Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
If they are expanding their programs, they’re obviously not going out of business soon. Still waiting for the update for TGA which I hope will be soon.
NEWS -- Provectus Biopharmaceuticals Expands Sponsored Research Collaboration with University of Calgary (Canada) to Investigate Systemic Administration of Pharmaceutical-Grade Rose Bengal for Treatment of Pediatric Leukemia
KNOXVILLE, TN, Aug. 17, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the Company has expanded its sponsored research program with Aru Narendran, MD, PhD, Professor, Departments of Pediatrics, Oncology, Biochemistry & Molecular Biology, and Physiology & Pharmacology at the Cumming School of Medicine of the University of Calgary in Calgary, Alberta, Canada to investigate systemic administration of Provectus’ pharmaceutical-grade rose bengal for the treatment of pediatric leukemia. The Company’s innovatively-assembled and proprietary rose bengal is the lead member of a class of small molecules called halogenated xanthenes.
As part of this new sponsored research, the Narendran team plans to:
NEWS -- Provectus Biopharmaceuticals Establishes Research Collaboration with University of Nevada, Las Vegas to Investigate Small Molecule Immunotherapy Rose Bengal for Tissue Regeneration and Repair
KNOXVILLE, TN, July 19, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the Company has initiated a new sponsored research program with Kelly Tseng, PhD, Associate Professor of Pathology and Lab Medicine, School of Life Sciences at the University of Nevada, Las Vegas (UNLV) to characterize the effects of Provectus’ pharmaceutical-grade rose bengal sodium (RBS) on vertebrate tissue regeneration and repair. RBS is the lead member of a class of small molecules called halogenated xanthenes that is entirely owned by Provectus.
The Tseng Lab at UNLV will assess the effects of RBS on animal development and tissue repair using the African clawed frog (Xenopus laevis), an established vertebrate model organism, and in vivo assays to evaluate key biological processes: embryo development, wound healing, and tissue regeneration.
Dr. Tseng is an expert in tissue regeneration and a leader in the regenerative biology and bioelectrical signaling fields. Her research group at UNLV seeks to elucidate the mechanisms of complex tissue regeneration in vertebrates using the highly regenerative clawed frog, with the goal of applying this knowledge to therapeutic strategies. Dr. Tseng has identified key factors that control limb and eye regeneration, and has also established a new model for studying embryonic eye stem cells.
She graduated from the Massachusetts Institute of Technology with a Bachelor of Science in Biology and Harvard University with a PhD, and was a Howard Hughes Medical Institute research fellow at Boston Children's Hospital. Dr. Tseng's work has been highlighted in books and media outlets including The New York Times.
Recent medical journal publications of hers include “From Cell Death to Regeneration: Rebuilding After Injury” (Front Cell Dev Biol 18;9:655048. 2021), “Studying in vivo Retinal Progenitor Cell Proliferation in Xenopus laevis” (Retinal Development. Methods in Molecular Biology, 2092:19-33. 2020), and “Using the Xenopus Developmental Eye Regrowth System to Distinguish the Role of Developmental Versus Regenerative Mechanisms” (Front Physiol 10:502. 2019).
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes (HXs). The Company’s lead molecule is RBS. A second HX molecule has been synthesized.
Provectus’ drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), animal health, and tissue regeneration and repair, and use multiple routes of administration, such as intralesional (IL), topical (.top), oral (P.O.), inhaled (.inh), intranasal (IN), and intravenous (IV).
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of:
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
I went to the 2022 Annual Meeting and here is my Summary
1. All motions passed by stockholders to include the Reverse Split. Interestingly, PRH only has the authority to implement the Reverse Split up to Dec 22, so this gives stockholders a little insight into timing for PRH to implement the RS and then move the company on to one of the major exchanges which will then open up institutional investors and hopefully start an upward projection of stock price.
2. Provectus has moved to a new office. A little less space but saves some money. Evidently the landlord had other uses for their previous location.
3. Dominic spent some time discussing their efforts to protect their intellectual property.
4. PRH has raised $27 million over the last 5 years. Dominic discussed the difficulties of raising funds in the future.
5. One investor asked about the TGA application. There was no comment from the company. The investor then asked if they would confirm that they had submitted an application to the TGA and they said no comment.
a. On a side note, although they will not confirm the application, most investors believe an application has been submitted and they are waiting on the results. See below for some previous post from Vorlon and LTG on potential TGA submittal.
b. https://www.investorvillage.com/groups.asp?mb=19572&mn=6358&pt=msg&mid=23031877 (LTG Post from IV Board)
c. https://www.investorvillage.com/groups.asp?mb=19572&mn=6424&pt=msg&mid=23108365 (Vorlon Post from IV Board)
6. Regarding PH-10, Dominic stated they need to accomplish more studies to demonstrate how PH-10 works, but they need to focus their available funds in other areas at this time.
7. There were some comments regarding the effectiveness of Rose Bengal in drug resistant staph infections.
8. One investor asked if the company was still following the business plan as provided below for your convenience. Dominic confirmed that this is still current.
a. https://www.provectusbio.com/media/presentation/2021.4/Provectus-Corporate-Deck-December2021-web.pdf
9. Dominic commented that Rose Bengal is proven to be a much more capable molecule for more applications than any of the PRH investors had imagined when they achieved control of the company.
Overall, there wasn’t a lot of new information provided at the meeting. I think we are in a wait and see mode at this time to see what happens with the TGA submittal. If that is approved, then a lot of doors will start opening up to include cash flow, major stock market listing, and potentially new partnerships. I would encourage other AM attendees to post their thoughts.
NEWS -- Provectus Biopharmaceuticals Presents Data from PV-10® Treatment of Metastatic Uveal Melanoma Patients in Two Oral Presentations at 2022 International Society of Ocular Oncology (ISOO) Congress
KNOXVILLE, TN, June 30, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that updated data from the Company’s initial expansion cohort of patients with uveal melanoma metastatic to the liver (mUM) in its cancers-of-the-liver Phase 1 trial of investigational immunocatalyst PV-10 (NCT00986661) were part of two oral presentations at the 20th Congress of the International Society of Ocular Oncology (ISOO), held June 17-21, 2022 in Leiden, The Netherlands.
The first presentation, given by Krysta McVay, Research Nurse, Department of Melanoma Medical Oncology, Division of Cancer Medicine at MD Anderson Cancer Center (MDACC), was entitled “A phase 1 study of percutaneous autolytic rose bengal disodium for metastatic uveal melanoma patients with hepatic metastases.” A copy of her presentation is available on Provectus’ website at: https://www.provectusbio.com/media/docs/publications/ISOO_PV-10_McVay-2022.pdf.
Sapna Patel, MD, Associate Professor, Department of Melanoma Medical Oncology, Division of Cancer Medicine and Director of the Uveal Melanoma Program at MDACC and Chair, Melanoma Committee, SWOG Cancer Research Network, made the second presentation entitled “Metabolic complete responses (mCR) in metastatic uveal melanoma (mUM) patients treated with image-guided injection of PV-10.” A copy of her presentation is available on the Company’s website at: https://www.provectusbio.com/media/docs/publications/ISOO_PV-10_mCR_Patel.ew16Jun22_v4.pdf.
