Register for free to join our community of investors and share your ideas. You will also get access to streaming quotes, interactive charts, trades, portfolio, live options flow and more tools.
I have a bid in at .0641 but tdameritrade is showing lowest bid at .0622. Strange
NEWS -- Provectus Receives Notice of Allowance for Fourth Cancer Combination Therapy Patent from the United States Patent and Trademark Office
First time in a long time that we had day with over a million in volume. Higher volume with price increase is a good sign.
News: $PVCT Provectus Provides Update on GI Tumor Program for Investigational Cancer Drug PV-10
Metastatic neuroendocrine tumors: Monotherapy data presentations planned. Phase 2 combination therapy trial designs with different classes of checkpoint inhibitor drugs being developed. Metastatic uveal melanoma: Data presentation planned. Patients have received PV-10 monotherapy and als...
Find out more Provectus Provides Update on GI Tumor Program for Investigational Cancer Drug PV-10
NEWS -- Provectus Provides Update on GI Tumor Program for Investigational Cancer Drug PV-10
NEWS -- Provectus Announces Preliminary Results from Phase 1 Trial of PV-10 for Treatment of Metastatic Neuroendocrine Tumors at ASCO 2019
NEWS -- Provectus Appoints Bruce Horowitz as Chief Operating Officer
KNOXVILLE, TN, May 09, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced the appointment of Bruce Horowitz as the Company’s Chief Operating Officer. Mr. Horowitz had previously served as Provectus’ Chief Operations Consultant since 2017. He has also served as a member of the Company’s Board of Directors (the “Board”) since 2017. Provectus is a clinical-stage biotechnology company developing a new class of drugs for oncology and dermatology based on a wholly-owned family of chemical small molecules called halogenated xanthenes.
Prior to working for Provectus, Mr. Horowitz has served as the Managing Director of Capital Strategists, LLC, which provides corporate, strategic, and financial consulting services, since 2006, and an advisor to family trusts and private individuals, with a focus on financial asset management, real estate management, and special situation investments. He has also served as the Managing Member of Plata LLC since 2017. Earlier in his career, Mr. Horowitz was a charter member of the New York Futures Exchange, was a Senior Vice President managing principal equity investment accounts, private equity investments and public offerings at Drake Capital Securities, and managed the trading department at the Los Angeles office of Laidlaw Equities. He was also a partner at Stanley Capital, a private equity buyout firm. Mr. Horowitz was the chairman and a member of two general obligation bond fund committees, raising more than $500 million in general revenue bonds for the Beverly Hills Unified School District (BHUSD). Subsequently, he was named the first chairman of both the state of California-mandated Citizens’ Oversight Committee and Facilities Advisory Committee, overseeing expenditure of all BHUSD general obligation bond funds. Mr. Horowitz is a founding member of the Los Angeles Chapter of the Positive Coaching Alliance. He founded and is currently the president of the Beverly Hills Basketball League, a youth basketball program that serves more than 35,000 families. Mr. Horowitz has also served as a member of the boards of directors of the American Youth Soccer Organization and Beverly Hills Little League. He holds a Juris Doctor degree from Benjamin N. Cardozo School of Law in New York City and a Bachelor of Arts degree from Washington University in St. Louis.
Ed Pershing, CPA, Chair of the Company's Board, said, “We are thrilled to have Bruce fill this critical leadership position at Provectus. Over the last couple of years, his contribution to reforming the Company’s legacy and inherited corporate governance, business management, and clinical operations issues has been quite consequential. This work ultimately led to realized operational savings of tens of millions of dollars as well as recovery efforts from realized and unrealized judgements and settlements of more than ten million dollars.”
Mr. Pershing added, “During the same period of time, Bruce’s influence has enabled Provectus to begin strategically pivoting the focuses of the clinical development program of the Company’s investigational cancer immunotherapy PV-10 towards neoadjuvant therapy for early-stage cancer where all disease can be treated swiftly with PV-10, and combination therapy with standard of care for later-stage disease where priming and activation of the functional immune response to PV-10 treatment can be augmented by another treatment or treatments.”
About PV-10
Provectus’ lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver (including metastatic symptomatic neuroendocrine tumors and metastatic uveal melanoma), and preclinical study for pediatric cancers. Orphan drug designation status has been granted to PV-10 by the U.S. Food and Drug Administration for the treatments of metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma in 2019.
