Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
They didn't call me!! lol voting closed Sept 14th looks like.
So, bottom around .50?
SNGX 50% hair-cut in past few weeks (very low volume) ???
And they called my cell direct about Shareholder vote yesterday ?
GLTA
My wife tells me the same thing. But I do like the technicals. As an engineering type I can really get into the charts. Sometimes the fundamentals are also worth the risk. We'll see. Soligenix posted an ROCE of -0.57%.
I was Carrier precom, sub tenders and shore sub/ship repair Pacific, East Coast and Scotland. I'll PM you during happy hour.
Well it's certainly nice to have a pension, but better you spend your money in Las Vegas than based on garbage here. They will give you free drinks.
What ships were you attached to?
Lol. I'll be collecting 2 retirements by that time. When this gets back over the $ it'll fatten the account just nicely.
You are buying penny stocks in your retirement account??
Should I go ahead and get you the link for Medicaid now?
I just put this in my ira on the 14th @ .695 based on what I though was worth the risk based on the chart. That's now shot to poop. I've been swinging this fairly regularly. Waiting for the chart to set up before I either average down or go ahead and trigger the S/L. .60 is a fairly decent support level IMO. So we'll see how she does. Like I said before, this market isn't for the weak. Especially with the massive volatility.
I'm not a short term investor, but on this stock, I'd be even less optimistic for short term than long.
I'm ticked at myself for being so stupid....well, I'm not mad as much as I'm disappointed.
Anywho, I only bought 700 shares as a trial, so this would be costing me a car payment if I had one.
Just starting to poke its nose under the 30 RSI. So much for my timing.
Yes! Kinda like that. It never surprises me what some can make a chart seem to look in whatever direction they want the reader to believe. A pos chart turning completely bullish by a bs artist.
I posted this as a potential pincher based on my charts yesterday. Then I saw this smiley face and tongue bs on twits today. I thought, why not!? This market is clueless lately. But at the same time, the same saying is the same. No risk, no reward.
Whatever floats the boat may be it's own torpedo to sink it. Anyhow, I'm short term optimistic.
I was just looking at different chart patterns. One called a "smiley face and tongue"? It's supposed to be the start of a bullish run. Never heard of that one till today. and IF it works out then great. This sideways crap sucks!! SNGX's daily mimics it imo. But, truth is, the market is killing just about everything all on its own with the volatility over the last couple months and looks to continue this way at least through the rest of this month and possibly much longer.
This ain't set up for jack squat! The possible uptrend just broke down again.
If nothing else this is set up nicely for a technical move.
SNGX daily
With all this good news and yet the stock price keeps tumbling. Kind'a makes ya go ....hmmmmm.
SNGX get's another $2.6m grant 9/12/2022:
"FDA Awards $2.6 Million Grant for Expanded Study of Soligenix's Hybryte(TM) for CTCL. 8:34 am ET September 12, 2022.
New York, New York--(Newsfile Corp. - September 12, 2022) - PCG Digital -- Late-stage biopharmaceutical company Soligenix, Inc. (NASDAQ: SNGX) has announced the award of a $2.6 million grant from the FDA to support a study of expanded HyBryteT treatment, including at-home usage. The FDA's support will give patients an opportunity to access Soligenix's HyBryteT therapy in an open-label setting. HyBryte is a novel skin directed photodynamic therapy for cutaneous T-cell lymphoma (CTCL), a rare skin disease and cancer.
GLTA
I'm surprised there isn't more of a spread considering the super low volume.
I have my first buy signal in a while. Whatever I do on the bid it seems like 2 other MM's are immediately on top of me.
SNGX only 4,000 traded today???!!!
GLTA
Need to see a solid reversal. We will imo.
SNGX less than 50k traded 2nd day in a row...
L2 is a joke.
When they gonna let SNGX loose? !!!
GLTA
Support (or resistance) is at .733....better hold your breath.
Might have to start warming up my crystal ball. Still needs time for the technical side but we've been clearly watching the fundamentals strengthen.
Clear manipulation.
About time for SNGX manipulation to stop & run UP again...
GLTA
FDA gives SNGX a bone!
