Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Nice bid support, this is going green from here.
13 years in this turd!!!!!!!!
Fck you Dees & Culpepper.
May God have mercy on your soul!!
Picked up some more shares, averaging down. I'm thinking tax loss selling is probably almost done. Up from here.
The PhII PH-10 trial has just been completed https://clinicaltrials.gov/ct2/show/study/NCT02322086
NEWS -- PROVECTUS BIOPHARMACEUTICALS PROVIDES UPDATE ON MELANOMA CLINICAL DEVELOPMENT PROGRAM FOR INVESTIGATIONAL DRUG PV-10
Videos of/about Ed P Accepting Haslam Award
Hello. Long time PVCT holder. I follow the other board quite frequently but do not have an account there.
I stumbled upon these 2 videos about/of Ed accepting his Haslam award, and I don't think they were posted over on IV. If they were already, I apologize. Here they are, and for the guys who are members of both boards, feel free to add them there as well. Thank you.
News Story
Search Company
11/14/2018 07:00:00 AM
PROVECTUS BIOPHARMACEUTICALS PROVIDES DRUG DEVELOPMENT UPDATE FOR IMMUNO-DERMATOLOGY ASSET PH-10
By GlobeNewswire
KNOXVILLE, TN, Nov. 14, 2018 (GLOBE NEWSWIRE) -- Provectus (OTCQB:PVCT) today provided an update on the Company's small molecule clinical development program for its investigational immuno-dermatology drug PH-10 for the treatments of psoriasis and atopic dermatitis. Pathways significantly improved by PH-10 use include published psoriasis transcriptomes and cellular responses mediated by IL-17, IL-22, and interferons.
Provectus has begun work in support of extended 12-week administration (proof-of-concept or POC) for topically applied, non-steroidal PH-10:
The Company has finished two toxicology-focused, non-clinical, single administration studies using carbon-14- (14C-) labeled rose bengal disodium (RB) to demonstrate lack of systemic uptake. RB is PH-10's active pharmaceutical ingredient. Radio-labeled RB, however, is easier to detect in plasma and tissues at very low levels than RB itself. These data suggest there is minimal potential of visceral target organ effects from topical application of PH-10.
The goal of a planned, non-clinical, toxicology-focused, 12-week administration study would be to demonstrate local effects in the skin from the extended use of PH-10 and identify any potential systemic toxicities.
When completed, the 12-week POC program may allow for direct comparison of PH-10 to approved topical treatments for psoriasis and atopic dermatitis.
PH-10's mechanism of action (MOA), as well as its implications for immunology, are beginning to be understood. James Krueger, MD, PhD and members of the Laboratory for Investigative Dermatology at The Rockefeller University have elucidated PH-10's MOA for psoriasis in a previous clinical study (NCT02322086).
A clinical study of PH-10's MOA for atopic dermatitis is planned for 2019, and would be undertaken by a different dermatology and immunology investigator.
Dominic Rodrigues, Vice Chair of the Company's Board of Directors, said, "PH-10 is a clinical-stage, multi-indication viable, and immuno-dermatology-capable pharmaceutical asset. It has a novel mechanism of action; exhibits attractive preliminary safety and efficacy properties; has compelling product, manufacturing, and pricing characteristics; and is based on a family of small molecules entirely and wholly owned by Provectus. We are committed to properly monetizing this important asset for shareholders by developing PH-10 into a Phase 3-ready candidate, for both psoriasis and atopic dermatitis, in an efficient, effective, and industry-recognized manner."
About PH-10
Provectus' investigational dermatology drug, PH-10, a topically applied, non-steroidal, small molecule-based gel, is undergoing clinical study for inflammatory dermatoses like psoriasis and atopic dermatitis. PH-10 is self-applied to affected skin with photoactivation via ambient exposure to visible light.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs for oncology and dermatology based on a family of molecules called halogenated xanthenes. The Company's lead investigational cancer drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers. Information about the Company's clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains "forward-looking statements" as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as "anticipate," "believe," "could," "estimate," "expect," "intend," "may," "plan," "predict," "project," "will," and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017 ).
###
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Source: Provectus Biopharmaceuticals Inc.
Home
News
Quotes
Settings
NEWS -- University of Tennessee Haslam College of Business Recognizes Provectus Biopharmaceuticals Chairman Ed Pershing as 2018 Outstanding Entrepreneur
KNOXVILLE, TN, Nov. 12, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that Ed Pershing, CPA, the Chairman of the Company’s board of directors, was recognized by the Haslam College of Business (HCB) at the University of Tennessee, Knoxville as HCB’s 2018 Outstanding Entrepreneur. Mr. Pershing also is Chief Executive Officer and co-founder of 35-year-old, privately-held, healthcare management consulting and accounting firm PYA, P.C.
The following is an excerpt from HCB’s alumni award gala’s program and website about him:
Ed Pershing (HCB, ’74) grew up in Alcoa, Tennessee, surrounded by a supportive family. His two older brothers attended the University of Tennessee, Knoxville, before he and his youngest brother followed three years later. “I witnessed firsthand how much they enjoyed and valued the experience,” Pershing says. “I never considered going anywhere else.”
An excellent student, Pershing received offers from each of the big eight accounting firms after graduation. He joined EY and was soon selected to participate in an education program on healthcare consulting. It was 1976, a time when consulting was not usually identified as a viable career, but Pershing had discovered his calling. “I was very blessed to participate in that program,” he says. “It set the foundation for my career and allowed me to pursue a path I’m very passionate about: healthcare and public health.”
Pershing co-founded Pershing Yoakley & Associates with fellow Haslam graduate Doug Yoakley (HCB, ’78) in 1983. “I started PYA with the idea of establishing a strong consulting practice but operating as a CPA firm to convey our commitment to sound business practices and ethical conduct,” Pershing says. Pershing’s focus on healthcare consulting led PYA to become a leader in the niche, recognized by Modern Healthcare as the ninth largest privately-held healthcare management consulting firm in the country. Today, approximately 80 percent of PYA’s practice is related to the healthcare industry, and consulting accounts for more than two-thirds of their work.
PYA also founded a number of affiliate companies in other sectors, including real estate, investment advisory services, healthcare claims analysis and auditing, and most recently, advanced analytics. “These ongoing opportunities to create new business enterprises have been extraordinarily satisfying for me,” says Pershing. “They allow us to expand our impact and our business footprint.”
Over the years, Pershing has retained firm ties to the Haslam College of Business, serving in the past as a member of the Chancellor’s Associates and currently on the Dean’s Advisory Council at Haslam. “Right here in our backyard, we have the ability to connect with students and professors in a way that enables us to attract the best and brightest to join us at PYA,” Pershing says. “Haslam is a pipeline for us for being able to identify and retain remarkable talent.”
The road to success as an entrepreneur can be summed up in one word, says Pershing: perseverance. “You need personal discipline and diligence each day,” he says. “Even when you don’t see progress, you’re building a foundation that will result in progress.”
About PV-10
Provectus’ investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and non-clinical study for pediatric cancers. Intratumoral injection of PV-10 can yield immunogenic cell death in solid tumor cancers and stimulate tumor-specific reactivity in circulating T cells.