This ongoing single-center mUM study at MDACC has been led since inception by Dr. Patel. Up to three hepatic mUM tumors can be injected per PV-10 treatment cycle. Response assessments are performed at Day 28, and then every three months. Patients with additional, injectable, visceral hepatic mUM disease may receive additional cycles of PV-10 after Day 28. Eligible patients may also receive standard of care immune checkpoint blockade during and after PV-10 treatment.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes (HXs). The Company’s lead molecule is RBS. A second HX molecule has been synthesized.
Provectus’ drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), and animal health, and use multiple routes of administration, such as intralesional (IL), topical (.top), oral (P.O.), inhaled (.inh), intranasal (IN), and intravenous (IV).
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of:
The Company’s Annual Report on Form 10-K for the period ended December 31, 2021, and
Provectus’ Quarterly Report on Form 10-Q for the period ended March 31, 2022.
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Announces Stockholder Approval to Undertake Reverse Split of Outstanding Equities and Reduce Number of Authorized Equities by Same Ratio
KNOXVILLE, TN, June 23, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the Company’s shareholders have approved the proposals of Provectus’ Board of Directors (Board) to seek the authority to undertake a reverse stock split and an authorized share reduction.
At the Company’s 2022 Annual Meeting of Stockholders held in Knoxville, Tennessee on June 22, shareholders also approved the Board’s recommendations of proposals for the election of directors, the approval of the compensation of Provectus’ named executive officers, and the ratification of the Company’s independent registered public accounting firm.
A copy of Provectus’ Form 8-K filed on June 23 that provided details of shareholder voting on the Board’s five proposals, including a brief description of and the tabulation of votes for each proposal, may be found here: https://www.sec.gov/ix?doc=/Archives/edgar/data/315545/000149315222017562/form8-k.htm.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes (HXs). The Company’s lead molecule is RBS. A second HX molecule has been synthesized.
Provectus’ drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), and animal health, and use multiple routes of administration, such as intralesional (IL), topical (.top), oral (P.O.), inhaled (.inh), intranasal (IN), and intravenous (IV).
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of:
#####
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Presents Multiple Metabolic Complete Responders from PV-10® Treatment of Early-Stage Metastatic Uveal Melanoma Patients at 2022 American Society of Clinical Oncology (ASCO) Annual Meeting
https://finance.yahoo.com/news/provectus-biopharmaceuticals-presents-multiple-metabolic-110000410.html
Do you believe co if they put proposal in doc they already know they have the votes for a yes? If so, won't that trigger people taking a tax loss they have thought was coming for years, longtime shareholders? I don't understand why companies do an RS, since it's the kiss of death in penny stocks. So, people have some thinking to do prior to June 22nd...
Reverse stock split proposal: Amend the Company’s Certificate of Incorporation (as amended by the Series D and D-1 Certificates of Designation) to effect a reverse stock split of the Company’s common stock, Series D Convertible Preferred Stock, and Series D-1 Convertible Preferred Stock at a ratio of between 1-for-10 and 1-for-50, and to make corresponding amendments to the Series D and D-1 Certificates of Designation to provide for the proportional adjustment of certain terms upon a reverse stock split, and
Has the announcement of a reverse split changed your thinking?
NEWS -- PVCT Quarterly Report (10-Q)
May 12, 2022 - 01:49PM
https://ih.advfn.com/stock-market/USOTC/provectus-biopharmaceuti-qb-PVCT/stock-news/88095039/quarterly-report-10-q
NEWS -- Provectus Biopharmaceuticals Announces Notice of Allowance of First U.S. Patent of PH-10 Immuno-Dermatology Agent for Treatment of Inflammatory Dermatoses and Epithelial Diseases
KNOXVILLE, TN, May 03, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed US patent (USP) application 16/204,832, “Combination of local and systemic therapies for enhanced treatment of dermatologic conditions,” covering the use of topical PH-10, a formulation of Provectus’ immunogenic-small molecule and pharmaceutical-grade rose bengal sodium (RBS), in combination with one or more systemic therapies for the treatment of various inflammatory dermatoses, such as psoriasis and atopic dermatitis, and various epithelial diseases. PH-10 is a clinical-stage, multi-indication viable, immuno-dermatology-capable pharmaceutical asset.
This allowed patent application will be the first Provectus patent award in dermatology from the USPTO. The Rockefeller University (TRU) is a co-assignee and James Krueger, MD, PhD of TRU’s Laboratory for Investigative Dermatology is a co-inventor.
Dominic Rodrigues, Vice Chair of Provectus’ Board of Directors, said, “We are grateful to Dr. Krueger and his research team for their key contributions to the innovation of the underlying halogenated xanthene medical science of this allowed patent application, and for their continued assistance in the Company’s prosecution efforts for this and other related Provectus intellectual property.”
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes (HXs). The Company’s lead molecule is RBS. A second HX molecule has been synthesized.
Provectus’ drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), and animal health, and use multiple routes of administration, such as intralesional (IL), topical (.top), oral (P.O.), inhaled (.inh), intranasal (IN), and intravenous (IV).
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the Securities and Exchange Commission (SEC), including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2021.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Immunotherapy Abstract for Metastatic Uveal Melanoma at American Society of Clinical Oncology (ASCO) 2022 Annual Meeting
KNOXVILLE, TN, April 27, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10, a formulation of Provectus’ immunogenic-small molecule and pharmaceutical-grade rose bengal sodium (RBS), for the treatment of uveal melanoma metastatic to the liver (mUM) (NCT00986661) will be presented in a poster session at the American Society of Clinical Oncology (ASCO) 2022 annual meeting, to be held June 3-7, 2022 in Chicago, Illinois.
ASCO Presentation Details:
NEWS -- Provectus Biopharmaceuticals Announces Notice of Allowance of First U.S. Patent Application for Use of PV-10® Immunotherapy in Hematology
KNOXVILLE, TN, April 26, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed US patent application 16/688,319, “Composition and Method for Treating Hematologic Cancers,” covering the use of PV-10, a formulation of Provectus’ immunogenic-small molecule and pharmaceutical-grade rose bengal sodium (RBS), for the treatment of hematologic diseases. In vivo data of mice with acute lymphoblastic leukemia receiving oral PV-10 showed increased survival compared to controls.1
This allowed patent application will be the first Provectus patent award in hematology from the USPTO. Innovate Calgary, the innovation company of the University of Calgary, is a co-assignee and Aru Narendran, MD, PhD of the University of Calgary’s Cumming School of Medicine is a co-inventor on the allowed patent application.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes (HXs). The Company’s lead molecule is RBS. A second HX molecule has been synthesized.
Provectus’ drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), and animal health, and use multiple routes of administration, such as intralesional (IL), topical (.top), oral (P.O.), inhaled (.inh), intranasal (IN), and intravenous (IV).
Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
Reference
1 “Provectus Biopharmaceuticals Provides Update on Research into Oral Administration of PV-10® Immunotherapy for Treatments of Hematology, Oncology, and Virology.” Provectus, 04 Oct. 2021, Press release.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the U.S. Securities and Exchange Commission (SEC), including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2021.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Seeks Stockholder Approval at 2022 Annual Meeting for Authority to Undertake Reverse Split of Outstanding Equities and Reduce Number of Authorized Equities by Same Ratio
KNOXVILLE, TN, April 19, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that, in conjunction with the filing of the Company’s preliminary 2022 proxy statement (Pre-14A), Provectus’ Board of Directors (Board) seeks approvals at the Company’s 2022 Annual Meeting of Stockholders on June 22nd for the authority to:
This has always been an obvious scam.. How do they pull it off for so long?
NEWS -- Provectus Biopharmaceuticals Names Aru Narendran, MD, PhD to Scientific Advisory Board
KNOXVILLE, TN, April 14, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced the addition of Aru Narendran, MD, PhD to the Company’s Scientific Advisory Board. Dr. Narendran is a professor in the departments of Pediatrics, Oncology, and Biochemistry & Molecular Biology at the University of Calgary’s Cumming School of Medicine (Calgary, Alberta, Canada), and holds the Kids Cancer Care Foundation Endowed Chair in Clinical and Translational Research in pediatric oncology.
Over the last five years, Provectus’ collaboration with Dr. Narendran and his lab research team at the University of Calgary has yielded:
NEWS -- Provectus Biopharmaceuticals Announces Presentation of Multiple Protein Kinase Signaling Targets, including WNK1, for PV-10® Immunotherapy at American Association for Cancer Research (AACR) 2022 Annual Meeting
KNOXVILLE, TN, April 11, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that preclinical research on systemic administration of investigational cancer immunotherapy PV-10 (rose bengal sodium) for the treatment of high-risk and refractory adult solid tumor cancers is being presented at the annual meeting of the American Association for Cancer Research (AACR), being held April 8-13, 2022 in New Orleans, Louisiana.
This PV-10 research has been led by Aru Narendran, MD, PhD and his team from the Cumming School of Medicine at the University of Calgary in Calgary, Alberta, Canada.
Dr. Narendran and his colleagues previously showed that PV-10 induced cell death at pharmacologically-relevant concentrations in a panel of phenotypically-diverse adult solid tumor cell lines comprising breast, colorectal, head and neck, and testicular cancers (Tran et al. 2021 ASCO). The University of Calgary team subsequently investigated target validation and modulation of PV-10 treatment on protein kinase signaling and associated impacts on specific oncogenic pathways in these adult solid tumor cancers.
A copy of the AACR poster presentation is available on Provectus’ website at https://www.provectusbio.com/media/docs/publications/AACR2022_PV-10_Adult_Tumor_Poster.pdf.
Highlights from the 2022 AACR Presentation:
NEWS -- Provectus Biopharmaceuticals Announces Synthesis of New Halogenated Xanthene Small Molecule
KNOXVILLE, TN, March 24, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the Company has successfully developed and manufactured a second halogenated xanthene (HX), adding to its lead, clinical-stage, HX small molecule called rose bengal sodium (RBS).
The molecular name of the newly synthesized HX is 4,5,6,7-tetrabromo-3',6'-dihydroxy-2',4',5',7'-tetraiodo-3H-spiro[isobenz- ofuran-1,9'-xanthen]-3-one.
Provectus’ intellectual property estate comprises an entire class of proprietary, related HX molecules that are proprietarily-synthesized using Quality-by-Design principles (QbD) under current Good Manufacturing Practice (cGMP) and International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines.
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors said, “Different permutations and combinations of halogen atoms on the backbone of this molecular class convey different biological, chemical, and/or physical properties to each class member. We are excited to have produced a new halogenated xanthene, further expanding our medical science platform. We look forward to developing this small molecule into a viable Provectus drug product candidate.”
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on a class of small molecules called halogenated xanthenes. Multi-route of administration drug discovery and development programs include investigational drugs and drug targets in oncology (clinical-stage), dermatology (clinical-stage), hematology, virology, microbiology, ophthalmology (clinical-stage), and animal health. Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the SEC, including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2020 and Provectus’ Quarterly Report on Form 10-Q for the quarter ended September 30, 2021.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Announces Oral Presentation of Updated Study Data from Metastatic Neuroendocrine Cancer Phase 1 Trial of PV-10® at 2022 European Neuroendocrine Tumor Society (ENETS) Annual Conference
KNOXVILLE, TN, March 16, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10 (rose bengal sodium) for the treatment of neuroendocrine tumors (NET) metastatic to the liver (mNET) refractory to somatostatin analogs (SSAs) and peptide receptor radionuclide therapy (PRRT) (NCT02693067) was presented at the annual conference of the European Neuroendocrine Tumor Society (ENETS), held from March 10-11, 2022 in a hybrid setting in Barcelona, Spain and online.
The oral presentation was made by the principal investigator of the clinical trial’s single center at The Queen Elizabeth Hospital (TQEH) in Adelaide, Australia: Tim Price, MBBS, DHlthSc (Medicine), FRACP, Head of Clinical Oncology Research and Chair of the combined Hematology and Medical Oncology Unit at TQEH, and Clinical Professor in the Faculty of Medicine at the University of Adelaide.
Highlights from the 2022 ENETS Presentation:
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Metastatic Uveal Melanoma Abstract for Oral Presentation at 2022 International Society of Ocular Oncology (ISOO) Congress
KNOXVILLE, TN, Feb. 16, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10 for the treatment of uveal melanoma metastatic to the liver (mUM) (NCT00986661) will be presented at the 20th Congress of the International Society of Ocular Oncology (ISOO), to be held June 17-21, 2022 in Leiden, The Netherlands.
The abstract that was accepted for oral presentation is entitled:
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Systemic Administration Abstract at American Association for Cancer Research (AACR) Annual Meeting 2022
KNOXVILLE, TN, Feb. 08, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that preclinical research on systemic administration of investigational cancer immunotherapy PV-10 (rose bengal sodium) for the treatment of high-risk and refractory adult solid tumor cancers will be presented at the annual meeting of the American Association for Cancer Research (AACR), to be held April 8-13, 2022 in New Orleans, Louisiana.
The abstract, which was accepted for poster presentation, is entitled:
Although still a long shot, and far from clinical trial derived results, PVCT sure has some shots on goal. Ophthalmology applications and/or veterinary medicine offshoots alone could provide some much needed cash flow for attempting to hunt down the main prize. Some biotech somewhere has to be taking notice.
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Neuroendocrine Cancer Abstract for Oral Presentation at 2022 European Neuroendocrine Tumor Society (ENETS) Annual Conference
KNOXVILLE, TN, Feb. 01, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10 (rose bengal sodium), which has completed enrollment for the treatment of neuroendocrine tumors (NET) metastatic to the liver (mNET) refractory to somatostatin analogs (SSAs) and peptide receptor radionuclide therapy (PRRT) (NCT02693067), will be presented at the annual conference of the European Neuroendocrine Tumor Society (ENETS), to be held in a hybrid setting from March 10-11, 2022 in Barcelona, Spain and online.