About PH-10
Provectus' lead investigational dermatology drug, PH-10, a topically applied, non-steroidal, small molecule-based gel, is undergoing clinical study for inflammatory dermatoses like psoriasis and atopic dermatitis. PH-10 is self-applied to affected skin with photoactivation via ambient exposure to visible light. Pathways significantly improved by PH-10 treatment include published psoriasis transcriptomes and cellular responses mediated by IL-17, IL-22 and interferons.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are small molecule chemical agents. Information about the Company’s clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2018).
Contact:
Provectus Biopharmaceuticals, Inc.
Heather Raines, CPA
Chief Financial Officer
Phone: (866) 594-5999
270 shares to drop this stock 75%? Something seems fishy about why someone would do that, right? Can anyone really buy this stock for a penny?
wow, 270 shares sold to drop this stock 40%. I wonder why games are being played with this stock...seems odd.
NEWS -- Provectus Biopharmaceuticals Announces Acceptances of PV-10 Poster Presentations at American Society of Clinical Oncology (ASCO) Annual Meeting
This actually saves the company money.
It all sounds good, but how much money can this company burn through w/o requiring more...
PV-10 Trial Updated
This is the Stage 3 patient cohort expansion we've been waiting on.
"The Company will further expand the Phase 1b portion of the PV-10-pembrolizumab combination study to include a cohort of up to 24 patients who have satellite or in-transit disease (Stage IIIB/C/D melanoma). A first expansion cohort currently is enrolling up to 24 patients with advanced melanoma (Stage IV) who are refractory to checkpoint inhibition."
NEWS -- Provectus Appoints Heather Raines, CPA as Chief Financial Officer
KNOXVILLE, TN, March 25, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced the appointment of Heather Raines as the Company’s Chief Financial Officer (CFO). Mrs. Raines had previously served as Provectus’ Controller since 2017, and now replaces the Company’s interim CFO. Provectus is a clinical-stage biotechnology company developing a new class of drugs for oncology and dermatology based on a wholly-owned family of chemical small molecules called halogenated xanthenes.
From 2007 to 2015, Mrs. Raines served as Controller of the Oak Ridge, Tennessee location of AMETEK, Inc. (AME), a global manufacturer of electronic instruments and electromechanical devices. Following AMETEK, from 2015 to 2017, she served as Vice President of Finance for BDry Waterproofing and Foundation Repair, a national supplier of waterproofing and foundation repair solutions. Mrs. Raines received a Master’s Degree in Accounting from Strayer University and a Bachelor’s Degree in Accounting from the University of Tennessee. She is a Certified Public Accountant (CPA), and a member of the American Institute of CPAs and the Tennessee Society of CPAs.
Ed Pershing, CPA, Chair of the Company's Board of Directors, said, “We are thrilled to have Heather assume this important leadership position at Provectus. She will continue her work integrating the finance and accounting function into all operational aspects of the Company as well as supporting our business planning and corporate development activities.”
Mr. Pershing added, “We have been very pleased with Mrs. Raines detailed approach to the business of biotechnology, the accountability to which she holds herself and those around her, and the passion she brings to Provectus’ goal of developing affordable and accessible oncology and dermatology drug therapies for patients in need.”
About PV-10
Provectus’ lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver (including metastatic symptomatic neuroendocrine tumors and metastatic uveal melanoma), and preclinical study for pediatric cancers. Orphan drug designation status has been granted to PV-10 by the U.S. Food and Drug Administration for the treatments of metastatic melanoma in 2006, hepatocellular carcinoma in 2011, neuroblastoma in 2018, and ocular melanoma in 2019.
About PH-10
Provectus' lead investigational dermatology drug, PH-10, a topically applied, non-steroidal, small molecule-based gel, is undergoing clinical study for inflammatory dermatoses like psoriasis and atopic dermatitis. PH-10 is self-applied to affected skin with photoactivation via ambient exposure to visible light. Pathways significantly improved by PH-10 treatment include published psoriasis transcriptomes and cellular responses mediated by IL-17, IL-22 and interferons.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are small molecule chemical agents. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
FORWARD-LOOKING STATEMENTS: This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2018).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
Provectus appoints Heather Raines as Chief Financial Officer (CFO). She had previously served as Provectus’ Controller since 2017, and now replaces the Company’s interim CFO.