About PH-10
Provectus’ investigational dermatology drug, PH-10, a topically applied, non-steroidal, small molecule-based gel, is undergoing clinical study for inflammatory dermatoses like psoriasis and atopic dermatitis. PH-10 is self-applied to affected skin with photoactivation via ambient exposure to visible light. Pathways significantly improved by PH-10 treatment include published psoriasis transcriptomes and cellular responses mediated by IL-17, IL-22 and interferons.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs for oncology and dermatology based on a family of molecules called halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
###
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Is this as big as I think it is?
$$$PVCT$$$
The sell on news crowd. Not worried.
Great news, and the market makers don't like it, must have a lot of shorts trying to cover.
News Story
Search Company
11/05/2018 08:00:00 AM
US FDA GRANTS PV-10 ORPHAN DRUG DESIGNATION FOR NEUROBLASTOMA
By GlobeNewswire
KNOXVILLE, TN, Nov. 05, 2018 (GLOBE NEWSWIRE) -- Provectus (OTCQB:PVCT) today announced that the Company was granted orphan drug designation (ODD) by the U.S. Food and Drug Administration (FDA) for small molecule oncolytic immunotherapy PV-10 for the treatment of neuroblastoma, a non-central nervous system (CNS) pediatric solid tumor. Intratumoral injection of PV-10 can yield immunogenic cell death (ICD) in solid tumor cancers and stimulate tumor-specific reactivity in circulating T cells.1-4
Neuroblastoma forms from immature nerve cells and usually begins in the adrenal glands. It may also begin in the abdomen, chest, or near the spine. Neuroblastoma most often occurs in children younger than 5 years of age, and presents when the tumor grows and causes symptoms. According to the National Cancer Institute SEER Cancer Statistics Review 1975-2015, the 5-year survival among children 0 to 19 years of age is 75.2%.5
Initial non-clinical testing of PV-10 in treatment-refractory neuroblastoma has closely paralleled previous non-clinical and clinical study of PV-10 for murine and human adult solid tumors, at both the tumor (selective destruction of injected tumors) and cellular (ICD) levels.6 Non-clinical investigation by member institutions of the Pediatric Oncology Experimental Therapeutics Investigators' Consortium (POETIC) has confirmed that ICD also occurs in neuroblastoma.
The FDA grants ODD status to medicines intended for the treatment, diagnosis or prevention of rare diseases or disorders that affect fewer than 200,000 people in the US. ODD status qualifies companies for benefits that include seven years of market exclusivity following marketing approval, tax credits on U.S. clinical trials, eligibility for orphan drug grants, and waiver of certain administrative fees.
ODD status previously was granted to PV-10 for the treatments of metastatic melanoma in 2007 and hepatocellular carcinoma (HCC) in 2013.
About Neuroblastoma
If detected at an early stage, surgery is the definitive treatment. Once neuroblastoma has recurred or spread from the primary site, therapeutic options are limited principally to chemotherapy or clinical trial.7 A recent review of emerging treatment options for neuroblastoma noted that "less than 50% of patients with high-risk neuroblastoma will survive long-term with current therapies, and survivors are at risk for serious treatment-related late toxicities."8
Therapeutic options include high-dose systemic chemotherapy with alkylating agents, radiation therapy, experimental treatments like regional therapy, or immunotherapy. Unlike many adult solid tumor types, pediatric solid tumors have largely proven unresponsive to immune checkpoint inhibitors such as anti-PD-1 antibodies. A recent Phase 2 study of pembrolizumab in pediatric solid tumors showed that less than 20% of patients tested were candidates for anti-PD-1 therapy based on low expression of the PD-L1 biomarker, and no candidates achieved an objective response.9
About PV-10
Provectus' lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About the Pediatric Oncology Experimental Therapeutics Investigators' Consortium
The Pediatric Oncology Experimental Therapeutics Investigators' Consortium (POETIC) was founded in February 2003 by Dr. Tanya Trippett at Memorial Sloan Kettering Cancer Center and Dr. Lia Gore at the University of Colorado Cancer Center. POETIC is composed of ten large academic medical centers in North America with a major emphasis on comprehensive cancer care and research that provide the collaborative and research strength needed to complete intensive phase I and II studies. Each of the institutions is uniquely suited to complete early studies in the pediatric and adolescent populations. POETIC's assets include membership in NCI-designated Comprehensive Cancer Centers, on-site NIH-funded pediatric and/or general clinical translational research centers (CTRCs/CTSAs), and active collaborations with developmental therapeutics programs for adults at a majority of its member institutions. The availability of strong basic science and translational research programs at the institutions allows focus on the development and evaluation of new therapeutic strategies for patients with cancer and related disorders. POETIC's pediatric oncology studies focus on the biologic basis for anti-cancer therapy, and in particular, attempt to explore and evaluate novel agents and/or combinations of therapies early in clinical development as well as new approaches to targeted delivery. For additional information about POETIC, please visit the Consortium's website at www.poeticphase1.org.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are chemical small molecules. Information about the Company's clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
5. Noone et al. (eds). SEER Cancer Statistics Review, 1975-2015, National Cancer Institute. Bethesda, MD, based on November 2017 SEER data submission, posted to the SEER web site, April 2018. Table 28.8.
6. Swift et al. In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors. Journal of Clinical Oncology 36, no. 15_suppl, 10557, 2018.
7. PDQ Pediatric Treatment Editorial Board. Neuroblastoma Treatment (PDQ®): Health Professional Version. 2018 Aug 17. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002-.
8. Applebaum et al. Emerging and investigational therapies for neuroblastoma. Expert Opinion on Orphan Drugs 5(4):355, 2017.
9. Geoerger et al. KEYNOTE-051: An update on the phase 2 results of pembrolizumab (pembro) in pediatric patients (pts) with advanced melanoma or a PD-L1-positive advanced, relapsed or refractory solid tumor or lymphoma. Journal of Clinical Oncology 36, no. 15_suppl, 10525, 2018.
FORWARD-LOOKING STATEMENTS: This release contains "forward-looking statements" as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as "anticipate," "believe," "could," "estimate," "expect," "intend," "may," "plan," "predict," "project," "will," and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017 ).
###
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Source: Provectus Biopharmaceuticals Inc.
Quotes From This Article
PVCT$0.0674 0.0016 2.32%
Provectus Biopharmaceuticals Inc
Home
News
Quotes
Settings
NEWS -- PROVECTUS ANNOUNCES PRESENTATION OF PHASE 1 TRIAL OF PV-10 FOR TREATMENT OF SYMPTOMATIC NEUROENDOCRINE TUMORS METASTATIC TO LIVER AT ESMO 2018 CONGRESS
KNOXVILLE, TN, Oct. 23, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that the Company's ongoing Phase 1 clinical trial of single agent PV-10 for the treatment of symptomatic neuroendocrine tumors (NETs) metastatic to the liver was the subject of a trials in progress (TIP) poster presented at the ESMO 2018 Congress (the European Society for Medical Oncology annual meeting), held in Munich, Germany from October 19-23, 2018. Intralesional injection of PV-10 can yield immunogenic cell death in solid tumor cancers that results in tumor-specific reactivity in circulating T cells.1,2,3,4
This single-center Phase 1 study is being conducted at The Queen Elizabeth Hospital in Adelaide, Australia to evaluate the potential safety, tolerability, and preliminary efficacy of PV-10 in patients with metastatic NETs (mNETs) of the liver (NCT02557321). The primary endpoint for the trial is safety, and secondary endpoints include objective response rate, target lesion somatostatin receptor (SSTR) expression, and biochemical response. Six patients in the first cohort each received one injection of PV-10 to one target lesion per treatment cycle. Patients are currently being enrolled into the second cohort that allows for injection of up to three lesions per cycle.
