Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
Oh how simple you must be. There has been a complete board change since then and there has also been an SEC investigation in which they specifically praised the changes made by the company. The two thieves are long gone and the only con artists left are people like you. Not one single person that works for the company has ever stolen for the company. This is fact as per the SEC.
Fool you once? Someone as simple as you sounds like you're fooled all the time.
Completion of enrollments ......ha 500:1 that they never evolve into completion of trials.
They may not be bankrupt yet but they sure aren't far from it.
Yeah let's all forget that those in-charge have stolen from investors. Fool me once shame on you, fool me twice same shame on me.
Ha! This idiot posted an article from 2014 saying that PVCT is about to go bankrupt! Ha!
Here we are mid way through 2018 and the company still isn't even close to going bankrupt.
It's funny of much people's credibility fades away when reading their articles in hindsight.
NEWS -- PROVECTUS BIOPHARMACEUTICALS COMPLETES ENROLLMENT OF PHASE 1B TRIAL OF PV-10 IN COMBINATION WITH KEYTRUDA® IN PATIENTS WITH METASTATIC MELANOMA
DO NOT DISMISS THIS ARTICLE! NEW INVESTORS PLEASE READ!
https://seekingalpha.com/article/2284173-provectus-strong-sell-on-unviable-phase-iii-trial-insufficient-cash-and-fraud-allegations
Additional research indicates PVCT's Phase 3 Trial is designed in such a way that it seems completely unviable and effectively impossible to enroll or execute.
Both industry data and PVCT's own internally generated documents show the current Phase 3 proposal seems too small, too short of duration, and far more expensive than guided.
PVCT seems impossibly low on cash to run a Phase 3 trial with more equity issuance imminent. Plus PVCT faces multiple fraud allegations from shareholders and lawsuits.
Recently settled shareholder lawsuit sheds interesting insight on how PVCT management views shareholders and potentially why the Phase 3 trial was designed as such.
New evidence connects PVCT to paid stock promoter "Hunter Marketing Group," contradicting PVCT management’s communication with investors.
In prior reports, I outlined my view on why I value shares of Provectus (NYSEMKT:PVCT) at $0. PVCT remains wildly overvalued with an estimated fully diluted market cap of ~$200m, which I believe creates at least -90% near-term downside risk before PVCT eventually goes bankrupt.
I think the case for PVCT hinges on the market's view of management's credibility and the outcome of the Phase 3 trial. Unfortunately, new research shows PVCT's Phase 3 trial seems clearly unviable (raising questions about management motives), PVCT's own documents indicate this Phase 3 trial is unviable1 and PVCT's cash seems woefully insufficient to complete the trial while management appears elusive on this topic as recently as last week's conference call. Most importantly, the study seems designed in such a way that it seems effectively incapable of enrollment and completion in my view. Keep in mind this is the same management team that has not started Phase 3 trials for 4 years (so far) while collecting an estimated ~$30m in compensation over that timeframe.
Perhaps most concerning, new research proves PVCT's has connections to paid stock promotion firm "Hunter Marketing", the firm which paid Small Cap Street $10k for promoting PVCT. I believe these connections appear irrefutable and seem contradictory to PVCT management's statements to investors. Could investors who PVCT raised money from while PVCT stock may have been artificially inflated due to paid stock promotion sue PVCT and demand their money back? Could this accelerate a potential PVCT bankruptcy?
NEWS -- STUDY OF T CELL MEDIATED IMMUNITY FROM COMBINATION OF PV-10 AND IMMUNE CHECKPOINT BLOCKADE IN MURINE MELANOMA MODELS PUBLISHED IN PLOS ONE
KNOXVILLE, TN, April 30, 2018 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, www.provectusbio.com) (“Provectus” or the “Company”), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, today announced that the open-access journal PLOS ONE published results from an H. Lee Moffitt Cancer Center and Research Institute (“Moffitt”) study investigating cancer combination therapy with intralesional (“IL”) PV-10 and immune checkpoint blockade (anti-CTLA-4, anti-PD-1 and anti-PD-L1 antibodies) in murine melanoma models. (Note1,2) The authors also examined the role of specific immune cell populations in eliciting and controlling tumor-specific response.