U.S. Food and Drug Administration Awards $2.6 Million Orphan Products Development Grant for Expanded Study of HyBryte in the Treatment of Cutaneous T-Cell Lymphoma PR Newswire
Money to support the study anyway.
https://finance.yahoo.com/news/u-food-drug-administration-awards-113000831.html
And now 78 goes through?!!
Blame it on the market! Everything is all broke imo.
Thanks Fed!
I'm BULLISH SNGX long though. Volatility creats opportunities. I have a decent position but if it wants to give me more (like double down) cheaper, I'll be ready.
That’s NOT what we want ?!
Nearing the .70 support level. 200 dma sitting @ .69. Still not pushing the oversolds yet. A little patience to see if this gets to the low .60's maybe.
SNGX chart
Just need the right news on the right day.
$1+++
![]()
This just popped up in my Twitter alert, and actually said 8/3.
Not sure why it alerted today?
Obviously didn't help the pps...
Ahem....that news actually hit last week, the day the stock topped out. If such great news, why is the stock price dropping since the release?
PRINCETON, N.J., July 27, 2022 /PRNewswire/ -- Soligenix, Inc
SNGX 8/2: "Study Reveals Successful Results for Treatment of Cutaneous T-Cell Lymphoma."
"August 2, 2022 Soligenix announces positive phase 3 results from FLASH study for the use of synthetic hypericin to reduce cutaneous T-cell lymphoma lesions.
Phase 3 results of the Fluorescent Light Activated Synthetic Hypericin (FLASH) study1 demonstrated significant reductions in cutaneous T-Cell lymphoma (CTCL) lesion sizes with synthetic hypericin (HyBryte; Soligenix) treatment. These findings provide hope for a new treatment to patients diagnosed with the life-altering disease. As a chronic type of cancer that severely impacts patients’ quality of life, there is an unmet need for a well-tolerated and safe treatment option. The FLASH study is the largest double-blind, randomized, placebo-controlled clinical trial for CTCL to date.
The study data also showed that the treatment response continued to improve over 6-week treatment cycles. The main end point of the study evaluated the Composite Assessment of Index Lesion Severity (CAILS) of three specific lesions and the success was determined as ≥50% reduction in CAILS score relative to baseline. Patients reported that their lesions continued to reduce throughout the treatment cycle. After the first 6-week treatment cycle, 16% of patients had a response (p=0.04 versus patients with 6 weeks of placebo treatment). The response rate continued to increase to 49% through 18 weeks of treatment (p<0.0001 versus patients with 6-week hypericin or placebo treatment).
Synthetic hypericin is a novel photodynamic therapy that uses safe, visible light for activation. The drug is topically applied to skin lesions and activated with visible light 16-24 hours after. The US Food and Drug Administration has granted orphan drug and fast track designations for the groundbreaking treatment.2.
Data throughout the study indicated that synthetic hypericin was safe and well-tolerated, while current treatment options for CTCL have significant safety concerns and black-box warnings. Synthetic hypericin will offer safer long-term options to patients. CTCL is a class of non-Hodgkin's lymphoma (NHL), a cancer of the white blood cells. Unlike other NIHLs, CTCL is caused by an expansion of malignant T-cell lymphocytes which normally migrate to the skin. There is currently no cure for CTCL.".
https://www.dermatologytimes.com/view/study-reveals-successful-results-for-treatment-of-cutaneous-t-cell-lymphoma
GLTA
...
This stock hits a resistance currently at $1.17. Newbies beware.
Your last statement is all you need to know.
Should be....but for some reason when we're holding it...it never is. Crazy how that 52 week high on any stock never seems to be when your holding it...WTF ? Or it's just a quick spike to that high. These penny stocks blow...
This puppy should be around $5 imo.
SNGX 7/27/2022: "Soligenix Receives Agreement from FDA on Initial Pediatric Study Plan for HyBryte(TM) for the Treatment of Cutaneous T-Cell Lymphoma.
Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that it has received agreement from the US Food & Drug Administration (FDA) on an initial pediatric study plan (iPSP) for HyBryte(TM) (synthetic hypericin) for the treatment of cutaneous T-cell lymphoma (CTCL). The agreed iPSP stipulates that Soligenix intends on requesting a full waiver of pediatric studies upon submission of a new drug application (NDA). Agreement with FDA on an iPSP is one of the regulatory requirements that must be met prior to submitting a NDA.
https://mma.prnewswire.com/media/1514137/HyBryte_High_Resolution_Logo.jpg
"We are pleased to have FDA's agreement on our proposal to request a full waiver of pediatric studies at the time of our HyBryte(TM) NDA filing later this year," stated Christopher J. Schaber, PhD, President & Chief Executive Officer of Soligenix. "This is consistent with decisions by the European Medicines Agency (EMA) and Medicines and Healthcare products Regulatory Agency (MHRA) in the United Kingdom which have previously granted product-specific waivers from the requirement for pediatric studies in applications for marketing authorization of HyBryte(TM) in the UK and Europe."
About HyBryte(TM)
HyBryte(TM) (research name SGX301) is a novel, first-in-class, photodynamic therapy utilizing safe, visible light for activation. The active ingredient in HyBryte(TM) is synthetic hypericin, a potent photosensitizer that is topically applied to skin lesions that is taken up by the malignant T-cells, and then activated by visible light 16 to 24 hours later. The use of visible light in the red-yellow spectrum has the advantage of penetrating more deeply into the skin (much more so than ultraviolet light) and therefore potentially treating deeper skin disease and thicker plaques and lesions. This treatment approach avoids the risk of secondary malignancies (including melanoma) inherent with the frequently employed DNA-damaging drugs and other phototherapy that are dependent on ultraviolet exposure. Combined with photoactivation, hypericin has demonstrated significant anti-proliferative effects on activated normal human lymphoid cells and inhibited growth of malignant T-cells isolated from CTCL patients. In a published Phase 2 clinical study in CTCL, patients experienced a statistically significant (p=0.04) improvement with topical hypericin treatment whereas the placebo was ineffective. HyBryte(TM) has received orphan drug and fast track designations from the FDA, as well as orphan designation from the European Medicines Agency (EMA).
The recently published Phase 3 FLASH trial trial enrolled a total of 169 patients (166 evaluable) with Stage IA, IB or IIA CTCL. The trial consisted of three treatment cycles. Treatments were administered twice weekly for the first 6 weeks and treatment response was determined at the end of the 8th week of each cycle. In the first double-blind treatment cycle, 116 patients received HyBryte(TM) treatment (0.25% synthetic hypericin) and 50 received placebo treatment of their index lesions. A total of 16% of the patients receiving HyBryte(TM) achieved at least a 50% reduction in their lesions (graded using a standard measurement of dermatologic lesions, the CAILS score) compared to only 4% of patients in the placebo group at 8 weeks (p=0.04) during the first treatment cycle (primary endpoint). HyBryte(TM) treatment in the first cycle was safe and well tolerated.
In the second open-label treatment cycle (Cycle 2), all patients received HyBryte(TM) treatment of their index lesions. Evaluation of 155 patients in this cycle (110 receiving 12 weeks of HyBryte(TM) treatment and 45 receiving 6 weeks of placebo treatment followed by 6 weeks of HyBryte(TM) treatment), demonstrated that the response rate among the 12-week treatment group was 40% (p<0.0001 vs the placebo treatment rate in Cycle 1). Comparison of the 12-week and 6-week treatment groups also revealed a statistically significant improvement (p<0.0001) between the two groups, indicating that continued treatment results in better outcomes. HyBryte(TM) continued to be safe and well tolerated. Additional analyses also indicated that HyBryte(TM) is equally effective in treating both plaque (response 42%, p<0.0001 relative to placebo treatment in Cycle 1) and patch (response 37%, p=0.0009 relative to placebo treatment in Cycle 1) lesions of CTCL, a particularly relevant finding given the historical difficulty in treating plaque lesions in particular.
The third (optional) treatment cycle (Cycle 3) was focused on safety and all patients could elect to receive HyBryte(TM) treatment of all their lesions. Of note, 66% of patients elected to continue with this optional compassionate use / safety cycle of the study. Of the subset of patients that received HyBryte(TM) throughout all 3 cycles of treatment, 49% of them demonstrated a positive treatment response (p<0.0001 vs patients receiving placebo in Cycle 1). Moreover, in a subset of patients evaluated in this cycle, it was demonstrated that HyBryte(TM) is not systemically available, consistent with the general safety of this topical product observed to date. At the end of Cycle 3, HyBryte(TM) continued to be well tolerated despite extended and increased use of the product to treat multiple lesions.