The Company currently plans to present comprehensive clinical data at a medical conference in the first quarter of 2019.
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “This poster describes Provectus’ continued progress investigating the use of PV-10, a small molecule oncolytic immunotherapy, for cancers of the liver. This work, like similar work underway in our companion Phase 1 basket study of hepatic tumors, builds on PV-10’s extensive use in clinical trial and expanded access settings to treat cutaneous and subcutaneous disease from melanoma and other cancers of the skin.”
Mr. Rodrigues added, “In addition to assessing the direct effect of PV-10 on injected visceral tumors, this neuroendocrine tumor work has the potential to extend our knowledge of PV-10’s systemic benefit resulting from adaptive immune system activation by oncolytic destruction of tumor tissue, which is fundamental to our work in melanoma. While melanoma has a high mutation frequency and is considered one of the most immunogenic types of cancer, neuroendocrine tumors have much lower mutation frequency, and thus are much less immunogenic. As such, this trial offers us insight into another dimension of PV-10’s use, the treatment of immunologically ‘cold’ cancers.”
A copy of the poster presentation is currently available on Provectus’ website at https://www.provectusbio.com/media/docs/publications/ESMO-2018_Abstract_1334TiP.pdf.
About Neuroendocrine Tumors
NETs associated with the gastrointestinal tract have endocrine secretory properties and a propensity for metastasis to the lungs, bronchi, and liver. mNETs located in the midgut and liver often secrete vasoactive products, giving rise to symptoms such as flushing and diarrhea, wheezing, abdominal cramps, and peripheral edema.
About PV-10
Provectus’ lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are small molecule chemical agents. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
NEWS -- PROVECTUS ANNOUNCES PRESENTATION OF UPDATED RESULTS FROM PHASE 1B TRIAL OF PV-10 IN COMBINATION WITH KEYTRUDA® (PEMBROLIZUMAB) FOR TREATMENT OF ADVANCED MELANOMA AT SMR 2018 CONGRESS
I guess someone knows something. I saw the volume so I bought 20,000 shares
Any hidden news that I am unable to find? A big surge in the volume. Already 5 times the 10 day average volume.
Here is the ESMO link http://cslide.ctimeetingtech.com/esmo2018/attendee/confcal/presentation/list?q=1334TiP The "TiP" means that it will likely be a "trial in progress" poster, which simply describes the trial and does not disclose any results.
NEWS -- PROVECTUS BIOPHARMACEUTICALS EXPANDS PATENT PORTFOLIO WITH ADOPTIVE CELL TRANSFER-BASED USE OF INVESTIGATIONAL CANCER DRUG
KNOXVILLE, TN, Sept. 18, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed the Company’s patent application for the use of adoptive cell transfer (ACT) of PV-10-induced T cells for the treatment of solid tumor cancers. Co-inventors include current and former members of a longstanding, PV-10-focused, translational research team at Moffitt Cancer Center (Moffitt) in Tampa, Florida, which has undertaken work in melanoma, breast cancer, and pancreatic cancer starting in 2011 1.
PV-10 is Provectus’ lead investigational drug for the treatment of adult and pediatric solid tumor cancers, and is the first small molecule oncolytic immunotherapy. It is administered via intralesional injection directly into superficial or visceral tumors to elicit immunogenic cell death of these injected tumors. PV-10 is the subject of several ongoing clinical trials of adults in melanoma 2,3 and cancers of the liver 4,5. PV-10 has also been shown to induce cell death in pediatric solid tumor cell lines derived from relapsed neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma 6.
Under the treatment concept of the allowed patent application, which is entitled “Method of Ex Vivo Enhancement of Immune Cell Activity for Cancer Immunotherapy with a Small Molecule Ablative Compound,” PV-10 is injected into solid tumors and the resulting immune products – T cells trained via PV-10 oncolytic immunotherapy to be functional against treated tumors – are harvested, banked, and amplified. Amplified T cells may be administered via ACT, if needed. The allowed patent application also covers the ACT treatment of either the original patient or other immunologically-suitable patients.
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “Hundreds of patients have received intralesional PV-10 for a variety of solid tumor types. One of several logical paths for expanding our clinical development program is to explore harnessing functional immunologic activation produced by PV-10 oncolytic immunotherapy via the use of adoptive cell transfer. Protecting Provectus’ intellectual property in this regard was an important and necessary task.”
About PV-10
Provectus’ lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are chemical small molecules. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
References
1. Moffitt’s translational research work on PV-10 (rose bengal) to date has resulted in three peer-reviewed publications:
I remember when this stock ran from .60 to $6 per share on pipe dreams
Now the stock has real promise with strong clinical trial results and a combination study with the best cancer drug on the planet (Keytruda) and we cant break this .067 to .074 range.
What gives?
That's the thing, could be any time. I'm hoping for anytime soon.
How much longer does everyone suspect this will wait on the launch pad? Wanting to see 0.10 before 2019 hits. Realistic or not?
NEWS -- PROVECTUS BIOPHARMACEUTICALS PROVIDES UPDATE ON GI CANCER PROGRAM FOR INVESTIGATIONAL DRUG PV-10
KNOXVILLE, TN, July 31, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today provided an update on the Company’s gastrointestinal (GI) cancer clinical development program for its lead investigational drug PV-10, which is administered percutaneously when targeting liver tumors.
Provectus’ ongoing, multi-center, open-label, Phase 1 clinical trial of PV-10 for patients with hepatic lesions (NCT00986661) – a “basket study” open to patients with different liver tumor types(1) – has expanded to include a single-site cohort of 10 uveal melanoma patients with hepatic metastases. This site is MD Anderson Cancer Center in Houston, Texas, and the principal investigator for the basket study’s new cohort is Sapna Patel, MD. Eligible metastatic uveal melanoma patients may receive either single agent PV-10 or PV-10 in combination with standard of care checkpoint inhibition.
The Company currently plans to present initial data from the metastatic uveal melanoma cohort at a medical conference in the second half of 2018, and also plans to present updated data from the entire basket study at a medical conference in the first half of 2019.
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “Uveal melanoma is a rare disease, where almost half of all patients develop metastases and the vast majority of them first present with liver metastasis. Treatments for metastatic uveal melanoma, like checkpoint inhibition, typically have very low response rates.”
Mr. Rodrigues added, “Our metastatic uveal melanoma cohort expansion of the Company’s liver basket study, like Provectus’ ongoing, parallel, Phase 1 clinical trial of symptomatic neuroendocrine tumors of the liver, continues to grow the GI cancer-focused drug development of PV-10. This new work further explores the use of the Company’s small molecule oncolytic immunotherapy for another cancer indication with substantial unmet need or that is a rare disease.”