This article may be accessed via PLOS ONE’s website at http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0196033.
The Moffitt authors noted, “In this study, we have shown the impact of combining systemic checkpoint blockade (PD-1, PD-L1) with the tumor-specific immune response induced by IL PV-10. Treatment with IL PV-10 and anti-PD-1 antibody resulted in a delay in tumor growth and enhanced T cell activation in the M05 tumor model. Similar effects were observed with IL PV-10 and anti-PD-L1 antibody in the B16 tumor model. The effect of combination therapy with IL PV-10 and PD-1 blockade is mediated by CD8+ T cells, and depletion of either CD4+ T cells or CD4+CD25+ Tregs enhances anti-tumor immunity in the M05 melanoma model. Together these results support further development of clinical trials to assess safety and anti-tumor T cell responses in patients after IL injection of PV-10 in combination with checkpoint blockade.”
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “We are grateful to our research collaborators, like those at Moffitt, who continue to independently establish PV-10 as an oncolytic immunotherapy in both monotherapy and combination therapy settings.”
Mr. Rodrigues added, “This PLOS ONE work established the rational clinical foundation for our ongoing Phase 1b/2 study PV-10 in combination with anti-PD-1 drug KEYTRUDA for patients with Stage IV melanoma. Moffit’s work also builds on the foundation for future combinations of PV-10 and anti-PD-L1 agents as well as other potential combinations and permutations of cancer therapies that include PV-10 as a key element.”
About PV-10
Provectus’ lead investigational oncology drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About our Phase 1b/2 Study of PV-10 + KEYTRUDA for Stage IV Melanoma
The Phase 1b portion of the study continues to enroll patients with metastatic melanoma at clinical sites in the U.S. (NCT02557321). Stage IV patients with at least one injectable lesion who are candidates for KEYTRUDA are eligible. A total of up to 24 patients would receive the combination of IL PV-10 and KEYTRUDA every three weeks for five cycles (i.e., for up to 12 weeks, with no further PV-10 administered after week 12), followed by only KEYTRUDA every three weeks for up to 24 months. The primary endpoint for the Phase 1b trial is safety and tolerability; objective response rate and progression-free survival are key secondary endpoints (both assessed via RECIST 1.1 after five treatment cycles, and then every 12 weeks thereafter).
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company’s website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
References:
1. The Society for Immunotherapy of Cancer (SITC) 29th Annual Meeting & Associated Programs (SITC 2014): Pilon-Thomas et al., Efficacy of intralesional injection with PV-10 in combination with co-inhibitory blockade in a murine model of melanoma. J Immunother Cancer. 2014; 2(Suppl 3): P120. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4288419/. Accessed April 16, 2018. (Poster).
2. The American Association for Cancer Research (AACR) Annual Meeting 2016: Weber et al., T cell mediated immunity after combination therapy with intralesional PV-10 and co-inhibitory blockade in a melanoma model. Cancer Res. July 15 2016 (76) (14 Supplement) 4978. http://cancerres.aacrjournals.org/content/76/14_Supplement/4978. Accessed April 16, 2018.
Trademark
KEYTRUDA® is a registered trademark of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. Kenilworth, New Jersey, U.S.A.
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
MM on IV is an AO and fraud. Was it an accident or homicide? We all know the truth.
NEWS -- PROVECTUS BIOPHARMACEUTICALS AND PEDIATRIC ONCOLOGY EXPERIMENTAL THERAPEUTICS INVESTIGATORS' CONSORTIUM (POETIC) ANNOUNCE ACCEPTANCE OF PV-10 POSTER PRESENTATION AT AMERICAN SOCIETY OF CLINICAL ONCOLOGY (ASCO) ANNUAL MEETING
KNOXVILLE, TN & NEW YORK, NY, April 25, 2018 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, www.provectusbio.com) (“Provectus” or the “Company”), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, and the Pediatric Oncology Experimental Therapeutics Investigators' Consortium (“POETIC”), a group of North American academic medical centers developing new pediatric cancer therapies, today announced that preclinical data from ongoing pediatric cancer research into PV-10 will be presented in a poster presentation at the American Society of Clinical Oncology (“ASCO”) Annual Meeting held in Chicago, IL from June 1-5, 2018. The presentation is entitled “In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors.”
