Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
$$$ SEEL $$$ on watch
https://schrts.co/HkSFDjxR
Hopefully, a reversal now.
Someone sold a 400k block into the open market
Seelos Therapeutics Announces Mid-Year 2021 Business and Clinical Update
August 09 2021 - 08:00AM
https://ih.advfn.com/p.php?pid=nmona&article=85786220
Wish we could track daily change in OS.
When OS stops increasing, time to buy.
Could easily be back to $4-5 by year end, or more depending on the catalyst.
IMO GLTA
I think dilution by company.
Why is this still down? TIA
GLTA
Absolutely massive news : NEW YORK, July 7, 2021 /PRNewswire/ -- Seelos Therapeutics, Inc. (Nasdaq: SEEL), a clinical-stage biopharmaceutical company focused on the development of therapies for central nervous system disorders and rare diseases, today announced positive in vivo data demonstrating down-regulation of SNCA mRNA and protein-expression from a study of SLS-004 in an in-vivo rodent model utilizing CRISPR-dCas9 gene therapy technology. A single dose of SLS-004 produced a therapeutically desirable 27% reduction on SNCA mRNA and a 40% reduction in SNCA protein expression.
(PRNewsfoto/Seelos Therapeutics, Inc.)
Parkinson's disease (PD) is the second most common neurodegenerative disorder in the world and currently there is no effective treatment to prevent PD or to halt its progression. The SNCA gene has been implicated as a highly significant genetic risk factor for PD. In addition, accumulating evidence has suggested that elevated levels of alpha-synuclein (a-synuclein) are causative in the pathogenesis of PD. Patients with impaired regulation of the SNCA gene show as high as 200% expression of a-synuclein protein. A reduction of 25%-50% in SNCA mRNA and protein expression should be sufficient to restore normal physiological levels of SNCA.
"We are highly encouraged by these preliminary findings demonstrating downregulation of SNCA mRNA and SNCA protein expression in this in vivo model," said Raj Mehra Ph.D., Chairman and CEO of Seelos. "Overexpression of alpha-synuclein has been implicated as a highly significant risk factor for Parkinson's and the accumulation of this protein is a pathological hallmark of synucleinopathies for additional diseases such as dementia with Lewy bodies and multiple system atrophy."
"The effect of SNCA downregulation achieved with our innovative CRISPR-dCas9 platform technology in this in vivo animal study is very promising and we plan to further validate the safety and efficacy of our lentivirus-based epigenome-editing approach in a full-range preclinical study under our Sponsored Research Agreement with Seelos, with the aim of reversing the Parkinson's disease-related pathology," said Boris Kantor, Ph.D., Associate Research Professor, Duke University School of Medicine.
Ornit Chiba-Falek, Division Chief, Translational Brain Sciences at Duke added, "With Seelos, we've moved forward with our innovative CRISPR-dCas9 based gene therapy approach onto in vivo studies. This pilot experiment in mice showed promising results with an effect on SNCA reduction within the presumed target therapeutic window. We are continuing the preclinical studies to further the development of epigenome-editing targeting SNCA towards precision medicine for Parkinson's disease."
Seelos Announces Positive In Vivo Data from a Gene Therapy Study of SLS-004 Utilizing CRISPR-dCas9 for Parkinson’s Disease Pathology
The PR this morning clearly states that the fda communicated with the company that they can move forward with approval after this part 2 of their Ketamine trial.
“We have received correspondence from the FDA that Part 2 will serve as a registrational study for the SLS-002 program and we will continue discussions with the FDA on the regulatory pathway going forward."
HUGE!
Seelos Therapeutics Doses First Patient in Part 2 of a Registrational Study of SLS-002 (Intranasal Racemic Ketamine) for Acute Suicidal Ideation and Behavior in Patients with Major Depressive Disorderhttps://t.co/VReQD02zFD#depression #SuicideAwareness
— Seelos Therapeutics (@seelostx) July 6, 2021
Reversal likely, yes.
This breaks out above $2.90
One of the most underrated patents for $SEEL.
Japan has a recent history for massive amounts of Phobias, depression and suicide.