Overall safety of HyBryte(TM) is a critical attribute of this treatment and was monitored throughout the three treatment cycles (Cycles 1, 2 and 3) and the 6-month follow-up period. HyBryte's(TM) mechanism of action is not associated with DNA damage, making it a safer alternative than currently available therapies, all of which are associated with significant and sometimes fatal, side effects. Predominantly these include the risk of melanoma and other malignancies, as well as the risk of significant skin damage and premature skin aging. Currently available treatments are only approved in the context of previous treatment failure with other modalities and there is no approved front-line therapy available. Within this landscape, treatment of CTCL is strongly motivated by the safety risk of each product. HyBryte(TM) potentially represents the safest available efficacious treatment for CTCL. With no systemic absorption, a compound that is not mutagenic and a light source that is not carcinogenic, there is no evidence to date of any potential safety issues.
The Phase 3 CTCL clinical study was partially funded by the National Cancer Institute via a Phase II SBIR grant (#1R44CA210848-01A1) awarded to Soligenix, Inc.
About Cutaneous T-Cell Lymphoma (CTCL)
CTCL is a class of non-Hodgkin's lymphoma (NHL), a type of cancer of the white blood cells that are an integral part of the immune system. Unlike most NHLs which generally involve B-cell lymphocytes (involved in producing antibodies), CTCL is caused by an expansion of malignant T-cell lymphocytes (involved in cell-mediated immunity) normally programmed to migrate to the skin. These malignant cells migrate to the skin where they form various lesions, typically beginning as patches and may progress to raised plaques and tumors. Mortality is related to the stage of CTCL, with median survival generally ranging from about 12 years in the early stages to only 2.5 years when the disease has advanced. There is currently no cure for CTCL. Typically, CTCL lesions are treated and regress but usually return either in the same part of the body or in new areas.
CTCL constitutes a rare group of NHLs, occurring in about 4% of the approximate 700,000 individuals living with the disease. It is estimated, based upon review of historic published studies and reports and an interpolation of data on the incidence of CTCL that it affects over 25,000 individuals in the U.S., with approximately 3,000 new cases seen annually."
GLTA
Just a matter of time before this pops. A lot of good things happening just needs to get some attention. Good luck all
SNGX new 7/26 proxy statement looks very positive as well.
Increase in Authorized shares is outlined “for potential mergers”. SNGX has been very good with their current Authorized shares 75m, and only 43m confirmed Outstanding as of 7/25/2022.
I’m usually not for increasing Authorized but this case seems best for near-term mergers, and ensures against threat of near-term reverse? IMO??
GLTA
SNGX 7/25/2022 announces Strategic Partnership with SERB Pharmaceuticals to Supply its Novel Ricin Antigen.
SERB Pharmaceuticals pursuing therapeutic treatment against ricin poisoning using Soligenix ricin antigen
"PRINCETON, N.J., July 25, 2022 -- Soligenix, Inc. (Nasdaq: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that it has signed a worldwide exclusive license to supply its ricin antigen to SERB Pharmaceuticals (SERB), for development of a novel therapeutic treatment against ricin toxin poisoning. There is an unmet need for protection against this highly potent toxin for which there is no vaccine or therapeutic intervention available.
"We are pleased to be partnering with Soligenix on the use of their antigen to accelerate our ricin therapeutic program. With no current therapeutic options, the threat of ricin represents a significant unmet need in the field of biodefense and medical countermeasures," said Anthony Higham, CEO of SERB Pharmaceuticals. "Our expertise in antibody development and the commercial scale manufacturing capabilities acquired with BTG together with SERB's track record of reliably providing a portfolio of high-quality Chemical, Biological, Radiological, and Nuclear (CBRN) antidotes, uniquely positions us to successfully deliver a solution."
"Beyond our own development of a heat stable ricin vaccine (RiVax®) to protect against lethal ricin poisoning, which has been supported with more than $30 million dollars to date by the U.S. government, we felt it important to also partner with SERB in the development of its ricin therapeutic drug candidate," stated Christopher J. Schaber, PhD, President and CEO of Soligenix. "SERB is a leader in the field of medical countermeasures to protect the public and military forces. By supplying our novel ricin antigen as an important component of their formulation, we are hopeful that it will assist in accelerating development of this early-stage program."