The abstract, which was accepted for oral presentation, is entitled:
NEWS -- Provectus Biopharmaceuticals Provides Update on Research into PV-10® Immunotherapy as an Immune Adjuvant for Making Vaccines Work Better
KNOXVILLE, TN, Jan. 27, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today provided an update on research of its investigational immunotherapy PV-10 (rose bengal sodium, a halogenated xanthene small molecule) as an immune adjuvant in vaccines for cancer and viruses to improve immune response upon vaccination, in conjunction with the Company’s ongoing efforts to expand its intellectual property.
On January 20, 2022, the U.S. Patent and Trademark Office (USPTO) published Provectus’ patent application entitled “Halogenated Xanthenes as Vaccine Adjuvants” (publication no. US 2022/0016242 A1), which contained among other things:
Excuse me
But doesn’t this appear to be fantastic news?
Stocks that never move and why the hell not?
Oh well
Good luck provectus.. seems like you have a great drug that will never actually help anybody that is alive today..
Maybe in 50 years they will let you save somebody.. meanwhile.. 5 year olds May mix’matxh mRNA “faux Vax” that has over 1 million negative Vares reports and over 20.000 deaths..
NEWS -- Provectus Biopharmaceuticals Announces Publication of Pharmaceutical-Grade Rose Bengal’s Activity Against Gram-Positive Bacteria
KNOXVILLE, TN, Jan. 10, 2022 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that preclinical research on a formulation of the Company’s pharmaceutical-grade rose bengal against Gram-positive (G+) bacteria was published in Molecules, an open-access journal of chemistry.
The article, entitled “Antibacterial Activity of Pharmaceutical-Grade Rose Bengal: An Application of a Synthetic Dye in Antibacterial Therapies,” may be accessed at: https://www.mdpi.com/1420-3049/27/1/322/htm.
This work, led by Michio Kurosu, PhD, Professor, Department of Pharmaceutical Sciences, College of Pharmacy, was conducted under a sponsored research agreement between the University of Tennessee Health Science Center in Memphis, Tennessee and Provectus.
Dr. Kurosu and his colleagues showed that:
I wish to read up on the liver cancer trial. Anyone know of any online info other than that provided at the link below? TIA
https://clinicaltrials.gov/ct2/show/NCT00986661?term=Provectus&draw=2&rank=10
NEWS -- Provectus Biopharmaceuticals Announces Presentation of Meta-Analysis Results of Single-Agent PV-10® in Stage III Cutaneous Melanoma at Society for Melanoma Research (SMR) 2021
KNOXVILLE, TN, Nov. 01, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that results from a meta-analysis of response, survival, and safety data from the Company’s Phase 2 and 3 clinical trials (NCT00521053 and NCT02288897, respectively) and expanded access program (EAP) (NCT01260779) of single-agent cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of Stage III cutaneous melanoma was presented at the SMR 2021 Virtual Congress (the Society for Melanoma Research annual meeting), which was held online from October 28th to 31st.
A copy of the poster presentation is available on Provectus’ website at https://www.provectusbio.com/media/docs/publications/SMR-2021-PV-10-in-Stage-III-Melanoma.21Oct2021.pdf.
Highlights of the SMR 2021 presentation:
Company has promise, no doubt about that. If this thing ever makes a break-out move, can't imagine how high it may go. Nonetheless, the stock is still a lottery ticket type investment. Their patent protection over Rose Bengal concerns me most. Not sure what type of `moat' they have for legal protection. Even if Rose Bengal is proven to be a highly effective (which I believe it will be proven to be), what prevents other BP's to just make clinical grade generic PV10 themselves?
The many potential uses of this beneficial drug is so impressive, for patient and investor sakes, I hope they are successful.
NEWS -- Provectus Biopharmaceuticals Announces Presentation of Updated Data from Combination Therapy Trial of PV-10® and Keytruda® for Treatment of Checkpoint-Refractory Advanced Cutaneous Melanoma at Society for Melanoma Research (SMR) 2021
NEWS -- Provectus Biopharmaceuticals Provides Update on Research into Oral Administration of PV-10® Immunotherapy for Treatments of Hematology, Oncology, and Virology
KNOXVILLE, TN, Oct. 04, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today provided an update on the Company’s research into the systemic administration of Provectus’ investigational immunotherapy, PV-10 (rose bengal disodium, a halogenated xanthene molecule), for the treatments of hematology, oncology, and virology indications, in conjunction with the Company’s efforts to expand its intellectual property.
Hematology: On August 5, 2021, the U.S. Patent and Trademark Office (USPTO) published Provectus’ patent application entitled “Composition and Method for Oral Treatment of Leukemia” (publication no. US 2021/0236418 A1), containing:
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10 Poster Presentations at Society for Melanoma Research (SMR) 2021 Congress
KNOXVILLE, TN, Sept. 27, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from historical clinical trials and expanded access studies of investigational cancer immunotherapy PV-10 (rose bengal disodium) for the single-agent treatment of Stage III melanoma (NCT00521053, NCT01260779, and NCT02288897) and an ongoing clinical trial of PV-10 combination therapy for the treatment of Stage IV melanoma (NCT02557321) will be presented on two poster presentations at the SMR 2021 Congress (the Society for Melanoma Research annual meeting) to be held online from October 28-31.
The two accepted abstracts are:
NEWS -- Provectus Biopharmaceuticals Announces Presentation of Full Study Data from Metastatic Neuroendocrine Cancer Phase 1 Trial of PV-10® at European Society for Medical Oncology (ESMO) Congress 2021
KNOXVILLE, TN, Sept. 21, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of neuroendocrine tumors (NET) metastatic to the liver (mNET) refractory to somatostatin analogs (SSAs) and peptide receptor radionuclide therapy (PRRT) (NCT02693067) was presented at the European Society for Medical Oncology (ESMO) Congress, held online from September 16-21, 2021.
Highlights from the ESMO 2021mNET Presentation:
Never happen until the warrants are gone end of August. Then we may see some selling as they blow out but then we should be free to finally trade normally.
Finally broke above .07... felt like we were hitting that number for months
Now lets see if we can break to a 52 week high and eclipse .10
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Neuroendocrine Cancer Abstract at European Society for Medical Oncology (ESMO) Congress 2021
KNOXVILLE, TN, July 19, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that data from an ongoing clinical trial of investigational cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of neuroendocrine tumors (NET) metastatic to the liver (mNET) refractory to somatostatin analogs (SSAs) and peptide receptor radionuclide therapy (PRRT) (NCT02693067) (https://clinicaltrials.gov/ct2/show/NCT02693067) will be presented at the European Society for Medical Oncology (ESMO) Congress, to be held online from September 16-21, 2021.
The abstract accepted for electronic poster presentation is entitled:
NEWS -- Provectus Biopharmaceuticals Announces Publication of In Vitro Data from Research on Oral Delivery of PV-10® at American Society of Clinical Oncology (ASCO) 2021 Annual Meeting
KNOXVILLE, TN, June 09, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that preclinical data from the Company’s research on oral delivery of investigational cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of adult solid tumors were published as an abstract as part at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting, held June 4-8 online.