This is larger than most realize.
https://psychedelicinvest.com/seelos-announces-issuance-of-a-new-patent-and-an-additional-notice-of-allowance-in-japan-for-intranasal-racemic-ketamine-sls-002/
Product Indication Development Phase Development Status
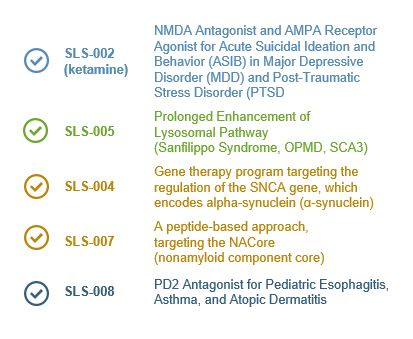
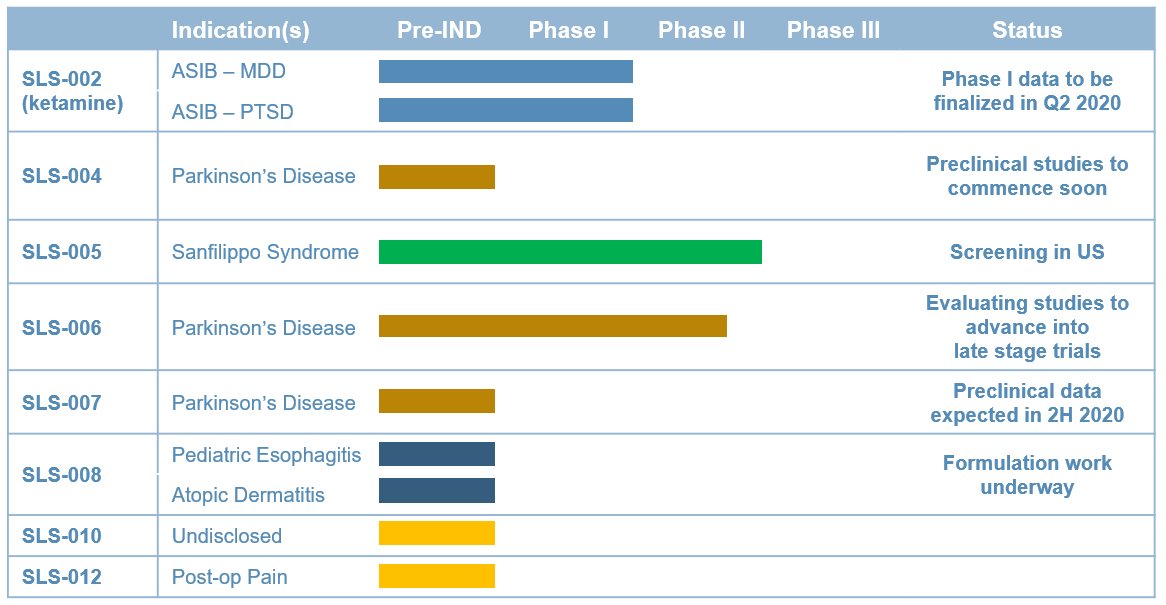
SLS-002 Acute Suicidal Ideation and Behavior (ASIB) in Phase II Completed open-label patient enrollment of Part 1 of proof-of-concept study; announced the initial topline data from Part 1 of the proof-of-concept study on May 17, 2021
Intranasal Racemic Ketamine Major Depressive Disorder (MDD)
SLS-005 Amyotrophic Lateral Sclerosis (ALS) Phase IIb/III Startup activities initiated
IV Trehalose Sanfilippo Syndrome Phase II Obtaining natural history data
SLS-004 Parkinson's Disease (PD) Pre-IND Preclinical studies ongoing
Gene Therapy
SLS-006 Parkinson's Disease (PD) Phase II/III Evaluating studies to advance into late-stage trials
Partial Dopamine Agonist
SLS-007 Parkinson's Disease (PD) Pre-IND Preclinical study ongoing
Peptide Inhibitor
SEEL is a great $$$$$ opportunity.