In pursuit of a ricin antidote, SERB will leverage its unique broad-spectrum polyclonal antibody platform, gained in its acquisition of BTG Specialty Pharmaceuticals. This specialized manufacturing process generates binding fragments from antibodies that are specific to a given antigen, helping to ensure potency and purity. This platform is currently used to manufacture two of the company's currently marketed products, CroFab® and DigiFab®.
The antibodies will be generated using a modified form of the ricin toxin, developed by Soligenix. The modifications have removed the biological activity of the protein so that it is not toxic, while still retaining its shape to trigger an effective antibody response.
The specific licensing terms have not been disclosed at this time, but consist of a manufacturing supply agreement and small royalty percentage upon commercialization.
The Ricin Threat
Ricin is a source of concern because it is a relatively easy to obtain, easy to weaponize and highly potent toxin. Ricin can be extracted from the seeds of the castor oil plant, Ricinus communis. Ricin is one of the most toxic biological agents known—a Category B bioterrorism agent and a Schedule number 1 chemical warfare agent.
Ricin has been a threat since governments began experimenting with it during World War I. Most famously used in the assassination of Bulgarian writer Georgi Markov in 1978, ricin has been developed and deployed with alarming frequency. Several ricin attacks have been prevented in Europe and the United States in recent years, ranging from a militant group in Germany prevented from launching a ricin attack by police in 2017 to the 2020 delivery of letters laced with ricin to the White House.
About SERB Pharmaceuticals
SERB is a growing pharmaceutical company and a dedicated ally to healthcare providers treating patients with critical conditions, focusing on emergency care and rare diseases. For over 30 years we have made treating these complex and life-threatening conditions possible, supporting clinicians, healthcare systems and governments while offering hope to patients and their families. As a fully integrated company, we have the experience and capabilities to acquire, develop, and manufacture our medicines to the highest standards, and make them available worldwide through our secure supply chain. SERB acquired BTG Specialty Pharmaceuticals in March of 2021."
GLTA
Finviz Target Price 3.88.
Actually, I was surprised to find there's very little short interest according to finviz.
Short Float 1.40% of a rather small float of Shs Float 42.82M. I still think this has a very good chance of getting back over the $.
Insiders accumulated: Insider Trans +51.41%
https://finviz.com/quote.ashx?t=SNGX
LOL! Shorts?!!!! Good one. The cry of any good penny stock.
A peer reviewed article reporting successful Phase 3 results in a JAMA publication is a reasonable predictor of eventual FDA approval. IMHO, the JAMA publication is a significant milestone and harbinger of good things to come for SNGX.
This'll get there imo. Doubt institutional interest will stay on the sidelines much longer.
Shorts trying to hold this back imo.
If all this were true, the stock would not be trading under a buck.
HyBryte(TM) Phase 3 FLASH Study for the Treatment of Cutaneous T-Cell Lymphoma Published in JAMA Dermatology
11:17 AM ET 7/20/22 | Dow Jones
-- Published findings demonstrate that HyBryte(TM) treatment statistically
significantly reduced CTCL lesion size
-- HyBryte(TM) has potential to address a critical gap in treatment of
early-stage CTCL
PRINCETON, N.J., July 20, 2022 /PRNewswire/ -- Soligenix, Inc. (NASDAQ: SNGX) (Soligenix or the Company), a late-stage biopharmaceutical company focused on developing and commercializing products to treat rare diseases where there is an unmet medical need, announced today that the results of its successful Phase 3 FLASH (Fluorescent Light Activated Synthetic Hypericin) study evaluating HyBryte(TM) (synthetic hypericin) for the treatment of cutaneous T-cell lymphoma (CTCL) has been published in the Journal of the American Medical Association (JAMA) Dermatology.