A copy of the abstract, which was first published by ASCO on May 19th, is available on its website at https://meetinglibrary.asco.org/record/200310/abstract.
This research on the potential oral administration of PV-10 for the prophylactic and/or therapeutic treatment of adult solid tumor cancers is led by Aru Narendran, MD, PhD, Professor, Departments of Pediatrics, Oncology, Biochemistry & Molecular Biology, and Physiology & Pharmacology in the Cumming School of Medicine at the University of Calgary (UCal) in Calgary, Alberta, Canada. In association with their ASCO abstract, Dr. Narendran’s team prepared a brief presentation for inclusion in this press release.
A copy of the presentation is available on Provectus’ website at https://www.provectusbio.com/media/docs/PV10-solid-tumor-in-vitro-2021.pdf.
Highlights from the ASCO 2021 abstract and UCal’s associated presentation:
NEWS -- Provectus Biopharmaceuticals Announces Presentation of Full Study Data from Metastatic Neuroendocrine Cancer Phase 1 Trial of PV-10® at American Society of Clinical Oncology (ASCO) 2021 Annual Meeting
KNOXVILLE, TN, June 04, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that preliminary full study data from the Company’s Phase 1 clinical trial of investigational cancer immunotherapy PV-10 (rose bengal disodium) for the treatment of neuroendocrine tumors (NET) metastatic to the liver (mNET) refractory to somatostatin analogs (SSAs) and peptide receptor radionuclide therapy (PRRT) (https://clinicaltrials.gov/ct2/show/NCT02693067) is be presented at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting, held June 4-8 online.
Highlights from the mNET Presentation at ASCO 2021:
NEWS -- State of Tennessee Awards Funding for Public-Private Partnership to Study Animal Cancers and Dermatological Disorders using Provectus’ Medical Science Platform
KNOXVILLE, TN, May 25, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the State of Tennessee, as part of its fiscal year 2021-2022 budget, has directed funding in the amount of $2.5 million to develop animal health drug products through partnerships with state universities that have agriculture and veterinary medicine programs and the Knoxville-headquartered biotechnology company.
This study will pursue oncology, hematology, and dermatology treatments for companion and production animals, furthering research in these areas and eventual drug development. The foundation of this study is the Company’s halogenated xanthene medical science platform that already includes:
Warrant overhang. 14 more weeks and they expire. Should be much better for us afterwards.
NEWS -- Provectus Biopharmaceuticals Announces Acceptance of PV-10® Immunotherapy Abstracts at American Society of Clinical Oncology (ASCO) 2021 Annual Meeting
never seen a company with more good news than this company and absolutely no move up... why cant this stock make a move?
NEWS -- Provectus Biopharmaceuticals Announces Publication of Australian Compassionate Use Patients Treated with PV-10® Immunotherapy for In-Transit Melanoma Lesions
NEWS -- Provectus Biopharmaceuticals Receives Notice of Allowance for Adult Solid Tumor Cancer Combination Therapy Patent for PV-10® Immunotherapy from United States Patent and Trademark Office
KNOXVILLE, TN, March 30, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed US patent (USP) application 16/678,133, which covers the use of intralesional (aka intratumoral) PV-10, an injectable formulation of Provectus’ proprietary small molecule rose bengal disodium (RBD), in concomitant combination with two immune checkpoint inhibitors (e.g., PV-10 + an anti-CTLA-4 agent + an anti-PD-(L)1 agent) for the treatment of adult solid tumor cancers such as melanoma, breast cancer, primary and metastatic liver cancers, prostate cancer, and small cell and non-small cell lung cancer.
The allowed patent application is the fourth continuation of USP 9,107,887, Provectus’ foundational cancer combination therapy patent granted by the USPTO in 2015. It is also related to USP 9,808,524, USP 9,839,688, and USP 10,471,144, which are also continuations. Provectus is the sole assignee on the allowed patent application.
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “This pending new patent further expands and enhances our intralesional oncology drug development program by protecting, in particular, the concomitant combination of PV-10 and the duo of anti-CTLA-4 and anti-PD-1 agents for the treatment of solid tumor cancers beyond melanoma, for which the combination of ipilimumab and nivolumab has been approved. There is emerging evidence of major differences in how the combination of anti-CTLA-4 and anti-PD-1 drugs acts on T cell populations and thus immune response, compared with either monotherapy.”
Mr. Rodrigues added, “Provectus has shown that single-agent PV-10 treatment of melanoma may lead to the maturation of immature T cells into functional ones, and that the anti-tumor activity of these T cells may be enhanced by the blockade of the PD-1 regulatory pathway. The Company’s clinical study of metastatic uveal melanoma is focused on showing that these T cells can be further enhanced, potentially synergistically so, by the blockade of both the CTLA-4 and PD-1 pathways.”
About PV-10
Intralesional (IL) administration of PV-10 for the treatment of solid tumor cancers can yield immunogenic cell death within hours of tumor injection, and induce tumor-specific reactivity in circulating T cells within days.1,2,3 This IL PV-10-induced functional T cell response may be enhanced and boosted in combination with immune checkpoint blockade (CB).4 In CB-refractory advanced cutaneous melanoma, IL PV-10 may restore disease-specific T cell function, which may also be prognostic of clinical response. IL PV-10 has been administered to over 450 patients with cancers of the skin and of the liver. It is administered under visual, tactile or ultrasound guidance to superficial malignancies, and under CT or ultrasound guidance to tumors of the liver.
About Rose Bengal Disodium
RBD is 4,5,6,7-tetrachloro-2',4',5',7'-tetraiodofluorescein disodium, a halogenated xanthene and Provectus’ proprietary lead molecule. Provectus' current Good Manufacturing Practices (cGMP) RBD is a proprietary pharmaceutical-grade drug substance produced by the Company's quality-by-design (QbD) manufacturing process to exacting regulatory standards that avoids the formation of uncontrolled impurities currently present in commercial-grade rose bengal. Provectus' RBD and cGMP RBD manufacturing process are protected by composition of matter and manufacturing patents as well as trade secrets.
An IL formulation (i.e., by direct injection) of cGMP RBD drug substance, cGMP PV-10, is being developed as an autolytic immunotherapy drug product for solid tumor cancers.
IL PV-10 is also undergoing preclinical study for relapsed and refractory pediatric solid tumor cancers, such as neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma.5,6
A topical formulation of cGMP RBD drug substance, PH-10®, is being developed as a clinical-stage immuno-dermatology drug product for inflammatory dermatoses, such as atopic dermatitis and psoriasis. RBD can modulate multiple interleukin and interferon pathways and key cytokine disease drivers.7
Oral formulations of cGMP RBD are undergoing preclinical study for relapsed and refractory pediatric blood cancers, such as acute lymphocytic leukemia and acute myelomonocytic leukemia.8,9
Oral formulations of cGMP RBD are also undergoing preclinical study as prophylactic and therapeutic treatments for high-risk adult solid tumor cancers, such as head and neck, breast, pancreatic, liver, and colorectal cancers.