NEWS -- Provectus Provides Update on Metastatic Uveal Melanoma Clinical Development Program for Investigational Drug PV-10
KNOXVILLE, TN, March 21, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today provided updates on the Company's metastatic uveal melanoma drug development program for its lead investigational cancer agent PV-10. Intratumoral injection of small molecule oncolytic immunotherapy PV-10 can yield immunogenic cell death in solid tumor cancers and stimulate tumor-specific reactivity in circulating T cells1-4, which may lead to a functional recruitment of the immune system.
Update #1. Presentations about PV-10 treatment of metastatic uveal melanoma will be given by Sapna Patel, MD, Associate Professor, Department of Melanoma Medical Oncology, Division of Cancer Medicine of The University of Texas MD Anderson Cancer Center at:
Massive bid support here now very unusual, perhaps someone knows something.
news and we hit .075 ish - .08 easy.
What an idiot, shorts are getting desperate
Except for the PR they just put out 2 weeks ago.
http://provectusbio.com/news/press-releases/provectus-pr-20190219-1
...and a week before that.
http://provectusbio.com/news/press-releases/provectus-pr-20190212-1
Make a phone call. If you reach someone who is an employee, that will be enough to double the stock.
Most are wondering if it exists anymore. There usual bi-monthly PR's are about 800 per cent overdue.
There are no life signs.
This stock looks ready to pop soon. Big time bid support, very thin on the ask.
Level 2 is very thin on the ask to .0725
Let's go slap the ask
Thanks for clarification
Alan Ross is a complete fraud. He's lying in order to create doubt. It's beyond obvious.
Where is this Alan Ross spreading this info?
PRH Group does NOT get $200M off the top of any sale.
On the other board Alan Ross keeps stating over and over that with any sale of the company the PRH Group gets the first $200 Million or 10X their initial investment.
I would encourage anyone to go back and read or re-read the 8K from March 23, 2017.
He harvested one line of information and is taking it completely out of context. The PRH Group is entitled to 10X their loan in the event of a Change of Control of the Board of Directors, aka a Change of Control Penalty. This is not the same as a buyout. It was done this way to protect the short term interest of their finances and of the companies well-being in the event of another group trying to take control. Again, not the same as a sale.
In actuality, in the event of a sale, PRH Group (with their Series D Stock that the loan is converted to) is entitled to priority proceeds in the amount of 4X their initial investment if the event happens within 2 years of issuance, and they are entitled to 6X if the event is after 2 years of issuance.
Still not a small number, but $80M and $120M is not $200M.
Far cry from 10X off the top.
Also, the lien on the IP gets released when the loan is converted to shares.
So, as far as concerning Alan Ross, 1 of 2 scenarios is possible:
Either Alan Ross understands this and is blatantly lying or Alan Ross doesn't understand this and honestly believes PRH gets 10X off the top.
In either scenario, you can't trust him or his financial acumen.
8K Below:
https://irdirect.net/filings/viewer/index/315545/000119312517093889/
Higher highs and higher lows today, .057-.0648 looking strong despite being down 4 percent.
NEWS -- Article on anti-tumor activity of PV-10 against relapsed and refractory neuroblastoma published in OncoTargets and Therapy
Here's why yesterday's news is significant:
http://www.pharmexec.com/are-right-drugs-getting-faster-fda-approval-0
How does this stock get off blue sky list?
NEWS -- PROVECTUS ANNOUNCES GRANT OF ORPHAN DRUG DESIGNATION IN U.S. TO PV-10 FOR TREATMENT OF OCULAR MELANOMA
KNOXVILLE, TN, Feb. 12, 2019 (GLOBE NEWSWIRE) -- Provectus (PVCT) today announced that orphan drug designation (ODD) status was granted by the U.S. Food and Drug Administration (FDA) to small molecule oncolytic immunotherapy PV-10 for the treatment of ocular melanoma (to include all melanoma disease affecting the eye and orbit). Intratumoral injection of small molecule oncolytic immunotherapy PV-10 can yield immunogenic cell death (ICD) in solid tumor cancers and stimulate tumor-specific reactivity in circulating T cells.(1-4)
Ocular melanoma is a general category of melanoma disease affecting the eye and orbit. Its most common form, uveal melanoma, is an intraocular affliction originating in melanocytes in the iris, ciliary body, or choroid. Together with melanomas that form in the conjunctiva, cornea, retina, and orbit, these melanomas constitute ocular melanoma. Approximately half of ocular melanoma patients develop metastatic disease despite successful treatment of their primary tumors. Metastatic disease has historically been, and remains, generally fatal.
