreak of Sudan virus in Uganda has been declared, with containment activities underway.
Transmission of filoviruses requires direct contact with bodily fluids from an infected person or contact with infected animals. The mortality rates following filovirus infections are extremely high, and in the absence of wide availability of effective therapeutics, are affected by the quality of supportive care available with a focus on early initiation of treatment. Resolution of the disease largely depends on the patient's own immune system. There are limited treatment options for Ebola Virus Disease and no available treatments for Sudan Virus or Marburg Virus Disease, although steady progress has also been made in development of immunotherapeutics for filoviruses beyond Ebola virus. There are approved vaccines for Ebola virus, requiring stringent ultra-low cold-chain storage, but no efficacious vaccines are yet available for Marburg virus or Sudan virus.
Filoviruses are one of the virus families identified as having the ability to cause pandemics. On the heels of the COVID-19 pandemic the US government is accelerating its investment in pandemic preparedness, including having "the ability to rapidly make vaccines effective against any virus family". Specific initiatives have been spear-headed by the White House and Biden-Harris administration, as evidenced by the "American Pandemic Preparedness: Transforming Our Capabilities" white paper released in September 2021.
About John A. Burns School of Medicine, University of Hawai'i at Manoa
The John A. Burns School Medicine (JABSOM) at the University of Hawai?i at Manoa is one of the leading medical institutions and one of the most ethnically diverse institutions in the United States. For more than a decade, JABSOM has ranked in the top 10% of allopathic medical schools for graduate retention with one of our UH-sponsored residency programs. Hawai?i's cultural diversity and geographical setting affords JABSOM a unique research environment to excel in research directed at eliminating diseases that disproportionately affect people in Hawaii and the Pacific region. JABSOM faculty bring more than $40 million in extramural funds into the state, annually. In addition, JABSOM was the first U.S. medical school to create a clinical department dedicated to the health and well-being of an indigenous population, Native Hawaiians.
About Soligenix, Inc.
Soligenix is a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need. Our Specialized BioTherapeutics business segment is developing and moving toward potential commercialization of HyBryte™ (SGX301 or synthetic hypericin) as a novel photodynamic therapy utilizing safe visible light for the treatment of cutaneous T-cell lymphoma (CTCL). With a successful Phase 3 study completed, regulatory approval is being sought and commercialization activities for this product candidate are being advanced initially in the U.S. Development programs in this business segment also include expansion of synthetic hypericin (SGX302) into psoriasis, our first-in-class innate defense regulator (IDR) technology, dusquetide (SGX942) for the treatment of inflammatory diseases, including oral mucositis in head and neck cancer, and proprietary formulations of oral beclomethasone 17,21-dipropionate (BDP) for the prevention/treatment of gastrointestinal (GI) disorders characterized by severe inflammation including pediatric Crohn's disease (SGX203).
Our Public Health Solutions business segment includes active development programs for RiVax®, our ricin toxin vaccine candidate, and SGX943, our therapeutic candidate for antibiotic resistant and emerging infectious disease, and our vaccine programs targeting filoviruses (such as Marburg and Ebola) and CiVax™, our vaccine candidate for the prevention of COVID-19 (caused by SARS-CoV-2). The development of our vaccine programs incorporates the use of our proprietary heat stabilization platform technology, known as ThermoVax®. To date, this business segment has been supported with government grant and contract funding from the National Institute of Allergy and Infectious Diseases (NIAID), the Defense Threat Reduction Agency (DTRA) and the Biomedical Advanced Research and Development Authority (BARDA).
For further information regarding Soligenix, Inc., please visit the Company's website at https://www.soligenix.com and follow us on LinkedIn and Twitter at @Soligenix_Inc.
This press release may contain forward-looking statements that reflect Soligenix, Inc.'s current expectations about its future results, performance, prospects and opportunities, including but not limited to, potential market sizes, patient populations and clinical trial enrollment. Statements that are not historical facts, such as "anticipates," "estimates," "believes," "hopes," "intends," "plans," "expects," "goal," "may," "suggest," "will," "potential," or similar expressions, are forward-looking statements. These statements are subject to a number of risks, uncertainties and other factors that could cause actual events or results in future periods to differ materially from what is expressed in, or implied by, these statements, such as experienced with the COVID-19 outbreak. Soligenix cannot assure you that it will be able to successfully develop, achieve regulatory approval for or commercialize products based on its technologies, particularly in light of the significant uncertainty inherent in developing therapeutics and vaccines against bioterror threats, conducting preclinical and clinical trials of therapeutics and vaccines, obtaining regulatory approvals and manufacturing therapeutics and vaccines, that product development and commercialization efforts will not be reduced or discontinued due to difficulties or delays in clinical trials or due to lack of progress or positive results from research and development efforts, that it will be able to successfully obtain any further funding to support product development and commercialization efforts, including grants and awards, maintain its existing grants which are subject to performance requirements, enter into any biodefense procurement contracts with the U.S. Government or other countries, that it will be able to compete with larger and better financed competitors in the biotechnology industry, that changes in health care practice, third party reimbursement limitations and Federal and/or state health care reform initiatives will not negatively affect its business, or that the U.S. Congress may not pass any legislation that would provide additional funding for the Project BioShield program. In addition, there can be no assurance as to the timing or success of any of its clinical/preclinical trials. Despite the statistically significant result achieved in the HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma, there can be no assurance that a marketing authorization from the FDA or EMA will be successful. Notwithstanding the result in the HyBryte™ (SGX301) Phase 3 clinical trial for the treatment of cutaneous T-cell lymphoma and the Phase 1/2 proof-of-concept clinical trial of SGX302 for the treatment of psoriasis, there can be no assurance as to the timing or success of the clinical trials of SGX302 for the treatment of psoriasis. Further, there can be no assurance that RiVax® will qualify for a biodefense Priority Review Voucher (PRV) or that the prior sales of PRVs will be indicative of any potential sales price for a PRV for RiVax®. Also, no assurance can be provided that the Company will receive or continue to receive non-dilutive government funding from grants and contracts that have been or may be awarded or for which the Company will apply in the future. These and other risk factors are described from time to time in filings with the Securities and Exchange Commission, including, but not limited to, Soligenix's reports on Forms 10-Q and 10-K. Unless required by law, Soligenix assumes no obligation to update or revise any forward-looking statements as a result of new information or future events."