The session title is Pediatric Oncology (Saturday, June 2, 8:00-11:30 AM), the topic is Pediatric Solid Tumors, the poster board number is 230, and the abstract number is 10557.
The preclinical pediatric cancer research was led by Aru Narendran, MD, PhD and researchers at the POETIC Laboratory for Pre-Clinical and Drug Discovery Studies at the University of Calgary (Canada), together with Tanya Trippett, MD, Director of POETIC, and researchers at Memorial Sloan Kettering Cancer Center. Further details regarding the presentation will be announced closer to the ASCO conference.
About PV-10
Provectus’ lead investigational cancer drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About the Pediatric Oncology Experimental Therapeutics Investigators' Consortium
The Pediatric Oncology Experimental Therapeutics Investigators’ Consortium (POETIC) was founded in February 2003 by Dr. Tanya Trippett at Memorial Sloan Kettering Cancer Center and Dr. Lia Gore at the University of Colorado Cancer Center. POETIC is composed of ten large academic medical centers in North America with a major emphasis on comprehensive cancer care and research that provide the collaborative and research strength needed to complete intensive phase I and II studies. Each of the institutions is uniquely suited to complete early studies in the pediatric and adolescent populations. POETIC’s assets include membership in NCI-designated Comprehensive Cancer Centers, on-site NIH-funded pediatric and/or general clinical translational research centers (CTRCs/CTSAs), and active collaborations with developmental therapeutics programs for adults at a majority of its member institutions. The availability of strong basic science and translational research programs at the institutions allows focus on the development and evaluation of new therapeutic strategies for patients with cancer and related disorders. POETIC’s pediatric oncology studies focus on the biologic basis for anti-cancer therapy, and in particular, attempt to explore and evaluate novel agents and/or combinations of therapies early in clinical development as well as new approaches to targeted delivery. For additional information about POETIC, please visit the Consortium’s website at http://www.poeticphase1.org.
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company’s website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Provectus is going to ASCO!
Provectus Biopharmaceuticals and Pediatric Oncology Experimental Therapeutics Investigators' Consortium (POETIC) Announce Acceptance of PV-10 Poster Presentation at American Society of Clinical Oncology (ASCO) Annual Meeting
Wednesday April 25, 2018
Preclinical pediatric cancer research undertaken at POETIC member institutions Alberta Children's Hospital (Canada) and Memorial Sloan Kettering Cancer Center
KNOXVILLE, TN & NEW YORK, NY, /GLOBE NEWSWIRE/ -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, www.provectusbio.com), ("Provectus" or the "Company"), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, and the Pediatric Oncology Experimental Therapeutics Investigators' Consortium ("POETIC"), a group of North American academic medical centers developing new pediatric cancer therapies, today announced that preclinical data from ongoing pediatric cancer research into PV-10 will be presented in a poster presentation at the American Society of Clinical Oncology ("ASCO") Annual Meeting held in Chicago, IL from June 1-5, 2018. The presentation is entitled "In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors."
The session title is Pediatric Oncology (Saturday, June 2, 8:00-11:30 AM), the topic is Pediatric Solid Tumors, the poster board number is 230, and the abstract number is 10557.
The preclinical pediatric cancer research was led by Aru Narendran, MD, PhD and researchers at the POETIC Laboratory for Pre-Clinical and Drug Discovery Studies at the University of Calgary (Canada), together with Tanya Trippett, MD, Director of POETIC, and researchers at Memorial Sloan Kettering Cancer Center. Further details regarding the presentation will be announced closer to the ASCO conference.