Preliminary data from Provectus’ uveal melanoma expansion cohort of its Phase 1 “basket study” of PV-10 for the treatment of cancers metastatic to the liver were presented in a poster presentation at the 15th International Congress of the Society for Melanoma Research (SMR 2018 Congress) in late-2018: a total of four patients had received PV-10 for at least one uveal melanoma liver tumor, two patients had received a second round of PV-10 treatment to an additional liver tumor, and one patient initiated standard of care immunotherapy (Opdivo® + Yervoy®) between PV-10 treatments; treatment-related adverse events were consistent with established patterns; and, tumor reduction was observed in 5 of 6 PV-10-injected tumors. A copy of the poster presentation is available on the Company’s website.
The FDA grants ODD status to medicines intended for the treatment, diagnosis or prevention of rare diseases or disorders that affect fewer than 200,000 people in the U.S. ODD status qualifies companies for benefits that include seven years of market exclusivity following marketing approval, tax credits on U.S. clinical trials, eligibility for orphan drug grants, and waiver of certain administrative fees.
ODD status previously was granted to PV-10 for the treatments of metastatic melanoma in 2006, hepatocellular carcinoma in 2011, and neuroblastoma in 2018.
About PV-10
Provectus’ lead investigational oncology drug, PV-10, the first small molecule oncolytic immunotherapy, can induce immunogenic cell death. PV-10 is undergoing clinical study for adult solid tumor cancers, like melanoma and cancers of the liver, and preclinical study for pediatric cancers.
About Provectus
Provectus Biopharmaceuticals, Inc. (Provectus or the Company) is a clinical-stage biotechnology company leading the development of a new class of drugs based on halogenated xanthenes, which are chemical small molecules. Information about the Company’s clinical trials can be found at the NIH registry, http://www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at http://www.provectusbio.com.
References
1. Wachter et al. Functional Imaging of Photosensitizers using Multiphoton Microscopy. Proceedings of SPIE 4620, 143, 2002.
2. Liu et al. Intralesional rose bengal in melanoma elicits tumor immunity via activation of dendritic cells by the release of high mobility group box 1. Oncotarget 7, 37893, 2016.
3. Qin et al. Colon cancer cell treatment with rose bengal generates a protective immune response via immunogenic cell death. Cell Death and Disease 8, e2584, 2017.
4. Liu et al. T cell mediated immunity after combination therapy with intralesional PV-10 and blockade of the PD-1/PD-L1 pathway in a murine melanoma model. PLoS One 13, e0196033, 2018.
Trademarks
OPDIVO® and YERVOY® are registered trademarks of Bristol-Myers Squibb, New York, New York, U.S.A.
FORWARD-LOOKING STATEMENTS
This release contains “forward-looking statements” as defined under U.S. federal securities laws. These statements reflect management's current knowledge, assumptions, beliefs, estimates, and expectations and express management's current views of future performance, results, and trends and may be identified by their use of terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “will,” and other similar terms. Forward-looking statements are subject to a number of risks and uncertainties that could cause our actual results to materially differ from those described in the forward-looking statements. Readers should not place undue reliance on forward-looking statements. Such statements are made as of the date hereof, and we undertake no obligation to update such statements after this date.
Risks and uncertainties that could cause our actual results to materially differ from those described in forward-looking statements include those discussed in our filings with the Securities and Exchange Commission (including those described in Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2017).
Contact:
Provectus Biopharmaceuticals, Inc.
Tim Scott, Ph.D.
President
Phone: (866) 594-5999
New Orphan Drug Designation for PVCT today 02/11/2019
https://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=670618
$PVCT 1.34 Million shares traded today vs. 134k average daily volume for last 10 days
New Orphan Drug Designation for PVCT today 02/11/2019
https://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=670618
$PVCT 1.34 Million shares traded today vs. 134k average daily volume for last 10 days
New Orphan Drug Designation for PVCT today 02/11/2019
https://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=670618
$PVCT 1.34 Million shares traded today vs. 134k average daily volume for last 10 days
Hope springs eternal, but having someone know something usually means more than $70K traded, never know though...