"The peer reviewed publication of these data in JAMA Dermatology is a testament and further validation to the importance of the findings for the scientific and CTCL disease communities," stated Ellen Kim, MD, Professor of Dermatology and Medical Director, Dermatology Clinic, Perelman Center for Advanced Medicine at the Hospital of the University of Pennsylvania, and the Lead Principal Investigator for the Phase 3 FLASH study. "With its chronic course and major impact on patient quality of life, CTCL is an orphan disease in urgent need of additional treatment options that are well-tolerated and safe over the long haul. The results from this Phase 3 study, which is the largest double-blind, randomized, placebo-controlled trial in CTCL to date, represents an important leap forward in the development of potential therapies to meet this unmet medical need."
The published findings demonstrate that HyBryte(TM) treatment statistically significantly reduced lesion size, with the treatment response further improving over successive 6-week treatment cycles. The primary endpoint evaluated the CAILS (Composite Assessment of Index Lesion Severity) score of three treated index lesions and success was defined as >=50% reduction in CAILS score relative to baseline. Lesion response continuously improved with treatment duration. After the first 6-week treatment window, 16% of patients had a response (p=0.04 versus patients with 6 weeks of placebo treatment; primary endpoint). This response rate continued to significantly increase to 49% through 18 weeks of treatment (p<0.0001 versus patients with 6-week hypericin or placebo treatment). Throughout the study, HyBryte(TM) was safe and well-tolerated. Importantly, HyBryte(TM) was observed to perform similarly against both patch and thicker plaque lesions characteristic of CTCL.
"In treating CTCL, which is a chronic cancer with no cure, long-term safety is of paramount concern. Most current treatment options for CTCL are associated with significant safety concerns, including black-box warnings. HyBryte(TM) treatment has demonstrated strong and rapid efficacy with a very benign safety profile," stated Dr. Richard Straube, MD, Senior Vice President and Chief Medical Officer of Soligenix. "This is of significant benefit to patients living with this difficult disease. The substantial increase in efficacy with longer treatment and the similar performance against both patch and plaque lesions are particularly encouraging. As one of the largest studies in CTCL, this study and this publication establishes a new benchmark in CTCL treatment."
About JAMA Dermatology
JAMA Dermatology is an international peer-reviewed journal published online weekly and in print/ online issue 12 times a year. It is one of the highest ranked journals in dermatology, with an acceptance rate of 9%. The journal, which has been in continuous publication since 1882, publishes studies in the areas of medical, surgical, pediatric, geriatric dermatology, oncologic and aesthetic dermatology. It prioritizes clinical and laboratory studies that reveal new information pertinent to the interests and needs of the medical dermatologist, dermatologic surgeon, and all those concerned with state-of-the-art care of cutaneous disease. The journal believes that knowledge derived from well-designed clinical trials and studies of cost-effectiveness are especially important for improving the practice of dermatology. JAMA Dermatology is a member of the JAMA Network family of journals, which includes JAMA, 11 JAMA Network specialty journals, and JAMA Network Open.
It is true, for longterm shareholders, the stock has performed poorly, however, the basic science is sound and if the company actually files the NDA this year, the future is promising. No illusions, the regulatory path ahead is no walk in the park, but it looks doable. With FDA approval and the extremely low "float", the risk/reward is quite attractive.
Resistance at .763. I'm betting it falls back once more. What an awful stock this has been.