About PV-10
Provectus' lead investigational cancer drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About the Pediatric Oncology Experimental Therapeutics Investigators' Consortium
The Pediatric Oncology Experimental Therapeutics Investigators' Consortium (POETIC) was founded in February 2003 by Dr. Tanya Trippett at Memorial Sloan Kettering Cancer Center and Dr. Lia Gore at the University of Colorado Cancer Center. POETIC is composed of ten large academic medical centers in North America with a major emphasis on comprehensive cancer care and research that provide the collaborative and research strength needed to complete intensive phase I and II studies. Each of the institutions is uniquely suited to complete early studies in the pediatric and adolescent populations. POETIC's assets include membership in NCI-designated Comprehensive Cancer Centers, on-site NIH-funded pediatric and/or general clinical translational research centers (CTRCs/CTSAs), and active collaborations with developmental therapeutics programs for adults at a majority of its member institutions. The availability of strong basic science and translational research programs at the institutions allows focus on the development and evaluation of new therapeutic strategies for patients with cancer and related disorders. POETIC's pediatric oncology studies focus on the biologic basis for anti-cancer therapy, and in particular, attempt to explore and evaluate novel agents and/or combinations of therapies early in clinical development as well as new approaches to targeted delivery. For additional information about POETIC, please visit the Consortium's website at www.poeticphase1.org.
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company's clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains "forward-looking statements" as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as "anticipate," "believe," "would," "could," "estimate," "expect," "intend," "may," "plan," "predict," "project," "will," and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: 866-594-5999
Press Releases
LT
NEWS -- PROVECTUS BIOPHARMACEUTICALS ANNOUNCES CHANGES TO BOARD OF DIRECTORS
FOR IMMEDIATE RELEASE
KNOXVILLE, TN, April 24, 2018 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, www.provectusbio.com) (“Provectus” or the “Company”), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, today announced changes to the Company’s Board of Directors (the “Board”) effective April 27, 2018.
Changes to Provectus’ Board
Nice to see you LT.
Been in this stock for many years and I hope your right LT. Have a great evening.
Pokerpro
Thanks, Pokerpro. I see ASITIS is here, too. How appropriate.
It was ASITIS, during the last week of this past January, who alerted me (on that other board) to the existence of iHub's trade-by-trade site and as I mentioned in my first post here, I've become addicted to following it on a daily basis.
I try to follow every day's trading reasonably closely. Anyone familiar with my posting history knows that I am fascinated with unusual/different/intriguing/anomalous activity with regard to that daily trading record. And something has come up recently in regard to that trade-by-trade record that has grabbed my attention.
When I began following the iHub site, my off-the-top-of-my-head "feeling" about the daily trading in this stock was confirmed. Notwithstanding whether the stock price closed on any day with a gain or a loss and notwithstanding the volume, I have believed for a long time that there seemed to be more selling activity trade-wise than buying activity on a daily basis. Because trading in this stock has ALWAYS been sporadic, even when the stock price was tens of times higher than it is now, I thought I could get a decent idea of how many of those trades were buys and how many were sells (e.g., was the trade at/near the Bid?; the Ask?) But, of course, it had to be just a WAG.
With the ihub site newly available to me, I've been able to make those guesses far, far more reliable. And for the first couple months, my intuitive belief was confirmed: on most days (not all but most) selling activity, as measured by the number of trades and the volume of those trades outdistanced buying activity whether the stock price closed with a loss or a gain.
Then, beginning on April 9th, that pattern dramatically reversed. For the past 11 trading days, buying activity - as measured by the number of trades and the volume of those trades, has overwhelmed selling activity on every one of those days. 7 gaining days, 3 losing days and today's even finish. And on most of those days, buying activity as measured by volume was 10 or more times the size of selling activity.
Intriguing? You betcha! What could it all mean? Something going on behind the curtains? Those in the know, know something?
I wouldn't be surprised if we find out in the pretty nearest future.
But that may just be me.
LT
Been reading your posts for years LT. I can tell you I welcome your wisdom and charting here. You may remember me as Itsallriggedanyways. Changed my moniker nearly 3 years ago to pokerpro05, but it is still Itsallriggedanyways on IV. Be nice to get Vorlon and the rest back here as well. I believe Vorlon could make nice with Ihub and get off of the Ihub death row. Anyways welcome.