Different formulations of cGMP RBD are also undergoing preclinical study as potential treatments for multi-drug resistant (MDR) bacteria, such as Gram-negative bacteria.
Topical formulations of cGMP RBD are also undergoing preclinical study as potential treatments for diseases of the eye, such as infectious keratitis
Tumor Cell Lysosomes as the Seminal Cancer Drug Target
Lysosomes are the central organelles for intracellular degradation of biological materials, and nearly all types of eukaryotic cells have them. Discovered by Christian de Duve, MD in 1955, lysosomes are linked to several biological processes, including cell death and immune response. In 1959, de Duve described them as ‘suicide bags’ because their rupture causes cell death and tissue autolysis. He was awarded the Nobel Prize in 1974 for discovering and characterizing lysosomes, which are also linked to each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Building on the Discovery, Exploration, and Characterization of Lysosomes
Cancer cells, particularly advanced cancer cells, are very dependent on effective lysosomal functioning.10 Cancer progression and metastasis are associated with lysosomal compartment changes11,12, which are closely correlated (among other things) with invasive growth, angiogenesis, and drug resistance13.
RBD selectively accumulates in the lysosomes of cancer cells upon contact, disrupting the lysosomes and causing the cells to die. Provectus2,14, external collaborators5, and other researchers15,16,17 have independently shown that RBD triggers each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Cancer Cell Autolytic Death via RBD: RBD-induced autolytic cell death, or death by self-digestion, in Hepa1-6 murine hepatocellular carcinoma (HCC) cells can be viewed in this Provectus video of the process (ethidium homodimer 1 [ED-1] (https://www.provectusbio.com/media/docs/videos/H4-RB-ED1-LSG-red-blue.mp4)stains DNA, but is excluded from intact nuclei; lysosensor green [LSG] stains intact lysosomes; the video is provided in 30-second frames, with a duration of approximately one hour). Exposure to RBD triggers the disruption of lysosomes, followed by nucleus failure and autolytic cell death. Identical responses have been shown by the Company in HTB-133 human breast carcinoma (which can be viewed in this Provectus video of the process, with a duration of approximately two hours)(https://www.provectusbio.com/media/docs/videos/HTB1-RB-LSG-EthD-523nm-red-blue.mp4) and H69Ar human multidrug-resistant small cell lung carcinoma. Cancer cell autolytic cell death was reproduced by research collaborators in neuroblastoma cells to show that lysosomes are disrupted upon exposure to RBD.5
Tumor Autolytic Death via RBD: RBD causes acute autolytic destruction of injected tumors (via autolytic cell death), mediating the release of danger-associated molecular pattern molecules (DAMPs) and tumor antigens; release of these signaling factors may initiate an immunologic cascade where local response by the innate immune system may facilitate systemic anti-tumor immunity by the adaptive immune system. The DAMP release-mediated adaptive immune response activates lymphocytes, including CD8+ T cells, CD4+ T cells, and NKT cells, based on clinical and preclinical experience in multiple tumor types. Mediated immune signaling pathways may include an effect on STING, which plays an important role in innate immunity.9
Orphan Drug Designations (ODDs)
ODD status has been granted to RBD by the U.S. Food and Drug Administration for metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma (including uveal melanoma) in 2019.
Intellectual Property
Provectus’ IP includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the process by which cGMP RBD and related halogenated xanthenes are produced, avoiding the formation of previously unknown impurities that exist in commercial-grade rose bengal in uncontrolled amounts. The requirement to control these impurities is in accordance with International Council on Harmonisation (ICH) guidelines for the manufacturing of an injectable pharmaceutical. US patent numbers are 8,530,675, 9,273,022, and 9,422,260, with expirations ranging from 2030 to 2031.
The Company's IP also includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the combination of RBD and CB (e.g., anti-CTLA-4, anti-PD-1, and anti-PD-L1 agents) for the treatment of a range of solid tumor cancers. US patent numbers are 9,107,887, 9,808,524, 9,839,688, and 10,471,144, with expirations ranging from 2032 to 2035; US patent application numbers include 20200138942 (i.e., 16/678,133).
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on an entirely- and wholly-owned family of small molecules called halogenated xanthenes. Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, https://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at https://www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
5. Swift et al. Potent in vitro and xenograft antitumor activity of a novel agent, PV-10, against relapsed and refractory neuroblastoma. OncoTargets and Therapy 12, 1293, 2019.
6. Swift et al. In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors. 2018 ASCO Annual Meeting, J Clin Oncol 36, 2018 (suppl; abstr 10557).
7. Krueger et al. Immune Modulation by Topical PH-10 Aqueous Hydrogel (Rose Bengal Disodium) in Psoriasis Lesions. Psoriasis Gene to Clinic, 8th International Congress, Br J Dermatol 177.
8. Swift et al. In Vitro Activity and Target Modulation of PV-10 Against Relapsed and Refractory Pediatric Leukemia. 2018 ASH Annual Meeting, Blood 132, 2018 (suppl; abstr 5207).
9. Thakur et al. Association of heat shock proteins as chaperone for STING: A potential link in a key immune activation mechanism revealed by the novel anti-cancer agent PV-10. 2020 AACR VAM II, (abstr 5393).
10. Piao et al. Targeting the lysosome in cancer. Annals of the New York Academy of Sciences. 2016; 1371(1): 45.
11. Nishimura et al. Malignant Transformation Alters Intracellular Trafficking of Lysosomal Cathespin D in Human Breast Epithelial Cells. Pathology Oncology Research. 1998; 4(4): 283.
12. Gocheva et al. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes & Development. 2006; 20(5): 543.
13. Fehrenbacher et al. Lysosomes as Targets for Cancer Therapy. Cancer Research. 2005; 65 (8): 2993.
14. Wachter et al. Imaging Photosensitizer Distribution and Pharmacology using Multiphoton Microscopy. Proceedings of SPIE 4622, 112, 2002.
15. Koevary. Selective toxicity of rose Bengal to ovarian cancer cells in vitro. International Journal of Physiology, Pathophysiology and Pharmacology 4(2), 99, 2012.
16. Zamani et al. Rose Bengal suppresses gastric cancer cell proliferation via apoptosis and inhibits nitric oxide formation in macrophages. Journal of Immunotoxicology, 11(4), 367, 2014.
17. Luciana et al. Rose Bengal Acetate photodynamic therapy-induced autophagy. Cancer Biology & Therapy, 10:10, 1048, 2010.