About Metastatic Uveal Melanoma
Uveal melanoma is a rare disease that is biologically and clinically distinct from cutaneous melanoma.(2,3) Nearly 50% of uveal melanoma patients develop metastatic disease, with 80-90% of them presenting with liver as the first site of disease involvement.(2,3,4) Outcomes of metastatic uveal melanoma are poor, with a median overall survival of 12 months and an 80% mortality rate within the first year.(5) A new set of National Comprehensive Cancer Network (NCCN) guidelines for the diagnosis and management of uveal melanoma were created in 2018.(6)
About PV-10
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
References
(1) Nine different hepatic tumor types have been treated with PV-10 to date: hepatocellular carcinoma (HCC), colorectal carcinoma, lung carcinoma, cutaneous and uveal melanomas, breast carcinoma, ovarian adenocarcinoma, and pancreatobiliary adenocarcinoma in the Phase 1 basket study (NCT00986661), and symptomatic neuroendocrine in the Phase 1 clinical trial (NCT02693067).
(2) Carvajal et al. Metastatic disease from uveal melanoma: treatment options and future prospects. The British Journal of Ophthalmology. 2017;101(1):38-44.
(3) Yang et al. Treatment of uveal melanoma: where are we now? Therapeutic Advances in Medical Oncology. 2018;10.
(4) Afzal et al. Metastatic uveal melanoma showing durable response to anti-CTLA-4 and anti-PD-1 combination therapy after experiencing progression on anti-PD-1 therapy alone. Journal for ImmunoTherapy of Cancer. 2018;6(1):13.
(5) Piperno-Neumann et al. Long-term survival in metastatic uveal melanoma (MUM). Journal of Clinical Oncology. 2018:36 (suppl; abstr e21501).
(6) National Comprehensive Cancer Network. Patients, Melanoma (Version 1.2018). https://www.nccn.org/professionals/physician_gls/pdf/uveal.pdf. Subscription required. Accessed July 11, 2018.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Just saw a story about liver cancer being on the rise in the US. Maybe we have the answer.
NEWS -- PROVECTUS BIOPHARMACEUTICALS ANNOUNCES ACCEPTANCE OF PV-10 POSTER PRESENTATION AT ESMO 2018 CONGRESS
FOR IMMEDIATE RELEASE
KNOXVILLE, TN, July 24, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that data from the Company’s ongoing clinical trial of PV-10 for the treatment of symptomatic metastatic neuroendocrine tumors of the liver (NCT02693067) will be presented in a poster presentation at the ESMO 2018 Congress (the European Society for Medical Oncology annual meeting), held in Munich, Germany from October 19-23, 2018.
The presentation is entitled “A Phase 1 Study of Oncolytic Immunotherapy of Metastatic Neuroendocrine Tumours using Intralesional Rose Bengal Disodium.”
Details regarding the presentation will be announced closer to the ESMO 2018 Congress.
About Neuroendocrine Tumors
Neuroendocrine tumors (NETs) associated with the gastrointestinal tract have endocrine secretory properties and a propensity for metastasis to the lungs, bronchi, and liver. Metastatic neuroendocrine tumors located in the midgut and liver often secrete vasoactive products, giving rise to symptoms such as flushing and diarrhea, wheezing, abdominal cramps, and peripheral oedema.
About PV-10
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Wouldnt it be nice if one day we wake up and Merck comes to their senses, buys PVCT for $4/share, all stock and cures 80% of cancer in this world!!!
Then MRK goes to $1000 a share :)!!!
NEWS -- PROVECTUS BIOPHARMACEUTICALS CLINICAL TRIALS UPDATE PROVIDED AT EUROPEAN POST-CHICAGO MELANOMA/SKIN CANCER MEETING
KNOXVILLE, TN, July 10, 2018 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that a clinical trials update was provided at the 8th European Post-Chicago Melanoma/Skin Cancer Meeting, held in Munich, Germany from June 28-29.
The presentation, entitled “Current Clinical Trials with PV-10 (Rose Bengal), The First Small Molecule Oncolytic Immunotherapy,” was made by Sanjiv Agarwala, MD, Chief of Medical Oncology & Hematology, St. Luke’s Cancer Center and Professor of Medicine, Temple/St. Luke’s School of Medicine, Bethlehem, Pennsylvania, during the meeting’s Intralesional Treatments session. A copy of the presentation is available on Provectus' website at https://www.provectusbio.com/media/docs/publications/2018-06-PV-10-Munich.pdf.
Dominic Rodrigues, Vice Chair of the Company's Board of Directors, said, “Of the several hundred patients who have received PV-10 in multiple treatment settings, the majority have been melanoma patients who received the investigational drug as a monotherapy or part of a combination therapy.”
Mr. Rodrigues added, “This substantial and substantive melanoma experience enabled Provectus to build a clinical development platform that has investigated PV-10’s anti-cancer potential in at least a dozen more types of solid tumor disease, in monotherapy and combination therapy settings, and in adults and children. These newer non-clinical and clinical areas, where PV-10 may exhibit similar immune activity, include non-melanoma skin cancers, hepatocellular carcinoma and other gastrointestinal cancers, women’s cancers, and pediatric cancers.”
About PV-10
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Rare Pediatric Disease Designation Priority Review Voucher
"The FDA grants RPDD for diseases that primarily affect children from birth to 18 years old, and affect fewer than 200,000 persons in the U.S. This program is intended to encourage development of new drugs and biologics for the prevention and treatment of rare pediatric diseases"
Here are the stats for each of the 4 cancer types that POETIC displayed this year for PV-10 at ASCO:
1. 800 new cases of neuroblastoma each year in the United States
2. Ewing sarcoma = 1 case per 1 million per year in the United States.
3. rhabdomyosarcoma = Approximately 250 new cases are diagnosed in the U.S. each year
4. Each year, about 800 to 900 new cases of osteosarcoma are diagnosed in the United States
Notice that every one of them fits within the requirements to get the FDA designation?
On September 30, 2016, the Advancing Hope Act of 2016 (Public Law No: 114-229) amended Section 529 of the FD&C Act. Among the changes, the term "rare pediatric disease" now means a disease that meets each of the following criteria:
A. The disease is a serious or life-threatening disease in which the serious or life-threatening manifestations primarily affect individuals aged from birth to 18 years, including age groups often called neonates, infants, children, and adolescents.
B. The disease is rare disease or conditions, within the meaning of Section 526.
The Act changed the language of Subsection (A) from, "The disease primarily affects individuals aged from birth to 18 years, including age groups often called neonates, infants, children, and adolescents." The full text of the Advancing Hope Act is available at: https://www.gpo.gov/fdsys/pkg/BILLS-114s1878enr/pdf/BILLS-114s1878enr.pdf
Effective 90 days after the enactment of the Advancing Hope Act of 2016, the sponsor of a rare pediatric disease product application that intends to request a priority review voucher must submit such request in a cover letter to their NDA/BLA submission.
https://www.fda.gov/forindustry/developingproductsforrarediseasesconditions/rarepediatricdiseasepriorityvoucherprogram/default.htm
SRPT sold their voucher to Gilead for $125M for a pediatric treatment and this analyst thinks they should have received more money.
https://www.bizjournals.com/boston/news/2017/02/21/sarepta-sells-fda-review-voucher-for-125m-but.html
"analyst Joseph Schwartz said that the price tag was lower than his expectation of $200 million. In recent years, such vouchers have fetched anywhere from $67.5 million to $350 million."