Watch for tutes: pension plans, University endowment funds, mutual funds, and hedge funds accumulating shares each session.
Various PT, ranging from 8$ to $15. > July going to be a share price transition well into the teens.
SEEL
Over $90M cash, huge pipeline, and numerous funds with $8+ PT’s
Getting some nice pre market action here.
Trading right at the resistance.
A close over $2.60 is very bullish
BTIG initiated shares as a buy with a price target of $14.
B. Riley said the results beat expectations, especially with regards to suicidality, leading to a buy reiteration and $15 price target.
Trades at $2.64.
Bounced off the bottom of $2.60.
Ketamine PH3 about to start
Seel Added to the Russell 2000,Russell 3000, and Russell Microcap.
-Seel Announces its Inclusion in the First U.S. Listed (ETF) Focused on Psychedelics
-Seel Receives European Orphan Drug Designation for SLS-005 (Trehalose) in Amyotrophic Lateral Sclerosis (ALS)
SEEL - some pretty decent market buys. They gotta be starting their SLS-002 Ketamine PH3 soon yeah? @seelostx
Priority drug voucher for SLS-005?
$56M cash?!
Massive pipeline…..
Seelos Therapeutics shares are trading higher after Cantor Fitzgerald initiated coverage on the stock with an Overweight rating and a $10 price target.
Seelos Therapeutics Announces its Inclusion in the First U.S. Listed Exchange Traded Fund (ETF) Focused on Psychedelics
- ETF Trades on the New York Stock Exchange Under the Symbol PSY
NEW YORK, June 1, 2021 -- Seelos Therapeutics, Inc. (Nasdaq: SEEL), a clinical-stage biopharmaceutical company focused on the development of therapies for central nervous system disorders and rare diseases, announced today that it has been included in the Defiance Next Gen Altered Experience ETF, the first U.S. listed Exchange Traded Fund (ETF) focused on psychedelics which began trading on Friday, May 28th on the New York Stock Exchange under the symbol PSY.
Seelos Therapeutics Receives European Orphan Drug Designation for SLS-005 (Trehalose) in Amyotrophic Lateral Sclerosis (ALS)
May 27 2021 - 07:00AM
PR Newswire (US)
https://ih.advfn.com/p.php?pid=nmona&article=85222524
News today.
It needs help.
They didn’t disclose the deets so chances are there is more downside. Par for the course with these guys....always raising money to cap momentum.
Company announced an offering today. If it’s priced in, SEEL moves. If not, there will likely be consolidation for the time being.
SEEL, Gap filled?
Seelos Therapeutics Inc (SEEL)
3.27 +0.08 (+2.51%) 05/19/21 [NASDAQ]
3.06 x 46 3.08 x 10
Post-market 3.06 -0.21 (-6.42%) 16:36 ET
Quote Overview for Wed, May 19th, 2021
was the downturn or breakdown happened to fill gap?, if so, the chart shows gap filled in last couple of sessions, Seelos will move higher, could be moving in dollars a day type move!?...will not be surprised if tomorrow gap up ride into mid 4s or 5s!?.
|
Followers
|
245
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
30373
|
|
Created
|
09/14/05
|
Type
|
Free
|
| Moderators | |||
Seelos Therapeutics is exploring new ways to think about and treat central nervous system (CNS) disorders in neurological diseases and disorders and in rare diseases. Our trials are designed to further evaluate the safety and efficacy of our acquired assets. These come to Seelos with well characterized efficacy and safety, thereby avoiding much of the lengthy and burdensome process of preclinical R&D.
By working with clinically ready novel and reformulated compounds we will move from asset acquisition to meaningful data readouts in an expedited and efficient fashion. It is our goal to develop unique and disruptive therapies and approaches to improve treatment of large known diseases, offer new treatments for large unmet or recently characterized indications as well as advance therapies for orphan indications.
Our approach to this process will require development that is appropriate and efficient for a company our size in the scope of work each program requires in regards to trial size and duration.







We invite you to stay update on the progress of our assets in our clinical development programs.





| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