Trademarks
PV-10® and PH-10® are registered trademarks of Provectus, Knoxville, Tennessee, U.S.A.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the SEC, including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2020 and Provectus’ Quarterly Report on Form 10-Q for the quarter ended September 30, 2020.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
NEWS -- Provectus Biopharmaceuticals Receives Notice of Allowance for Refractory Pediatric Solid Tumor Cancer Monotherapy and Combination Therapy Patent for PV-10® Immunotherapy from United States Patent and Trademark Office
KNOXVILLE, TN, March 23, 2021 (GLOBE NEWSWIRE) -- Provectus (OTCQB: PVCT) today announced that the United States Patent and Trademark Office (USPTO) has allowed US patent application 16/412,872, which covers the use of intralesional (aka intratumoral) PV-10, an injectable formulation of Provectus’ proprietary small molecule rose bengal disodium (RBD), as both a monotherapy and in combination with immune checkpoint blockade (CB) or radiotherapy for the treatment of relapsed and refractory pediatric solid tumor cancers.
The allowed patent application recognizes the first intellectual property (IP) generated from the Company’s sponsored research of and collaboration with Aru Narendran, MD, PhD, Professor, Departments of Pediatrics, Oncology, Biochemistry & Molecular Biology, and Physiology & Pharmacology at the Cumming School of Medicine of the University of Calgary in Calgary, Alberta, Canada.
The foundation of this oncology-focused IP includes work described by the Narendran research team in its paper entitled “Potent in vitro and xenograft antitumor activity of a novel agent, PV-10, against relapsed and refractory neuroblastoma” (OncoTargets and Therapy 2019) and on its medical conference poster entitled “In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors” (2018 American Society of Clinical Oncology annual meeting).
Dominic Rodrigues, Vice Chair of the Company’s Board of Directors, said, “This pending new patent is another example of the expansion and enhancement of our intralesional oncology drug development program, as we continue to explore systemic administration of rose bengal disodium to treat cancer.”
Mr. Rodrigues added, “Intralesional administration of PV-10 in solid tumor cancers has shown efficacy and systemic activity in clinical settings covering cutaneous and visceral disease, immunologically hot and cold tumor types such as melanoma and cancers of the liver, and in combinations with checkpoint inhibition and radiation.”
About PV-10
Intralesional (IL) administration of PV-10 for the treatment of solid tumor cancers can yield immunogenic cell death within hours of tumor injection, and induce tumor-specific reactivity in circulating T cells within days.1,2,3 This IL PV-10-induced functional T cell response may be enhanced and boosted in combination with CB.4 In CB-refractory advanced cutaneous melanoma, IL PV-10 may restore disease-specific T cell function, which may also be prognostic of clinical response. IL PV-10 has been administered to over 450 patients with cancers of the skin and of the liver. It is administered under visual, tactile or ultrasound guidance to superficial malignancies, and under CT or ultrasound guidance to tumors of the liver.
About Rose Bengal Disodium
RBD is 4,5,6,7-tetrachloro-2',4',5',7'-tetraiodofluorescein disodium, a halogenated xanthene and Provectus’ proprietary lead molecule. Provectus' current Good Manufacturing Practices (cGMP) RBD is a proprietary pharmaceutical-grade drug substance produced by the Company's quality-by-design (QbD) manufacturing process to exacting regulatory standards that avoids the formation of uncontrolled impurities currently present in commercial-grade rose bengal. Provectus' RBD and cGMP RBD manufacturing process are protected by composition of matter and manufacturing patents as well as trade secrets.
An IL formulation (i.e., by direct injection) of cGMP RBD drug substance, cGMP PV-10, is being developed as an autolytic immunotherapy drug product for solid tumor cancers.
IL PV-10 is also undergoing preclinical study for relapsed and refractory pediatric solid tumor cancers, such as neuroblastoma, Ewing sarcoma, rhabdomyosarcoma, and osteosarcoma.5,6
A topical formulation of cGMP RBD drug substance, PH-10®, is being developed as a clinical-stage immuno-dermatology drug product for inflammatory dermatoses, such as atopic dermatitis and psoriasis. RBD can modulate multiple interleukin and interferon pathways and key cytokine disease drivers.7
Oral formulations of cGMP RBD are undergoing preclinical study for relapsed and refractory pediatric blood cancers, such as acute lymphocytic leukemia and acute myelomonocytic leukemia.8,9
Oral formulations of cGMP RBD are also undergoing preclinical study as prophylactic and therapeutic treatments for high-risk adult solid tumor cancers, such as head and neck, breast, pancreatic, liver, and colorectal cancers.
Different formulations of cGMP RBD are also undergoing preclinical study as potential treatments for multi-drug resistant (MDR) bacteria, such as Gram-negative bacteria.
Topical formulations of cGMP RBD are also undergoing preclinical study as potential treatments for diseases of the eye, such as infectious keratitis
Tumor Cell Lysosomes as the Seminal Cancer Drug Target
Lysosomes are the central organelles for intracellular degradation of biological materials, and nearly all types of eukaryotic cells have them. Discovered by Christian de Duve, MD in 1955, lysosomes are linked to several biological processes, including cell death and immune response. In 1959, de Duve described them as ‘suicide bags’ because their rupture causes cell death and tissue autolysis. He was awarded the Nobel Prize in 1974 for discovering and characterizing lysosomes, which are also linked to each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Building on the Discovery, Exploration, and Characterization of Lysosomes
Cancer cells, particularly advanced cancer cells, are very dependent on effective lysosomal functioning.10 Cancer progression and metastasis are associated with lysosomal compartment changes11,12, which are closely correlated (among other things) with invasive growth, angiogenesis, and drug resistance13.
RBD selectively accumulates in the lysosomes of cancer cells upon contact, disrupting the lysosomes and causing the cells to die. Provectus2,14, external collaborators5, and other researchers15,16,17 have independently shown that RBD triggers each of the three primary cell death pathways: apoptosis, autophagy, and necrosis.
Cancer Cell Autolytic Death via RBD: RBD-induced autolytic cell death, or death by self-digestion, in Hepa1-6 murine hepatocellular carcinoma (HCC) cells can be viewed in this Provectus video of the process (ethidium homodimer 1 [ED-1] stains DNA, but is excluded from intact nuclei; lysosensor green [LSG] stains intact lysosomes; the video is provided in 30-second frames, with a duration of approximately one hour). Exposure to RBD triggers the disruption of lysosomes, followed by nucleus failure and autolytic cell death. Identical responses have been shown by the Company in HTB-133 human breast carcinoma (which can be viewed in this Provectus video of the process, with a duration of approximately two hours) and H69Ar human multidrug-resistant small cell lung carcinoma. Cancer cell autolytic cell death was reproduced by research collaborators in neuroblastoma cells to show that lysosomes are disrupted upon exposure to RBD.5
Tumor Autolytic Death via RBD: RBD causes acute autolytic destruction of injected tumors (via autolytic cell death), mediating the release of danger-associated molecular pattern molecules (DAMPs) and tumor antigens; release of these signaling factors may initiate an immunologic cascade where local response by the innate immune system may facilitate systemic anti-tumor immunity by the adaptive immune system. The DAMP release-mediated adaptive immune response activates lymphocytes, including CD8+ T cells, CD4+ T cells, and NKT cells, based on clinical and preclinical experience in multiple tumor types. Mediated immune signaling pathways may include an effect on STING, which plays an important role in innate immunity.9
Orphan Drug Designations (ODDs)
ODD status has been granted to RBD by the U.S. Food and Drug Administration for metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma (including uveal melanoma) in 2019.