PVCT could be starting 4 (possibly more) P1 trials that each could qualify for a PPDD and thus, a voucher for possibly each one.
Within a one month time frame, the FDA granted two PPDD to Cellectar Biosciences for the same drug treating both neuroblastoma and rhabdomyosarcoma; two of the cancers POETIC just presented on behalf of PV-10 and PVCT at ASCO.
If this is the precedent being set by the FDA, things could start moving very quickly in the near future for PVCT.
This is good news, but I do wish there was more discussion of the science here.
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
NEWS -- PROVECTUS BIOPHARMACEUTICALS EXPANDS GLOBAL PATENT PORTFOLIO FOR ACTIVE PHARMACEUTICAL INGREDIENT MANUFACTURE
FOR IMMEDIATE RELEASE
KNOXVILLE, TN, June 19, 2018 (GLOBE NEWSWIRE) -- Provectus today announced that the Indian Patent Office (IPO) has granted the Company’s patent application for the synthetic process used to produce the active pharmaceutical ingredient (API) in PV-10, which is Provectus’ lead investigational oncology drug. The Company’s patent estate associated with API manufacturing provides intellectual property protection in a number of countries in Asia, Europe, and North America into the 2030s.
IPO patent 297453 is linked to U.S. patent (USP) 8,530,675, entitled “Process for the Synthesis of 4,5,6,7-tetrachloro-3',6'-dihydroxy-2',4',5',7'-tetraiodo-3H-spiro[isobenzofuran-1,9-xanthen]-3-one (Rose Bengal) and Related Xanthenes,” which was awarded by the United States Patent and Trademark Office (USPTO) in 2013. The Company received related awards from, among others, the European Patent Office (EPO), the Japan Patent Office (JPO), and the State Intellectual Property Office (SIPO or the Chinese Patent Office) in 2015.
Continuations of Provectus’ foundational manufacturing patent include USP 9,273,022 and USP 9,422,260, which were awarded by the USPTO in 2016.
Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, http://www.provectusbio.com) (Provectus or the Company) is a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers in adults and children.
About PV-10
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
no longer relevant
SEC co-Director of Enforcement Division praises Provectus' new leadership in SEC press release.
In the SEC’s press release on this matter, Steven Peikin, Co-Director of the SEC’s Enforcement Division said, “The SEC’s settlement with Provectus – which does not include any penalty – takes into account the proactive remediation and cooperation by the company’s new leadership. Provectus fired wrongdoers, took other steps to remedy its controls, and provided SEC staff with critical information regarding its former executives’ expense reimbursement abuses.”
The SEC also noted that following the Company’s investigation of Dees’ travel expenses, a Special Committee of Provectus’ Board of Directors with the help of independent counsel and a forensic accounting firm, reviewed executive expenses in general, and subsequently devoted several months to investigate Culpepper’s travel expenses. Provectus voluntarily shared the results and details of these investigations with the SEC, “reducing the time and resources necessary for the Commission staff to conclude their investigation.”
Dominic Rodrigues, Chairman of the Company’s Board of Directors, said, “We are grateful to Commission staff for working with Provectus to conclude the SEC investigation of the Company. We also appreciate that the SEC recognized prompt and specific remedial acts by Provectus as well as the cooperation afforded to Commission staff by the Company during the investigation.”
https://globenewswire.com/news-release/2017/12/12/1253450/0/en/PROVECTUS-Announces-Resolution-of-SEC-Investigation.html
2017 Article
Federal investigators accuse two former Provectus Biopharmaceuticals Inc. executives of defrauding investors of millions.
A lawsuit filed Dec. 12 in Knoxville’s U.S. District Court accuses Provectus co-founder, former CEO and former chairman Craig Dees of taking at least $3.2 million between January 2011 and February 2016. He resigned from the company Feb. 29, 2016, but now faces 10 counts of fraud.
“Although Dees represented that the funds he obtained were to be used for expenses that he incurred on behalf of Provectus, in reality Dees was treating Provectus as his personal piggy bank and used the Company’s funds to pay for his personal expenses, including but not limited to, entertainment, clothing, cosmetic surgeries for female friends, large tips at Hooters and other restaurants, as well as personal travel,” the Securities and Exchange Commission asserts in the lawsuit.
He did so by submitting “hundreds of false cash advance requests and expense reimbursements,” failing to provide documentation for most and falsifying those he did provide, according to the suit. “For example, in December 2015, Dees paid over $13,000 to cover breast enhancement surgery and other cosmetic procedures on behalf of female acquaintances,” the suit says.
Other Provectus executives were supposed to review that spending, but one of those responsible — former chief financial officer and later interim CEO Peter Culpepper — is alleged to have taken nearly $200,000 himself in a similar manner, according to the SEC.
Further allegations
Knoxville lawyer Jeffrey Whitt, representing Dees, didn’t return a request for comment Friday. On Dec. 12 he told Law 360 news service via email that his firm made no statements on pending litigation.
Provectus and Culpepper consented to separate SEC orders, without admitting or denying investigators’ findings, according to an SEC news release.
Peter R. Culpepper (Photo: Amy Smotherman Burgess/News Sentinel)
In the Dec. 12 order Culpepper is described as Provectus CFO from February 2004 to April 2016, chief operating officer from February 2008 to December 2016, and interim CEO from February 2016 to December 2016.
The order says Culpepper also got cash advances with little documentation, including $13,103 for “hotel stays, spa services, and products for Culpepper and his wife, and meals for his family.”
Altogether the SEC said he got $199,194 from 2013 to 2015 for unauthorized personal expenses. In 2016 and 2017, Culpepper cut his claims by $59,079 after questions emerged.
In his settlement with the SEC, Culpepper agreed to pay $152,376 in reimbursement and interest, plus a civil penalty above $90,000, according to Law 360. He is also barred from working as an accountant before the SEC for at least three years.
Culpepper’s attorney, Tera Rica Murdock of Nashville, also did not respond Friday to a request for comment.
The Provectus board voted unanimously Dec. 27, 2016, to fire Culpepper. President Tim Scott was appointed interim CEO. In a letter to the board, Culpepper denied the allegations, disputing an auditor’s finding that he owed Provectus $294,255, but said he couldn’t document $39,000 in expenses and would reimburse the company for them.
Company response
Provectus board chairman Dominic Rodrigues said Friday the company itself faced no monetary penalty, and is still fixing and testing its financial controls
"We are grateful to Commission staff for working with Provectus to conclude the SEC investigation of the Company,” he said in a Dec. 12 news release. “We also appreciate that the SEC recognized prompt and specific remedial acts by Provectus as well as the cooperation afforded to Commission staff by the Company during the investigation."
Rodrigues highlighted a quote from Steven Peikin, co-director of the SEC’s enforcement division.
“The SEC’s settlement with Provectus – which does not include any penalty – takes into account the proactive remediation and cooperation by the company’s new leadership,” Peikin said in a news release. “Provectus fired wrongdoers, took other steps to remedy its controls, and provided SEC staff with critical information regarding its former executives’ expense reimbursement abuses.”
The SEC noted the company hired outside counsel and a forensic accountant, replaced executives, created new internal controllers, and shared all results with investigators, "reducing the time and resources necessary for the Commission staff to conclude their investigation."