Pokerpro
On IV there is a poster Market Maker who has to be the dumbest AH poster I have ever seen. Truly knows nothing about markets or market making.
Thanks LT. I also like the PVCT inputs on facebook.
I think I'm going to try posting here. I've been a PVCT shareholder for 7 years and for almost all that time I've been posting on Investor Village (IV). As a matter of fact, in November of 2011, I rescued the then dormant IV PVCT board from oblivion and started posting there on my lonesome until a couple of friends joined me. Now there are 3 IV PVCT boards and angry clashes have arisen between two of them. I am interested in PVCT, not angry clashes amongst message boarders. I recently stumbled upon the iHub trade-by-trade site and have become addicted to it since my primary interest is technical analysis and all the things that can be learned from a close examination of everyday trading. If posting here works for me I may try to convince unhappy and dissatisfied fellow IV'ers to join me.
LT
NEWS -- SECOND STUDY OF EXPANDED ACCESS PATIENTS TREATED WITH PV-10 FOR IN-TRANSIT MELANOMA PUBLISHED IN JOURNAL OF SURGICAL ONCOLOGY
FOR IMMEDIATE RELEASE
KNOXVILLE, TN, April 19, 2018 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, http://www.provectusbio.com) (“Provectus” or the “Company”), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, today announced that the Journal of Surgical Oncology (“JSO”) had published results from an investigator-led, single-center study of patients with in-transit melanoma who received intralesional PV-10 under expanded access. The JSO article detailed the experience of investigators at the Princess Alexandra Hospital in Brisbane, Australia treating 45 patients with almost exclusively Stage III disease from 2008 to 2015.
The current article may be accessed via the JSO’s website at https://onlinelibrary.wiley.com/doi/10.1002/jso.24921. (1)
Patients and Key Results
NEWS -- PROVECTUS BIOPHARMACEUTICALS EXPANDS GLOBAL PATENT PORTFOLIO FOR CANCER COMBINATION THERAPY
KNOXVILLE, TN, April 18, 2018 (GLOBE NEWSWIRE) -- Provectus Biopharmaceuticals, Inc. (OTCQB: PVCT, http://www.provectusbio.com) (“Provectus” or the “Company”), a clinical-stage biotechnology company developing PV-10 as the first small molecule oncolytic immunotherapy for solid tumor cancers, today announced that the Japan Patent Office (JPO) had granted and the European Patent Office (EPO) had allowed the Company’s patent application for the combination of PV-10 with systemic immunomodulatory therapy (i.e., immune checkpoint inhibition). Pfizer, Inc. is a co-assignee on the award and allowance.
The JPO patent and EPO patent allowance are related to U.S. patent (USP) 9,839,688, “Combination of rose bengal and systemic immunomodulative therapies for enhanced treatment of cancer,” which was awarded by the United States Patent and Trademark Office (USPTO) in December 2017. USP 9,839,688 is one of the continuations of the Company’s foundational cancer combination therapy patent, USP 9,107,887, which was awarded by the USPTO in August 2015.
Provectus’ patent portfolio provides global intellectual property protection into the 2030s for the synthesis and use of PV-10 and other halogenated xanthene-based therapeutics as monotherapies and part of combination therapies for cancer.
About PV-10
Provectus’ lead investigational cancer drug product, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. It elicits tumor immunity via activation of dendritic cells mediated by the release of damage-associated molecular pattern molecules. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company’s website at www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-599
https://finance.yahoo.com/news/provectus-biopharmaceuticals-expands-global-patent-100000327.html?.tsrc=applewf
News is out....thoughts? Besides black magic.
Yeah I feel it. This one is quite the waiting game, but I'm very confident. Hopefully the volume keeps up.
I feel some energy going on. Hope at least one more person feels it.
.85 strike mature date 2020. I called the company.
did you check the terms and strike price on the warrants? all I could find was a strike price of .75 cents and 80 cents with a 2018 expiration. I hope you found something better.