|
Followers
|
208
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9772
|
|
Created
|
06/06/06
|
Type
|
Free
|
| Moderators | |||
| SNGX [NASD] |
| Soligenix, Inc. |
| Healthcare | Biotechnology | USA |
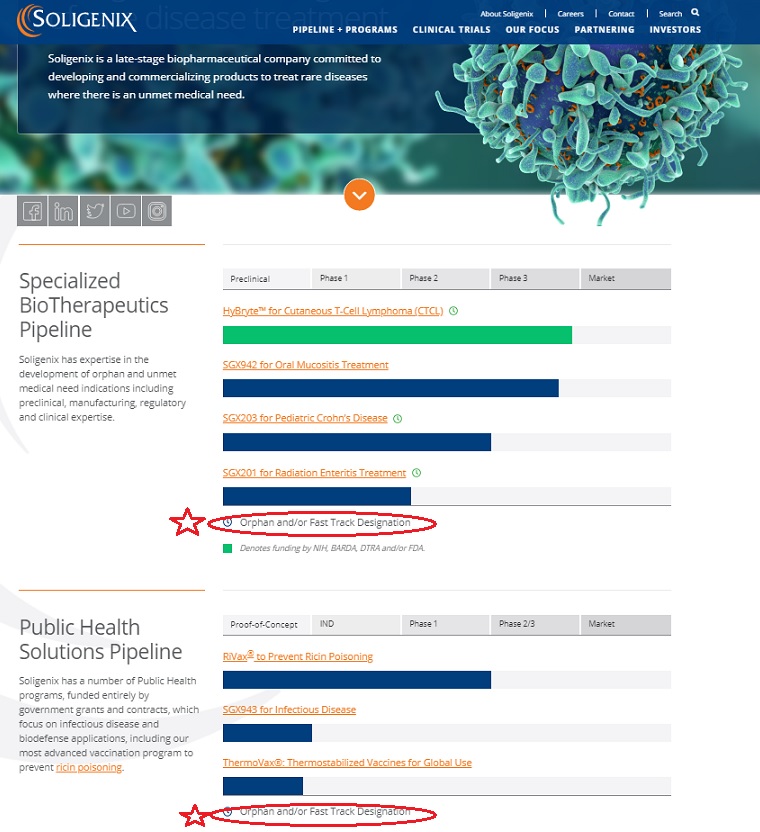
Soligenix, Inc. (Soligenix) is a late-stage biopharmaceutical company.
The Company is focused on developing and commercializing products to treat rare diseases.
The Company operates through two business segments:
Specialized BioTherapeutics and Public Health Solution. Soligenix’s Specialized BioTherapeutics business segment is developing
a photodynamic therapy (SGX301) utilizing topical synthetic hypericin activated with safe visible fluorescent light for the treatment of cutaneous T-cell lymphoma (CTCL).
The Public Health Solutions business segment includes active development programs for RiVax,
its ricin toxin vaccine candidate and SGX943, its therapeutic candidate for antibiotic-resistant and emerging infectious disease.
CTCL is its innate defense regulator technology, dusquetide (SGX942) for the treatment of oral mucositis in head and neck cancer.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
Soligenix has expertise in the development of orphan and unmet medical need indications including preclinical, manufacturing, regulatory and clinical expertise.
| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment | ||||
| | | | | |
Orphan and/or Fast Track Designation
Soligenix has a number of Public Health programs, funded entirely by government grants and contracts, which focus on infectious disease and biodefense applications, including our most advanced vaccination program to prevent ricin poisoning.
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation


| Preclinical | Phase 1 | Phase 2 | Phase 3 | Market |
|---|---|---|---|---|
| HyBryte™ for Cutaneous T-Cell Lymphoma (CTCL) almost done [in phase 3] | ||||
| | | | | |
| SGX942 for Oral Mucositis Treatment almost done [in phase 3] | ||||
| | | | | |
| SGX203 for Pediatric Crohn’s Disease [phase 2 almost done] | ||||
| | | | | |
| SGX201 for Radiation Enteritis Treatment [phase 1 going into phase 2] | ||||
| Proof-of-Concept | IND | Phase 1 | Phase 2/3 | Market |
|---|---|---|---|---|
| RiVax® to Prevent Ricin Poisoning | ||||
| | | | | |
| SGX943 for Infectious Disease | ||||
| | | | | |
| ThermoVax®: Thermostabilized Vaccines for Global Use | ||||
| | | | | |
Orphan and/or Fast Track Designation
| PER IHUB MGMT |
02-07-2021
DISCLAIMER: ONLY FOR MICK
https://investorshub.advfn.com/boards/profilea.aspx?user=1012
*The Board Monitor and herewithin , are not licensed brokers and assume NO responsibility for actions,
investments,decisions, or messages posted on this forum.
CONTENT ON THIS FORUM SHOULD NOT BE CONSIDERED ADVISORY NOR SOLICITATION
AUTHORS MAY HAVE BUYS OR SELLS WITH THE COMPANIES MENTIONED IN TRADING POSTERS SHOULD DUE DILIGENT BUYING OR SELLING.
ALL POSTING SHOULD BE CONSIDERED FOR INFORMATION ONLY. WE DO NOT RECOMMEND ANYONE BUY OR SELL ANY SECURITIES POSTED HEREWITHIN.
ANY trade entered into risks the possibility of losing the funds invested.
• There are no guarantees when buying or selling any security.Any
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