Rodrigues said Provectus is suing its former bookkeeper Bible Harris Smith PC and former internal auditor RSM US LLP, alleging negligence and breach of fiduciary duty against both for not catching Dees and Culpepper. The suits seek more than $3 million from Bible Harris Smith and more than $10 million from RSM US, Rodrigues said.
Joint history
Dees, Scott and Eric Wachter all worked at Oak Ridge National Laboratory, leaving to found Photogen Technologies Inc. in 1996, which initially sought to develop a laser therapy for cancer. Provectus split off from Photogen in 2002 to focus on drug development.
From at least 2011 until 2016, Provectus directly employed only its three founders and Culpepper as its CFO, though it retained consultants and contractors, according to the SEC.
Craig Dees (Photo: Clay Owen)
The court filing says Provectus has “no revenue.” The company repeatedly sought funding from investors to develop its drugs.
Dees, described in the suit as a 65-year-old resident of Hillsborough, N.C., was supposed to have a base salary of $500,000 per year, but got substantially more in perks and bonuses, according to the SEC: a total of $3 million in 2011, $1.2 million in 2012, $642,654 in 2013, and $637,692 in 2014.
But in response to a 2013 shareholder lawsuit alleging excessive executive pay, Provectus “significantly curtailed Dees’ discretion to award bonuses to himself and other executives and required Dees and other Provectus executives to repay bonuses from 2010 and 2011.” The 2014 agreement called for Dees, Scott, Wachter and Culpepper to repay $2.24 million.
So, the SEC charges, Dees made up for those losses through cash advances, growing from $238,423 in 2011 to $885,808 in 2015, and $67,261 in the first two months of 2016.
“The undisclosed amounts received by Dees supplemented his annual salary by 48 percent to 164 percent from 2011 to 2014 and exceeded his total compensation in 2013 and 2014,” the suit says.
In November 2015, an internal auditor raised questions on those cash advances. In response Dees submitted fabricated receipts which didn’t even add up correctly, according to the SEC.
Board fallout
On Feb. 29, 2016, Dees said he was leaving Provectus for “health and personal reasons,” at the same time announcing a review of company policies including executive compensation.
Alfred Smith IV replaced Dees as board chairman, while Culpepper became interim CEO; and Wachter, the chief technology officer, occupied Dees’ board seat.
In March 2017, Provectus announced it would restructure its board and seek up to $20 million in new financing. When stockholders PRH Group paid $2.5 million into an escrow account, Smith, Scott and Kelly McMasters would resign from the board, according to an agreement at the time. They would be replaced by Dominic Rodrigues, Bruce Horowitz, and a PRH appointee.
After another $5 million was raised – expected in June 2017 – Wachter and Jan Koe were to resign from the board as well. At that time Edward Pershing – who is also a PRH member – and another PRH nominee would join the board.
According to Provectus’ website, that first round of changes took place, but the second apparently didn’t: Smith, McMasters and Scott are off the board, but Wachter and Koe are still members, while Pershing is listed as “observer.”
Scott remains as president and Wachter is still chief technology officer, while Horowitz is chief operations consultant.
https://www.knoxnews.com/story/money/business/2017/12/29/hooters-provectus-investors-money-cancer-drugs-sec/988404001/
2016 article
https://www.thestreet.com/story/13519277/1/this-drug-company-loaned-its-now-fired-ceo-2-5m-for-made-up-travel-advances.html
Provectus Biopharmaceuticals (PVCT) Chairman and CEO H. Craig Dees was fired after the company's board discovered he allegedly pocketed almost $2.5 million in unsubstantiated travel expenses over the past three years.
The extravagant travel-expense payouts to Dees are real but the actual travel likely isn't, Provectus says, which now leaves the company at risk of criminal or civil investigations by the Securities and Exchange Commission.
The full extent of Dees' alleged embezzlement of company funds -- and the trouble Provectus finds itself in due to his alleged misdeeds -- were disclosed in the company's 10-K filed on March 30.
Provectus is a penny stock that has operated on the dark side of biotech legitimacy for years. The company's long-delayed effort to develop a melanoma treatment made from Rose Bengal, a common stain used to diagnose eye disease, borders on the ridiculous. Most biotech companies try to develop drugs to help patients. A truthful rendering of Provectus' mission statement, on the other hand, would read like, "Striving everyday to transfer as much shareholder cash to executives' pockets as possible."
On this score, Dees was very good at his job.
Soon after Dees was fired in February, Provectus' board hired outside accountants to look into his travel expenses and reimbursements. The investigation found the following:
In 2015, Dees received $898,430 in travel expense "advances." He submitted receipts for that "travel" totaling only $297,170, and most of those receipts "did not appear to be authentic."
In 2014, Dees received $819,000 for travel expense "advances" but didn't submit any travel receipts at all.
In 2013, Dees received $752,034 for travel expenses "advances." He submitted receipts to account for $54,034 of the travel but again, Provectus' investigation found those receipts to be faked.
The amount of money allegedly embezzled by Dees from Provectus over three years is staggering, especially for a company with three full-time employees which has struggled to raise small amounts of money to remain solvent.
Provectus, in its 10-K, does not say where the money given to Dees for "travel" actually ended up. [We can guess the obvious answer.] However, the company does consider all the travel-related expenses of Dees to be "theft loss" and the missing money is being reported to the Internal Revenue Service as extra income to Dees.
That'll be a Form 1099 MISC Dees will have trouble explaining.
Provectus is not the innocent victim of Dees' alleged plundering. Another executive at the company (not identified) approved the travel expense advances given to Dees without any supportive documentation, according to Provectus' 10-K. In essence, Dees asked one of his fellow company executives to write him big checks for travel advances, and that someone just wrote the checks, no questions asked.
In the arcane language of federal securities law, this means Provectus has "material weakness over its internal controls of financial reporting." It's illegal to make a personal loan to an executive or director of a public company.
When the feds call, and they will, Provectus has to explain how $2.5 million in personal loans -- "advances" -- to its now-fired CEO just walked out the door.
2016 article
https://www.thestreet.com/story/13519277/1/this-drug-company-loaned-its-now-fired-ceo-2-5m-for-made-up-travel-advances.html
Provectus Biopharmaceuticals (PVCT) Chairman and CEO H. Craig Dees was fired after the company's board discovered he allegedly pocketed almost $2.5 million in unsubstantiated travel expenses over the past three years.
The extravagant travel-expense payouts to Dees are real but the actual travel likely isn't, Provectus says, which now leaves the company at risk of criminal or civil investigations by the Securities and Exchange Commission.
The full extent of Dees' alleged embezzlement of company funds -- and the trouble Provectus finds itself in due to his alleged misdeeds -- were disclosed in the company's 10-K filed on March 30.
Provectus is a penny stock that has operated on the dark side of biotech legitimacy for years. The company's long-delayed effort to develop a melanoma treatment made from Rose Bengal, a common stain used to diagnose eye disease, borders on the ridiculous. Most biotech companies try to develop drugs to help patients. A truthful rendering of Provectus' mission statement, on the other hand, would read like, "Striving everyday to transfer as much shareholder cash to executives' pockets as possible."
On this score, Dees was very good at his job.
Soon after Dees was fired in February, Provectus' board hired outside accountants to look into his travel expenses and reimbursements. The investigation found the following:
In 2015, Dees received $898,430 in travel expense "advances." He submitted receipts for that "travel" totaling only $297,170, and most of those receipts "did not appear to be authentic."