What's going on here? Nice volume coming in. Perhaps someone knows something brewing.
Watch them lining up on the bid at .056
Just slap the ask!
Short squeeze!!!!.
Northbound to Greenland
Stop smoking the crack
Can this stock do a LOXO move?
Wow trading in the $230s now
Yes, green again
Haha yeah right
|
Followers
|
185
|
Posters
|
|
|
Posts (Today)
|
0
|
Posts (Total)
|
9952
|
|
Created
|
08/09/02
|
Type
|
Free
|
| Moderators | |||
.png)

Provectus Biopharmaceuticals is investigating new therapies for the treatment of skin cancer and liver cancer. Provectus investigational oncology drug, PV-10, is an ablative immunotherapy under investigation in solid tumor cancers. The Company has received orphan drug designations from the FDA for its melanoma and hepatocellular carcinoma indications. PH-10, its topical investigational drug for dermatology, is undergoing clinical testing for psoriasis and atopic dermatitis. Provectus has completed Phase 2 trials of PV-10 as a therapy for metastatic melanoma, and of PH-10 as a topical treatment for psoriasis. In addition, Provectus has begun a Phase 3 trial as a therapy for metastatic melanoma. Information about these and the Company's other clinical trials can be found at the NIH registry, www.clinicaltrials.gov. For additional information about Provectus, please visit the Company's website at www.provectusbio.com.
The red synthetic dye Rose Bengal is shown in a bottle at Provectus Pharmaceuticals, Inc.
CURRENT PVCT PIPELINE (as of May 23, 2017)
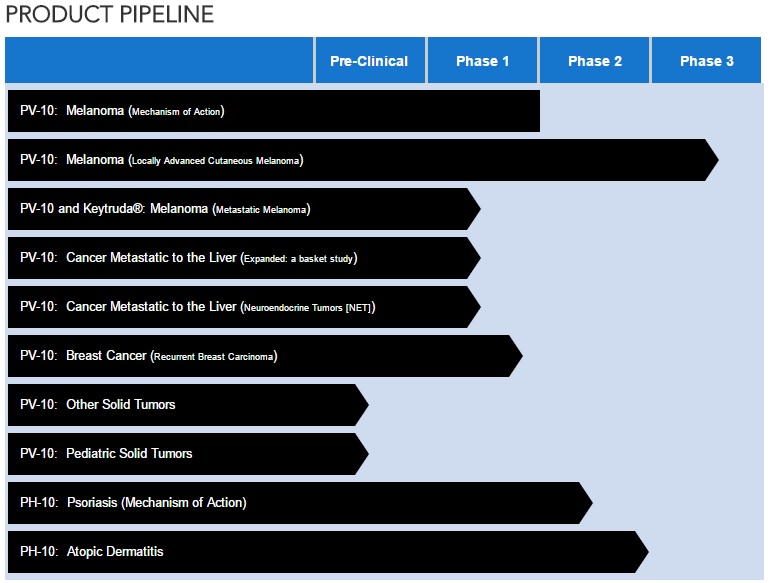
--------------------------------------------------------------
Charts and Technical:
PVCT 6 months chart:
http://stockcharts.com/h-sc/ui?s=PVCT&p=D&yr=0&mn=6&dy=0&id=p03754661229
Technical analysis:
http://www.stockta.com/cgi-bin/analysis.pl?symb=PVCT&num1=7&cobrand=&mode=stock
PVCT News and Analysis:
PVCT News Blog with the latest news and analysis:
http://provectuspharmaceuticalsinc.blogspot.ca/p/news.html
CONNECTING THE DOTS - CURRENT NEWS PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/p/current-news_22.html
CONNECTING THE DOTS - BLOG PAGE
http://provectuspharmaceuticalsinc.blogspot.ca/
PVCT News at OTC
http://www.otcmarkets.com/stock/PVCT/news
CLINICAL TRIALS Updates and Info:
| Short Interest | 167,323 (69.16%) Apr 13, 2017 |
| Significant Failures to Deliver | No |
| Volume | |
| Day Range: | |
| Bid Price | |
| Ask Price | |
| Last Trade Time: |