Intellectual Property
Provectus’ IP includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the process by which cGMP RBD and related halogenated xanthenes are produced, avoiding the formation of previously unknown impurities that exist in commercial-grade rose bengal in uncontrolled amounts. The requirement to control these impurities is in accordance with International Council on Harmonisation (ICH) guidelines for the manufacturing of an injectable pharmaceutical. US patent numbers are 8,530,675, 9,273,022, and 9,422,260, with expirations ranging from 2030 to 2031.
The Company's IP also includes a family of US and international (a number of countries in Asia, Europe, and North America) patents that protect the combination of RBD and CB (e.g., anti-CTLA-4, anti-PD-1, and anti-PD-L1 agents) for the treatment of a range of solid tumor cancers. US patent numbers are 9,107,887, 9,808,524, 9,839,688, and 10,471,144, with expirations ranging from 2032 to 2035; US patent application numbers include 20200138942.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company developing immunotherapy medicines for different disease areas based on an entirely- and wholly-owned family of small molecules called halogenated xanthenes. Information about the Company’s clinical trials can be found at the National Institutes of Health (NIH) registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
5. Swift et al. Potent in vitro and xenograft antitumor activity of a novel agent, PV-10, against relapsed and refractory neuroblastoma. OncoTargets and Therapy 12, 1293, 2019.
6. Swift et al. In vitro and xenograft anti-tumor activity, target modulation and drug synergy studies of PV-10 against refractory pediatric solid tumors. 2018 ASCO Annual Meeting, J Clin Oncol 36, 2018 (suppl; abstr 10557).
7. Krueger et al. Immune Modulation by Topical PH-10 Aqueous Hydrogel (Rose Bengal Disodium) in Psoriasis Lesions. Psoriasis Gene to Clinic, 8th International Congress, Br J Dermatol 177.
8. Swift et al. In Vitro Activity and Target Modulation of PV-10 Against Relapsed and Refractory Pediatric Leukemia. 2018 ASH Annual Meeting, Blood 132, 2018 (suppl; abstr 5207).
9. Thakur et al. Association of heat shock proteins as chaperone for STING: A potential link in a key immune activation mechanism revealed by the novel anti-cancer agent PV-10. 2020 AACR VAM II, (abstr 5393).
10. Piao et al. Targeting the lysosome in cancer. Annals of the New York Academy of Sciences. 2016; 1371(1): 45.
11. Nishimura et al. Malignant Transformation Alters Intracellular Trafficking of Lysosomal Cathespin D in Human Breast Epithelial Cells. Pathology Oncology Research. 1998; 4(4): 283.
12. Gocheva et al. Distinct roles for cysteine cathepsin genes in multistage tumorigenesis. Genes & Development. 2006; 20(5): 543.
13. Fehrenbacher et al. Lysosomes as Targets for Cancer Therapy. Cancer Research. 2005; 65 (8): 2993.
14. Wachter et al. Imaging Photosensitizer Distribution and Pharmacology using Multiphoton Microscopy. Proceedings of SPIE 4622, 112, 2002.
15. Koevary. Selective toxicity of rose Bengal to ovarian cancer cells in vitro. International Journal of Physiology, Pathophysiology and Pharmacology 4(2), 99, 2012.
16. Zamani et al. Rose Bengal suppresses gastric cancer cell proliferation via apoptosis and inhibits nitric oxide formation in macrophages. Journal of Immunotoxicology, 11(4), 367, 2014.
17. Luciana et al. Rose Bengal Acetate photodynamic therapy-induced autophagy. Cancer Biology & Therapy, 10:10, 1048, 2010.
Trademarks
PV-10® and PH-10® are registered trademarks of Provectus, Knoxville, Tennessee, U.S.A.
FORWARD-LOOKING STATEMENTS: The information in this press release may include “forward-looking statements,” within the meaning of U.S. securities legislation, relating to the business of Provectus and its affiliates, which are based on the opinions and estimates of Company management and are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those projected in the forward-looking statements. Forward-looking statements are often, but not always, identified by the use of words such as “seek,” “anticipate,” “budget,” “plan,” “continue,” “estimate,” “expect,” “forecast,” “may,” “will,” “project,” “predict,” “potential,” “targeting,” “intend,” “could,” “might,” “should,” “believe,” and similar words suggesting future outcomes or statements regarding an outlook.
The safety and efficacy of the agents and/or uses under investigation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated or that such agents as products will achieve any particular revenue levels.
Due to the risks, uncertainties, and assumptions inherent in forward-looking statements, readers should not place undue reliance on these forward-looking statements. The forward-looking statements contained in this press release are made as of the date hereof or as of the date specifically specified herein, and Provectus undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except in accordance with applicable securities laws. The forward-looking statements are expressly qualified by this cautionary statement.
Risks, uncertainties, and assumptions include those discussed in the Company’s filings with the SEC, including those described in Item 1A of the Company’s Annual Report on Form 10-K for the year ended December 31, 2020 and Provectus’ Quarterly Report on Form 10-Q for the quarter ended September 30, 2020.
###
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
more good news today as the company keeps trekking forward... can someone just buy them for a $1 a share already :)
Mark could use some good news
Thanks FDT! I've put it on my watch list. I'll be cheerin for ya!
Yes Slambino, just a hope
Have friends that work at MRK that have heard about PV10 but no real insight other than the good publications about the positive results combining with Keytruda.
|
Followers
|
186
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9950
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
CURRENT PVCT PIPELINE (as of May 23, 2017)
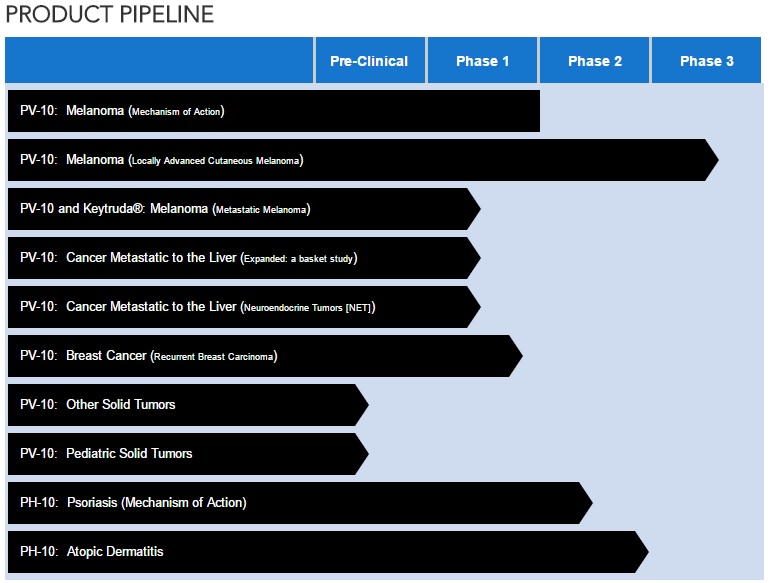
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