In 2014, Dees received $819,000 for travel expense "advances" but didn't submit any travel receipts at all.
In 2013, Dees received $752,034 for travel expenses "advances." He submitted receipts to account for $54,034 of the travel but again, Provectus' investigation found those receipts to be faked.
The amount of money allegedly embezzled by Dees from Provectus over three years is staggering, especially for a company with three full-time employees which has struggled to raise small amounts of money to remain solvent.
Provectus, in its 10-K, does not say where the money given to Dees for "travel" actually ended up. [We can guess the obvious answer.] However, the company does consider all the travel-related expenses of Dees to be "theft loss" and the missing money is being reported to the Internal Revenue Service as extra income to Dees.
That'll be a Form 1099 MISC Dees will have trouble explaining.
Provectus is not the innocent victim of Dees' alleged plundering. Another executive at the company (not identified) approved the travel expense advances given to Dees without any supportive documentation, according to Provectus' 10-K. In essence, Dees asked one of his fellow company executives to write him big checks for travel advances, and that someone just wrote the checks, no questions asked.
In the arcane language of federal securities law, this means Provectus has "material weakness over its internal controls of financial reporting." It's illegal to make a personal loan to an executive or director of a public company.
When the feds call, and they will, Provectus has to explain how $2.5 million in personal loans -- "advances" -- to its now-fired CEO just walked out the door.
A few repeats? lol I bet you can't. You're a fool and your ignorance is on full display.
BOARD OF DIRECTORS
Ed Pershing, CPA - Chairman of the Board
Mr. Pershing is Chief Executive Officer of Pershing Yoakley & Associates (“PYA”), a top 20 healthcare consulting and top 100 accounting firm in the U.S. PYA, which he co-founded more than 30 years ago, expanded from a three-employee office to more than 270 employees and five affiliate companies serving more than 3,500 clients in 50 states. Mr. Pershing’s healthcare experience and expertise include turnaround/performance improvement initiatives, long-range planning studies, development of numerous hospital and medical office projects, restructuring of healthcare organizations, liaison between boards of directors and management teams to craft corporate visions and strategies, mergers, acquisitions, divestitures, and leasing arrangements. He has served as an expert witness on healthcare industry matters and in several Certificate of Need appeals. Mr. Pershing also has represented healthcare organizations before regulatory agencies such as the Centers for Medicare & Medicaid Services, Internal Revenue Service, and Departments of Mental Health, Insurance, and Medicaid. He graduated from the University of Tennessee with a Bachelor of Science in Accounting, and was one of 18 professionals from the U.S. and Great Britain to participate in the first Ernst & Ernst Accelerated Healthcare Program, a one-year full-time education and work-study program in healthcare industry matters. Mr. Pershing is a Certified Professional Accountant (CPA).
Dominic Rodrigues - Vice Chairman of the Board
Mr. Rodrigues has served in leadership roles at Provectus since 2017, including former Chairman of the Board. He previously served as President of Rhisk Capital, which provided management consulting, corporate development, and portfolio management services, since 2005. Project industries and technologies included aerospace & defense (a technology-focused investment capital pool; an operational role in a related data communications solutions company), biotechnology, financial services (a capital markets-focused technology company), gaming, healthcare, life sciences, nanotechnology (a venture capital fund investment), wealth management (a start-up private wealth office), and restaurants. Since 2013 Mr. Rodrigues has been an Adjunct Professor of Finance at the Lee Business School of the University of Nevada, Las Vegas, where he teaches valuation and CFA exam preparation courses. His business development, corporate development, finance, and leadership experiences at various companies include SAIC Venture Capital Corporation, the multi-billion dollar subsidiary of research and engineering company SAIC, where he was an observer or member of the board of directors of a number of portfolio companies. Mr. Rodrigues currently serves as a member of the audit & finance committee of Three Square Food Bank. He holds business, economics, and engineering degrees from The Wharton School, the London School of Economics, the Massachusetts Institute of Technology, and the University of Toronto. Mr. Rodrigues also is a Chartered Financial Analyst (CFA).
Bruce Horowitz - Board Member
Mr. Horowitz has served in a leadership role at Provectus since 2017. He previously served as the Managing Director of Capital Strategists, LLC, which provides corporate, strategic, and financial consulting services, since September 2006. He also serves as a trusted advisor to family trusts and private individuals, with a focus on financial asset management, real estate management and special situation investments. Earlier in his career Mr. Horowitz was a charter member of the New York Futures Exchange, a Senior Vice President managing principal equity investment accounts, private equity investments and public offerings at Drake Capital Securities, and managed the trading department at Laidlaw Equities. He was also a partner at Stanley Capital, a private equity buyout firm. Mr. Horowitz was the chairman and a member of two general obligation bond fund committees, raising more than $500 million in general revenue bonds for the Beverly Hills Unified School District. Subsequently, he was named the first chairman of both the state of California-mandated Citizens’ Oversight Committee and Facilities Advisory Committee, overseeing expenditure of all BHUSD general obligation bond funds. Mr. Horowitz is a founding member of the Los Angeles Chapter of the Positive Coaching Alliance. He founded and is currently the president of the Beverly Hills Basketball League, a youth basketball program that serves more than 35,000 families. Mr. Horowitz has also served as a member of the board of directors of the American Youth Soccer Organization and Beverly Hills Little League. He holds a Juris Doctor degree from Benjamin N. Cardozo School of Law in New York City and Bachelor of Arts degree from Washington University in St. Louis.
John Lacey, III, M.D. - Board Member
Dr. Lacey is the former Chief Medical Officer and Senior Vice President of University Health System d/b/a University of Tennessee Medical Center (“UTMC”), a 600+ bed academic medical center based in Knoxville since 1999. Dr. Lacey served continuously in this capacity from 1999 until retirement from UTMC in 2016. He also operated an internal medicine practice for 39 years. Dr. Lacey graduated from the University of Tennessee with a Bachelor’s degree in Nuclear Engineering and the University of Tennessee Medical School (Memphis) with a Doctor of Medicine degree. Dr. Lacey helped create Knoxville Area Project Access, a partnership with the Knoxville Academy of Medicine and providers to give primary and specialty health services to the uninsured and medically underserved, and was the inaugural chair of the Governor’s Health and Wellness Task Force, which focused on improving Tennessee’s national health ranking. Dr. Lacey has been recognized by several entities for contributions to population health.
Jan E. Koe - Board Member
Mr. Koe has a 30-year track record of success in consulting, asset management, real estate and public company governance, and has represented major insurance firms, national retailers and Fortune 500 companies. He is President of GoStar, which is the manager of Real Solutions Opportunity Fund 2005-I and Real Solutions Fund Management LLC and Real Solutions Investment LLC. He is also Principal of Method K Partners, Inc., a commercial real estate firm, which he founded in 1988. He has served on the Board of Directors of ONE Bio, Corp. where he was Chair of the Compensation Committee and a member of the Financial Audit Committee. He holds a degree in Business Administration and Psychology from Luther College.
Hey Fred, how's business been lately? You have got to be the biggest fool I've ever seen on any of these board. And your multiple accounts on the other board are too funny!
I think it's time to start exposing you.