Do you have info on the patents with Pfizer. Maybe a link? Thanks man
Quiet here. New investor. Bought a nice pot of options.
Fak!! Well I own 50,000 warrants then! Funny they trade like a stock..
Does anyone know the difference between ticket PVCT and PVCTW? Both are listed on QB
No argument from me about the efficacy of PV-10.
However, I think the PPS may eventually be headed back to 5's. I lightened my load around .077-.08, but I will add more if we dip below .06
added today at 7
This can be a simple and effective product administered in a doctors office as easy as getting a simple shot. Since it is safe, might find a lot of combos
No I.V., no radiation... No massive bills.
Phase 3 needs to meet endpoints. FDA approval should not be a problem after that.
Where are you on this now?
.80 just 3 years ago and prior to that it was higher. A high Overall Survival(OS) rate using drug is what will get volume behind this stock, and in a way the longer we wait to hear the results the better since we hope people are living longer after using it, right?
If only the volume would pick up! Once it does, I expect this to grasp at 0.10.
But signal generated today at Americanbulls,com
Tell Dr. Predicto thanks and godspeed!
Dr. Predicto speaks": very quick upside coming soon will challenge old highs and breakthrough to new highs.
8-K out. Nothing special, just another PRH note issued, this time for $250,000. PRH is being careful not to dole out one cent more than is needed, it seems. However, they are still backing PVCT, which is good.
https://ih.advfn.com/p.php?pid=nmona&article=76799133
I think it got a little ahead of itself in the last rally, plus the Maxim ratchets started hitting heavily once we were over .07 Its RSI reached the solidly overbought zone, but is approaching neutral now.
We saw this same kind of softening between hard news a few times last year, and we came out ahead each time. I think this has been setting up for another little mini-pop in the next week, although I think we're still gonna hit strong resistance at .09 Can we break though? It will probably take a couple of 5M volume days.
Had a chance to home in on the 9's but never got there even with the news, which I thought was very good. Previously I had thought the scope was limited to external melanoma lesions, still a very big market. This PR however, expanded usage to other cancers.
Don't think enough people saw it or follow this company.
Breaking through the low 9's and new highs quick to come.
What might be holding it back is the fact it has come from 2 cents, still don't see this overbought.
Now that’s what I call investing! Great job.
Out of PVCT with a 20%+ gain.
Hey thanks again, not posting just reading and finally some sanity!!!
Thanks, will do and much appreciated! On another note, I wonder why my post was deleted, did I say something wrong?
|
Followers
|
185
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9952
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
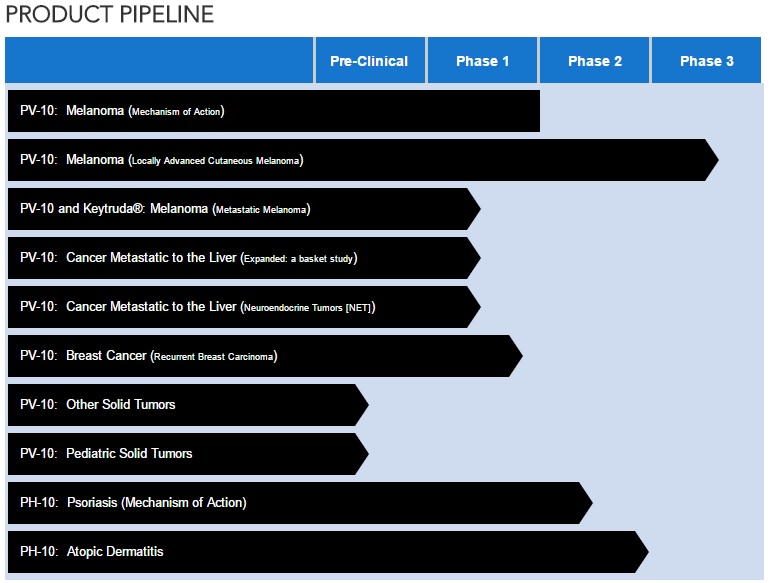
CURRENT PVCT PIPELINE (as of May 23, 2017)
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