SOURCE Soligenix, Inc.
GLTA !
Shhhh.. let them play.
SNGX spring loaded
Soligenix Enters Q4 With Multiple Catalysts in Play
Newsfile Corp.
Thu, October 13, 2022 at 5:30 AM·4 min read
In this article:
New York, New York--(Newsfile Corp. - October 13, 2022) - PCG Digital -- Late-stage biopharmaceutical company, Soligenix, Inc. (NASDAQ: SNGX) is anticipating significant catalysts in Q4 and a potentially transformational year in 2023.
https://finance.yahoo.com/news/soligenix-enters-q4-multiple-catalysts-123000437.html
This HARDLY has the volume to attract that BS. But still, the MM's are letting it happen.
%$#@ers!!!
F’n algo trading take downs…
Gotta crack down on that.
GLTA
|
Followers
|
208
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9772
|
|
Created
|
06/06/06
|
Type
|
Free
|
| Moderators | |||
| SNGX [NASD] |
| Soligenix, Inc. |
| Healthcare | Biotechnology | USA |
Soligenix, Inc. (Soligenix) is a late-stage biopharmaceutical company.
The Company is focused on developing and commercializing products to treat rare diseases.
The Company operates through two business segments:
Specialized BioTherapeutics and Public Health Solution. Soligenix’s Specialized BioTherapeutics business segment is developing
a photodynamic therapy (SGX301) utilizing topical synthetic hypericin activated with safe visible fluorescent light for the treatment of cutaneous T-cell lymphoma (CTCL).
The Public Health Solutions business segment includes active development programs for RiVax,
its ricin toxin vaccine candidate and SGX943, its therapeutic candidate for antibiotic-resistant and emerging infectious disease.
CTCL is its innate defense regulator technology, dusquetide (SGX942) for the treatment of oral mucositis in head and neck cancer.
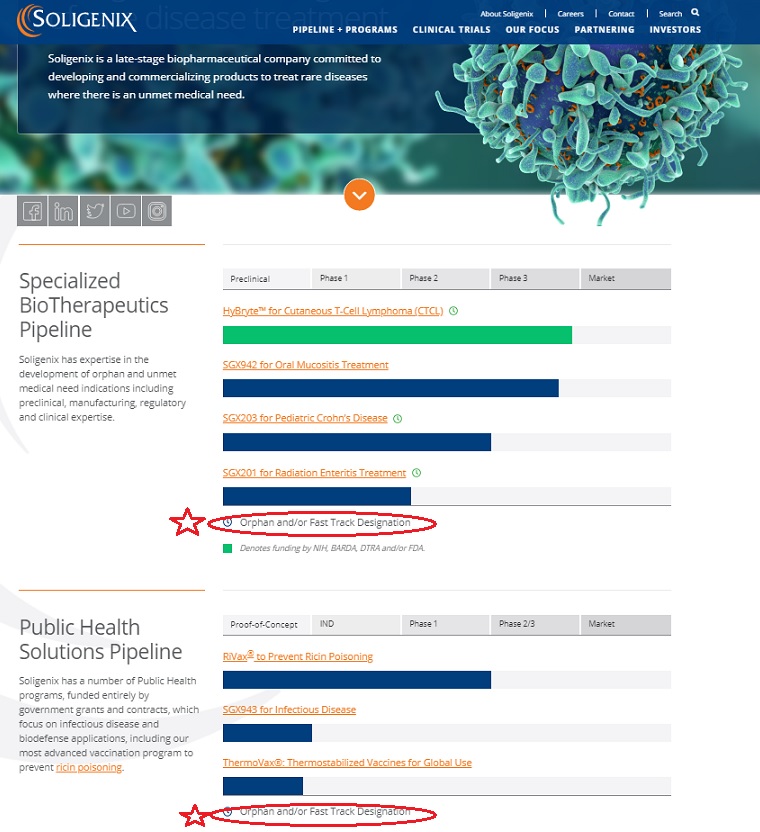
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment | ||||
| | | | | |
Orphan and/or Fast Track Designation
Soligenix has a number of Public Health programs, funded entirely by government grants and contracts, which focus on infectious disease and biodefense applications, including our most advanced vaccination program to prevent ricin poisoning.
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation


| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) almost done [in phase 3] | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment almost done [in phase 3] | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease [phase 2 almost done] | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment [phase 1 going into phase 2] | ||||
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation
| PER IHUB MGMT |
02-07-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