Medcowboy....please take your meds. You are Mr. Irrelevant here. GL
You've already displayed a lack of knowledge on this board and by most of your statements, I find it hard to take anything you say seriously. You're just toxic buddy. Your claims have flimsy evidence if any. Idk why you waste your time here tbh. It's a penny stock, there's risk.
Ha I ask that very same question of pvct management.
Complete board change hahahaha okay can you list who was previously on the board and then list who is currently aboard? I bet we see a few repeats. Also if youd like I can post an article from just this last year which talks of their PAST and PRESENT theft convictions.
Ha or Maxim saw the red flags and are getting the hell out of dodge. This "penny stock" will continue to float above .053 while those in the know sell off and then it's all down hill from there. Hahaha I still cannot believe people still buy into the idea that diluted red dye somehow can cure cancer! Wake up people!
PVCT will present at ASCO next week. Here is the abstract. http://abstracts.asco.org/214/AbstView_214_230375.html
I think it's just a sore spot to buy because of the volatility. People want in, trying to accumulate below .08, but I know I'd be antsy about buying at 8. Long run it is a safe bet, but short term sees pretty major moves up and down. Any day now it should start hitting past 8, maybe reach mid 8s by June- one can hope. I have no doubt this will reach double digits, just have to wait it out.
Agree.. what is so big about .08?
Has hit it 20 times over the past few months and cant get past.
If it does, should rapidly move higher.
Have owned this stock on and off for 6 years and do believe they are in a much better place than they have been in a long time.
Just need the Keytruda combination to really work in this trial!
Hoping to see this break 8 soon. A lot of good news lately, such a big change since last year- they've really stepped up their game and I think investors have noticed.
NEWS -- PROVECTUS BIOPHARMACEUTICALS AND PEDIATRIC ONCOLOGY EXPERIMENTAL THERAPEUTICS INVESTIGATORS' CONSORTIUM (POETIC) ANNOUNCE PV-10 ABSTRACT AT AMERICAN SOCIETY OF CLINICAL ONCOLOGY (ASCO) ANNUAL MEETING
KNOXVILLE, TN & NEW YORK, NY, May 17, 2018 (GLOBE NEWSWIRE) -- Provectus and POETIC today announced preclinical PV-10 data from pediatric cancer research that will be presented at the 2018 American Society of Clinical Oncology (“ASCO”) Annual Meeting in Chicago, IL.
PV-10 induced cell death in pediatric solid tumor cell lines derived from relapsed pediatric neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma with a measurable therapeutic window compared to normal cells. Western blot analyses of cells treated with PV-10 indicated induction of apoptosis. Drug combination studies showed synergy with radiation and agents that target mitosis. Xenograft studies showed significant reduction of tumor burden in PV-10-treated mice compared to control animals, with a corresponding increase in overall survival.
Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, http://www.provectusbio.com) (“Provectus” or the “Company”) is a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for adult and pediatric solid tumor cancers. The Pediatric Oncology Experimental Therapeutics Investigators' Consortium (“POETIC”) is a group of North American academic medical centers developing new pediatric cancer therapies.
Findings will be highlighted in a poster presentation on Saturday, June 2 from 8:00 to 11:30 a.m. CDT in Hall A as part of the Pediatric Solid Tumors topic during the Pediatric Oncology session. The POETIC poster title is “In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors,” the poster board number is 230, and the abstract number is 10557.
This preclinical pediatric cancer research was led by Aru Narendran, MD, PhD and researchers at the POETIC Laboratory for Pre-Clinical and Drug Discovery Studies at the University of Calgary (Canada), together with Tanya Trippett, MD, Director of POETIC and researchers at Memorial Sloan Kettering Cancer Center.
Dr. Trippett said, “POETIC's mission is to promote the early clinical development of promising therapies for the treatment of children, adolescents, and young adults with cancer. We hope our collaboration with Provectus on PV-10 leads to an opportunity to improve cancer care for children around the world.”
Ed Pershing, Chair of Provectus’ Board of Directors, said, “We are thrilled to work alongside POETIC and its member institutions in Canada and the U.S. in an effort to jointly advance options for pediatric cancer patients around the globe.”
About PV-10
Provectus’ lead investigational cancer drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers such as Ewing sarcoma, neuroblastoma, osteosarcoma, and rhabdomyosarcoma.
About the Pediatric Oncology Experimental Therapeutics Investigators' Consortium
The Pediatric Oncology Experimental Therapeutics Investigators’ Consortium (POETIC) was founded in February 2003 by Dr. Tanya Trippett at Memorial Sloan Kettering Cancer Center and Dr. Lia Gore at the University of Colorado Cancer Center. POETIC is composed of ten large academic medical centers in North America with a major emphasis on comprehensive cancer care and research that provide the collaborative and research strength needed to complete intensive phase I and II studies. Each of the institutions is uniquely suited to complete early studies in the pediatric and adolescent populations. POETIC’s assets include membership in NCI-designated Comprehensive Cancer Centers, on-site NIH-funded pediatric and/or general clinical translational research centers (CTRCs/CTSAs), and active collaborations with developmental therapeutics programs for adults at a majority of its member institutions. The availability of strong basic science and translational research programs at the institutions allows focus on the development and evaluation of new therapeutic strategies for patients with cancer and related disorders. POETIC’s pediatric oncology studies focus on the biologic basis for anti-cancer therapy, and in particular, attempt to explore and evaluate novel agents and/or combinations of therapies early in clinical development as well as new approaches to targeted delivery. For additional information about POETIC, please visit the Consortium’s website at http://www.poeticphase1.org.
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company’s website at www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Yeah, you've been saying that for over 4 years now. Maybe one day, if you keep posting enough bull****, you might actually get something right! How many more years do you need? lol
|
Followers
|
185
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9952
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
CURRENT PVCT PIPELINE (as of May 23, 2017)
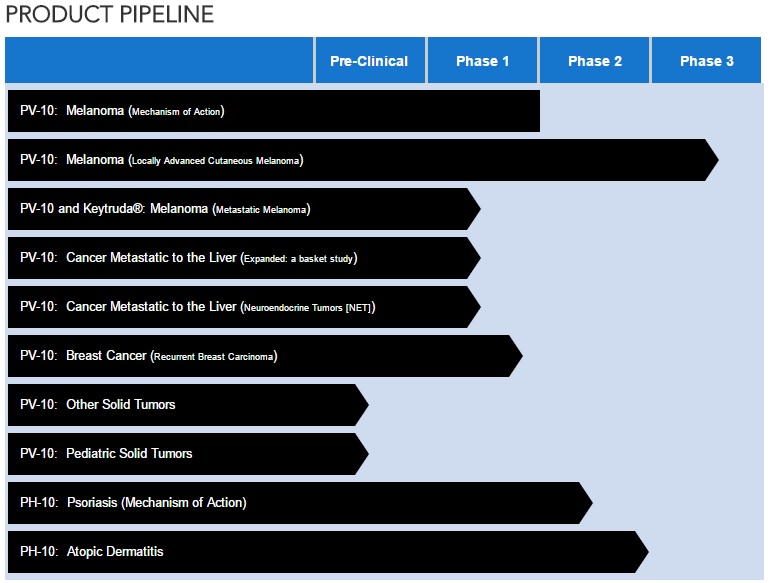
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
